Abstract
Scopoletin is a phytochemical of coumarins distributed in some plants. The aim of this study was the evaluation of scopoletin content and antioxidant activity of nettle, mallow, purslane, knot-grass, dill and coriander encountered in Black Sea regions. Analyses were conducted on the leaves and stems of these plants, except knot-grass. The lowest scopoletin content was found in nettle leaves (11.48 mg/kg) and the highest amount was found in knot-grass (6708.37 mg/kg). Scopoletin was not detected in stems of mallow, purslane and dill plants. The lowest total phenolic content was found in mallow stem (0.86 mg GAE/g) and the highest value was in knot-grass (67.74 mg GAE/g). The lowest ferric reducing antioxidant power (FRAP) was ascertained in purslane stem (1571.58 µmol Fe2+/g) and the highest value in coriander leaves (64905.90 µmol Fe2+/g). The mallow stem displayed the lowest DPPH radical scavenging activity of 48.63 µmol TE/g, while knot-grass provided the highest DPPH radical scavenging activity of 5261.56 µmol TE/g. These results revealed that knot-grass was the richest source of scopoletin and antioxidants. The studied plants exhibited a high interest in food, cosmetic and pharmaceutical industries.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introductıon
More than ten thousand phytochemicals have been figured out in food products. The use, importance and efficacy of these phytochemicals have been largely studied. The phytochemicals from different parts of fruits and vegetables can be ranged into chemical classes such as isoflavones, phytates, indoles, flavonoids, terpenes, phenolic acids, coumarins, polyphenols, lycopene, isothiocyanates, carotenoids and sulfides.
Coumarins are one of the subgroups of phytochemicals known as heterocycle benzopyran compounds resulted from the fusion of the pyrone ring and benzene ring. Two groups of the heterocyclic rings are identified depending on the position of the carbonyl group. The first group is coumarins which are resulted from the combination of α-pyrone and benzene rings, while the second one is chromon formed by fusing γ-pyrone and benzene rings. Coumarins have been largely reported in plants, however, their functions have not been completely defined. They are considered as a fungicide and bactericide as well as plant growth regulator [1].
Scopoletin and its β-D-glycoside form, scopolin, belong to coumarins. They are distinguished from other coumarins by their physicochemical properties and healing applications [2]. The synthesis of scopoletin is related to stress factors such as mechanic injury and drying, and it defends plants against microbiological attacks [2]. Scopoletin is a phytoalexin found in several kinds of plants in nature [3]. Plants containin scopoletin have been involved in the management of many symptoms and diseases such as convulsions, inflammation, rheumatic pains, leprosy, cardiovascular and neuromuscular diseases, asthma, hypertension, hyperthyroidism, cancer and hyperglycemia [4, 5].
Although fruits and vegetables from different kinds of families including Geraniaceae, Liliaceae, Musaceae, Compositae, Convolvulaceae, Loasaceae, Urticaceae, Apiaceae, Fabaceae, Malvaceae and Solanaceae have been reported to contain numerous phytochemicals, no study has investigated the scopoletin contents of nettle (Urtica diocia L.), mallow (Malva sylvestris L.), purslane (Portulaca oleracea L.), knot-grass (Polygonum cognatum Meissn), dill (Anethum graveolens L.) and coriander (Coriandrum sativum L.) grown in Turkey. Thus, the main aim of the present study was the evaluation the scopoletin contents of plants found wild or cultivated and prevalently consumed in Turkey. In addition, the antioxidant properties of plants were determined.
2 Materıals and methods
2.1 Material
Six different edible plants including nettle (Urtica diocia L.), common mallow (Malva sylvestris L.), purslane (Portulaca oleracea L.), knot-grass (Polygonum cognatum Meissn), dill (Anethum graveolens L.) and coriander (Coriandrum sativum L.) were collected from ten locations of Black sea Region (mainly from Samsun and Tokat), Turkey. In this study, the leaves and young stems of these plants were investigated, except knot-grass. Knot-grass was analysed as a whole plant due to its structure, as it has far smaller leaves than other plants. Prior to the analyses, the collected plants were washed with distilled water. Afterward, they were dried at room temperature and then the leaves and stems were separated. The leaves and stems were separately ground and stored in dark for further analyses.
