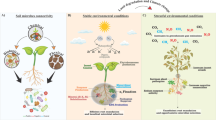
Abstract
Land consolidation of dryland-to-paddy conversion (LC-DtPC) has rapidly changed the farmland ecological environment in the short term, thereby affecting the microbial community and its mediated soil carbon sequestration. It is crucial for the improvement and stabilization of food production capacity. However, there is currently a lack of necessary knowledge about the short-term effects of LC-DtPC on the ecological processes of abundant and rare microbial taxa as well as the formation of soil carbon pool. Therefore, this study used field experiments and high-throughput sequencing technology to explore the short-term effects of LC-DtPC on abundant and rare microbial taxa and the mechanism driving soil carbon pool formation. Our results revealed that (1) the alpha diversity of abundant and rare bacteria and rare fungi was decreased by 1.14%, 6.74% and 12.84%, respectively, in the process of LC-DtPC, but the diversity of abundant fungi was increased by 14.09%. LC-DtPC significantly altered the β-diversity of abundant and rare bacterial, and rare fungal communities (p < 0.05); (2) LC-DtPC lowered the number of core microbes in the network, and reduced the robustness and vulnerability of abundant and rare microbial networks; (3) LC-DtPC significantly reduced soil carbon pool index by 28.75% in the short term (p < 0.05), and it was closely correlated with bacterial exogenous degradation and metabolism, carbohydrate metabolism, amino acid metabolism, lipid metabolism, and saprophytic fungal abundance; and (4) structural equation modeling revealed that rare bacteria played a bigger role in carbon pool formation than abundant bacteria, and bacteria played a greater role than fungi in the process of carbon pool formation under the synergistic effect of bacteria and fungi. This study explored the changes in abundant and rare microbial taxa and functions, and provided a theoretical basis for an in-depth understanding of soil carbon pool formation and mechanism during the LC-DtPC process.
Highlights
1. Dryland-to-paddy conversion (LC-DtPC) altered the diversity and function of abunant and rare taxa.
2. LC-DtPC significantly reduced soil carbon pool index by 28.75%.
3. Rare bacteria played a greater role in carbon pool formation during LC-DtPC.
AbstractSection Graphical Abstract
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Irrigated agriculture has globally become an important trend to replace the rainfed agriculture, so as to alleviate the threat of drought and to increase food production (Rosa et al. 2020). In the past decade, driven by food demand and policy guidance, China has carried out large-scale farmland improvement projects on the land consolidation of dryland-to-paddy conversion (LC-DtPC), with a cumulative area of more than 4 million hectares (Li et al. 2021a, b). Such a large-scale rapid LC-DtPC elevated the water and oxygen stress on soil microorganisms, led to their metabolic dysfunctions and affected the farmland ecosystems (Wu et al. 2018), which may cause a series of ecological issues, such as surface warming, groundwater overexploitation, and alterations in the form of soil heavy metal pollutants (Kang and Eltahir 2018; Wen et al. 2021). During the implementation of the land consolidation project, it is stipulated that the follow-up management period of the project shall not be less than 3 years, during which the transformation of land use types makes the soil environment extremely unstable. Therefore, three years is generally categorized into short-term land consolidation (Li et al. 2021a, b). In other words, during the first one to two years of LC-DtPC process, the effect of this conversion on the abundant and rare microbial taxa and their functions, and manipulation of the formation of soil carbon pool has not been studied extensively. This is crucial for the improvement and stabilization of food production. Also, it contributes to a comprehensive understanding of the ecological process of LC-DtPC and of the resulting changes in ecosystem functions.
In the process of LC-DtPC, the redox environment of soil changed after 15 days of flooding. pH values of both acidic and alkaline soils tended to be neutral, and the reduction potential decreased, while the content of Fe2+ and amorphous iron oxide increased (Jiao et al. 2019a, b). Research has found that LC-DtPC can increase soil nutrient heterogeneity after one year, resulting in varying degrees of spatial variability in soil pH, organic matter, salinity, and nitrogen, phosphorus, and potassium (Liang et al. 2020). Microbes play a pivotal role in regulating the global biogeochemical cycle and maintaining soil carbon pools (Kong et al. 2022). Previous studies have shown that wet and dry events significantly affect the soil microbial diversity, community structure, and community function (Qi et al. 2021). The soil organic carbon content of the paddy field was 12 ~ 58% higher than that of dryland soil at the same latitude (Qi et al. 2021). Oxygen limitation caused by the changes in water content can inhibit the activity of aerobic microorganisms, and further trigger the downregulation of carbon cycle-related genes and enzyme activities, resulting in a decreased rate of soil organic matter decomposition (Chen et al. 2021; Fuhrmann et al. 2018). Therefore, the residence time of the organic matter in rice gets higher than that in the dryland (Chen et al. 2021). In addition, species richness and distribution in microbial communities are non-uniform, and different microorganisms exhibit different sensitivities to environmental perturbations (Zhu et al. 2022a). Therefore, the abundant and rare microorganisms tend to exhibit differentiated ecosystem functions while resisting the perturbations, especially the changes of soil physicochemical properties, water, oxygen and other microenvironment disturbed by the LC-DtPC (Liang et al. 2020). Some studies have confirmed that even for highly redundant microorganisms with high abundance and diversity, the changes in precipitation profoundly affected the carbon and nitrogen cycling functions of the these microorganisms (Jiao et al. 2019a, b; Kuypers et al. 2018). Another study demonstrated that in paddy fields, the soil solution phosphorus turnover was regulated by the rare taxa (Wei et al. 2019). In general, the abundant and rare species are known to maintain their ecosystem versatility by forming complex ecological networks through intraspecific or interspecific interactions (Shu et al. 2021). It has been reported that the nitrogen-transforming microbial network in paddy fields was much more complex than that in the drylands (Kuypers et al. 2018). Overall, although previous studies effectively linked the abundant microbial taxa, microbial networks, and nutrient cycling in soil, they did not pay much attention to explain the contribution of the soil abundant and rare taxa to the formation of soil carbon pool regarding LC-DtPC. Also, they did not clarify the shifts in the microbial metabolic trait and functional changes triggered by LC-DtPC.
