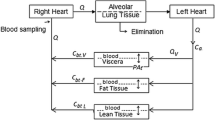
Abstract
Conventional pharmacokinetic (PK) concepts fail to describe the long-term pharmacokinetics of the extremely cationic amphiphilic drug amiodarone. A nonclassical model based on the phenomenon of trapping at tissue binding sites with very long release times is presented, which implies that a volume of distribution and a steady-state level cannot be defined. In agreement with clinical PK data available in the literature, the model well describes not only single-dose disposition curves but also the persistently increasing plasma concentration–time curve during long-term treatment (up to 5 years) and the washout curve following cessation of therapy. The novel aspect is a long-tailed tissue residence time distribution which is incorporated into a recirculatory model leaving the initial distribution process and the clearance concept unchanged. The underlying theoretical approach, which is known as “strange or anomalous” kinetics in physical sciences, and the fractal scaling property of the model may enhance our understanding of the PK of extremely hydrophobic xenobiotics.
Similar content being viewed by others
REFERENCES
B. N. Singh. Amiodarone: the expanding antiarrhythmic role and how to follow a patient on chronic therapy. Clin. Cardiol. 20:608–618 (1997).
P. C. Adams, D. W. Holt, G. C. A. Storey, A. R. Morley, J. Callaghan, M. R. C. Path, and R. W. F. Campbell. Amiodarone and its desethyl metabolite: tissue distribution and morphologic changes during long-term therapy. Ther. Prev. Pharmacol. 72:1064–1075 (1985).
D. W. Holt, G. T. Tucker, P. R. Jackson, and G. C. A. Storey. Amiodarone pharmacokinetics. Am. Heart J. 106:840–847 (1983).
R. Candinas, J. Frielingsdorf, H. R. Ha, T. Carrel, M. Turina, and F. Follath. Myocardial amiodarone concentrations after short-and long-term treatment in patients with end-stage heart failure. Eur. J. Clin. Pharmacol. 53:331–336 (1998).
R. Latini, G. Tognoni, and R. E. Kates. Clinical pharmacokinetics of amiodarone. Clin. Pharmacokin. 9:136–156 (1984).
M. Weiss. Generalizations in linear pharmacokinetics using properties of certain classes of residence time distributions. I. Log-convex drug disposition curves. J. Pharmacokin. Biopharm. 14:635–657 (1986).
G. Pfister and H. Scher. Dispersive (non-Gaussian) transient transport in disordered solids. Adv. Phys. 27:747–798 (1978).
M. F. Shlesinger, G. M. Zaslavsky, and J. Klafter. Strange kinetics. Nature 363:31–37 (1978).
M. Weiss and W. Förster. Pharmacokinetic model based on circulatory transport. Eur. J. Clin. Pharmacol. 16:287–293 (1979).
M. Weiss, G. H. Hübner, G. I. Hübner, and W. Teichmann. Effects of cardiac output on disposition kinetics of sorbitol: recirculatory modelling. Br. J. Clin. Pharmacol. 41:261–268 (1996).
M. Weiss and M. S. Roberts. Tissue distribution kinetics as determinant of transit time dispersion of drugs in organs: Application of a stochastic model to the rat hindlimb. J. Pharmacokin. Biopharm. 24:173–196 (1996).
J.-P. Bouchaud and A. Georges. Anomalous diffusion in disordered media: statistical mechanisms, models and physical applications. Phys. Report 195:127–193 (1990).
M. Gitterman and G. H. Weiss. Generalized theory of the kinetics of tracers in biological systems. Bull. Math. Biol. 56:171–186 (1994).
G. T. Tucker, P. R. Jackson, G. C. A. Storey, and D. W. Holt. Amiodarone disposition: polyexponential, power and gamma functions. Eur. J. Clin. Pharmacol. 26:655–656 (1984).
J. B. Bassingthwaighte, L. S. Liebovich, and B. J. West. Fractal Physiology. Oxford University Press, New York, 1994.
J. B. Bassingthwaighte and D. A. Beard. Fractal 15O-labeled water washout from the heart. Circ. Res. 77:1212–1220 (1995).
L. Bass. Heterogeneity within observed regions: physiologic basis and effects on estimation of rates of biodynamic processes. Circulation 72:47–52 (1985).
L. Bass, J. Aisbett, and A. J. Bracken. Asymptotic forms of tracer clearance curves: theory and applications of improved extrapolations. J. Theorel. Biol. 111:755–785 (1984).
G. H. Weiss, R. E. Goans, M. Gitterman, S. A. Abrams, N. E. Vieira, and A. L. Yergey. A non-Markovian model for calcium kinetics in the body. J. Pharmacokin. Biopharm. 22:367–379 (1994).
J. H. Matis and T. E. Wehrly. A general approach to non-Markovian compartmental models. J. Pharmacokin. Biopharm. 26:437–456 (1998).
M. Weiss. Pharmacokinetics in organs and the intact body: model validation and reduction. Eur. J. Pharm. Sci. 7:119–127 (1998).
S. A. Gross and P. Somani. Amiodarone-induces ultrastructural changes in canine myocardial fibers. Am. Heart J. 112:771–779 (1998).
H. Lüllmann, R. Lüllmann-Rauch, and O. Wassermann. Drug-induced phospholipidoses. CRC Crit. Rev. Toxicol. 4:185 (1975).
L. G. Herbette, M. Trumbore, D. W. Chester, and A. M. Katz. Possible molecular basis for the pharmacokinetics and pharmacodynamics of three membrane-active drugs: propranolol, nimodipine and amiodarone. J. Mol. Cell. Cardiol. 20:373–378 (1988).
J. S. Beck and A. Rescigno. Calcium kinetics: the philosophy and practice of science. Phys. Med. Biol. 15:566–567 (1970).
S. D. Beder, M. H. Cohen, and G. BenShachar. Time course of myocardial amiodarone uptake in the piglet heart using a chronic animal model. Pediat. Cardiol. 19:204–211 (1998).
C. I. Haffajee, J. C. Love, A. T. Canada, L. J. Lesko, G. Asdourian, and J. S. Alpert. Clinical pharmacokinetics and efficacy of amiodarone for refractory tachyarrhythmias. Circulation 67:1347–1355 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Weiss, M. The Anomalous Pharmacokinetics of Amiodarone Explained by Nonexponential Tissue Trapping. J Pharmacokinet Pharmacodyn 27, 383–396 (1999). https://doi.org/10.1023/A:1020965005254
Published:
Issue Date:
DOI: https://doi.org/10.1023/A:1020965005254