Abstract
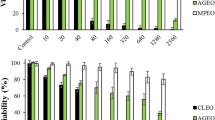
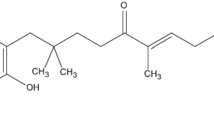
This study aimed to prepare hydrogel containing Cymbopogon citratus (DC.) Stapf, Poaceae, volatile oil encapsulated in poly (d, l-lactide-co-glycolide) nanoparticles and to evaluate its in vitro anti-herpetic activity. Polymeric nanoparticles were prepared by solvent emulsification-diffusion method and incorporated in carbomer hydrogels. In vitro release profiles for the nanogel, loaded nanoparticles and hydrogel containing free oil were evaluated by dialysis. Inhibitory activities against Herpes simplex for the formulations were investigated in Vero cells. Hydrogel was developed using nanoparticles with mean diameter of 217.1 nm and negative Zeta potential (−20.5mV). Volatile oil release profile showed a biphasic pattern with an initial faster release and subsequent sustained phase in all formulations. Nanogel strongly inhibited virus in a non-cytotoxic concentration, 42.16 times lower than free oil, 8.76 and 2.23 times than loaded nanoparticles and hydrogel containing free oil, respectively. These results highlight the potential of nanogel to protect oil against volatilization, control release and improve its anti-herpetic activity.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Abreu, F.O.M.S., Oliveira, E.F., Paula, H.C.B., Paula, R.C.M., 2012. Chitosan/cashewgum nanogels for essential oil encapsulation. Carbohydr. Polym. 89, 1277–1282.
Adams, R.P., 2007. Identification of Essential Oils Components by Gas Chromatography/quadrupole Mass Spectroscopy, 4th ed. Allured Publishing Corporation.
Anvisa, 2010. Farmacopeia Brasileira, 5a ed. Agência Nacional de Vigilância Sanitária, Ministério da Saúde, Brasília, DF, pp. 189–204.
Aiemsaard, J., Aiumlamai, S., Aromdee, C., Taweechaisupapong, S., Khunkitti, W., 2011. The effect of lemongrass oil and its major components on clinical isolate mastitis pathogens and their mechanisms of action on Staphylococcus aureus DMST 4745. Res. Vet. Sci. 91, e31–e37.
Astani, A., Reichling, J., Schnitzler, P., 2011. Screening for antiviral activities of isolated compounds from essential oils. Evid. Based Complem. Alternat. Med., https://doi.org/10.1093/ecam/nep187.
Bernstein, D.I., Bellamy, A.R., Hook, E.W., Levin, M.J., Wald, A., Ewell, M.G., Wolff, P.A., Deal, C.D., Heineman, T.C., Dubin, G., Belshe, R.B., 2013. Epidemiology, clinical presentation, and antibody response to primary infection with herpes simplex virus type 1 and type 2 in young women. Clin. Infect. Dis. 56, 344–351.
Borenfreund, E., Puerner, J., 1985. Toxicity determined in vitro by morphological alterations and neutral red absorption. Toxicol. Lett. 24, 119–124.
Bouchemal, K., Briançon, S., Perrier, E., Fessi, H., Bonnet, I., Zydowicz, N., 2004. Synthesis and characterization of polyurethane and poli (ether urethane) nanocapsules using a new technique of interfacial polycondensation combined of spontaneous emulsification. Int. J. Pharm. 269, 89–100.
Bourne, K.Z., Bourne, N., Reising, S.F., Stanberry, L.R., 1999. Plant products as topical microbicide candidates: assessment of in vitro and in vivo activity against herpes simplex virus type 2. Antiviral Res. 42, 219–226.
Chen, X., Peng, LH., Shan, Y.H., Li, N., Wei, W., Yu, L., Li, Q.M., Liang, W.Q., Gao, J.Q., 2013. Astragaloside IV-loaded nanoparticle-enriched hydrogel induces wound healing and anti-scar activity through topical delivery. Int. J. Pharm. 447, 171–181.
Clogston, J.D., Patri, A.K., 2011. Zeta potential measurement. Methods Mol. Biol. 697, 63–70.
Cohen-Sela, E., Teitlboim, S., Chorny, M., Koroukhov, N., Danenberg, H.D., Gao, J., Golomb, G., 2009. Single and double emulsion manufacturing techniques of an amphiphilic drug in PLGA nanoparticles: formulations of mithramycin and bioactivity. J. Pharm. Sci. 98, 1452–1462.