It is worth noting that in the current research, it seems unscientific to simply use the dryland and paddy field as the object of alternating dry–wet research, because the course of natural rewetting or re-drying is much slower (Schimel 2018). Related research showed that direct watering of disturbed soils can mobilize more carbon than natural rewetting (Smith et al. 2017), while the slow natural infiltration reduced the mass transport between the soil organic molecules and microorganisms (Schimel 2018). Hence, there is a need to distinguish between the wet-dry alternation and LC-DtPC. Shifts between the paddy field and dryland may result in dramatically different soil microbial diversity patterns and functions from the natural wet-dry events. Therefore, a comprehensive understanding of the microbial response to LC-DtPC and an exploration of the environmental factors driving their community changes is very important for accelerating and deepening the LC-DtPC. Based on this, the present study explored the response pattern and the function of soil abundant and rare microbial communities to LC-DtPC at the field experiment scale. Furthermore, the mutual feedback mechanism of the soil microorganisms, soil carbon pool and environmental factors was explored. Specifically, the aims of this study were as follows: (1) to clarify the diversity of soil abundant and rare microorganisms and their relationship with soil carbon pool before and after LC-DtPC, (2) to explore the characteristics and stability of the co-occurrence networks of abundant and rare microorganisms, and (3) to reveal the LC-DtPC-driven functional and metabolic changes of the abundant and rare microorganisms and their mechanism driving the formation of soil carbon pool. This study will help understand the response of the soil abundant and rare microorganisms to LC-DtPC, predict the carbon sequestration capacity and mechanism of LC-DtPC-type farmland, and provide a scientific basis for promoting the land consolidation, farmland management and low-carbon agricultural development.
2 Materials and methods
2.1 Field experiments and soil sample collection
The field experiment of this study was carried out in the Land Science Research Center of the China University of Mining and Technology, Xuzhou, Jiangsu. The region exhibited a temperate continental monsoon climate, with an annual precipitation of 800 ~ 930 mm and an annual average temperature of 14.2 °C (Li et al. 2021a, b). The soil type was cinnamon fluvo-aquic soil, and the planting method was wheat-corn rotation. In the field experiment, 10 experimental plots of 15 m × 20 m were used, and 1.2 m anti-seepage ditches were set up in adjacent experimental plots. Among them, 5 plots were planted with corn (DL, variety: Nonghua 101), and 5 plots were subjected to flooding treatment for 5 days and then transplanted with rice (PF, variety: Wuyugeng 31). The seeds were sterilized with 2% H2O2 before sowing, and the rice plants were shallow irrigated according to the local agricultural water quota of 425 m3 during the growth period. The experiment was carried out between June 2019 and November 2019 (a corn and rice season as a short-term dryland-to-paddy conversion).
Before harvest, two topsoil (0 ~ 20 cm) samples were collected at the same selected sampling site in each plot using the 5-point sampling method, resulting 10 samples of PF and 10 samples of DL samples. One of the collected soil samples was stored at -80 °C for DNA extraction and subsequent sequencing analysis. The other soil samples were air-dried, with the gravel, animal and plant residues removed, grounded, and filtered through a 2 mm sieve for the determination of soil physicochemical properties. The changes in the soil physicochemical properties of LC-DtPC farmland are listed in Table S1.
2.2 Soil physico-chemical analysis and carbon pool index calculation
Soil physico-chemical properties and carbon pool index were determined according to previous studies (Li et al. 2021a, b; Zhu et al. 2022a, b, c), including soil pH, electrical conductivity (EC), total nitrogen (TN), nitrate nitrogen (NN), ammonium nitrogen (AN), available phosphorus (AP), available potassium (AK), soil organic matter (SOM), soil organic carbon (SOC), microbial biomass carbon (MBC), easily oxidizable organic carbon (EOC), dissolved organic carbon (DOC) and recalcitrant organic carbon (ROC). Details of the determination method are shown in Supplementary Information. Blair et al. (1995) found that the organic matter that could be oxidized by 333 mmol·L−1 KMnO4 changed the most during crop cultivation. Therefore, the organic matter that can be oxidized by 333 mmol·L−1 KMnO4 is called active organic matter, and the organic matter that cannot be oxidized is called non-active organic matter. They also proposed the Soil Carbon Pool Management Index (CPMI) for the first time. The CPMI is an indicator of changes in soil organic matter caused by soil management measures. It is a systematic and sensitive monitoring method for soil carbon changes, and can reflect the degree to which agricultural measures reduce or update soil quality. In addition, the calculations of CPMI are also shown in the Supplementary Information.
2.3 DNA extraction, sequencing and division of microbial taxa
DNA was extracted from soil samples using FastDNA™ SPIN Kit for Soil (MP Biomedicals, USA) using the manufacturer’s protocol. The V4-V5 region of bacterial 16S rRNA gene was amplified by PCR with primers 515F (5'- GTGCCAGCMGCCGCGGTAA-3') and 907R (CCGTCAATTCMTTTRAGTTT). The fungal ITS1 was amplified using primers ITS5F (5'-GGAAGTAAGTCGTAACAAGG-3') and ITS1R (5'-GCTGCGTTCTTCTCGATGC-3'). The PCR amplification procedure was as follows: pre-denaturation at 95 °C for 3 min, followed by a total of 27 cycles of denaturation at 95 °C for 30 s, annealing at 55 °C for 30 s, and extension at 72 °C for 30 s, and a final extension at 72 °C for 5 min and then holding at 10 °C. The PCR amplification products were detected by 2% agarose gel electrophoresis, and the target fragments were recovered using a DNA gel extraction kit (Axygen, USA). Based on the preliminary quantitative results of electrophoresis, the recovered PCR amplification products were subjected to fluorescence quantification using a Quant-iT™ PicoGreen™ dsDNA Assay Kit (MP Biomedicals, USA) and read by a microplate reader (BioTek FLx800, USA). Subsequently, the samples were mixed in a ratio according to the sequencing amount required for each sample. A TruSeq Nano DNA LT Library Prep Kit (Illumina, USA) was adopted to construct the soil microbial sequencing library (Ma et al. 2021; Zhu, Y. et al. 2022b). After quantification with Qubit and quantitative PCR (Q-PCR), the constructed library was sequenced with HiSeq2500 PE2500 (Illumina, USA) by Shanghai Personalbio Technology Co, Ltd.
