Abstract
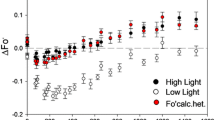
High-temperature-induced inhibition of the acceptor side of Photosystem II (PS II) was studied in tobacco thylakoids using oxygen evolution, chlorophyll a (Chl a) fluorescence and redox potential measurements. When thylakoids were heated at 2 °C/min from 25 to 50 °C, the oxygen evolving complex became inhibited between 32 and 45 °C, whereas the acceptor side of PS II tolerated higher temperatures. Variable Chl a fluorescence decreased more slowly than oxygen evolution, suggesting that transitions between some S-states occurred even after heat-induced inhibition of the oxygen evolving activity. 77 K emission spectroscopy reveals that heating does not cause detachment of the light-harvesting complex II from PS II, and thus the heat-induced increase in the initial F0 fluorescence is due to loss of exciton trapping in the heated PS II centers. Redox titrations showed a heat-induced increase in the midpoint potential of the QA/QA -) couple from the control value of –80 mV to +40 mV at 50 °C, indicating a loss of the reducing power of QA -). When its driving force thus decreased, electron transfer from QA -) to QB in the PS II centers that still could reduce QA became gradually inhibited, as shown by measurements of the decay of Chl a fluorescence yield after a single turnover flash. Interestingly, the heat-induced loss of variable fluorescence and inhibition of electron transfer from QA -) to QB could be partially prevented by the presence of 5 mM bicarbonate during heating, suggesting that high temperatures cause release of the bicarbonate bound to PS II. We speculate that both the upshift in the redox potential of the QA/QA -) couple and the release of bicarbonate may be caused by a heat-induced structural change in the transmembrane D1 or D2 proteins. This structural change may, in turn, be caused by the inhibition of the oxygen evolving complex during heating.
Similar content being viewed by others
References
Andréasson L-E, Vass I and Styring S (1995) Ca2+ depletion modifies the electron transfer on the both donor and acceptor sides in Photosystem II from spinach. Biochim Biophys Acta 1230: 155–164
Blubaugh DJ and Govindjee (1988) The molecular mechanism of bicarbonate effect at the plastoquinone reductase site of photosynthesis. Photosynth Res 19: 85–128
Berry J and Björkmann O (1980) Photosynthetic responses and adaptation to temperature in higher plant. Ann Rev Plant Physiol 31: 491–543
Bukhov NG, Sabat SC and Mohanty P (1990) Analysis of chlorophyll a fluorescence changes in weak ligh in heat treated Amaranthus chloroplasts. Photosynth Res 23: 81–87
Cao J and Govindjee (1990) Chlorophyll a fluorescence transient as an indicator of active and inactive Photosystem II in thylakoid membranes. Biochim Biophys Acta 1015: 180–188
Cheniae GM and Martin IF (1970) Site and function of manganese within Photosystem II. Role in O2 evolution and system II. Biochim Biophys Acta 197: 219–239
Coleman WJ, Govindjee and Gutowsky HS (1988) The effect of chloride on the thermal inactivation of oxygen evolution. Photosynth Res 16: 261–276
Crofts AR and Wraight CA (1983) The electrochemical domain of photosynthesis. Biochim Biophys Acta 726: 149–185
Dau H (1994) Molecular mechanisms and quantitative models of variable Photosystem II fluorescence. Photochem Photobiol 60: 1–23
De Las Rivas J and Barber J (1997) Structure and thermal stability of Photosystem II reaction centers studied by infrared spectroscopy. Biochemistry 36: 8897–8903
Diner BA (1977) Dependence of the deactivation reactions of Photosystem II on the redox state of the plastoquinone pool A varied under anaerobic conditions. Equilibria on the acceptor side of Photosystem II. Biochim Biophys Acta 460: 247–258
Diner BA, Petrouleas V and Wendoloski JJ (1991) The iron-quinone electron-acceptor complex of Photosystem II. Physiol Plant 81: 423–436