2.2 Methods
2.2.1 Preparation of plant extracts
The dried and ground samples were soaked in 40% ethyl alcohol and extracted in an ultrasonic bath at 80–85 °C for 15 min. The extract was filtered and the solid phase was re-extracted 3–4 times following the same procedure. The extract was then concentrated with a rotary evaporator.
2.2.2 Moisture content (moist)
The moisture content was measured with a vacuum oven (NÜVE, FN 500P, Turkey) at 70 °C for 24 h according to the preliminary tests [6].
2.2.3 Determination of scopoletin content (SCO)
The analysis of scopoletin the same method as in Vipul et al. [7] was used with some modifications. Briefly, the extracts were filtered through a 0.45 μm Millipore filter and isocratically injected into the HPLC system. The isocratic elution was conducted according to chromatographic conditions. A Phenomenex C18 column (250 mm × 4.6 mm, 5 μm) (California, USA) was used. The eluent consisted of an isocratic mixture of methanol and water 30: 70 ratio containing 0.1% v/v formic acid. The mobile phase was filtered through a 0.45 μm Millipore filter and degassed by sonication for 30 min. A total of 20 μl of each extract was injected and HPLC operation was run at a flow rate of 1.0 ml/min and a wavelength of 366 nm.
Five different concentrations (20, 40, 60, 80, 100 ppm) were prepared from the scopoletin stock solution after dilution with the mobile phase and injected in triplicates. Thanks to the regression equation obtained from the standard curve, the concentrations were calculated with y = 4906× + 79,399, R2 = 0.996 equation. Results were expressed as mg/kg unit in dry matter.
2.2.4 Determination of total phenolic content (TP)
The total phenolic content was determined in the ethanolic extracts according to the method of Zannou et al. [8]. The total phenolic content was calculated and expressed as gallic acid equivalents per 100 g of (mg GAE/100 g) based on the gallic acid standard curve.
2.2.5 Determination of antioxidant activities
The antioxidant activity was evaluated with two different techniques including Ferric Reducing Antioxidant Power (FRAP) and DPPH radical scavenging activity.
2.2.5.1 Ferric reducing antioxidant power (FRAP)
FRAP analysis were conducted following the method described in Pashazadeh et al. [9]. The FRAP values were calculated by standard curves prepared from FeSO47H2O and expressed as µmol Fe2+ per gram of dry matter.
2.2.5.2 DPPH free radical scavenging activity
DPPH radical scavenging capacity of samples was determined according to the procedure reported in Pashazadeh et al. [9]. The Trolox calibration curve was plotted as a function of the percentage of DPPH radical scavenging activity. The results were expressed as µmol Trolox (TE) per gram of dry matter.
2.2.6 Statistical analyses
The relation between total phenolic content and antioxidant activity of plants was determined with the help of the SPSS 16 statistical software. Differences between averages were detected with a Duncan multiple comparison test (p < 0.05). In addition, correlation analysis was conducted with the Pearson method (p < 0.05).
3 Results and discussion
The scopoletin content and antioxidant activity of six different plants including nettle (Urtica diocia L.), mallow (Malva sylvestris L.), purslane (Portulaca oleracea), knot-grass (Polygonum cognatum), dill (Anethum graveolens) and coriander (Coriandrum sativum) were investigated in the present study. The results have shown diversity in scopoletin content and antioxidant activity depending on the type and parts of plants.
3.1 Scopoletin content and antioxidant activity of nettle
The results of scopoletin content and antioxidant activity determined in the leaves and stems of nettle were presented in Table 1. As can be seen, the scopoletin content was evaluated in ten different samples of nettle leaves and seven of them contained scopoletin. The lowest amount of scopoletin in nettle leaves was 11.48 mg/kg dry basis, whereas, the highest value in nettle leafy part was 79.35 mg/kg dry basis). It was remarked that the scopoletin content of some samples in nettle leafy was below the detection limits. A range of 15.51–150.44 mg/kg dry basis of scopoletin content was found in the stem. The nettle stem displayed higher amount of scopoletin when compared to the leaf part.