By default, the sequences were clustered into operational taxonomic units (OTUs) with 97% similarity. To evaluate the responses of abundant and rare microbial taxa to LC-DtPC, all OTUs were divided into six categories following the criteria adopted in Dai’s study (Dai et al. 2016): (i) always abundant taxa (AAT); (ii) conditionally abundant taxa (CAT); (iii) always rare taxa (ART); (iv) conditionally rare taxa (CRT); (v) moderate taxa (MT); (vi) conditionally rare and abundant taxa (CRAT). AAT, CAT, and CRAT were collectively referred to as abundant taxa, and ART and CRT were collectively referred to as rare taxa. The detailed classification criteria are listed in the Supplementary Information.
2.4 Statistical analysis
The SPSS 22.0 software was used for statistical analysis. One-way analysis of variance (ANOVA) and honestly significant difference (HSD) test were performed to analyze the data of different treatment groups under the condition of p < 0.05. The microbial α-diversity index was calculated using the vegan package in R. Based on the Bray–Curtis dissimilarity matrix, principal coordinate analysis (PCoA) was used to visualize the differences in the microbial communities with the vegan package in R. The Kruskal–Wallis rank-sum test was used to evaluate the differences in the microbial sub-community structure after LC-DtPC. The abundance of microbial communities at the phylum level was obtained using the plyr package in R. The ggClusterNet package in R was used to construct the co-occurrence network of the microbial subcommunities, with parameters set as r > 0.6 and p < 0.05. The network robustness and vulnerability were analyzed to obtain the network topology characteristics degree centrality and closeness centrality (Wen et al. 2022). The keystone OTUs of the network were determined by intra-module connectivity (Zi) and inter-module connectivity (Pi) that nodes with Zi ≥ 2.5 or Pi ≥ 0.62 were classified as keystone OTUs. Furthermore, a random forest model was used to predict the top 20 abundant OTUs and soil carbon indexes and the main drivers of physicochemical properties. Bacterial and fungal function predictions were performed using the PICRUSt2 and FUNGuild tools, respectively, which were implemented at http://mem.rcees.ac.cn/, and visualized with the pheatmap package in R. Finally, the structural equation model was constructed using the piecewiseSEM package in R (Liu et al. 2022). Shipley’s d-separation test was used to verify whether a path was missing from the model, with p > 0.05 indicating no missing path and a good model fit.
3 Results
3.1 Response of the abundant and rare microbial communities to LC-DtPC
After quality control, there were 354,141, 81,968 and 12,113, 7,758 OTUs for the bacteria and fungi in the paddy field and dryland soil samples, respectively, according to 97% sequence similarly. The number of OTUs in the abundant and rare bacterial taxa was 196 and 121,406, respectively, and the number of OTUs of abundant and rare fungal taxa was 5 and 6,576, respectively (as shown in the Supplementary Information). In the PF and DL treatments, the dominant phyla in both the abundant and rare taxa were Proteobacteria, Actinobacteria, and Chloroflexi, while the ratio of rare Acidobacteria was increased in the DL treatment. Ascomycota accounted for 76% and 35.74% of the relative abundance of abundant fungi in the PF and DL treatments, respectively, and Ascomycota and Basidiomycota were the dominant ones in rare fungi (Fig. 1c). LC-DtPC significantly reduced the Shannon index of rare bacteria by 6.74% (p < 0.001), whereas no significant difference in the Shannon index was seen in the abundant bacteria and abundant and rare fungi (Fig. 1a). PCoA analysis based on the Bray–Curtis similarity demonstrated that the abundant and rare bacteria and rare fungi were significantly different in the ordinal spatial clustering (pANOSIM < 0.001). This result was further confirmed by the Kruskal–Wallis test. The variation degrees of rare bacteria and fungi were both higher than those of abundant ones (R2abundant bacteria = 0.195, R2bare bacteria = 0.050, R2abundant fungus = 0.195, and R2bare fungus = 0.050), indicating that the response of rare microbial taxa was less stable than that of the abundant taxa to LC-DtPC (Fig. 1b).
The alpha diversity of DL and PF treatments (a, d, g, j). Principal coordinate analysis (PCoA) results showing beta-diversity patterns between the DL and PF treatments (b, e, h, k). ANOSIM is similarity analysis. Distribution of dominant phylum of abundant and rare communities in DL and PF treatments (c, f, i, l). Abundant bacteria: a, b, c; Rare bacteria: d, e, f; abundant fungus: g, h, i; rare fungus: j, k, l. Asterisks denote significant correlations. * p < 0.05, *** p < 0.001 and NS p > 0.05
3.2 Co-occurrence networks of the abundant and rare microbial communities and their robustness
The co-occurrence networks for the abundant and rare microbes were constructed based on the Spearman correlation between the OTUs to explore the interconnection of microorganisms after LC-DtPC (Fig. 2). In general, compared with the DL treatment, the PF treatment reduced the number of nodes and edges of the overall bacterial network, and the degrees of abundant, rare, and overall networks were significantly reduced by 52.05%, 20.43% and 84.13%, respectively. On the contrary, an increase in the network closeness was noted. The decrease in these attributes of nodes indicated that LC-DtPC reduced the stability of the bacterial network. In contrast, the LC-DtPC reduced the number of nodes and edges of soil fungi networks. However, the degrees of rare and overall fungal networks of the PF treatment were significantly increased compared to the DL treatment. In addition, due to the small amount of OTUs of abundant fungi, the co-occurrence network could not be built. Nevertheless, the mutual connection between the abundant fungi might still exist. Regarding the core species of the network, LC-DtPC exhibited a small impact on the fungal network, but it significantly reduced the number of core species of the abundant and rare bacterial networks (Figure S6). LC-DtPC significantly lowered the robustness and vulnerability, indicating a decrease in co-occurrence network complexity and stability (Figure S7).
