Abstract
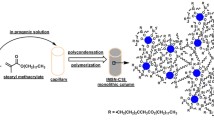
This article describes the fabrication of porous nicotinic acid-functionalized chito-oligosaccharide-bonded titania/silica hybrid monoliths (TiO2/SiO2@ChO-N) through a co-gelation sol–gel process. A capillary monolith with a well-defined and homogeneous structure was obtained by controlling the hydrolysis speed of titanium alkoxides in a sol mixture by using glycerol and acetylacetone. As a result of the functionalization with chito-oligosaccharides (ChO)-modified nicotinic acid, the obtained stationary phase provides superior physiochemical properties, such as a cationic hydrophilic surface, porosity, and mechanical strength. Scanning electron microscope and attenuated total reflectance-infrared spectroscopy were used to characterize the functionalized monolithic columns. The produced capillary columns showed high chromatographic performance with acceptable selectivity for charged analytes as well as organic polar compounds such as nucleic bases, nucleosides, carbamate pesticides, and strobilurin fungicides. The obtained results also indicated that the functionalized ChO’s amino, amide, hydroxyl, and pyridinium ring moieties served as hydrophilic electrostatic traps for charged substances, in addition to stroing π–π interaction with the carbamate pesticides and strobilurin fungicides analytes via hydrogen bonding.
Graphical abstract










Similar content being viewed by others
Availability of data and materials
The datasets used and analyzed during the current study are available.
References
T. Tennikova, F. Svec, B. Belenkii, J. Liq. Chromatogr. (1990). https://doi.org/10.1080/01483919008051787
H. Zou, X. Huang, M. Ye, Q. Luo, J. Chromatogr. A (2002). https://doi.org/10.1016/S0021-9673(02)00072-9
R.A. Wu, L. Hu, F. Wang, M. Ye, H. Zou, J. Chromatogr. A (2008). https://doi.org/10.1016/j.chroma.2007.09.022
H. Kobayashi, T. Ikegami, H. Kimura, T. Hara, D. Tokuda, N. Tanaka, Anal. Sci. (2006). https://doi.org/10.2116/analsci.22.491
I. de Paula Lima, S.P. Valle, M.A.L. de Oliveira, F.F. de Carvalho Marques, F.A.S. Vaz, Microchem. J. (2023). https://doi.org/10.1016/j.microc.2023.108598
K. Tsuge, L.W. Lim, T. Takeuchi, Anal. Sci. (2021). https://doi.org/10.2116/analsci.20P279
L.W. Lim, L. Rong, T. Takeuchi, Anal. Sci. (2012). https://doi.org/10.2116/analsci.28.205
M. Hefnawy, J. Chromatogr. A (2023). https://doi.org/10.1016/j.chroma.2023.463819
F. Svec, J.M. Frechet, Sci. (1996). https://doi.org/10.1126/science.273.5272.205
F. Furqani, L.W. Lim, T. Takeuchi, J. Indones. Chem. Soc. (2019). https://doi.org/10.34311/jics.2019.02.2.81
Z. Jiang, N.W. Smith, Z. Liu, J. Chromatogr. A (2011). https://doi.org/10.1016/j.chroma.2011.02.024
A. Khodabandeh, R.D. Arrua, S.C. Thickett, E.F. Hilder, A.C.S. Appl, Mater. Interfaces (2021). https://doi.org/10.1021/acsami.1c03542
L. Han, H. Liu, J. Zhang, J. Zhou, T. Jiang, Appl. Surf. Sci. (2022). https://doi.org/10.1016/j.apsusc.2022.154456
