Abstract
Ficus auriculata (Timla) is an underutilized wild fruit compared to the more commonly consumed fruit of the same genus, Ficus carica (Anjeer). This review focuses on the industrial and pharmaceutical benefits of F. auriculata while highlighting its potential as a valuable alternative to F. carica. Based on the vast literature found in Scopus database, both fruits have significant nutritional and medicinal value; however, due to the rarity and lack of widespread knowledge, F. auriculata still remains underutilized, unlike F. carica, which is well known and more popular in the human diet. In terms of nutrition, F. auriculata is superior in various aspects like protein (3–5.32%), magnesium (68.0 mg/100 g), potassium (329.0 mg/100 g), and iron (5.0 mg/100 g). As a result, it plays a pivotal role for addressing nutrient deficiencies. Besides, it has rich history of many noteworthy applications in traditional remedies which are also recognized in modern pharmaceutical research. This could be attributed to the presence of phytocompounds. Ficus auriculata needs to be promoted, not only as a nutritional addition to the plate but also as an economically viable fruit. Based on its considerable potential, it is possible that F. auriculata can also be used as an alternative to F. carica; however, further research and promotion of the benefits of F. auriculata could help to increase its utilization and provide additional health and economic benefits to communities. Moreover, omics technologies based comprehensive study of fruits can assist in the identification of bioactive metabolites, nutritional content, and possible health benefits. It offers insights into the genetic composition, gene expression patterns, protein activities, and metabolic pathways of fruits thereby revealing the mechanisms driving flavor, color, and disease resistance.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Wild fruits are an important resource for many communities around the world, as they provide not only health benefits but also seasonal income. Despite being rare, wild fruits are known for their nutritional value and health benefits. Phytochemicals present in wild fruits, such as flavonoids, anthocyanins and phenolics, can be attributed to their different genotypes, environmental factors and growth conditions [1].
Ficus auriculata is one such underutilized wild fruit that belongs to the Moraceae family, which consists of more than 700 species; widely distributed throughout the tropics and subtropics. Ficus auriculata plants are commonly known as “Roxburg Fig” or “Elephant ear fig”. A trait specific to F. auriculata, i.e., produces fruit that changes color from green to red or purple as it ripens, in addition to its towering height and broad leaves. These fruits are edible and can be incorporated into the human diet, while the leaves can be a good source of fodder for animals [2]. These fruits are commonly known as ‘Trimul’ in Hindi inhabiting the hilly areas. Ficus auriculata is also known as Timbal, Timla, Timal, Tirmal, Tremal, Trimmal and Gular udumbara in India [3]. The fruits are available twice a year and known for their delectable, sweet taste when fully ripe. Fresh consumption of fruits is a popular choice due to their high content of dietary fiber, natural sugar, and minerals, making them a nutritious appetizer. In addition, it possesses diuretic, laxative, and digestive regulatory properties, providing relief against diabetes and constipation [4]. The health benefits of fruits are attributed to their diverse phytochemical content, including anthocyanins, antioxidants, flavonoids, tannins, and phenolic compounds although it was found that the plant did not contain alkaloids [5]. Its medicinal properties are not limited to the fruit, other parts, such as roots, leaves, stems, bark, and latex, are traditionally used by various communities [6] as therapeutics.
The most common and famous fruit of the Ficus genus is F. carica, commonly known as Fig or Anjeer. Sometimes it is also referred to as “the fruit of the heaven” [7]. It contains high amounts of bioactive compounds such as cyanidin, rutin, luteolin, chlorogenic acid, and ( +)-catechin, which are not only limited to its fruits but can also be found in the peel and leaves [8]. Numerous researchers have identified bioactive compounds in a wide range of fruit species that exhibit a variety of beneficial effects on the management and prevention of various diseases, including antidiabetic, anticancer, and anti-inflammatory effects. Fig fruits are widely recognized for their delectable taste and impressive nutritional profile. They are an abundant source of proteins, amino acids, carbohydrates, minerals, vitamins, and dietary fiber. In terms of the nutritive index, fig fruits had notably greater values than other fruits, such as apple, raisin, and date fruits, with values of 11, 9, 8 and 6, respectively. Dried figs are a nutrient-rich food and are often regarded as a poor man’s diet in Mediterranean countries [7]. Red wine and coffee are known for their phenolic compounds, but it was found that fig fruits contain more phenolic compounds [9]. To fill in the gaps and find new potential scope for F. auricualta, this review focuses on the comparative metabolic profiles as well as industrial and pharmaceutical benefits of F. auriculata and F. carica.