3.3 Impact of LC-DtPC on soil carbon pool
It can be seen from Fig. 3 that after LC-DtPC, the SOC content was significantly reduced (p < 0.05), the standard deviations of carbon pool index were decreased, and the soil carbon pool was relatively uniform. The soil active organic carbon was significantly reduced (p < 0.05). MBC and EOC were decreased by 28.50% and 29.09%, respectively, both of which were highly significantly different (p < 0.01). However, the LC-DtPC barely affected the ROC content. Compared with paddy field, the CPMI in dryland showed a larger change range, and the dispersion was also large. The average CPMI dropped 11.65% after LC-DtPC which was significantly different.
3.4 Relationship between the microbial community function and soil carbon pool
KEGG (level 2) functional information was obtained by the functional prediction through PICRUSt2, and 41 potential metabolic functions of the bacterial community were obtained. The results demonstrated that LC-DtPC led to a decline in most of the bacterial metabolic functions (Fig. 4a). Among them, the relative abundance related to carbohydrate metabolism, amino acid metabolism, lipid metabolism and exogenous degradation and metabolism was decreased significantly (p < 0.05). Furthermore, most of these bacterial metabolic functions were significantly associated with EOC and CPMI in the DL treatment (Fig. 4b). On the contrary, in the PF treatment, only DOC was associated with these functions, and all other carbon pool indexes were not significantly affected (Fig. 4c). Compared with the DL treatment, the PF treatment reduced the microbial decomposition function by 56.46%.
Based on the OTUs data, the ecological community functions of the fungi were predicted in FUNGuild v1.0 into three categories: pathogenic, saprotrophic, and symbiotic, and were finally refined into 21 fungal functions. The LC-DtPC boosted 11 fungal functions, while reduced 10 functions (Fig. 4d). In the DL samples, the fungi were mainly saprotroph-symbiotroph (18.53%) and pathotrophic-saprotrophic-symbiotrophic (11.01%). In the PF samples, the fungi were mainly Saprotrophic (27.49%), while saprotrophic-symbiotrophic (1.58%) type fungi were decreased. Correlation analysis showed that the soil carbon pool exhibited a greater impact on fungal functions in the PF treatment than in the DL treatment (Fig. 4e, f). Moreover, the fungal function of paddy field was more affected by the soil nutrients than that of dryland, especially by TN. Among them, DOC, EOC and ROC in the PF treatment were significantly related to animal pathogens, saprophytic fungi, and lichen symbiotic fungi, respectively (Fig. 4f).
3.5 Mechanism of the soil carbon pool formation by abundant and rare subcommunity
The soil carbon pool was closely related to NN, AN, and AK in dryland soils, whereas it was closely related to AP in paddy fields (Figure S1). The multiple regression model and random forest were adopted to verify the relative contributions of the soil variables to the top 20 abundant OTUs of abundant and rare microorganisms (Figure S2). ROC and pH could explain the relative abundances of the major OTUs of abundant bacteria in the DL treatment. The OTU abundances including OTU1987 (Proteobacteria) and OTU36032 (Actinobacteria) were found to be the important factors predicting the soil carbon pool level in abundant bacteria in the PF treatment. While in the PF treatment, CPMI and AP could explain the abundance of major OTUs in abundant bacteria. OTU114437 (Rokubacteria), OTU106927 (Chloroflexi) and OTU98 (Proteobacteria) were the major predictors of the soil carbon pool in abundant bacteria. In contrast, OTU99876 (Proteobacteria) was closely related to the soil carbon pool level of rare bacteria in the DL treatment. MBC and EOC significantly affected the rare bacteria OTUs in the DL and PF treatments, respectively. All carbon pool indexes, except MBC, were key variables in determining the OTU abundance of rare bacteria in the PF treatment. MBC and EOC also displayed a similar importance for rare fungi in the DL and PF treatments as for rare bacteria.
Structural equation modeling was employed to simulate the impact of synergy of the abundant and rare bacteria and fungi on soil carbon pool index (Fig. 5). The results showed that under the synergy of abundant and rare bacteria, the carbon pool directly affected the composition of the rare bacteria, while the carbon pool index was mainly affected by the composition of the soil carbon pool and rare bacteria. Abundant and rare fungi exhibited a direct impact on the carbon pool index at the same time. In addition, during the synergy of the rare bacteria and fungi, the carbon pool index was directly affected by rare bacteria, whereas the effect of fungi was not significant. Similarly, in the synergy of abundant bacteria and fungi, fungi did not affect the carbon pool index significantly, while the abundant bacteria directly affected the carbon pool index. LC-DtPC directly affected the soil carbon pool. In conclusion, the role played by the rare bacteria in the formation of carbon pool could be greater than that of the abundant bacteria. Both abundant and rare fungi affected the formation of soil carbon pool. In the process of carbon pool formation affected by both bacteria and fungi, bacteria contributed to a greater extent than fungi.
Composite structural equation model (SEM) assessing the direct and indirect effects of LD-DtPC, soil nutrient (the first axis of the principal component analysis based on TN, NN, AN, AP and AK), soil physiochemical (the first axis of the principal component analysis based on pH and EC), carbon pool (the first axis of the principal component analysis based on DOC, MBC, EOC, ROC, SOM and SOC), abundant and rare microbial community composition (first axis of alpha index and top 3 phylums) on CPMI. Numbers adjacent to arrows show standardized path coefficients. Blue and red lines indicate positive and negative relationships, respectively. The R.2 values indicate the proportion of variance explained. Asterisks denote significant differences based on the Wilcoxon test. *p < 0.05, **p < 0.01, and ***p < 0.001
4 Discussion
4.1 Soil carbon pool changes and microbial diversity response patterns during LC-DtPC
The present study revealed that the LC-DtPC resulted in a decrease in the soil carbon pool and concomitant reduction in the soil nutrient content. This is consistent with the previous observations that LC-DtPC directly affected the carbon pool composition of dryland and paddy soils (Li et al. 2021a, b). LC-DtPC exhibited a direct impact on soil carbon pool composition. The decrease in the soil carbon pool and soil nutrients was due to the changes in the soil oxygen limitation caused by flooding treatment, which in turn affected the microbial activity and caused differences in the accumulation of organic matter in the soil (Dong et al. 2021). This ultimately led to a reduction in the amount of organic carbon and nutrients released into the soil. Studies have shown that the oxygen limitation of organic matter lowered the activity of oxidases involved in the decomposition of plant residues and inhibited the transformation turnover and nutrient cycling of organic matter (Keiluweit et al. 2017). In addition, oxygen limitation in paddy fields hindered the contribution of fungi to soil carbon pool compared to that of the drylands because most fungi are aerobic, whereas bacteria tolerate anaerobic conditions better (Luo et al. 2020; Zhang et al. 2021). This result was further verified by the analysis of the effect of fungi-bacteria synergy on the soil carbon pool using the structural equation modeling.
