Abstract
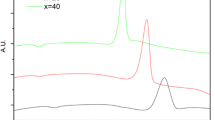
The crystallization kinetics of Ca1-xMgx(PO3)2glasses was studied for powder and bulk samples of the compositions x=0, 0.5 and 1.0 by means of DSC. The activation energy calculated by the Freedman method or the Kissinger method is smaller for the mixed glass C0.5Mg0.5(PO3)2than for the glasses with x=0 and 1.0. For most of the samples, the two-parameter Šesták-Berggren equation gives a better description of the crystallization kinetics; only for powder Ca(PO3)2and bulk Mg(PO3)2does the Johnson-Mehl-Avrami model fit better with the experimental data.
Similar content being viewed by others
References
M. Jarcho, Clin. Orthop. Rel. Res., 157 (1981) 259.
A. Bertoluzza, S. Cacciari, A. Tinti and M. Vasina, J. Mat. Sci: Materials in Medicine, 6 (1995) 76.
A. Bertoluzza, Spectroscopy of Biological Molecules, C. Sandorfy and T. Theopanides (eds.), D Reider Publ., New York 1984. p. 191.
P. Prokůpková, P. Mošner, L. Koudelka and M. Vlček, J. Mater. Sci. Lett. 33 (1998) 743.
J. Malek, Thermochim. Acta, 200 (1992) 257.
M. J. Richardson and N. G. Savill, Polymer, 16 (1975) 753.
A. G. Nord and K. B. Lindberg, Acta Chem. Scand., A29 (1975) 1.
Handbook of Chemistry and Physics, CRC Press, Inc., Boca Raton 1983.
J. Malek, Y. Messaddeq, S. Inoue and T. Mitsuhashi, J. Mater. Sci. Lett., 30 (1995) 3082.
W. Johnson and R. Mehl, Trans. Am. Inst. Min. Metall. Pet. Eng., 135 (1939) 416.
M. Avrami, J. Chem. Phys., 9 (1941) 177.
J. Šesták and M. Berggren, Thermochim. Acta, 3 (1971) 1.
H. L. Friedmann, J. Polym. Sci., C6 (1964) 183.
H. E. Kissinger, Anal. Chem., 29 (1957) 1702.
J. C. Perng, J. S. Lee, T. J. Lin and C. W. Huang, Thermochim. Acta, 177 (1991) 1.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mošner, P., Prokupková, P. & Koudelka, L. Kinetic Studies of Crystallization in Ca1-xMgx(PO3)2Glasses. Journal of Thermal Analysis and Calorimetry 54, 937–945 (1998). https://doi.org/10.1023/A:1010124811597
Issue Date:
DOI: https://doi.org/10.1023/A:1010124811597