Sajfrtova et al. [10] have determined the scopoletin contents ranged from 16 mg/kg to 56 mg/kg in nettle roots. Likewise, Orčić et al. [11] have assessed that the roots and flowers of nettle had the highest scopoletin contents of 91.42 µg/g and 180.4 µg/g, respectively. Francišković et al. [12] have reported the amounts of scopoletin ranged from 13.42 to 34.38 µg/g and 52.61 and 111.5 µg/g in nettle. These findings were in accordance with the results of the present study. It has been reported that the extraction techniques, solvents and cultivation conditions could affect the scopoletin content of nettle [10, 11]. The moisture contents varied between 74.81–87.38% and 86.40, and 91.79% for nettle leaves and stems, respectively. Similarly Shonte et al. [13] have found a moisture content of 85.00% in the nettle collected from South Africa. As can be seen in Table 1, the nettle leaves displayed the highest total phenolic content (10.00–31.45 mg GAE/g) and antioxidant activity when compared to nettle stems. Kőszegi et al. [14] have reported less total phenolic content of 0.7–1.6 mg GAE/g in nettle. Previously, many studies have investigated the effects of the phenolic content of fruits and vegetables on antioxidant activity. With respect to our results, nettle leaves displayed significant relations (p < 0.01) between phenolic content, FRAP (r = 0.847**) and DPPH (r = 0.957**), as well as FRAP and DPPH (r = 0.810**). Similarly, in nettle stems, the correlations between phenolic content and FRAP (r = 0.728*) as well as FRAP and DPPH (r = 0.881**) were significant (p < 0.05). However, the scopoletin contents of both leaf and stem of nettle have not an effect on the total phenolic content and antioxidant activity.
3.2 Scopoletin content and antioxidant properties of purslane
As can be seen in Table 2, scopoletin was found in 4 samples purslane leaves. No scopoletin was detected in purslane stems. The level of scopoletin in purslane leaves was ranged from 44.19 mg/kg to 162.18 mg/kg dry matter basis. The reason of the absence of scopoletin in some purslane samples has been thought to be environmental conditions, as this metabolite is mostly secreted in the plants under stress. Furthermore, the accumulation of scopoletin in plants has shown significant changes in respect with habitats.
Xin et al. [15] have tentatively identified scopoletin together with two terpenoids in purslane. They reported that terpenoids isolated from purslane have low cytotoxic effects. According to the findings of Ai et al. [16], purslane plants had scopoletin contents varying between 0.121 µg/ml and 81.89 µg/ml. They have notified that the difference in the quantity of scopoletin content of purslane samples might be linked to the origin of the plant, suggesting that this diversity could be used to assess the quality of purslane. The moisture contents of purslane leaves and stems were found between of 91.10–94.17% and 88.50–93.64%, respectively. The lowest total phenolic contents were 3.52 and 1.54 mg/g and the highest were 9.35 and 3.96 mg/g in leaves and stems, respectively. FRAP determined in leaves and stems were 3845.49–14,084.04 µmol/g and 1571.58–3883.11 µmol/g, respectively. The lowest values of DPPH radical scavenging activity were 174.75 and 65.78 µmol/g, and the highest values were 631.67 and 214.92 µmol/g in leaves and stems, respectively. As can be seen, the values of total phenolic content and antioxidant activity of purslane were higher in leaves. Moukette et al. [17] have reported in purslane DPPH radical scavenging activity, FRAP and total phenolic compounds of 3.07 µg/ml, 0.14 µg/ml in 50 µg/ml and 127.5 mg/ g, respectively. There was a significant relationship between total phenolic content, FRAP (r = 0.894**) and DPPH (r = 0.788**) as well as FRAP and DPPH (r = 0.785**) (p < 0.01) of purslane leaves. Similarly, in the stems, a correlation was found between total phenolic content and FRAP (r = 0.703*, p < 0.05) and between total phenolic content and DPPH (r = 0.799**, p < 0.01) as well.