It was found that LC-DtPC directly affected the soil carbon pool composition, resulting in dramatic differences in the structures, diversity patterns, and functional characteristics between the abundant and rare microbial taxa. Compared with the DL treatment, the PF treatment significantly decreased the diversity of the rare bacteria. The diversity of rare bacteria affected the changes of ROC and SOC, while the content of EOC was related to the diversity of abundant and rare fungi (Figure S3). This could be caused by a direct change in soil water content that increased the anaerobic niche and reduced soil nutrient content, resulting in increased anaerobic taxa and decreased diversity (Evans and Wallenstein 2014; Jansson and Hofmockel 2020). The changes in the species composition supported this hypothesis. After LC-DtPC, the abundance of the aerobic abundant and rare bacteria Actinobacteria decreased sharply, while the abundances of anaerobic bacteria Chloroflexi and Firmicutes significantly increased. Chloroflexi is a facultative anaerobic organism and can be converted to EOC under low nutrient conditions (Fierer et al. 2007). Studies revealed that the abundance of Actinobacteria was likely to be associated with soil moisture, and its relative abundance increased in the conditions of insufficient soil moisture (Banerjee et al. 2016). However, this is inconsistent with our results which suggest that the effects of oxygen and nutrient limitations on Actinobacteria may be larger than soil moisture. In contrast, most species of the abundant and rare fungi exhibited decreased abundances after LC-DtPC, suggesting that oxygen limitation reduced the sensitivity of aerobic fungi to environmental conditions (Ochoa-Hueso et al. 2018). In addition, the β-diversity of the abundant and rare bacteria and rare fungi in the PF treated soil was significantly lower than that in the DL treated soil, indicating that LC-DtPC resulted in big differences in the bacterial and rare fungi communities in the DL treatment. In contrast, the PF treatment made these microbial communities less diverse. Furthermore, rare microbial taxa were more unstable in response to LC-DtPC than the abundant taxa.
4.2 Mechanisms of soil carbon pool formation and changes of microbial co-occurrence networks during LC-DtPC
Co-occurrence networks not only can reveal the inter-community relationships, but also provide unique perspectives on the response of the species to habitat changes using different selection mechanisms (Zhu et al. 2022b). A decrease in the robustness and vulnerability of the co-occurrence networks of abundant and rare species was noted after LC-DtPC. This decline can be attributed to the higher environmental pressures resulting from the LC-DtPC. Researches have shown that LC-DtPC reduced the soil carbon pool and nutrient content, making the niches of microorganisms narrower and sensitive to environmental changes (Wu et al. 2021), which could cause the instability. On the other hand, LC-DtPC lowered the degree and elevated the closeness of the network topological properties, thus verifying that LC-DtPC made the microbial co-occurrence network unstable. Furthermore, the rare bacterial and fungal taxa occupied the network module hubs and link nodes, and changes in these taxa might promote the robustness of the co-occurrence network after LC-DtPC more than abundant taxa. Moreover, microorganisms in dryland were sensitive to soil carbon pools, while the paddy field taxa were far more sensitive to soil nutrients (especially TN) and soil physicochemical properties (EC), than to the soil carbon pool (Figure S5). This can explain the difference in the microbial robustness changes in dryland and paddy fields after LC-DtPC. In drylands, the increased robustness of co-occurrence networks of microorganisms in soil could be due to the organic matter accumulation, which provides SOC and stable nutrients for microbial activity (Wei et al. 2012). After the flooding treatment, redox potential affected the transformation and turnover of the soil organic matter. Water stress reduced the nitrogen availability to soil microbes and inhibited the microbial metabolism (de Vries et al. 2018). As a result, the network robustness of soil carbon pool was smaller in paddy fields. In summary, the changes in soil carbon pool led to a decrease in the robustness of the microbial co-occurrence networks, while elevated the soil nutrients. Meanwhile, the redox potential in paddy fields contributed to the increased robustness of the network after LC-DtPC. It was also noted that in the DL treatment, the community differences among the abundant and rare bacteria and rare fungi decreased with an increase in the carbon pool, while the community differences increased with an increase in the carbon pool in the PF treatment (Figure S4). Such differentiation may be due to the differences in the ability of the abundant and rare species to compete for limited resources in different environments (Jiao et al. 2019a, b). Dry soil reduces the microbial access to soluble carbon resources, while water stress reduces the efficiency of microbial utilization of carbon resources (Schimel 2018). Meanwhile, the same taxonomic differences demonstrated opposite responses to carbon pool and nutrients, and this could be because of the differences in acquisition strategies of the species for different resources.
There is a nonlinear cascade relationship between the microbial communities and ecosystem properties. Recent studies have shown that abundant and rare microbial taxa have different metabolic potentials and ecological roles (Jiao and Lu 2020). The results demonstrated that LC-DtPC reduced the decomposition function and exogenous degradation and metabolism of the soil microorganisms, indicating that LC-DtPC limited the microbial activity and possibly inhibited the decomposition of plant residues by microorganisms (Chen et al. 2012; Yang et al. 2022). In addition, LC-DtPC reduced the relative abundance of genes related to carbohydrate metabolism, amino acid metabolism, and lipid metabolism. Studies have shown that the abundances of functional genes were significantly positively correlated with their specific enzyme activities. Therefore, LC-DtPC make microorganisms lose their ability to degrade complex substrates, thereby contributing to changes in soil carbon pool (Li et al. 2021a, b). RDA showed that the effect of LC-DtPC on rare bacteria was greater than that on the abundant bacteria, and its effect on abundant fungi was larger than that on rare fungi (Figure S5). This study also revealed that the core species for predicting the soil carbon pool level in the DL treatment were aerobic microorganisms, while those in the PF treatment were anaerobic microorganisms (Fig. 4d). The utilization rate of carbon nutrients by anaerobic microorganisms was lower than that by aerobic microorganisms, leading to an enhanced carbon mineralization and shrinking the soil carbon pool during LC-DtPC (Chen et al. 2021). In particular, the interactions between the bacteria and fungi were pervasive and diverse. The structural equation modeling showed that in the synergy of rare bacteria and fungi, the carbon pool index was directly affected by rare bacteria, while the effects of fungi were not significant. In the synergy of abundant bacteria and fungi, fungi had no significant effect on the carbon pool index, while the abundant bacteria directly affected the carbon pool index. This could be because of the differences in the limiting microbial survival and metabolic activities by resource (substrate) availability and environmental heterogeneity (Shu et al. 2021; Wu et al. 2021; Zhang et al. 2022). In conclusion, rare bacteria played a bigger role in the carbon pool formation than the abundant bacteria. Both the abundant and rare fungi affected the formation of soil carbon pool. In the process of carbon pool formation under the synergistic effect of bacteria and fungi, bacteria played a more important role than fungi.
