Abstract
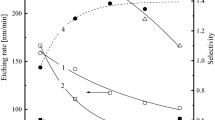
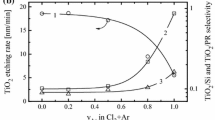
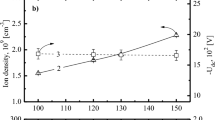
The drift of TEOS etch rate has been observed during MERIE oxide etch for the damascene process. The etch rate typically fluctuates between 5300 Å/min and 6000 Å/min. Studies using fluorocarbon-based chemistry show a normal TEOS etch rate when the chamber wall is heavily coated with polymer deposition. On the other hand, a lower etch rate appears when the chamber has less deposition. Hysteresis behavior has been observed during the etch rate of TEOS, as well as emission intensity trends of F, CF x (x=1~3), and SiF. From the observed emission intensity variation of F, CF x , and SiF, a model is proposed to explain the impact of chamber wall polymer deposition on the etch rate of TEOS. This model includes a mechanism of etch rate enhancement by embedding oxygen in the chamber wall polymer. From the correlation between etch rate and emission intensity, it clearly shows that F is directly responsible for the etch of TEOS. Compared to F, CF x plasma chemistry has a closer link to chamber wall polymer formation, but contributes less in the etch of TEOS.
Similar content being viewed by others
REFERENCES
D. M. Manas and D. L. Flamm, eds., Plasma Etching, An Introduction, Academic Press, San Diego (1989).
S. M. Rossnagel, J. J. Cuomo, and W. D. Westwood, eds., Handbook of Plasma Processing Technology: Fundamentals, Etching, and Surface Interactions, Noyes Publications, Park Ridge, New Jersey (1990).
C. J. Mogab, A. C. Adams, and D. L. Flamm, J. Appl. Phys. 49, 3796 (1978).
R. A. Heinecke, Solid State Electron. 18, 1146 (1975).
S. Joyce, J. G. Langan, and J. I. Steinfeld, J. Chem. Phys. 88, 2027 (1988).
R. D'Agostino, F. Cramarossa, S. De Benedictis, and G. Ferraro, J. Appl. Phys. 52, 1259 (1981).
H. Sugai, K. Nakamura, Y. Hikosaka, and M. Nakamura, J. Vac. Sci. Technol. A13, 887 (1995).
C. J. Liu, S. Lee, S. K. Tai, Y. C. Tien, C. F. Hsu, and J. Su, Plasma Processing XII, 193rd Meeting of the Electrochemical Society, San Diego, May 4–8 (1998).
T. Akimoto, S. Furuoya, K. Harashima, and E. Ikawa, Proc. 10th Symp. Plasma Process., G. S. Mathad and D. W. Hess, eds., Vol. 94-20 (1994), p. 311.
T. Akimoto, S. Furuoya, K. Harashima, and E. Ikawa, Jpn. J. Appl. Phys. 33, Pt. 1, No. 4B, 2151 (1994).
D. S. King, P. K. Schenck, and J. C. Stephenson, J. Mol. Spectrosc. 78, 1 (1979).
W. R. Harshbarger, R. A. Porter, T. A. Miller, and P. Norton, Appl. Spectrosc. 31, 201 (1977).
P. K. Carroll and T. P. Grennar, J. Phys. B3, 865 (1970).
M. Suto and N. Washida, J. Chem. Phys. 78, 1012 (1983).
C. Larrieu, M. Chaillet, and A. Dargelos, J. Chem. Phys. 96, 3732 (1992).
S. Dzioba and H. M. Naguib, J. Appl. Phys. 53, 4389 (1982).
G. Herzberg, Molecular Spectra and Molecular Structure: Constants of Diatomic Molecules, Vol 4, Academic Press, New York, (1979).
V. M. Donnelly, D. L. Flamm, W. C. Dautremont-Smith, and D. J. Werder, J. Appl. Phys. 55, 242 (1984).
G. S. Oehrlein, S. W. Robey, and J. L. Lindstrom, Appl. Phys. Lett. 52, 1170 (1988).
M. L. Burke, W. L. Dimpfl, P. M. Sheaffer, P. F. Zittle, and L. S. Bernstein, J. Chem. Phys. 100, 138 (1996).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lee, S., Tien, YC. & Hsu, CF. Direct Spectroscopic Evidence of the Influence of Chamber Wall Condition on Oxide Etch Rate. Plasma Chemistry and Plasma Processing 19, 285–298 (1999). https://doi.org/10.1023/A:1021699826946
Issue Date:
DOI: https://doi.org/10.1023/A:1021699826946