1.1 Approaches for data collection
The initial search for the current review is done based on the keywords searched within databases and resulted in a total number of 4,832 articles. The keywords used for the initial search were “Ficus auriculata”, “Ficus carica”, “Underutilized”, “Bioactive”, “nutritional security”. These keywords were used individually and with the combination of others. All the articles were manually scrutinized. The further screening process is mentioned in Fig. 1.
1.2 Characteristics of the plant
Ficus auriculata is an evergreen tree with a height of 4–10 m that is scattered in tropical, subtropical, and temperate regions at altitudes of 1800–2600 m [10]. It is a native plant of Asia and is widely distributed in Nepal, India, Bhutan, Myanmar, China, Vietnam, Pakistan, Thailand, and Malaysia [10]. Morphologically, with spreading limbs lifted by roots, the tree has rough-textured, grayish-brown bark. The leaves are deciduous and attached to the stem, which is 8.5 cm long and oval-shaped with a rounded tip and heart-shaped base. The leaves are 21.5 cm long and 23.5 cm wide, with vein patterns similar to those of a net. It has unisexual flowers; male flowers are approximately 4 mm long, while female flowers are 6 mm long. Both types of flowers have modified petals and sepals that look like thread-like scales. The fruits are approximately 30.55 g in weight and pear-shaped, and they have a diameter of 4.5 cm and a stalk that is also 4.5 cm long. The fruit has an opening at the top, which is guarded by scales and turns yellowish to purple as it matures. It contains enclosed small seeds that are eaten along with the fruit. Edible fruits can be processed into juices, jams, and curries in India. In Vietnam, unripe fruits are used in salads. The color of the ripened and mature fruit is dark red [3, 6, 11]. Similarly, F. carica is a rapidly growing tree with varying heights depending on the genotype, ranging from 1 m (3 feet) to 10–12 m tall [12]. The tree’s bark is silver-gray and smooth, and the leaves have 3–5 lobes and are approximately 4–6 cm long. The fruits are known as Anjeer, are peer shaped, and can range in length from 1 to 4 in, with varying colors as they ripen from yellowish green to dark purple [13].
1.3 Phytochemistry
Phytochemical screening revealed the presence of flavonoids, phenols, tannins, resins, and glycosides, while alkaloids were absent in F. auriculata. Leaf extracts contain carbohydrates, flavonoids, phenols, terpenoids, glycosides, tannins, and alkaloids, while saponins are absent. The leaves were found to contain phenolic compounds that exhibit a broad range of bioactive properties. The total phenol content of the leaves was 21.404 mg GAE/mg dry weight, while the total flavonoid content was 50.83 ± 1.32 mg GAE/mg dry weight.
The stem bark extract of F. auriculata contains alkaloids, fatty oils, saponins, carbohydrates, glycosides, resins, phytosterols, tannins, phenols, flavonoids, amino acids, and proteins [14]. The phytocompounds and nutritional profiles (fruit) of the different parts of the tree are listed in Tables 1 and 2. Amino acid composition analysis of F. auriculata fruit revealed the presence of l-hydroxyproline, DL-isoleucine, DL-valine, DL-2-aminobutyric acid, DL-tryptophan, DL-alanine, l-arginine, DL-aspartic acid, l-cysteine hydroxychloride, l-threonine, and l-tyrosine [15]. Zeaxanthin, lutein, beta-cryptoxanthin, Beta-carotene, alpha-cryptoxanthin, and lycopene are some of the common carotenoid found in the Ficus genus, gallic acid, syringic acid, chlorogenic acid, epicatechin, catechin, 3-caffeolyquinic acid, rutin, 5-caffeolyquinic acid, kaempferol-3-rutinoside, quercetein-3-glucoside, quercetein-3-acetylglycoside, qurcetein-3-rutinoside, ferulic acid, luteolin-8-c-glucoside, bergapten, kaempferol-3-glucoside, cyanidin-3-rutinoside, Protocatechnic acid, psoralen, catechol, vanillin, syringaldehyde, Syringol, P-vinyl glyaiacol, P-propyl phenol, Naringenin, Naringin, Isoquercitrin, Quercetin-3-galactoside, and vitexin, are some of the common phenolic compounds in the Ficus genus and kaempferol, Quercetin, luteolin, Rutin, apigenin, catechin, luteolin-7-o-glucoside, epicatechin, myricetin, naringenin, Hesperetin, daidzein, biochanin, genistein, and taxifolin are some of the common flavonoid compounds found in the Ficus genus [16].