5 Conclusions
In the present study, LC-DtPC decreased the diversity of abundant and rare bacteria and rare fungi, but increased the diversity of abundant fungi. The rare taxa communities were more sensitive to LC-DtPC than the abundant taxa. Soil nutrients and electrical conductivity were found to be the important factors affecting the microbial β-diversity in paddy fields, and soil carbon pool was the main factor affecting the microbial β-diversity in dryland. LC-DtPC reduced the robustness and vulnerability of co-occurrence networks of the abundant and rare bacteria and fungi. LC-DtPC significantly lowered the soil carbon pool capacity in the short term. In addition, LC-DtPC weakened the functions of bacterial exogenous degradation and metabolism, carbohydrate metabolism, amino acid metabolism, and lipid metabolism, increased the abundance of saprophytic fungi, and affected soil carbon pool capacity. Moreover, the role of rare bacteria in the formation of carbon pool was found to be greater than that of the abundant bacteria. Both abundant and rare fungi affected the formation of the soil carbon pool. In the process of carbon pool formation under the synergistic effect of bacteria and fungi, it was found that bacteria contributed to a greater extent than fungi. Overall, the present study clarified the responses of the abundant and rare microbial taxa in LC-DtPC, revealed LC-DtPC-driven functional and metabolic changes of the abundant and rare microorganisms, and the mechanism driving the formation of soil carbon pool. Our study can help the prediction of carbon sequestration capacity of LC-DtPC-type farmland, providing a new perspective for scientific farmland management and land consolidation.
Availability of data and materials
Data used in this study are stored in a public data repository and can be made available upon reasonable request following data-sharing regulations. The R scripts used in data analyses are available from the corresponding author upon request.
References
Banerjee S, Helgason B, Wang L, Winsley T, Ferrari BC, Siciliano SD (2016) Legacy effects of soil moisture on microbial community structure and N2O emissions. Soil Biol Biochemistry 95:40–50. https://doi.org/10.1016/j.soilbio.2015.12.004
Blair GJ, Lefroy R, Lisle L (1995) Soil carbon fractions based on their degree of oxidation, and the development of a carbon management index for agricultural systems. Aust J Agr Res 46:1459–1466. https://doi.org/10.1071/AR9951459
Chen L, Yang Y, Deng S, Xu Y, Wang G, Liu Y (2012) The response of carbohydrate metabolism to the fluctuation of relative humidity (RH) in the desert soil cyanobacterium Phormidium tenue. Eur J Soil Biol 48:11–16. https://doi.org/10.1016/j.ejsobi.2011.10.002
Chen X, Hu Y, Xia Y, Zheng S, Ma C, Rui Y, He H, Huang D, Zhang Z, Ge T, Wu J, Guggenberger G, Kuzyakov Y, Su Y (2021) Contrasting pathways of carbon sequestration in paddy and upland soils. Glob Chang Biol 27(11):2478–2490. https://doi.org/10.1111/gcb.15595
Dai T, Zhang Y, Tang Y, Bai Y, Tao Y, Huang B, Wen D (2016) Identifying the key taxonomic categories that characterize microbial community diversity using full-scale classification: a case study of microbial communities in the sediments of Hangzhou Bay. FEMS Microbiol Ecol 92(10):1–11. https://doi.org/10.1093/femsec/fiw150
de Vries FT, Griffiths RI, Bailey M, Craig H, Girlanda M, Gweon HS, Hallin S, Kaisermann A, Keith AM, Kretzschmar M, Lemanceau P, Lumini E, Mason KE, Oliver A, Ostle N, Prosser JI, Thion C, Thomson B, Bardgett RD (2018) Soil bacterial networks are less stable under drought than fungal networks. Nat Commun 9(1):3033. https://doi.org/10.1038/s41467-018-05516-7
Dong H, Zhang S, Lin J, Zhu B (2021) Responses of soil microbial biomass carbon and dissolved organic carbon to drying-rewetting cycles: A meta-analysis. Catena. 207: 105610. https://doi.org/10.1016/j.catena.2021.105610
Evans SE, Wallenstein MD (2014) Climate change alters ecological strategies of soil bacteria. Ecol Lett 17(2):155–164. https://doi.org/10.1111/ele.12206
Fierer N, Bradford MA, Jackson RB (2007) Toward an ecological classification of soil bacteria. Ecology 88(6):1354–1364. https://doi.org/10.1890/05-1839
Fuhrmann I, He Y, Lehndorff E, Brüggemann N, Amelung W, Wassmann R, Siemens J (2018) Nitrogen fertilizer fate after introducing maize and upland-rice into continuous paddy rice cropping systems. Agr Ecosyst Environ 258:162–171. https://doi.org/10.1016/j.agee.2018.02.021
Jansson JK, Hofmockel KS (2020) Soil microbiomes and climate change. Nat Rev Microbiol 18(1):35–46. https://doi.org/10.1146/annurev-environ-012320-082720
Jiao S, Lu Y (2020) Soil pH and temperature regulate assembly processes of abundant and rare bacterial communities in agricultural ecosystems. Environ Microbiol 22(3):1052–1065. https://doi.org/10.1111/1462-2920.14815
Jiao S, Wang J, Wei G, Chen W, Lu Y (2019a) Dominant role of abundant rather than rare bacterial taxa in maintaining agro-soil microbiomes under environmental disturbances. Chemosphere 235:248–259. https://doi.org/10.1016/j.chemosphere.2019.06.174
Jiao S, Xu Y, Zhang J, Lu Y (2019b) Environmental filtering drives distinct continental atlases of soil archaea between dryland and wetland agricultural ecosystems. Microbiome 7(1):15. https://doi.org/10.1186/s40168-019-0630-9