Costa, P., Lobo, J.M.S., 2001. Modeling and comparison of dissolution profiles. Eur. J. Pharm. Sci. 13, 123–133.
Falcão, D.Q., Mourão, S.C., Araujo, J.L., Pereira, P.A.K, Cardoso, A.C.A., Almeida, K.B., Zibetti, F.M., Lima, B.G., 2015. Challenges in development of essential oil nanodelivery systems and future prospects. In: Naik, J. (Ed.), Nano Based Drug Delivery. IAPC Publishing, Zagreb, pp. 557–578.
Falcão, D.Q., Santos, A.R., Ortiz-Silva, B., Louro, R.P., Seiceira, R., Finotelli, P.V., Ferreira, J.L.P., De Simone, S.G., Amaral, A.C.F., 2011. Development of nanoencapsulation forms from Cymbopogon citratus essential oil. Lat. Am. J. Pharm. 30, 765–772.
Farag, R.S., Shalaby, A.S., El-Baroty, G.A., Ibrahim, N.A., Ali, M.A., Hassan, E.M., 2004. Chemical and biological evaluation of the essential oils of different Melaleuca species. Phytother. Res. 18, 30–35.
Fatahzadeh, M., Schwartz, RA, 2007. Human herpes simplex virus infections: epidemiology, pathogenesis, symptomatology, diagnosis, and management. J. Am. Acad. Dermatol. 57, 737–763.
Fernandes, C.P., Mascarenhas, M.P., Zibetti, F.M., Lima, B.G., Oliveira, R.P.R.F., Rocha, L., Falcão, D.Q., 2013. HLB value, an important parameter for the development of essential oil phytopharmaceuticals. Rev. Bras. Farmacogn. 23, 108–114.
Flores, F.C., Lima, JA, Ribeiro, R.F., Alves, S.H., Rolim, C.M.B., Beck, R.C.R., Silva, C.B., 2013. Antifungal activity of nanocapsule suspensions containing tea tree oil on the growth of Trichophyton rubrum. Mycopathologia 175, 281–286.
Fontana, M.C., Rezer, J.F.P., Coradini, K., Leal, D.B.R., Beck, R.C.R., 2011. Improved efficacy in the treatment of contact dermatitis in rats by a dermatological nanomedicine containing clobetasol propionate. Eur. J. Pharm. Biopharm. 79, 241–249.
Förster, M., Bolzinger, M.A., Fessi, H., Briançon, S., 2009. Topical delivery of cosmetics and drugs. Molecular aspects of percutaneous absorption and delivery. Eur. J. Dermatol. 19, 309–323.
Gomes, C., Moreira, R.G., Castell-Perez, E., 2011. Poly (DL-lactide-co-glycolide) (PLGA) nanoparticles with entrapped trans-cinnamaldehyde and eugenol for antimicrobial delivery applications. J. Food Sci. 76, 16–24.
Guterres, S.S., Alves, M.P., Pohlmann, A.R., 2007. Polymeric nanoparticles, nanospheres and nanocapsules, for cutaneous applications. Drug Target Insights 2, 147–157.
Iannitelli, A., Grande, R., Di Stefano, A., Di Giulio, M., Sozio, P., Bessa, L.J., Laserra, S., Paolini, C., Protasi, F., Cellini, L., 2011. Potential antibacterial activity of carvacrol-loaded poly (DL-lactide-co-glycolide) (PLGA) nanoparticles against microbial biofilm. Int. J. Mol. Sci. 12, 5039–5051.
Ibrahim, M.M., Abd-Elgawad, A.E.H., Soliman, O.A.E., Jablonski, M.M., 2013. Pharmaceutical nanotechnology nanoparticle-based topical ophthalmic formulations for sustained celecoxib release. J. Pharm. Sci. 102, 1036–1053.
Jeong, Y., Na, H., Seoa, D., Kim, D., Lee, H., Jang, M., Na, S., Roh, S., Kim, S., Nah, J., 2008. Ciprofloxacin-encapsulated poly (DL-lactide-co-glycolide) nanoparticles and its antibacterial activity. Int. J. Pharm. 352, 317–323.