Both F. auriculata and F. carica are known to contain various phytochemicals, including flavonoids, terpenoids, and phenolic acids. Some of the similar compounds found in both plants include quercetin, rutin, kaempferol, and beta-sitosterol.
Tables 1 and 2 show the comparison of phytocompounds and methods of their isolation in various studies of both species; however, it would be early to conclude that one of the species lacked specific phytocompounds because variations in the extraction procedure may have affected the results; therefore, a comparative analysis of the phytocompounds in both species is necessary to identify the missing phytocompounds in a specific plant.
1.4 Nutrient and mineral composition
The composition of nutrients and minerals found in fruits can differ depending on their geographical location. This can be observed at various scales, from variations between countries and continents to differences between regions within the same country. These differences may be a result of diverse environmental factors, including changes in climate, soil properties, and other ecological influences [36]. The results of various studies on nutrient and mineral composition are listed in Table 3.
According to Idrus et al. [38], the mineral composition of fig closely resembles that of human milk, with a particular emphasis on iron, which is reportedly present at 50% of the level found in beef liver. Furthermore, figs can be an important source of dietary calcium, and from Table 3, it can be concluded that there are significant variations in nutrient and mineral composition between F. auriculata and F. carica among different studies. Two studies on the same fruit (F. auriculata) or F. carica differed significantly in their reported values, which highlights the importance of conducting multiple studies for accurate nutritional analysis. Khatun et al. [36] suggested that F. auricualta could serve as a viable alternative to F. carica because it contains important nutrients such as protein, fat, Mg, K, and Vit. C and Fe. Therefore, including F. auriculata in one’s diet could provide nutritional benefits like those of F. carica while also offering unique advantages due to its specific nutrient profile.
1.5 Traditional and modern applications
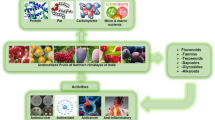
The medicinal properties of the Ficus genus are not only limited to F. auriculata, but a variety of plants in this genus (Fig. 2). The leaves of Ficus species are utilized in the treatment of numerous diseases, such as skin wounds and gastrointestinal problems, including dysentery and diarrhea. The bark and stems of F. arnottiana and F. hispida exhibit hypoglycemic activity, while stem juice is effective for treating wounds, cuts, and diarrhea. Additionally, the roots of F. begalensis have been found to possess anthelmintic activity, and latex is obtained from the roots used to treat mumps, cholera, diarrhea, and vomiting. Furthermore, a mixture of the root powder of F. auriculata and the bark of Oroxylum indicum can be used to treat jaundice [3, 6, 11]. The fruits are consumed as vegetables and are said to be beneficial for curing diabetes and improving blood pressure. The latex of the plant is used for treating wounds [39]. Fresh juice from the leaves of F. racemose L. is used to treat gastrointestinal problems. Roots of F. bengalensis have been reported to exhibit anthelmintic activity and to inhibit insulinase activity in the liver and kidney, while fruits exhibit antitumor properties [40]. Ficus auriculata fruits are traditionally utilized to treat mumps, jaundice, and cholera. In addition, they have been found to possess wound healing properties [41].
The role of F. carica has been described by various researchers for its antioxidant, anticancer, antidiabetic, hepatoprotective, and antibacterial activities [42]. While both F. carica and F. auricualta have their own unique benefits, the current review focuses more on modern applications of F. auriculata, as it is underutilized.
1.6 Modern applications of Ficus auricualta
1.6.1 Antibacterial activity
A study by Tamta et al. [43] compared the antibacterial activities of F. auriculata from two regions of Uttarakhand. The chloroform extract obtained from Almora species exhibited the highest zone of inhibition, while the methanol extract obtained from the fruit of Haldwani (Nainital) species exhibited the strongest inhibition against human pathogenic bacteria. Similarly, a study conducted by Shao et al. [25] showed that isoflavones from the fruits of F. auriculata exhibit antibacterial activity against P. euruginosa, B. cereus, S. albus, E. coli, and S. epidermidis, with MIC values of 10,555 and 1.25 µg/ml, respectively [35]. Alcoholic extracts obtained from the leaves and fruits of trees were found to have antimicrobial effects against Staphylococcus aureus, Bacillus subtilis, Escherichia coli, Bacillus aureus, and Pseudomonas aeruginosa. Similarly, the fruit extract clearly inhibited Klebsiella pneumoniae, Staphylococcus epidermidis, Proteus vulgaris, Mycoplasma putida, and E. coli in a concentration-dependent manner according to the disc diffusion method. Ata concentration of 60 µg, it has the highest activity against Staphylococcus epidermis and Mycoplasma genetalium [44]. Research has shown that F. auriculata can be a promising fruit with antibacterial properties, although further research is needed.