3.3 Scopoletin content and antioxidant properties of dill
Ten samples of dill were investigated and only dill leaves displayed scopoletin (Table 3). As can be seen, scopoletin was detected in 9 out of 10 dill leaves samples investigated. The lowest scopoletin content was 27.81 mg/kg, whereas the highest value was 241.54 mg/kg dry matter.
Scopoletin was detected in 9 samples out of 10 dill leaves samples. But scopoletin was not found in dill stems. The lowest and highest amounts of scopoletin in dill leaves were 27.81 mg/kg and 241.54 mg/kg (dry matter), respectively. Scopoletin has been previously reported in dill leaves, flowers and fruits [18, 19]. In this study, the scopoletin content detected in dill were higher than the values identified previously in 50% ethanol, methanol and water extracts of C. pluricaulis, evaluated as 0.1738%, 0.0932%, 0.0435%, respectively [7].
The moisture contents of dill leaves and stems were 81.83–88.13% and 87.15–91.77%, respectively. The lowest total phenolic contents were 11.27 and 2.83 mg/g, and the highest were 34.99 and 17.76 mg/g in leaves and stems, respectively. In contrast, Wasli et al. [20] have previously reported less total phenolic content ranging from 2.34 to 9.97 mg/g. The lowest FRAP in leaves and stems were 16037.59 and 2111.88 µmol/g and the highest FRAP were 51310.04 and 18616.85 µmol/g, respectively. For DPPH, the lowest values were found to be 667.10 and 150.40 µmol/g, while the highest values were 2503.31 and 609.14 µmol/g in leaves and stems, respectively. It was noticed that total phenolic content and antioxidant activity values of dill were higher in leaves than in stems. Similar results have been found by Ortan et al. [19] who affirmed that the polyphenol content of flower and leaves of dill are higher than the values detected in its fruits. Significant relation (p < 0.01) was found between FRAP and DPPH values of dill leaves (r = 0.950**).
3.4 Scopoletin content and antioxidant properties of coriander
Scopoletin content and antioxidant activity of coriander were given in Table 4. Scopoletin was analysed in ten samples of coriander leaves and stems. As shown in Table 4, scopoletin was detected in five leaf samples and three stems samples. The lowest amounts of scopoletin were 24.79 and 25.68 mg/kg, and the highest 155.4 and 37.37 mg/kg in leaves and stems, respectively. The differences observed were considered to depend on various stress factors and environmental conditions.
Scopoletin were subsequently reported in the fruits of coriander [18, 21]. Similarly, Aissaoui et al. [22] have identified umbelliferone and scopoletin as the hydroxy coumarins isolated from extracts of coriander seed samples. Oganesyan [23] have found scopoletin content of 1.79% in 96% ethyl alcohol extract and 2.03% in 40% ethyl alcohol extract of coriander green parts. Their findings were higher than our results. The moisture of coriander leaves was ranged between 83.29 and 89.85%. The lowest total phenolic content, FRAP and DPPH were 13.85 mg/g, 23297.07 µmol/g and 573.75 µmol/g, respectively. The highest values were 32.70 mg/g, 64905.90 µmol/g and 4580.67 µmol/g for total phenolic content, FRAP and DPPH, respectively. Rathore et al. [24] have noticed that coriander leaves displayed stronger antioxidant activity than the seeds and remarked a positive correlation between total phenolic content and antioxidant activity. Saxena et al. [25] have reported the maximum values of total phenolic content and DPPH as 92.99 mg/g and 10.85 mg/g in coriander seeds, respectively. The moisture of coriander stem was found between 87.58 and 93.34%. Total phenolic content, FRAP and DPPH were found in the ranges of 3.07–27.39 mg/g, 3766.40–33917.58 µmol/g and 159.99–828.04 µmol/g, respectively. As can be seen, total phenolic content and antioxidant values of coriander are higher in leaves. In coriander leaves a significant relation between the content of phenolic compounds and FRAP (r = 0.849**) was determined. In stem parts, a relationship between phenolic content, FRAP (r = 0.938**) and DPPH (r = 0.874**) values was determined (p < 0.01). Furthermore, in the stems, the correlation was significant (p < 0.01) between FRAP and DPPH (r = 0.878**).
