Abstract
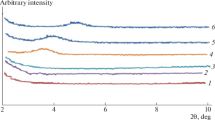
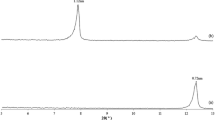
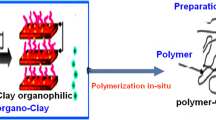
The preparation of new hybrid nanocomposites, by emulsion polymerization of styrene in the presence of a reactive organophilic clay, and their characterization are described. The reactive organophilic clay is synthesized by exchanging the inorganic cations in the interlayer structure of a natural clay with the quaternary salt of the aminomethylstyrene. This salt is prepared by a Gabriel reaction starting from the (chloromethyl)styrene. The polymeric matrix of the resulting materials was found to be constituted by polystyrene homopolymer and by a blocky copolymer of styrene and quaternary salt of the (aminomethyl)styrene units. The glass transition temperature of the nanocomposites increase as the percentage of organophilic clay increases, although the average molar masses of the polymeric matrix decrease, because of termination and/or chain-transfer reactions caused by the organophilic clay during the polymerization. Accordingly, the inherent reinforcing action due to the presence of the reactive organophilic clay is higher than the observed one.
Similar content being viewed by others
References
J. A. MANSON and L. H. SPERLING, in “Polymer Blends and Composites” (Plenum Press, New York, London, 1976).
B. K. G. THENG, Clay Miner. 18 (1970) 357.
M. AJAYAN, O. STEPHAN, C. COLLIEX and D. TRAUTH, Science 265 (1994) 1212.
H. GOLDEN, H. DENG, F. J. DISALVO, J. M. J. FRÉCHET and P. M. THOMPSON, ibid. 268 (1995) 1463.
L. N. LEWIS, Chem. Rev. 93 (1993) 2693.
P. COUVREUR and C. VAUTHIER, J. Control. Release 17 (1991) 187.
A. C. D. NEWMAN, in “Chemistry of Clays and Clay Minerals, Mineralogical Society Monograph no 6” (Longman Scientific and Technical, London, 1987).
M. J. WILSON, in “A Handbook of Determinative Methods for Clay Minerals” (Blackie, Glasgow, London, 1987).
R. A. VAIA, R. K. TEUKOLSKY and E. P. GIANNELIS, Chem. Mater. 5 (1994) 1017.
D. H. SOLOMON and D. G. HAWTHORNE, in “Chemistry of Pigments and Fillers” (Krieger, Malabar, FL, 1991).
P. M. MESSERSMITH and S. I. STUPP, J. Mater. Res. 7 (1992) 1395.
J. WU and M. M. LERNER, Chem. Mater. 5 (1993) 838.
J. T. LANDRY, B. K. COLTRAIN, M. L. LANDRY, J. J. FITZGERAD and V. K. LONG, Macromolecules 26 (1993) 3702.
A. BLUMSTEIN, J. Polym. Sci. Part A 3 (1965) 2653.
A. BLUMSTEIN, S. L. MALHOTRA and A. C. WATTERSON, J. Polym. Sci. Part A-2 8 (1970) 1599.
C. KATO, K. KURODA and H. TAKAHARA, Clays Clay Miner 29 (1981) 294.
M. OGAWA, K. KURODA and H. TAKAHARA, Clay Sci. 7 (1989) 243.
Y. FUKUSHIMA, A. OKADA, M. KAWASUMI, T. KURAUCHI and O. KAMIGAITO, Clay Miner 23 (1988) 27.
Y. KOJIMA, A. USUKI, M. KAWASUMI, A. OKADA and T. KURAUCHI, O. KAMIGAITO, J. Polym. Sci. Polym. Chem. 31 (1993) 983.
K. YANO, A. USUKI, A. OKADA, T. KURAUCHI and O. KAMIGAITO, J. Polym. Sci. 31 (1993) 2493.
R. A. VAIA, H. ISHII and E. P. GIANNELIS, Chem. Mater. 5 (1993) 1694.
L. BIASCI, M. AGLIETTO, G. RUGGERI and F. CIARDELLI, Polymer 35(15) (1994) 3296.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Laus, M., Camerani, M., Lelli, M. et al. Hybrid nanocomposites based on polystyrene and a reactive organophilic clay. Journal of Materials Science 33, 2883–2888 (1998). https://doi.org/10.1023/A:1017550206613
Issue Date:
DOI: https://doi.org/10.1023/A:1017550206613