1.6.2 Antioxidant activity
Several studies have reported the antioxidant potential of F. auriculata. The methanol and chloroform extracts of F. auriculata stems exhibit the highest scavenging activities of 84.088% and 83.864%, respectively, at a concentration of 0.1 mg/ml, while the hexane extract shows a lower scavenging activity of 42% in the DPPH assay [3]. The DPPH EC50 activity of the leaves was 251.33 mg/ml, and that of the reducing power assay (RPA) was 54.40 [13]. Using the ultrasonic-assisted extraction technique, Shahinuzzaman et al. [45] determined that the optimum extraction conditions for F. auriculata (Fruits) to achieve the highest antioxidant activity (85.20 ± 0.96%) using the DPPH assay were 52.5% ethanol (v/v), 40 °C and an extraction time of 22 min. A study conducted by Akhtar et al. [46] showed the antioxidant properties of 12 Ficus sp. Ficus auricualta exhibited the highest (29.18 ± 0.16 mg TE/g DL) antioxidant activity in leaves among all species. Using an ultrasonic-assisted extraction process, the DPPH activity of the leaves, bark, and fruits of F. auriculata ranged from 87.30 ± 1.29% to 93.78 ± 0.83%, and the ABTS scavenging activity ranged from 96.47 ± 0.83% to 99.66 ± 0.55%. The inhibitory effects of DPPH on the bark, leaves, and fruit were 87.30 ± 1.29%, 93.78 ± 0.83% and 89 ± 0.83%, respectively [45, 46]. In conclusion, various studies have indicated that F. auriculata has significant potential as an antioxidant. These findings contribute to the growing body of research on the potential health benefits of F. auriculata and provide opportunities for future exploration of its antioxidant properties.
1.6.3 Antidiabetic activity
F. auriculata fruits have shown potential as a natural remedy for type 2 diabetes mellitus (T2DM). The inhibitory effects of these compounds on α-glucosidase may be attributed to their phenolic compounds [47]. In particular, the methanol extract of fruit was observed to exhibit the greatest dose-dependent inhibitory activity against α-amylase and α-glucosidase when capered to acarbose [48]. Furthermore, the bark extract of Ficus auricualta has shown potential in the treatment of postprandial hyperglycemia and T2DM through its observed α-amylase inhibition activity [49]. It is necessary to identify the specific phytoconstituents responsible for this activity and to confirm its efficacy in living organisms.
1.7 Hepatoprotective
Doley and Kakoti [50] reported the hepatoprotective effects of Ficus auricualta and Sarcohlamys pulcherrima, and their findings suggested that liver damage caused by carbon tetrachloride (CCl4, 1 ml/kg, 50% v/v with olive oil) was caused by increased levels of SGOT (serum glutamate oxaloacetate transaminase), ALP (alkaline phosphatase), bilirubin, and SGPT (serum glutamate pyruvate transaminase). However, pretreatment with both plant extracts at dosages of 200 and 400 mg/kg resulted in a reduction in these levels; interestingly, the results showed that a dose of 400 mg/kg of both plant extracts yielded results similar to those of silymarin, a well-known hepatoprotective drug with a dosage of 25 mg/kg.