S.T. Wang, M.Y. Wang, X. Su, B.F. Yuan, Y.Q. Feng, Anal. Chem. (2012). https://doi.org/10.1021/ac301258q
L. Zhang, Y. Wang, W. Zhang, Y.I. Hsu, T.A. Asoh, B. Qi, H. Uyama, A.C.S. Biomater, Sci. Eng. (2022). https://doi.org/10.1021/acsbiomaterials.2c00385
M. Abi Jaoudé, J. Randon, Anal. Bioanal. Chem. (2011). https://doi.org/10.1007/s00216-011-4681
Y. Chen, Y. Yi, J.D. Brennan, M.A. Brook, Chem. Mater. (2006). https://doi.org/10.1021/cm060948d
G. Hasegawa, A. Kitada, S. Kawasaki, K. Kanamori, K. Nakanishi, Y. Kobayashi, H. Kageyama, T. Abe, J. Electrochem. Soc. (2014). https://doi.org/10.1149/2.0491501jes
S.-T. Wang, W. Huang, W. Lu, B.-F. Yuan, Y.-Q. Feng, Anal. Chem. (2013). https://doi.org/10.1021/ac4025297
S. Ma, Y. Li, C. Ma, Y. Wang, J. Ou, M. Ye, Adv. Mater. (2019). https://doi.org/10.1002/adma.201902023
X. Lv, ChemNanoMat (2022). https://doi.org/10.1002/cnma.202100460
B.H. Fumes, M.R. Silva, F.N. Andrade, C.E.D. Nazario, F.M. Lanças, TrAC Trends Anal. Chem. (2015). https://doi.org/10.1016/j.trac.2015.04.011
M. Sato, H. Hara, T. Nishida, Y. Sawada, J. Mater. Chem. (1996). https://doi.org/10.1039/JM9960601767
M.E.I. Badawy, M.A.M. El-Nouby, A.E.M. Marei, Int. J. Anal. Chem. (2018). https://doi.org/10.1155/2018/3640691
M.E.I. Badawy, A.E.-S.M. Marei, M.A.M. El-Nouby, Sep. Sci. Plus (2018). https://doi.org/10.1002/sscp.201800084
M.A.M. El-Nouby, M.E.I. Badawy, A.E.-S.M. Marei, Nanotechnol. Environ. Eng. (2021). https://doi.org/10.1007/s41204-021-00139-8
X. Huang, F.W. Foss Jr., P.K. Dasgupta, Anal. Chim. Acta (2011). https://doi.org/10.1016/j.aca.2011.09.028
W. Bragg, S.A. Shamsi, J. Chromatogr. A (2012). https://doi.org/10.1016/j.chroma.2012.08.002
Y. Jiang, B. Zhang, J. Li, Y. Sun, X. Wang, P. Ma, D. Song, Talanta (2021). https://doi.org/10.1016/j.talanta.2020.121799
Z. Lü, P. Zhang, L. Jia, J. Chromatogr. A (2010). https://doi.org/10.1016/j.chroma.2010.05.051
M.M. Jaworska, D. Antos, A. Górak, React. Funct. Polym. (2020). https://doi.org/10.1016/j.reactfunctpolym.2020.104606
C. Qiao, X. Ma, X. Wang, L. Liu, Lwt (2021). https://doi.org/10.1016/j.lwt.2020.109984
O. Ruzimuradov, S. Nurmanov, Y. Kodani, R. Takahashi, I. Yamada, J. Sol Gel Sci. Technol. (2012). https://doi.org/10.1007/s10971-012-2903-7
Y.A. Chesalov, G.B. Chernobay, T.V. Andrushkevich, J. Mol. Catal. A Chem. (2013). https://doi.org/10.1016/j.molcata.2013.03.007
J. Randon, J.-F. Guerrin, J.-L. Rocca, J. Chromatogr. A (2008). https://doi.org/10.1016/j.chroma.2008.10.108
F. Schindler, H. Schmidbaur, Angew. Chem. Int. Ed. (1967). https://doi.org/10.1002/anie.196706831
A.B. Zimmerman, A.M. Nelson, E.G. Gillan, Chem. Mater. (2012). https://doi.org/10.1021/cm3016534
G.K. Tat’yana, A.P. Safronov, E.V. Shadrina, M.V. Ivanenko, A.I. Suvorova, O.N. Chupakhin, J. Colloid Interface Sci (2012). https://doi.org/10.1016/j.jcis.2011.09.018