3.5 Scopoletin content and antioxidant properties of common mallow
Scopoletin content and antioxidant activity of common mallow were summarized in Table 5. In the leaves, the lowest amount of scopoletin was 38.93 mg/kg, while, the highest was 103.56 mg/kg. Scopoletin was not found in stems. In the previous studies, scopoletin has been reported in the aerial parts of mallow [26, 27]. Gasparetto et al. [28] have identified scopoletin and 5,7-dimethylhydroxycoumarin in the leaves of mallow.
The moisture contents leaves were found between 82.05 and 91.65%. The total phenolic content, FRAP and DPPH were ranked as 8.62–20.84 mg/g, 15529.89–31905.62 µmol/g and 633.70–1314.15 µmol/g, respectively. DellaGreca et al. [29] have reported 24% of radical scavenging capacity in mallow leaves. Barros et al. [30] have fond the highest antioxidant activity in mallow leaves when compared to fruits. Gasparetto et al. [28] have found total phenolic contents of 386.5 mg/g, 317.0 mg/g, 258.7 mg/g and 56.8 mg/g in mallow leaves, flowered stems, flower and immature fruits. These values were higher than those found in this study. This difference is obviously linked to the origin of plant material and experimentation conditions. The moisture of mallow stems was detected between 86.40% and 91.79%. The total phenolic content, FRAP and DPPH were found in the ranges of 0.86–17.25 mg/g, 1653.57–5129.87 µmol/g, 48.63–273.79 µmol/g, respectively. Beghdad et al. [31] have reported the total phenolic content of 24.123 mg/g and 2.173 mg/g in mallow leaves and stems was 2.173 mg/g, respectively. These results are higher than our findings when considered the leaves, however, stems showed higher values. In the leaves, a correlation was found between phenolic content, FRAP (r = 0.646*) and DPPH values (r = 0.714*) in (p < 0.05), and between FRAP and DPPH (r = 0.845**) (p < 0.01). In its stems, a significant correlation between phenolic content and FRAP (r = 0.751*) and between FRAP and DPPH (r = 0.672*, p < 0.05) were found.
3.6 Scopoletin content and antioxidant properties of Knot-grass
Knot-grass was analysed as a whole plant due to its structure, as it has far smaller leaves than other leaves investigated in the present study. Scopoletin contents and antioxidant activity of knot-grass plants were given in Table 6.
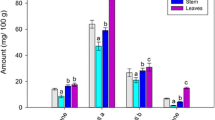
The amounts of scopoletin in knot-grass were ranged from 1833.87 mg/kg and 6708.37 mg/kg. Scopoletin was detected in 8 knot-grass samples out of 10 studied samples. Knot-grass displayed the highest amount of scopoletin amongst the investigated plants. Knot-grass is likely the most under stress due to its creeping abilities. To the authors’ knowledge, no literature on the scopoletin content of knot-grass has been published. However, other species from the same family, namely P. aviculare were reported to contain scopoletin [32]. Likewise, Dudek-Makuch and Mat£awska [33] have recorded 0.4133% of scopoletin in chestnut. The moisture of knot-grass leaves was detected between of 81.83 and 88.13%. The total phenolic content, FRAP and DPPH were ranked as 8.11–67.74 mg/g, 10983.58–48354.09 µmol/g and 654.67–5261.56 µmol/g, respectively. Yildirim et al. [34] have recorded a range of 0.01–0.50 μg/ml of total phenolic in knot-grass. A correlation was found between phenolic content and FRAP (r = 0.984**) and DPPH values (r = 0.913**) (p < 0.01), and between FRAP and DPPH (r = 0.860**) (p < 0.01). Scopoletin content of knot-grass plant did not show a correlation between the total phenolic content and antioxidant activity. The average values of moisture, total phenolic content, FRAP and DPPH of all the investigated plants in the present study and their statistical differences were given in Table 7. As can be seen, a significant difference was observed between moisture, total phenolic content, FRAP and DPPH these plants. Purslane had the highest moisture content, while the highest total phenolic content, FRAP and DPPH free radical scavenging activity was determined in coriander leaves. When considered total phenolic content, no statistical difference was detected between the results of knot-grass, nettle leaves and dill leaves.
