Abstract
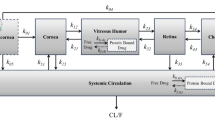
Methods of calculating the area under the concentration–time curve and the associated standard error are proposed for studies in which each animal contributes one independent data point to a pool of data. This approach can be used for data analysis in bioequivalence studies employing tissue sampling compartments. Application of this method indicated that an azone-containing ophthalmic formulation of levobunolol did not produce better ocular bioavailability than a formulation containing no penetration enhancer.
Similar content being viewed by others
REFERENCES
American Pharmaceutical Association—Academy of Pharmaceutical Sciences. Guidelines for Biopharmaceutical Studies in Man, Am. Pharm. Assoc. Acad. Pharm. Sci., Washington, D.C., 1972.
D. D-S. Tang-Liu, S. S. Liu, and R. J. Weinkam. Ocular and systemic bioavailability of ophthalmic flurbiprofen. J. Pharmacokin. Biopharm. 12:611–626 (1984).
J. M. Conrad and J. R. Robinson. Aqueous chamber drug distribution volume measurement in rabbits. J. Pharm. Sci. 66:219–224 (1977).
K. Himmelstein, I. Guvenir, and T. F. Patton. Preliminary pharmacokinetic model of pilocarpine uptake and distribution in the eye. J. Pharm. Sci. 67:603–606 (1978).
V. F. Smolen and R. D. Shoenwald. Drug absorption analysis from pharmacological data. I. Method and confirmation exemplified for the mydriatic drug tropicamide. J. Pharm. Sci. 60:96–103 (1971).
M. Eller and R. D. Schoenwald. Determination of ethoxzolamide in the iris/ciliary body of the rabbit eye by high performance liquid chromatography. Comparison of tissue levels following intravenous and topical administrations. J. Pharm. Sci. 73:1261–1264 (1984).
L. B. Sheiner, B. Rosenberg, and V. V. Marathe. Estimation of population characteristics of pharmacokinetic parameters from routine clinical data. J. Pharmacokin. Biopharm. 5:445–479 (1977).
D. D.-S. Tang-Liu, S. S. Liu, J. Neff, and R. Sandri. Levobunolol disposition after ophthalmic doses to rabbits. J. Pharm. Sci. 76:780–783.
D. D.-S. Tang-Liu, S. S. Liu, J. Richman, and R. J. Weinkam. HPLC quantitation of levobunolol and its metabolite, dihydrolevobunolol, in biological fluids. J. Liq. Chromatogr. 9:2237–2252.
F. E. Satterthwaite. Synthesis of variance. Psychometrika 6:309–316 (1941).
D. J. Best and J. C. W. Rayner. Welch's approximate solution for the Behrens-Fisher problem. Technometrics 29:205–210 (1987).
G. A. Milliken and D. E. Johnson. Analysis of Messy Data, Vol. 1. Designed Experments, Lifetime Learning, Belmont, Calif., 1984, p. 19.
R. D. Schoenwald and H.-S. Huang. Corneal penetration behavior of β-blocking agents. I. Physicochemical factors. J. Pharm. Sci. 72:1266–1271 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tang-Liu, D.DS., Burke, P.J. The Effect of Azone on Ocular Levobunolol Absorption: Calculating the Area Under the Curve and Its Standard Error Using Tissue Sampling Compartments. Pharm Res 5, 238–241 (1988). https://doi.org/10.1023/A:1015997814379
Issue Date:
DOI: https://doi.org/10.1023/A:1015997814379