Abstract
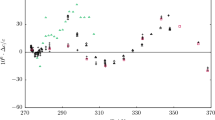
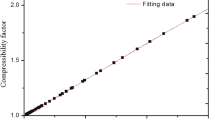
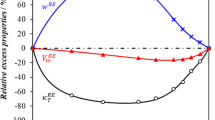
The speed of sound in gaseous hydrogen bromide (HBr) and boron trichloride (BCl3) was measured using a highly precise acoustic resonance technique. The HBr speed-of-sound measurements span the temperature range 230 to 440 K and the pressure range from 0.05 to 1.5 MPa. The BCl3 speed-of-sound measurements span the temperature range 290 to 460 K and the pressure range from 0.05 MPa to 0.40 MPa. The pressure range in each fluid was limited to 80% of the sample vapor pressure at each temperature. The speed-of-sound data have a relative standard uncertainty of 0.01%. The data were analyzed to obtain the ideal-gas heat capacities as a function of temperature with a relative standard uncertainty of 0.1%. The heat capacities agree with those calculated from spectroscopic data within their combined uncertainties. The speeds of sound were fitted with the virial equation of state to obtain the temperature-dependent density virial coefficients. Two virial coefficient models were employed, one based on the hard-core square-well intermolecular potential model and the second based on the hard-core Lennard–Jones intermolecular potential model. The resulting virial equations of state reproduced the speed-of-sound measurements to 0.01% and can be expected to calculate vapor densities with a relative standard uncertainty of 0.1%. Transport properties calculated from the hard-core Lennard–Jones potential model should have a relative standard uncertainty of 10% or less.
Similar content being viewed by others
REFERENCES
A. R. H. Goodwin and M. R. Moldover, J. Chem. Phys. 95:5236 (1991).
K. A. Gillis, Int. J. Thermophys. 18:73 (1997).
J. J. Hurly, Int. J. Thermophys. 20:455 (1999).
K. A. Gillis, Int. J. Thermophys. 15:821 (1994).
K. A. Gillis, A. R. H. Goodwin, and M. R. Moldover, Rev. Sci. Instrum. 62:2213 (1991).
J. J. Hurly, submitted for publication (1999).
D. R. Stull, Ind. Eng. Chem. 39:517 (1947).
J. F. Mathews, Chem. Rev. 72:71 (1972).
R. E. Lynn, Jr. and K. A. Kobe, Chem. Rev. 52:117 (1953).
J. E. Mayer and M. G. Mayer, Statistical Mechanics (John Wiley & Sons, New York, 1959), p. 469.
K. A. Gillis and M. R. Moldover, Int. J. Thermophys. 17:1305 (1996).
T. Kihara, Rev. Mod. Phys. 25:831 (1953).
C. G. Maitland and E. B. Smith, Chem. Phys. Lett. 22:443 (1973).
J. P. M. Trusler, Int. J. Thermophys. 18:635 (1997).
E. A. Mason and T. H. Spurling, The Virial Equation of State (Pergamaon Press, Oxford, 1969).
R. J. Dulla, J. S. Rowlinson, and W. R. Smith, Mol. Phys. 21:229 (1971).
B. M. Axilrod and E. J. Teller, J. Chem. Phys. 11:299 (1943).
L. A. Weber, Int. J. Thermophys. 15:461 (1994).
K. S. Pitzer and R. F. Curl, Jr., J. Am. Chem. Soc. 79:2369 (1957).
Tsonopolulos, AIChE J. 24:1112 (1978).
N. Van Nhu, G. A. Iglesias-Silva, and F. Kohler, Ber. Bunsenges. Phys. Chem. 93:526 (1989).
N. Van Nhu and F. Kohler, Ber Bunsenges Phys. Chem. 92:1129 (1988).
J. O. Hirschfelder, C. F. Curtiss, and R. B. Bird, Molecular Theory of Gases and Liquids (John Wiley & Sons, New York, 1954).
A. E. Schuil, Phil. Mag. 28:679 (1939).
Matheson Gas Data Book, unabridged ed. (Matheson Company, East Rutherford, NJ, 1974).
R. A. Suehla, NASA-TR-R-132 116:57, 79 (1962).
T. S. Ro, J. Kestin, and J. M. Hellemans, Physica 65 (1973).
K. Veda and K. Kigoshi, J. Inorg. Nucl. Chem. 36:989 (1974).
W. A. Wakeham, H. E. Khalifa, and J. K. Esti, J. Chem. Phys. 65:5186 (1976).
A. Eucken, Phys. Z. 14:324 (1913).
JANAF Thermochemical Tables, 2nd ed., NSRDS-NBS 37 (U.S. Department of Commerce, Washington, DC, 1971).
R. C. Reid, J. M. Prausnitz, and T. K. Sherwood, The Properties of Gases and Liquids, 3rd ed. (McGraw-Hill, New York, 1977).
K. A. Gillis, J. B. Mehl, and M. R. Moldover, Rev. Sci. Instrum. 67:1850 (1996).
BYU DIPPR801 Thermophysical Properties Database, Spring 1998 Preview Release (Brigham Young University, Provo, UT 84602).
E. W. Lemmon, M. O. McLinden, and D. G. Friend, in NIST Chemistry WebBook, NIST Standard Reference Database Number 69, W. G. Mallard and P. J. Linstrom, eds. (National Institute of Standards and Technology, Gaithersburg, MD, Nov. 1998) (http://webbook.nist.gov).
B. J. McBride, S. Gordon, and M. A. Reno, Coefficients for Calculating Thermodynamic and Transport Properties of Individual Species, NASA Report TM-8209 4513 (1993).
E. F. Westrum, D. R. Stull, and G. C. Sinke, The Chemical Thermodynamics of Organic Compounds (John Wiley and Sons, New York, 1969).
A. R. Gordon and C. Barnes, J. Chem. Phys. 1:692 (1933).
E. G. Long and K. A. Kobe, Petrol. Refin. 29:124 (1950).
J. B. Austin, J. Am. Chem. Soc. 54:3459 (1932).
JANAF Thermochemical Tables, 3rd ed., J. Phys. Chem. Ref. Data Suppl. 1:14 (1985).
Thermodynamic Properties of Individual Substances, 4th ed., Vol 2. Elements Carbon, Silicon, Germanium, Tin, Lead, and Their Compounds, Pt. 2. Tables (Hemisphere, New York, 1991).
D. Wagman, R. Schumm, V. Parker, R. Nuttall, I. Halow, W. Evans, K. Churney, and S. Bailey, J. Phys. Chem. Ref. Data Suppl. 2 (1982).
D. R. Lide (ed.), Handbook of Chemistry and Physics, 75th ed. (CRC Press, Boca Raton, FL, 1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hurly, J.J. Thermophysical Properties of Gaseous HBr and BCl3 from Speed-of-Sound Measurements. International Journal of Thermophysics 21, 805–829 (2000). https://doi.org/10.1023/A:1006627306105
Issue Date:
DOI: https://doi.org/10.1023/A:1006627306105