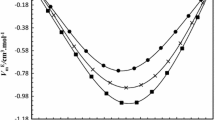
Abstract
The refractive indices of mixtures of ethanol+(1-propanol, 2-propanol, 1,2-propanediol, and 1,3-propanediol) have been measured at temperatures of 288.15, 293.15, 298.15, and 303.15 K at atmospheric pressure. In this work, an analysis of the mixing properties gives valuable information on packing trends, showing the interactions of ethanol in a hydroxilic environment. From the measured refractive indices on mixing, the parameters of a set of analytical expressions, which represent the composition and temperature dependences of the derived property, are reported. The excess molar volumes are estimated using derivations of the Heller equation, which are dependent on the refractive indices on mixing; accurate values are obtained for the alcohol mixtures, using literature data. The effect of temperature on the mixing and derived properties is analyzed in terms of the relative position of hydroxyl groups, the alkyl chain length, and the available hydrogen-bond interactions between solute and solvent molecules.
Similar content being viewed by others
REFERENCES
M. Iglesias, B. Orge, G. Marino, and J. Tojo, J. Chem. Eng. Data 44:661 (1999).
C. Gonzalez, M. Iglesias, J. Lanz, and J. M. Resa, Thermochim. Acta 328:277 (1999).
M. J. Fontao and M. Iglesias, Phys. Chem. Liq. 38:623 (2000).
S. Y. Gerlsma, J. Biol. Chem. 243:957 (1968).
A. R. Fersht, Enzyme Structure and Mechanism (W. H. Freeman, Reading, UK, 1977).
T. Arakawa and S. N. Timasheff, Biochemistry 21:6536 (1982).
J. F. Back, D. Oakenfull, and B. Smith, J. Am. Chem. Soc. 18:5191 (1979).
O. Redlich and A. Kister, Ind. Eng. Chem. 40:345 (1948).
M. Iglesias, B. Orge, and J. Tojo, Fluid Phase Equil. 126:203 (1996).
W. Heller, J. Phys. Chem. 69:1123 (1965).
TRC Thermodynamic Tables (Thermodynamics Research Center, Texas A&M University, College Station, Texas, 1994).
J. A. Riddick, W. B. Bunger, and T. K. Sakano, Organic Solvents, Physical Properties and Methods of Purification, 4th ed. (Wiley–Interscience, New York, 1986).
L. Alburquerque, C. Ventura, and R. Gonçalves, J. Chem. Eng. Data 41:685 (1996).
P. Bevington, Data Reduction and Error Analysis for the Physical Sciences (McGraw–Hill, New York, 1969).
D. W. Marquardt, J. Soc. Indust. Appl. Math. 2:431 (1963).
B. Orge, M. Iglesias, G. Marino, B. E. de Cominges, M. M. Piñeiro, and J. Vijande, J. Therm. Anal. Calorimetry 56:381 (1999).
M. Iglesias, B. Orge, and J. Tojo, J. Chem. Thermodyn. 27:1161 (1995).
M. Nakata and M. Sakurai, J. Chem. Soc. Faraday Trans. I 83:2449 (1987).
G. C. Benson and H. D. Pflug, J. Chem. Eng. Data 15:382 (1970).
M. García and M. I. Paz Andrade, Anal. Quím. 70:489 (1974).
I. Ch. Wei and L. Rowley, J. Chem. Eng. Data 29:332 (1984).
J. Zielkiewicz, J. Chem. Thermodyn. 25:1243 (1993).
R. Haase and W. Tillman, Z. Phys. Chem. 196:271 (1996).
H. Lee, J. Chem. Thermodyn. 22:463 (1990).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fontao, M.J., Iglesias, M. Effect of Temperature on the Refractive Index of Aliphatic Hydroxilic Mixtures (C2–C3). International Journal of Thermophysics 23, 513–527 (2002). https://doi.org/10.1023/A:1015113604024
Issue Date:
DOI: https://doi.org/10.1023/A:1015113604024