Abstract
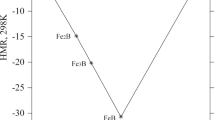
Polytherm diagram of the ternary system KCl–FeCl2 –H2 O between 0 and 70°C. Phase equilibria in the KCl–FeCl2 –H2 O system were studied over the temperature range 0–70°C by conductimetric and analytical methods.
A solubility polytherm of the system was constructed. We have observed the crystallization fields of the KCl and FeCl2 6H2 O (at 0°C), KCl and FeCl2 4H2 O (at 15, 30 and 40°C) and KCl, FeCl2 4H2 O and of a double salt KClFeCl2 2H2 O are obtained at 70°C.
Similar content being viewed by others
Références
L. S. Ruby, B. G. Zabransky and J. G. Stevens, J. Chem. Phys., 54 (1971) 4559.
R. Cohen-Adad and J. W. Lorimer, I.U.P.A.C. Solubility Data-Series, Alkali Metal and Ammonium Chlorides in Water and D2O, Pergamon Press, Oxford 1991, Vol. 47.
H. E. Boeke, Neues Jahrib. Min., 1 (1911) 48.
Wilke-Dorfurte and G. Heyne, Ber., 45 (1912) 1013.
R. Quan, P. L. Coke and S. E. Kesler, Geol. Soc. Am. Abstr. Prog., 16 (1984) 629.
M. Legrand and R. S. Paris, Bull. Soc. Chim. Fr., 11 (1967) 4283.
I. M. Chou and L. D. Phan, J. Chem. Eng. Data, 31 (1986) 154.
M. Bouchacourt, Thèse de 3ème cycle, Lyon, 1976, no 494.
A. Chretien, Ann. Chem. Fr., 12 (1929) 26.
F. A. H. Schreinemakers, J. Phys. Chem., 11 (1893) 76.
G. Charlot, Chimie Analytique Quantitative II, Ed. Masson et Cie, Paris 1974.
M. Pinta, Spectrophotométrie d'Absorption Atomique, Application à l'Analyse Chimique, Ed. Masson et Cie, Paris 1971.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Atbir, A., Aneflous, L., Marrouche, A. et al. Diagramme Polythermique Du Systeme Ternaire KCl–FeCl2–H2O Entre 0 Et 70°C. Journal of Thermal Analysis and Calorimetry 59, 893–900 (2000). https://doi.org/10.1023/A:1010178426237
Issue Date:
DOI: https://doi.org/10.1023/A:1010178426237