Ducruet JM and Lemoine Y (1985) Increased heat sensitivity of photosynthetic aparatus in triazine-resistant biotypes from different plant species. Plant Cell Physiol 26: 419–429
Gounaris K, Brain ARR, Quinn PJ and Williams WP (1983) Structural and functional changes associated with heat-induced phase-separations of non-bilayer lipids in chloroplast thylakoid membranes. FEBS Lett 153: 47–52
Gounaris K, Brain AP, Quinn PJ and Williams W (1984) Structural reorganization of chloroplast thylakoid membranes in response to heat stress. Biochim Biophys Acta 766: 198–208
Govindjee and Van Rensen JJS (1993) Photosystem II reaction center and bicarbonate. In: Deisenhofer J and Noris JR (eds) The Photosynthetic Reaction Center, pp 357–389. Academic Press, San Diego, CA
Johnson GN, Rutherford AW and Krieger A (1995) A change in the midpoint potential of the quinone QA in Photosystem II associated with photoactivation of oxygen evolution. Biochim Biophys Acta 1229: 202–207
Joliot A and Joliot P (1964) Etude cinétique de la réaction photochimique libérant l+oxygene au cours de la photosynthese. C R Acad Sci Paris 258: 4622–4625
Joliot P, Lavergne J and Béal D (1992) Plastoquinone compartmentation in chloroplasts. I. Evidence for domains with different rates of photo-reduction. Biochim Biophys Acta 1101: 1–12
Katoh S and San Pietro A (1967) Ascorbate-supported NADP photoreduction by heated Euglena chloroplasts. Arch Biochem Biophys 122: 144–152
Kashino Y, Koike H and Satoh K (1992) Isolation of oxygenevolving Photosystem II particles with the intact QB site. In Murata N (ed) Research in Photosynthesis, Vol II, pp 163–166. Kluwer Academic Publishers, Dordrecht, The Netherlands
Klimov VV, Baranov SV and Allakhverdiev SI (1997) Bicarbonate protects the donor side of Photosystem II against photoinhibition and thermoinactivation. FEBS Lett 418: 243–246
Krause GH and Santarius K (1975) Relative permeability of the chloroplast envelope. Planta 127: 285–299
Krieger A, Weis E and Demeter S. (1993) Low-pH-induced Ca2+ ion release in the water-splitting system is accompanied by a shift in the midpoint redox potential of the primary quinone acceptor QA. Biochim Biophys Acta 1144: 411–418
Krieger A, Rutherford AW and Johnson GN (1995) On the determination of redox midpoint potential of the primary quinone electron acceptor, QA, in Photosystem II. Biochim Biophys Acta 1229: 193–201
Porra RJ, Thompson WA and Kriedemann PE (1989) Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim Biophys Acta 975: 384–394
Rova M, Mamedov F, Magnuson A, Freriksson P-O and Styring S (1998) Coupled activation of the donor and acceptor side of Photosystem II during photoactivation of the oxygen evolving cluster. Biochemistry 37: 11039–11045
Santarius KA (1975) Site of heat sensitivity in chloroplasts and differential inactivation of cyclic and noncyclic photophoshorylation by heating. J Therm Biol 1: 101–107
Schreiber U and Armond PA (1978) Heat-induced changes of chlorophyll fluorescence in isolated chloroplasts and related heat-damage at the pigment level. Biochim Biophys Acta 502: 138–151
Weis E and Berry JS (1988) Plants and high temperature stress. In: Long SP and Noris Woodward FI (eds) Plants and Temperature, pp 329–346. The Company of Biologists Limited, Cambridge, UK
Woodbury NW, Parson WW, Gunner MR, Prince RC and Dutton, PL (1986) Radical-pair energetics and decay mechanisms in reaction centers containing anthraquinones, naphtoquinones or benzoquinones in place of ubiquinone. Biochim Biophys Acta 851: 6–22
Yamashita S and Butler WL (1968) Inhibition of chloroplasts by UV-irradiation and heat treatment. Plant Physiol 43: 2037–2040
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pospíšil, P., Tyystjärvi, E. Molecular mechanism of high-temperature-induced inhibition of acceptor side of Photosystem II. Photosynthesis Research 62, 55–66 (1999). https://doi.org/10.1023/A:1006369009170
Issue Date:
DOI: https://doi.org/10.1023/A:1006369009170