Abstract
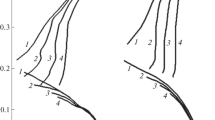
The polarization curves of low carbon steel in deaerated carbonate bicarbonate buffers (pH 10.3) show three distinct regions: (a) an active region characterized by a Tafel slope of 125 ± 5 mV which is independent of the chloride concentration, (b) a gradual active to passive transition, and (c) a broad passive region. The addition of NaCl has a stronger effect on the passive than on the active region. For NaCl concentrations of 0.1 to 2 M, NaCl has only a small promoting effect (a reaction order of 0.1) on the anodic dissolution of the steel in the active region. Measurements of electrochemical impedance under free corrosion conditions confirm the above findings. The results are explained in terms of the more favourable formation of an iron hydroxy-carbonate complex (called green rust carbonate) than the formation of the corresponding green rust chloride complex. Passivity is attributed to the formation of a protective film containing both FeOOH and Fe2O3. It deteriorates with increasing chloride concentration and potential and improves with increase of buffer concentration. Evidence is also presented for metastable pitting, particularly in the presence of the lower chloride concentration, for example, 0.1, 0.2 and 0.5 M NaCl.
Similar content being viewed by others
References
L. Garverik (Ed.), Corrosion in Petroleum Production Operations, in ‘Corrosion in the Petrochemical Industry', (ASM International, Metals Park, OH, 1994), p. 279.
L.E. Newton and R.H. Hausler, (Eds), Houston, CO2 Corrosion in Oil and Gas Production', (NACE, Houstan, TX 1984).
Y.J. Tan, S. Bailey, B. Kinsella and A. Lowe, J. Electrochem. Soc.,147 (2000) 530.
C. de Waard, U. Lotz and D.E. Milliams, Corrosion 47 (1991) 976.
Z. Xia, K.C. Chou and Z. Szklarska-Smialowska, Corros. Sci. 45 (1989) 636.
R.N. Tuttle and R.D. Kane, ‘H 2S Corrosion in Oil and Gas Production: A Compilation of Classic Papers’ (NACE, Houson, TX 1981).
L. Bertolini, F. Bolzani, T. Pastore and P. Pedeferri, Br. Corr. J. 31 (1996) 218.
G. Nakayama, Y. Fukaya and M. Akashi, in ‘Electrochemical Synthesis and Modi.cation of Materials', Material Research Society Symposia (MRS) Proceedings, Vol. 451, (MRS, Pittsburg,1997), p. 567.
R.N. Parkins, C.S. O'Dell and R.R. Fessler, Corros. Sci. 24 (1984)34.
M. Odziemkowski, J. Flis and D.E. Irish, Electrochim. Acta 39 (1994) 2225.
J.K. Heuer and J.F. Stubbins, Corros. Sci. 41 (1999) 1231.
J.M. Blengino, M. Keddam, J.P. Kabbe and L. Robbiola, Corros.Sci. 37 (1995) 621.
L.J. Simpson and C.A. Melendres, J. Electrochem, Soc. 143 (1996)2146.
E.B. Castro, S.G. Real, R.H. Milocco and J.R. Vilche, Electrchim.Acta 36 (1991) 117.
L.J. Oblonsky and T.M. Devine, J. Electrochem. Soc. 144 (1997) 1252.
G. Fierro, G.M. Ingo and F. Mancia, Corrosion 45 (1989) 814.
M. Abdelmoula, Ph. Refait, S.H. Drissi, J.P. Mihe and J.M.R. Genin, Corros. Sci. 38 (1996) 623.
S.L. Hirnyi, Mater. Sci. (Russia) 37 (2001) 87.
Y.F. Cheng and J.L. Luo, J. Electrochem. Soc. 146 (1999) 970.
B.R. Linter and G.T. Burstein, Corros. Sci. 41 (1999) 117.