4 Conclusion
Coumarin is one of the subgroups of phytochemicals which are commonly used especially in medicine. Scopoletin is also a kind of coumarin whose bio-activity has been proved by several medicinal studies.Scopoletin content and antioxidant activity of six different edible plants collected from ten locations were determined. At the end of the conducted study, scopoletin was detected in all leaves of nettle, purslane, dill, coriander and mallow. Scopoletin wasn’t determined in stem parts of purslane, dill and mallow. Knot-grass was analysed as a whole plant due to has far smaller leaves. When the scopoletin content are considered, the lowest quantity of scopoletin was determined content in nettle leaves and the highest quantity of scopoletin was detected in the knot-grass. The lowest total phenolic content, FRAP and DPPH free radical scavenging power values were as certained in stem parts of purslane, the highest total phenolic matter content and DPPH free radical scavenging power values were determined in knot-grass plant. While the highest FRAP value was detected in leaves of coriander. Few studies have reported scopoletin content in foods. Therefore, the findings of the present study contributed to the literature in terms of scopoletin content and antioxidant activity of the plants investigated. In addition, the studied plants can be used to produce antioxidant-based medicaments and antioxidant-enriched foods.
Data availability
The dataset generated for this study are available on request to the corresponding author.
References
Venugopala KN, Rashmi V, Odhav B. Review on natural coumarin lead compounds for their pharmacological activity. Biomed Res Int. 2013. https://doi.org/10.1155/2013/963248.
Shaw CY, Chen CH, Hsu CC, Chen CC, Tsai YC. Antioxidant properties of scopoletin isolated from Sinomonium acutum. Phyther Res. 2003;17:823–5. https://doi.org/10.1002/ptr.1170.
Siwinska J, Kadzinski L, Banasiuk R, Gwizdek-Wisniewska A, Olry A, Banecki B, Lojkowska E, Ihnatowicz A. Identification of QTLs affecting scopolin and scopoletin biosynthesis in Arabidopsis thaliana. BMC Plant Biol. 2014;14:1–14. https://doi.org/10.1186/s12870-014-0280-9.
Gnonlonfin GJB, Sanni A, Brimer L. Review Scopoletin—a Coumarin Phytoalexin with medicinal properties. CRC Crit Rev Plant Sci. 2012;31:47–56. https://doi.org/10.1080/07352689.2011.616039.
Yuan C, Wang MH, Wang F, Chen PY, Ke XG, Yu B, Yang YF, You PT, Wu HZ. Network pharmacology and molecular docking reveal the mechanism of Scopoletin against non-small cell lung cancer. Life Sci. 2021. https://doi.org/10.1016/j.lfs.2021.119105.
Pashazadeh H, Zannou O, Galanakis CM, Aldawoud TMS, Ibrahim SA, Koca I. Optimization of drying process for Rosa pimpinellifolia L. fruit (black rose hips) based on bioactive compounds and modeling of drying process. Int J Food Prop. 2021;24:1367–86. https://doi.org/10.1080/10942912.2021.1967384.
Vipul U, Neeru S, Amit TK, Amreesh M, Brijpal S, Singh B. Standardization of HPLC method of scopoletin in different extracts of Convolvulus pluricaulis. Int J Pharm Sci Drug Res. 2013;5:28–31.
Zannou O, Koca I, Aldawoud TMS, Galanakis CM. Recovery and stabilization of anthocyanins and phenolic antioxidants of Roselle (Hibiscus sabdariffa L.) with hydrophilic deep eutectic solvents. Molecules. 2020;25:3715. https://doi.org/10.3390/molecules25163715.
