Abstract
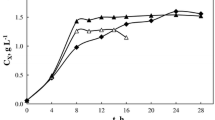
The metabolism of hybridoma cells was controlled to reduce metabolic formation in fed-batch cultures by dynamically feeding a salt-free nutrient concentrate. For this purpose, on-line oxygen uptake rate (OUR) measurement was used to estimate the metabolic demand of hybridoma cells and to determine the feeding rate of a concentrated solution of salt-free DMEM/F12 medium supplemented with other medium components. The ratios among glucose, glutamine and other medium components in the feeding nutrient concentrate were adjusted stoichiometrically to provide balanced nutrient conditions for cell growth. Through on-line control of the feeding rate of the nutrient concentrate, both glucose and glutamine concentrations were maintained at low levels of 0.5 and 0.2 mM respectively during the growth stage. The concentrations of the other essential amino acids were also maintained without large fluctuations. The cell metabolism was altered from that observed in batch cultures resulting in a significant reduction of lactate, ammonia and alanine production. Compared to a previously reported fed-batch culture in which only glucose was maintained at a low level and only a reduced lactate production was observed, this culture has also reduced the production of other metabolites, such as ammonium and alanine. As a result, a high viable cell concentration of more than 1.0 × 107 cells/mL was achieved and sustained over an extended period. The results demonstrate an efficient nutrient feeding strategy for controlling cell metabolism to achieve and sustain a high viable cell concentration in fed-batch mammalian cell cultures in order to enhance the productivity.
Similar content being viewed by others
References
Bebbington CR, Renner G, Thompson S, King D, Abrams D and Yarranton GT (1992) High level expression of a recombinant antibody from myeloma cells using a glutamine synthetase gene as an amplifiable selectable marker. Bio/technology 10: 169–175.
Bibila TA, Ranucci CS, Glazomietsky K, Buckland BC and Aunins JG (1994) Monoclonal process development using medium concentrates. Biotechnol Progress 10: 87–96.
Fleischaker RT (1982) An experimental study in the use of instrumentation to analyze metabolism and product formation in cell culture. Thesis, Massachusetts Institute of Technology, Cambridge, MA.
Frame KK and Hu W-S (1985) Oxygen uptake of mammalian cells in microcarrier culture: Response to changes of glucose concentration. Biotechnol Lett 7: 147–152.
Glacken MW, Fleischaker RJ and Sinskey AJ (1986) Reduction of waste product excretion via nutrient control: Possible strategies for maximizing product and cell yields on serum in cultures of mammalian cells. Biotechnol Bioeng 28: 1376–1389.
Hu W-S and Demain AL (1979) Regulation of antibiotic biosynthesis by utilizable carbon sources. Proc Biochem 14(9): 2–6.
Hu W-S, Dodge TC, Frame KK and Himes VB (1987) Effect of glucose and oxygen on the cultivation of mammalian cells. Dev Biol Standard 66: 279–290.
Hu W-S and Himes VB (1989) Stoichiometric considerations of mammalian cell metabolism in bioreactors. In: Fiechter A, Okada H and Tanner, RD (eds) Bioproducts and Bioprocesses, Springer-Verlag Publishers, pp. 33–45.
Hu W-S and Oberg MG (1990) Monitoring and control of animal cell bioreactors: Biochemical engineering considerations. In: Lubineicky AS (ed) Large Scale Mammalian Cell Culture Technology, Marcel Dekker, Inc., New York, NY.
Konstantinov KB, Nishio N, Seki T and Yoshida T (1991) Physiologically motivated strategies for control of the fed-batch cultivation of recombinant Escherichia coli for phenylalanine production. J Ferm Bioeng 71: 350–355.
Savinell JM and Palsson BO (1992) Network analysis of intermediary metabolism using linear optimization. II. Interpretation of hybridoma cell metabolism. J Theor Biol 154: 455–473.
Tritsch TL and Moore GE (1962) Spontaneous decomposition of glutamine in cell culture media. Exp Cell Res 28: 360–364.
Wang H, Cooney CL and Wang DIC (1977) Computer-aided baker's yeast fermentation. Biotechnol Bioeng 19: 68–86.
Xie L and Wang DIC (1994) Fed-batch cultivation of animal cells using different medium design concepts and feeding strategies. Biotechnol Bioeng 43: 1175–1189.
Zhou W and Hu W-S (1994a) On-line characterization of a hybridoma cell culture process. Biotechnol Bioeng 44: 170–177.
Zhou W and Hu W-S (1994b) Optimization of fed-batch mammalian cell culture processes. In: Galindo E and Ramirez OT (eds) Advances in Bioprocess Engineering. Proceedings, Intemational Symposium on Bioprocess Engineering, Cuernavaca, Mexico, June 20–24, 1994, Kluwer Acad. Publishers, pp. 109–114.
Zhou W, Rehm J and Hu W-S (1995) High viable cell concentration fed-batch cultures of hybridoma cells through on-line nutrient feeding. Biotechnol Bioeng 46: 579–587.
Zielke HR, Ozand PT, Tildon JT, Sevdalian DA and Cornblath M (1978) Reciprocal regulation of glucose and glutamine utilization by cultured human diploid fibroblasts. J Cell Physiol 95: 41–48.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhou, W., Rehm, J., Europa, A. et al. Alteration of mammalian cell metabolism by dynamic nutrient feeding. Cytotechnology 24, 99–108 (1997). https://doi.org/10.1023/A:1007945826228
Issue Date:
DOI: https://doi.org/10.1023/A:1007945826228