Abstract
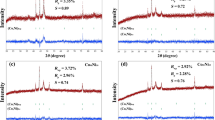
The surface oxygen distribution the active oxygen species for CO on the perovskite-type catalyst La1-xSrxCo1-xMnxO3 and its catalytic oxidation activity with CO as probe were investigated by means of XRD, TPD and XPS in a continuous flow microreactor. Results showed that different adsorbed oxygen species and lattice oxygen were distributed on the catalyst surface. Meanwhile, the surface lattice oxygen of the oxides was reacting in the course of CO oxidation. This leads to the conclusion that, when x=0.6, the catalyst shows the best oxidative activity and lower starting temperature.
Similar content being viewed by others
REFERENCES
S. Rajaadurai, J.J. Carberry, B. Li, C.B. Alcock: J. Catal., 131, 582 (1991).
M. Chen, A.J. Tench: Adv. Catal., 32, 117 (1983).
N. Yamazoe, Y. Teraoka, T. Seiyama: Chem. Lett., 1767 (1981).
T. Nakamura, M. Misono, Y. Yoneda: Bull. Chem. Soc. Jpn., 55, 394 (1982).
T. Nakamura, M. Misono, Y. Yoneda: J. Catal., 83, 51 (1983).
Li Xuyuan, Zhang Ziping, Ma Jiantai: WuLiHuaXueXueBao., 12, 502 (1996).
T. Nakamura, M. Misono, Y. Yoneda: Chem. Lett., 1589 (1981).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Li, R., Ma, J., Xu, J. et al. Studies on the Surface Oxygen Species and the Catalytic Activity for CO Oxidation of La1-xSrxCo1-xMnxO3, a Perovskite-Type Oxides Catalyst. Reaction Kinetics and Catalysis Letters 70, 363–370 (2000). https://doi.org/10.1023/A:1010365622170
Issue Date:
DOI: https://doi.org/10.1023/A:1010365622170