Kahan, M.T.H., Ather, A., Thompson, K.D., Gambari, R., 2005. Extracts and molecules from medicinal plants against herpes simplex viruses. Antiviral Res. 67, 107–119.
Khuroo, T., Verma, D., Talegaonkar, S., Padhi, S., Panda, A.K., Iqbal, Z., 2014. Topotecan-tamoxifen duple PLGA polymeric nanoparticles: investigation of in vitro, in vivo and cellular uptake potential. Int. J. Pharm. 473, 384–394.
Koch, C., Reichling, J., Schneele, J., Schnitzler, P., 2008. Inhibitory effect of essential oils against herpes simplex virus type 2. Phytomedicine 15, 71–78.
Markoulatos, P., Georgopoulou, A., Siafakas, N., Plakokefalos, E., Tzanakaki, G., Kourea-Kremastinou, J., 2001. Laboratory diagnosis of common herpes virus infections of the central nervous system by a multiplex PCR assay. J. Clin. Microbiol. 39, 4426–4432.
Minami, M., Kita, M., Nakaya, T., Yamamoto, T., Kuriyama, H., Imanishi, J., 2003. The inhibitory effect of essential oils on herpes simplex virus type-1 replication in vitro. Microbiol. Immun. 47, 681–684.
Moinard-Chécot, D., Chevalier, Y., Briançon, S., Beney, L., Fessi, H., 2008. Mechanism of nanocapsules formation by the emulsion-diffusion process. J. Colloid. Interface Sci. 317, 458–468.
Mora-Huertas, C.E., Fessi, H., Elaissari, A., 2010. Polymer-based nanocapsules for drug delivery. Int. J. Pharm. 385, 113–142.
Mu, L., Feng, S.S., 2003. PLGA/TPGS Nanoparticles for controlled release of paclitaxel: effects ofthe emulsifierand drug loading ratio. Pharm. Res. 20, 1864–1872.
Naik, M.I., Fomda, BA. Jaykumar, E., Bhat, JA, 2010. Antibacterial activity of lemon-grass (Cymbopogon citratus) oil against some selected pathogenic bacteria. Asian Pac. J. Trop. Med. 3, 535–538.
Negrelle, R.R.B., Gomes, E.C., 2007. Cymbopogon citratus (DC.) Stapf: chemical composition and biological activities. Rev. Bras. Pl. Med. 9, 80–92.
Nishimura, T., Toku, K., Fukuyasu, H., 1977. Antiviral compounds XII. Antiviral activity of amino hydrazones of alkoxyphenyl substituted carbonyl compounds against influenza virus in eggs and mice. Kitasato Arch. Exp. Med. 50, 39–46.
Oloyede, O.I., 2009. Chemical profile and antimicrobial activity of Cymbopogon citratus leaves. J. Nat. Prod. 2, 98–103.
Orhan, I.E., Özçelik, B., Kartal, M., Kan, Y., 2012. Antimicrobial and antiviral effects of essential oils from selected Umbelliferae and Labiatae plants and individual essential oil components. Turkish J. Biol. 36, 239–246.
Pereira, P.P., Puntel, R.L., Boschetti, T.K., Morel, A.F., 2009. Antioxidant effects of different extracts from Melissa officinalis, Matricaria recutita and Cymbopogon citratus. Neurochem. Res. 34, 973–983.
Pereira, R.S., Sumita, T.C., Furlan, M.R., Jorge, A.O.C., Ueno, M., 2004. Atividade antibacteriana de óleos essenciais em cepas isoladas de infecção urinária. Rev. Saúde Pública 38, 326–328.
Pinto, Z.T., Sánchez, F.F., Santos, A.R., Amaral, A.CF., Ferreira, J.L.P., Escalona-Arranz, J.C., Queiroz, M.M.C., 2015. Chemical composition and insecticidal activity of Cymbopogon citratus essential oil from Cuba and Brazil against housefly. Braz. J. Vet. Parasitol. 24, 36–44.
Reed, L.J., Müench, H., 1938. A simple method of estimating fifty percent endpoints. Am. J. Hyg. 27, 493–497.
Rodriguez, D.J., Chulia, J., Simões, C.M.O., Amoros, M., Mariotte, A.M., Girre, L., 1990. Search for in vitro antiviral activity of a new isoflavonic glycoside from Ulex europaeus. Planta Med. 56, 59–62.