Kang S, Eltahir EAB (2018) North China Plain threatened by deadly heatwaves due to climate change and irrigation. Nat Commun. 9(1):2894. https://doi.org/10.1038/s41467-018-05252-y
Keiluweit M, Wanzek T, Kleber M, Nico P, Fendorf S (2017) Anaerobic microsites have an unaccounted role in soil carbon stabilization. Nat Commun 8(1):1771. https://doi.org/10.1038/s41467-017-01406-6
Kong W, Wei X, Wu Y, Shao M, Zhang Q, Sadowsky MJ, Ishii S, Reich PB, Wei G, Jiao S, Qiu L, Liu L (2022) Afforestation can lower microbial diversity and functionality in deep soil layers in a semiarid region. Glob Chang Biol 28(20):6086–6101. https://doi.org/10.1111/gcb.16334
Kuypers MMM, Marchant HK, Kartal B (2018) The microbial nitrogen-cycling network. Nat Rev Microbiol 16(5):263–276. https://doi.org/10.1038/nrmicro.2018.9
Li B, Li P, Zhang W, Ji J, Liu G, Xu M (2021) Deep soil moisture limits the sustainable vegetation restoration in arid and semi-arid Loess Plateau. Geoderma. 399:115122. https://doi.org/10.1016/j.geoderma.2021.115122
Li X, Hui N, Yang Y, Ma J, Luo Z, Chen F (2021) Short-term effects of land consolidation of dryland-to-paddy conversion on soil CO2 flux. J Environ Manage. 292:112691. https://doi.org/10.1016/j.jenvman.2021.112691
Liang Y, Xiao X, Nuccio EE, Yuan M, Zhang N, Xue K, Cohan FM, Zhou J, Sun B (2020) Differentiation strategies of soil rare and abundant microbial taxa in response to changing climatic regimes. Environ Microbiol 22(4):1327–1340. https://doi.org/10.1111/1462-2920.14945
Liu S, García-Palacios P, Tedersoo L, Guirado E, van der Heijden MGA, Wagg C, Chen D, Wang Q, Wang J, Singh BK, Delgado-Baquerizo M (2022) Phylotype diversity within soil fungal functional groups drives ecosystem stability. Nat Ecol Evol 6(7):900–909. https://doi.org/10.1038/s41559-022-01756-5
Luo Z, Ma J, Chen F, Li X, Zhang Q, Yang Y (2020) Adaptive Development of Soil Bacterial Communities to Ecological Processes Caused by Mining Activities in the Loess Plateau. China Microorganisms 8(4):477. https://doi.org/10.3390/microorganisms8040477
Ma J, Gonzalez-Ollauri A, Zhang Q, Xiao D, Chen F (2021) Ecological network analysis to assess the restoration success of disturbed mine soil in Zoucheng. China Land Degrad Dev 32(18):5393–5411. https://doi.org/10.1002/ldr.4116
Ochoa-Hueso R, Collins SL, Delgado-Baquerizo M, Hamonts K, Pockman WT, Sinsabaugh RL, Smith MD, Knapp AK, Power SA (2018) Drought consistently alters the composition of soil fungal and bacterial communities in grasslands from two continents. Glob Chang Biol 24(7):2818–2827. https://doi.org/10.1111/gcb.14113
Qi J, Chen B, Gao J, Peng Z, Jiao S, Wei G, Kivlin S (2021) Responses of soil bacterial community structure and function to dry–wet cycles more stable in paddy than in dryland agricultural ecosystems. Global Ecol Biogeogr 31(2):362–377. https://doi.org/10.1111/geb.13433
Rosa L, Chiarelli DD, Rulli MC, Dell’Angelo J, D’Odorico P (2020) Global agricultural economic water scarcity. Sci Adv. 6(18):e6031. https://doi.org/10.1126/sciadv.aaz6031
Schimel JP (2018) Life in Dry Soils: Effects of Drought on Soil Microbial Communities and Processes. Annu Rev Ecol S 49(1):409–432. https://doi.org/10.1146/annurev-ecolsys-110617-062614
Shu D, Guo Y, Zhang B, Zhang C, Van Nostrand JD, Lin Y, Zhou J, Wei G (2021) Rare prokaryotic sub-communities dominate the complexity of ecological networks and soil multinutrient cycling during long-term secondary succession in China’s Loess Plateau. Sci Total Environ. 774:145737. https://doi.org/10.1016/j.scitotenv.2021.145737
Smith AP, Bond-Lamberty B, Benscoter BW, Tfaily MM, Hinkle CR, Liu C, Bailey VL (2017) Shifts in pore connectivity from precipitation versus groundwater rewetting increases soil carbon loss after drought. Nat Commun 8(1):1335. https://doi.org/10.1038/s41467-017-01320-x
Wei X, Qiu L, Shao M, Zhang X, Gale WJ (2012) The accumulation of organic carbon in mineral soils by afforestation of abandoned farmland. PLoS One. 7(3):e32054. https://doi.org/10.1371/journal.pone.0032054
Wei X, Hu Y, Razavi BS, Zhou J, Shen J, Nannipieri P, Wu J, Ge T (2019) Rare taxa of alkaline phosphomonoesterase-harboring microorganisms mediate soil phosphorus mineralization. Soil Biol Biochemistry 131:62–70. https://doi.org/10.1016/j.soilbio.2018.12.025
Wen E, Yang X, Chen H, Shaheen SM, Sarkar B, Xu S, Song H, Liang Y, Rinklebe J, Hou D, Li Y, Wu F, Pohorely M, Wong JWC, Wang H (2021) Iron-modified biochar and water management regime-induced changes in plant growth, enzyme activities, and phytoavailability of arsenic, cadmium and lead in a paddy soil. J Hazard Mater. 407:124344. https://doi.org/10.1016/j.jhazmat.2020.124344
Wen T, Xie P, Yang S, Niu G, Liu X, Ding Z, Xue C, Liu YX, Shen Q, Yuan J (2022) ggClusterNet: An R package for microbiome network analysis and modularity-based multiple network layouts. iMeta. 1(3):e23. https://doi.org/10.1002/imt2.32
Wu X, Wang S, Fu B, Liu Y, Zhu Y (2018) Land use optimization based on ecosystem service assessment: A case study in the Yanhe watershed. Land Use Policy 72:303–312. https://doi.org/10.1016/j.landusepol.2018.01.003
Wu MH, Chen SY, Chen JW, Xue K, Chen SL, Wang XM, Chen T, Kang SC, Rui JP, Thies JE, Bardgett RD, Wang YF (2021) Reduced microbial stability in the active layer is associated with carbon loss under alpine permafrost degradation. P Natl Acad Sci USA 118(25):e2025321118. https://doi.org/10.1073/pnas.2025321118