J. Ou, Z. Liu, H. Wang, H. Lin, J. Dong, H. Zou, Electrophoresis (2015). https://doi.org/10.1002/elps.201400316
W. Rupp, N. Hüsing, U. Schubert, J. Mater. Chem. (2002). https://doi.org/10.1039/B204956B
H.-J. Chen, L. Wang, W.-Y. Chiu, Mater. Chem. Phys. (2007). https://doi.org/10.1016/j.matchemphys.2006.02.007
C. Aydoğan, B. Beltekin, N. Demir, B. Yurt, Z. El Rassi, Molecules (2023). https://doi.org/10.3390/molecules28031423
Y. Chen, Q. Chen, L. Song, H.-P. Li, F.-Z. Hou, Microporous Mesoporous Mater. (2009). https://doi.org/10.1016/j.micromeso.2008.12.021
T. Hara, G. Desmet, G.V. Baron, H. Minakuchi, S. Eeltink, J. Chromatogr. A (2016). https://doi.org/10.1016/j.chroma.2016.03.009
S.-H. Jun, E.-J. Lee, S.-W. Yook, H.-E. Kim, H.-W. Kim, Y.-H. Koh, Acta Biomater. (2010). https://doi.org/10.1016/j.actbio.2009.06.024
P. Renoud, B. Toury, S. Benayoun, G. Attik, B. Grosgogeat, PLoS One (2012). https://doi.org/10.1371/journal.pone.0039367
A.J. Alpert, J. Chromatogr. A (1990). https://doi.org/10.1016/s0021-9673(00)96972-3
Z. Li, S. Li, F. Zhang, H. Geng, B. Yang, Talanta (2021). https://doi.org/10.1016/j.talanta.2021.122340
T. Takeuchi, T. Kawasaki, L.W. Lim, Anal. Sci. (2010). https://doi.org/10.2116/analsci.26.511
S. Jena, J. Dutta, K.D. Tulsiyan, A.K. Sahu, S.S. Choudhury, H.S. Biswal, Chem. Soc. Rev. (2022). https://doi.org/10.1039/D2CS00133K
M. Shahid, S. Manoharadas, H. Chakdar, A.F. Alrefaei, M.F. Albeshr, M.H. Almutairi, Chemosphere (2021). https://doi.org/10.1016/j.chemosphere.2021.130372
T. Inoue, Y. Nagatomi, K. Suga, A. Uyama, N. Mochizuki, J. Agric. Food. Chem. (2011). https://doi.org/10.1021/jf104421q
A.H. Hara, H.K. Kaya, Environ. Entomol. (1983). https://doi.org/10.1093/ee/12.2.496
M. Yaşar Mumin, H. Aral, M. Sunkur, T. Aral, Chem. Sel. (2022). https://doi.org/10.1002/slct.202204069
S. Noga, A. Felinger, B. Buszewski, J. AOAC Int. (2012). https://doi.org/10.5740/jaoacint.SGE_Noga
D.W. Bartlett, J.M. Clough, J.R. Godwin, A.A. Hall, M. Hamer, B. Parr-Dobrzanski, Pest Manag. Sci. (2002). https://doi.org/10.1002/ps.520
X. Wang, X. Li, Y. Wang, Y. Qin, B. Yan, C.J. Martyniuk, Environ. Pollut. (2021). https://doi.org/10.1016/j.envpol.2021.116671
Acknowledgements
This study was partly supported by JSPS Grants-in-Aid for Scientific Research (KAKENHI, Grant number: 18K05168), Japan. The authors acknowledge the MEXT scholarship from the Japanese government and instrumentation assistance from Prof. Fusheng Li.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of the author, the corresponding author states that there is no conflict of interest.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
El-Nouby, M.A.M., Lim, L.W. Mixed-mode chromatographic performance using nicotinic acid-functionalized chito-oligosaccharide-bonded Ti/Si hybrid monolithic capillary columns. ANAL. SCI. 39, 2019–2029 (2023). https://doi.org/10.1007/s44211-023-00416-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s44211-023-00416-3