Sahana, D.K., Mittal, G., Bhardwaj, V., Ravi Kumar, M.N.V., 2008. PLGA Nanoparticles for oral delivery of hydrophobic drugs: influence of organic solvent on nanoparticle formation and release behavior in vitro and in vivo using estradiol as a model drug. J. Pharm. Sci., 1530–1542.
Sahoo, S.K., Panyam, J., Prabha, S., Labhasetwar, V., 2002. Residual polyvinyl alcohol associated with poly (DL-lactide-co-glycolide) nanoparticles affects their physical properties and cellular uptake. J. Control. Release 82, 105–114.
Schaffazick, S.R., Guterres, S.S., 2003. Caracterizaçãoeestabilidade físico-químicade sistemas poliméricos nanoparticulados para administração de fármacos. Quim. Nova 26, 726–737.
Schaneberg, B.T., Khan, I.A., 2002. Comparison of extraction methods for marker compounds in the essential oil of lemon grass by GC J. Agric. Food Chem. 50, 1345–1349.
Schnitzler, P., Koch, C., Reichling, J., 2007. Susceptibility of drug-resistant clinical herpes simplex virus type 1 strains to essential oils of ginger, thyme, hyssop, and sandalwood. Antimicrob. Agents Chemother. 51, 1859–1862.
Schnitzler, P., Schuhmacher, A., Astani, A., Reichling, J., 2008. Melissa officinalis oil affects infectivity of enveloped Herpes viruses. Phytomedicine 15, 734–740.
Schuhmacher, A., Reichling, J., Schnitzler, P., 2003. Virucidal effect of peppermint oil on the enveloped viruses herpes simplex virus type 1 and type 2 in vitro. Phytomedicine 10, 504–510.
Silva, C.B., Guterres, S.S., Weisheimer, V., Schapoval, E.E.S., 2009. Antifungal activity ofthe lemongrass oil and citral against Candida spp. Braz.J. Infect. Dis. 12, 63–66.
Tajidin, N.E., Ahamad, S.H., Rosenani, A.B., Azimah, H., Munirah, M., 2012. Chemical composition and citral content in lemongrass (Cymbopogon citratus) essential oil at three maturity stages. Afr. J. Biotechnol. 11, 2685–2693.
Thompson, K.D., 2006. Herbal extracts and compounds active against herpes simplex virus. Adv. Phytomed. 2, 65–86.
United States Pharmacopeia (USP), 2008. USP 31; The National Formulary: NF 26, vol. 1. United States Pharmacopeial Convention, Rockville, pp. 817.
Viana, G.S.B., Vale, T.G., Pinho, R.S.N., Matos, F.JA, 2000. Antinociceptive effect ofthe essential oil from Cymbopogon citratus in mice. J. Ethnopharmacol. 70, 323–327.
Weisheimer, V., Miron, D., Silva, C.B., Guterres, S.S., Schapoval, E.E.S., 2010. Microparticles containing lemongrass volatile oil: preparation, characterization and thermal stability. Pharmazie 65, 885–890.
Wu, L., Zhang, J., Watanabe, W., 2011. Physical and chemical stability of drug nanoparticles. Adv. Drug Deliv. Rev. 63, 456–469.
Zhang, T., Sturgis, T.F., Youan, B.C., 2011. pH-responsive nanoparticles releasing tenofovir intended for the prevention of HIV transmission. Eur. J. Pharm. Biopharm. 79, 526–536.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ contributions
KBA contributed running the laboratory work, analysis of the data and drafted the paper. ACFA contributed in collecting plant sample and identification, and confection of herbarium. JLA contributed to development and characterization of the formulations. SCM contributed to in vitro CcVO release studies. JFC and MTVR contributed to antiviral activity studies. DQF designed the study, supervised the laboratory work and contributed to critical reading of the manuscript. All the authors have read the final manuscript and approved the submission.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Almeida, K.B., Araujo, J.L., Cavalcanti, J.F. et al. In vitro release and anti-herpetic activity of Cymbopogon citratus volatile oil-loaded nanogel. Rev. Bras. Farmacogn. 28, 495–502 (2018). https://doi.org/10.1016/j.bjp.2018.05.007
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2018.05.007