Yang Y, Dou Y, Wang B, Wang Y, Liang C, An S, Soromotin A, Kuzyakov Y (2022) Increasing contribution of microbial residues to soil organic carbon in grassland restoration chronosequence. Soil Biol Biochem. 170:108688. https://doi.org/10.1016/j.soilbio.2022.108688
Zhang Q, Ma J, Yang Y, Luo Z, Wang Y, Chen F (2021) Mining Subsidence-Induced Microtopographic Effects Alter the Interaction of Soil Bacteria in the Sandy Pasture. China. Front Env Sci. 9:656708. https://doi.org/10.3389/fenvs.2021.656708
Zhang Q, Ma J, Gonzalez-Ollauri A, Yang Y, Chen F (2022) Soil microbes-mediated enzymes promoted the secondary succession in post-mining plantations on the Loess Plateau. China Soil Ecol Lett 5(1):79–93. https://doi.org/10.1007/s42832-022-0148-0
Zhu Y, Ge X, Wang L, You Y, Cheng Y, Ma J, Chen F (2022) Biochar rebuilds the network complexity of rare and abundant microbial taxa in reclaimed soil of mining areas to cooperatively avert cadmium stress. Front Microbiol. 13:972300. https://doi.org/10.3389/fmicb.2022.972300
Zhu Y, Ma J, Chen F, Yu R, Hu G, Yang Y (2022b) Effective alleviation of Cd stress to microbial communities in mining reclamation soils by thiourea-modified biochar amendment. Pedosphere 32:866–875. https://doi.org/10.1016/j.pedsph.2022.06.017
Zhu Y, Wang L, You Y, Cheng Y, Ma J, Chen F (2022) Enhancing network complexity and function of soil bacteria by thiourea-modified biochar under cadmium stress in post-mining area. Chemosphere. 302:134811. https://doi.org/10.1016/j.chemosphere.2022.134811
Funding
This work was supported by the National Natural Science Foundation of China (Nos. 51974313, 52270171 and 41907405). We gratefully acknowledge the financial support from these programs.
Author information
Authors and Affiliations
Contributions
FC performed the supervision, conceptualization, methodology, project administration and funding acquisition. YZ and XS collected the samples. YZ, XS and XL performed the experiments. YZ performed the data analyses and wrote the manuscript. FC, JM and LW helped perform the analysis with constructive discussions. All authors contributed the revisions during the editing process. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Competing interests
The authors have no financial or proprietary interests in any material discussed in this article.
Additional information
Communicated by Fangbai Li.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1:
Table S1. Soil physicochemical properties of DL and PF treatments. Figure S1. Correlations between soil physicochemical properties and soil carbon pool. (a): DL treatment; (b): PFtreatment. DOC, dissolved organic carbon; MBC, dissolved organic carbon; EOC, easily oxidizable organic carbon; ROC, recalcitrant organic carbon; SOC, total organiccarbon; SOM, soil organic matter; CPMI, carbon pool management index. Asterisks denote significant correlation. * p < 0.05, ** p <0.01 and *** p < 0.001. The same below. Figure S2. Contribution of soil carbon pools and soil physicochemical properties to the top 20 OTUs of microorganisms based on correlation and random forest models. Circle size represents the variables’ importance (percentage of increase of mean square error calculated via random forest model). Colours represent Spearman correlations. Asterisks denote significant correlation. DL: (a, c, e, g); PF: (b, d, f, h). Abundant and rare bacteria: (a, b, c, d); Abundant and rare fungus: (e, f, g, h) Figure S3. Relationship between Shannon index and soil carbon pool (CPMI, DOC, MBC, EOC, SOC and ROC)in DL and PF treatments. Figure S4. Relationship of beta-diversity of abundant (a, b) and rare (c, d) bacterial communities and abundant (e, f) and rare (g, h) fungal communities (Bray-Curtis dissimilarity) to soil nutrients (a, c, e and g) and carbon pool (b, d, f and h) dissimilarities (Euclidean distance) in DL and PF treatments, respectively. Soil nutrients include TN, AN, NN, AP, AK. Circle in Figure represents Bary-Curtis weight. Figure S5. Redundancy analysis (RDA) showing the major drivers influencing abundant bacteria (a), rare bacteria (b), abundant fungus (c) and rare fungus taxa in DL and PF treatments at phylum levels. The individual effect of each soil property was calculated by the rdacca.hppackage. The length of the line connecting the arrow to the origin represents the degree of correlation between a certain environmental factor and community distribution and species distribution. The longer theline, the greater the correlation, and vice versa. Figure S6. Z-P plot of co-occurrence networks in DL (a, b, c) and PF (d, e, f) treatments. Abundant bacteria: (a), (d); Rare bacteria: (b), (e); Rare fungus: (c), (d). Figure S7. Co-occurrence network robustness, including natural connectivity and vulnerability in DL and PF treatments. Abundant bacteria: (a), rare bacteria: (b), rare fungus: (c).
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhu, Y., Wang, L., Song, X. et al. Changes in abundant and rare microbial taxa that dominated the formation of soil carbon pool during short-term dryland-to-paddy conversion. Carbon Res. 2, 26 (2023). https://doi.org/10.1007/s44246-023-00060-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44246-023-00060-6