Pashazadeh H, Özdemir N, Zannou O, Koca I. Antioxidant capacity, phytochemical compounds, and volatile compounds related to aromatic property of vinegar produced from black rosehip (Rosa pimpinellifolia L.) juice. Food Biosci. 2021. https://doi.org/10.1016/j.fbio.2021.101318.
Sajfrtová M, Sovová H, Opletal L, Bártlová M. Near-critical extraction of β-sitosterol and scopoletin from stinging nettle roots. J Supercrit Fluids. 2005;35:111–8. https://doi.org/10.1016/j.supflu.2004.12.008.
Orcic D, Franciškovic M, Bekvalac K, Svircev E, Beara I, Lesjak M, Mimica-Dukic N. Quantitative determination of plant phenolics in Urtica dioica extracts by high-performance liquid chromatography coupled with tandem mass spectrometric detection. Food Chem. 2014;143:48–53. https://doi.org/10.1016/j.foodchem.2013.07.097.
Francišković M, Gonzalez-Pérez R, Orčić D, Sánchez de Medina F, Martínez-Augustin O, Svirčev E, Simin N, Mimica-Dukić N. Chemical composition and immuno-modulatory effects of Urtica dioica L. (stinging nettle) extracts. Phyther Res. 2017;31:1183–91. https://doi.org/10.1002/ptr.5836.
Shonte TT, Duodu KG, de Kock HL. Effect of drying methods on chemical composition and antioxidant activity of underutilized stinging nettle leaves. Heliyon. 2020. https://doi.org/10.1016/j.heliyon.2020.e03938.
Kőszegi K, Vatai G, Békássy-Molnár E. Comparison the soxhlet and supercritical fluid extraction of nettle root (Urtica dioica L.). Period Polytech Chem Eng. 2015;59:168–73. https://doi.org/10.3311/PPch.
Xin H-L, Hou Y-H, Xu Y-F, Yue X-Q, Li M, Lu J-C, Ling C-Q. Portulacerebroside a: new cerebroside from Portulaca oleracea L. Chin J Nat Med. 2008;6:401–3. https://doi.org/10.1016/s1875-5364(09)60033-9.
Ai J, Leng A, Gao X, Zhang W, Li D, Xu L, Ying X. HPLC determination of the eight constitutes in Portulaca oleracea L. from different locations. Eur J Med Plants. 2015;5:156–64. https://doi.org/10.9734/ejmp/2015/13253.
Moukette BM, Ama Moor VJ, Biapa Nya CP, Nanfack P, Nzufo FT, Kenfack MA, Ngogang JY, Pieme CA. Antioxidant and synergistic antidiabetic activities of a three-plant preparation used in Cameroon folk medicine. Int Sch Res Not. 2017;2017:1–7. https://doi.org/10.1155/2017/9501675.
Suhaj M. Spice antioxidants isolation and their antiradical activity: a review. J Food Compos Anal. 2006;19:531–7. https://doi.org/10.1016/j.jfca.2004.11.005.
Ortan A, Popescu ML, Gaita AL, Dinu-Pîrvu C, Câmpeanu GH. Contributions to the pharmacognostical study on Anethum graveolens, Dill (apiaceae). Rom Biotechnol Lett. 2009;14:4342–8.
Wasli H, Jelali N, Silva AMS, Ksouri R, Cardoso SM. Variation of polyphenolic composition, antioxidants and physiological characteristics of dill (Anethum graveolens L.) as affected by bicarbonate-induced iron deficiency conditions. Ind Crops Prod. 2018;126:466–76. https://doi.org/10.1016/j.indcrop.2018.10.007.
Adam M, Dobiáš P, Eisner A, Ventura K. Extraction of antioxidants fom plants using ultrasonic methods and their antioxidant capacity. J Sep Sci. 2009;32:288–94. https://doi.org/10.1002/jssc.200800543.