E.B. Castro, J.R. Vilche and A.J. Arvia, Corros. Sci. 32 (1991) 37.
J. O'M Bockris and S.U. Khan, ‘Surface Electrochemistry: A Molecular Level Approach’ (Plenum Press, New York, 1993), p. 756.
W.J. Lorenz and K.E. Heusler, Anodic Dissolution of Iron Group Metals, in F. Mans.eld (Ed.), ‘Corrosion Mechanism', (Marcel Dekker, New York, 1987), p. 1.
D.M. Drazic, The electrochemistry of iron in an active state, in J.O'M. Bockris and B.E. Conway, (Ed.), ‘Modern Aspects of Electrochemistry', Vol. 19, (Plenum Press, New York, 1989), p. 69.
M. Keddam, Anodic dissolution, in P. Marcus and J. Oudar, (Ed.), ‘Corrosion Mechanisms in Theory and Practice’, (Marcel Dekker, New York, 1995), p. 55.
K.E. Heusler, in A.J. Bard (Ed.), ‘Encyclopedia of the Electrochemistry of the Elements’ Vol. 9A, (Marcel Dekker, New York,1982), p. 230.
Ph. Refait and J.M.R. Genin, Corros. Sci. 34 (1993) 797.
S.H. Drissi, Ph. Refait, M. Abdelmoula and J.M.R. Genin, Corros.Sci. 37 (1995) 2025.
J.M.R. Genin, A.A. Olowe, Ph. Refait and L. Simon, Corros. Sci. 38 (1996) 1751.
Ph. Refait, S.H. Drissi, J. Pytkiewicz and J.M.R. Genin, Corros.Sci. 39 (1997) 1710.
Ph. Refait, M. Abdelmoula and J.M.R. Genin, Corros. Sci. 40 (1998) 1560.
J. Stewart, P.H. Balkwill and D.E. Williams, Corros. Sci. 36 (1994)1213.
G.S. Frankel, J. Electrochem Soc. 45 (1998) 2186.
Z. Szklarska-Smialowska, ‘Pitting Corrosion of Metals’ (NACE, Houston, TX, 1986), p. 204 and 347.
D. Sazou, A. Diamantopoulou and M. Pagitsas, J. Electroanal.Chem. 499 (2000) 1.
L.F. Gar.as-Mesias and J.M. Sykes, Corros. Sci. 41 (1999) 959.
T.T. Lunt, S.T. Pride, J.R. Scully, J.L. Hudson and A.S. Mikhailov, J. Electrochem. Soc. 144 (1997) 1620; B. Wu, J.R. Scully, J.L. Hudson and A.S. Mikhailov, Ibid 144 (1997) 1614.
Y. Zuo, H. Wang, J. Zhao and J. Xiong, Corros. Sci. 44 (2002) 13.
Y.F. Cheng, M. Wilmott and J.L. Lou, Br. Corr. J. 34 (1999)280.
Carmel B. Breslin, Digby D. Macdonald, Janusz Sikora and Elzbieta Sikora, Electrochim. Acta 42 (1997) 127.
P.C. Pistorius and G.T. Burstein, Corros. Sci. 36 (1994) 525; G.T. Burstein, P.C. Pistorius and S.P. Mattin, Ibid 35 (1993) 57.
G.L. Makar and D. Tromans, Corrosion 52 (1996) 250.
R.J. Chen and K. Nobe, J. Electrochem. Soc. 119 (1972) 1457.
A.J. Bard and L.R. Faulkner, ‘Electrochemical Methods: Fundamentals and Applications', (Wiley, New York, 1980), p. 218.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Al-Kharafi, F., Ateya, B. & Abdallah, R. Electrochemical behaviour of low carbon steel in concentrated carbonate chloride brines. Journal of Applied Electrochemistry 32, 1363–1370 (2002). https://doi.org/10.1023/A:1022684930409
Issue Date:
DOI: https://doi.org/10.1023/A:1022684930409