Aissaoui A, Zizi S, Israili ZH, Lyoussi B. Hypoglycemic and hypolipidemic effects of Coriandrum sativum L. in Meriones shawi rats. J Ethnopharmacol. 2011;137:652–61. https://doi.org/10.1016/j.jep.2011.06.019.
Oganesyan ET, Nersesyan ZM, Parkhomenko AY. Chemical composition of the above-ground part of Coriandrum sativum. Pharm Chem J. 2007;41:149–53. https://doi.org/10.1007/s11094-007-0033-2.
Rathore SS, Saxena SN, Singh B. Potential health benefits of major seed spices. Int J Seed Spices. 2013;3:1–12.
Saxena SN, Sharma YK, Rathore SS, Singh KK, Barnwal P, Saxena R, Upadhyaya P, Anwer MM. Effect of cryogenic grinding on volatile oil, oleoresin content and anti-oxidant properties of coriander (Coriandrum sativum L.) genotypes. J Food Sci Technol. 2015;52:568–73. https://doi.org/10.1007/s13197-013-1004-0.
Prudente AS, Loddi AMV, Duarte MR, Santos ARS, Pochapski MT, Pizzolatti MG, Hayashi SS, Campos FR, Pontarolo R, Santos FA, et al. Pre-clinical anti-inflammatory aspects of a cuisine and medicinal millennial herb: Malva sylvestris L. Food Chem Toxicol. 2013;58:324–31. https://doi.org/10.1016/j.fct.2013.04.042.
Zakhireh S, Jalilzadeh Hedayati M, Bahari S. Identification of non-polar compounds of aerial organs of Malva sylvestris L. and its antioxidant effects. Life Sci J. 2013;10:2934–7.
Gasparetto JC, Martins CAF, Hayashi SS, Otuky MF, Pontarolo R. Ethnobotanical and scientific aspects of Malva sylvestris L.: a millennial herbal medicine. J Pharm Pharmacol. 2012;64:172–89. https://doi.org/10.1111/j.2042-7158.2011.01383.x.
DellaGreca M, Cutillo F, D’Abrosca B, Fiorentino A, Pacifico S, Zarrelli A. Antioxidant and radical scavenging properties of Malva sylvestris. Nat Prod Commun. 2009;4:893–6. https://doi.org/10.1177/1934578x0900400702.
Barros L, Carvalho AM, Ferreira ICFR. Leaves, flowers, immature fruits and leafy flowered stems of Malva sylvestris: a comparative study of the nutraceutical potential and composition. Food Chem Toxicol. 2010;48:1466–72. https://doi.org/10.1016/j.fct.2010.03.012.
Mohammed CB, Chahid B, Fatima B, Fatima-Zohra S, Meriem B, Farid C. Antioxidant activity, phenolic and flavonoid content in leaves, flowers, stems and seeds of mallow (Malva sylvestris L.) from North Western of Algeria. Afr J Biotechnol. 2014;13:486–91. https://doi.org/10.5897/ajb2013.12833.
Costea M, Tardif FJ. The biology of Canadian weeds. 131. Polygonum aviculare L. Can J Plant Sci. 2005;85:481–506. https://doi.org/10.4141/P03-187.
Dudek-Makuch M, Matławska I. Coumarins in horse chestnut flowers: isolation and quantification by UPLC method. Acta Pol Pharm Drug Res. 2013;70:517–22.
Yildirim A, Mavi A, Kara AA. Antioxidant and antimicrobial activities of Polygonum cognatum Meissn extracts. J Sci Food Agric. 2003;83:64–9. https://doi.org/10.1002/jsfa.1288.
Acknowledgements
We thank the Ondokuz Mayis University Scientific Research Projects Office (Grant Number PYO. MUH.1904.16.012) for funding this project.
Author information
Authors and Affiliations
Contributions
I.K assured the project management and founding acquisition. All authors made the formal analysis, wrote and reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Çakır, D.K., Zannou, O. & Koca, I. Scopoletin contents and antioxidant properties of some edible plants of Black Sea regions. Discov Food 2, 7 (2022). https://doi.org/10.1007/s44187-022-00010-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44187-022-00010-y