1.8 Industrial aspects
F. auriculata can be used to make jam, curries, and juice [51]. However, the process of making these items has not been mentioned by researchers. With its diverse application, it is a versatile raw material for various industries. On the other hand, F. carica, a widely recognized species within the Ficus genus, has many industrial applications. Various parts of plants, including the peel, leaves, and whole fruit, possess valuable properties that can be harnessed in multiple ways. For instance, the lyophilized powder of the peel can serve as a natural colorant; similarly, the leaves can be powdered to prolong the shelf life of pasteurized milk and aid in the clotting of fresh goat cheese [8]. The whole fig can be used as a sugar replacement for ice cream and to produce pullulan gum. In particular, Pullulan gum was produced by fermenting fig syrup in the presence of Aureobasidium pullulans under aerobic conditions, and it was concluded that Fig syrup is better than sucrose in fermentation as a carbon source [52]. Dried figs, jam, juice, preserved fruits, and wine can be made from fruits of F. carica [53]. Bakery products, which are primarily made from cereals and have a prolonged shelf life, constitute a significant portion of the human diet. Khapre et al. made cookies by incorporating Dinkar variety fresh Fig fruit powder [54]. These fig-enriched cookies contained 12% fig powder and displayed satisfactory sensory attributes while providing good nutritional value. The cookies had a protein content of 6.9% dietary fiber content of 3.1%, and a potassium content of 1.1%. Furthermore, the cookies contained 53.1% reducing sugars and 22.9% non-reducing sugars. Despite being highly explored figs (F. carica) several of its parts like leaves remain underutilized. To fill in the gaps Shiraishi et al. [55] conducted a study to evaluate hydroethanolic extract of leaves from five fig varieties. The result of their study shows that some of the varieties have good antimicrobial activity suggesting their potential for industrial application such as food, nutraceutical, and cosmetics. Given that F. auriculata is a member of the same genus as F. carica, it is possible that this plant may also possess similar properties and be useful in these applications. Further studies are needed to explore the full potential and industrial applications of F. auriculata.
The quality of any fruit declines after the harvesting which can be due to various metabolic activities. Understanding the molecular and biochemical parameters responsible for the ripening of fruits and their related traits opens possibilities for genetic modifications through genetic engineering. Omics based technologies including transcriptomics, proteomics and metabolomics can provide a deeper understanding of these postharvest changes. These methods include tools such as RNAi, CRISPR or antisense RNA technologies to produce more advanced and enhanced versions of fruits. This can also be applied to increase the shelf life. Many proven concepts are available for models such as tomatoes and apples [56, 57]. It might open some new pathways for the commercialization of underutilized plants while helping to enhance their shelf life.
2 Conclusion and future prospects
Generally, fruits are high in vitamins, minerals, fiber, and antioxidants, which make them vital for healthy health. Vitamin C, potassium, and folate are among the vital elements found in fruits that promote heart health, the immune system, and cellular repair. Their high fiber content facilitates satiety, assists in digestion, and helps control blood sugar levels. Fruits include antioxidants that fight oxidative stress and lower the risk of chronic illnesses including diabetes, cancer, and heart disease. Fruit is an essential part of a balanced diet since they promote overall wellbeing, help with weight control, and can enhance skin health. Both F. auriculata and F. carica are rich sources for all these nutritional values. Although F. carica is commonly used in various industries, such as baking, dairy and beverage, F. auriculata is often overlooked due to a lack of knowledge and awareness of its potential. There are possibilities for considering this fruit as an alternative to F. carica and raising awareness about this fruit could lead to its increased consumption and utilization. Further research on the benefits of F. auriculata could help increase its utilization and provide additional health and economic benefits to communities that have access to this wild fruit. Additionally, some potential research gaps include the need for detailed comparative study on metabolic profiling of edible fruits belonging to Ficus genus various fruits of this genus have very low shelf life demanding for the proper research on the method for post harvesting processes, thereby increasing their market value and economic potential. Besides, literature dominated the potential applications of these fruits, however the market availability of products is still nowhere to be found as per consumer demand.
Data availability
No datasets were generated or analysed during the current study.
References
Li Y, Zhang JJ, Xu DP, Zhou T, Zhou Y, Li S, Li HB. Bioactivities and health benefits of wild fruits. Int J Mol Sci. 2016;17(8):1258. https://doi.org/10.3390/IJMS17081258.
Zhang Z, Wang XM, Liao S, Tian HZ, Li HQ. Taxonomic treatment of the Ficus auriculata complex (Moraceae) and typification of some related names. Phytotaxa. 2019;399(3):203–8. https://doi.org/10.11646/phytotaxa.399.3.4.
Mehra N, Tandon S. Traditional uses, phytochemical and pharmacological properties of Ficus auriculata: a review. J Drug Deliv Ther. 2021;11(3):163–9. https://doi.org/10.22270/jddt.v11i3.4853.
Bhutia R, Kumar Rai Y, Kumar Rai BLG. Wild edible plant used by ethnic peoples on the boundary of subtropical temperate natural forest, Mamlay watershed, South Sikkim. Indian J Nat Prod Resour. 2020. https://doi.org/10.56042/ijnpr.v11i4.32496.
Baluni P, Kuriyal SK, Dobriyal K. Folk culture of garhwal himalaya: ethnic food and its medicinal value—a brief analysis. J Mt Res. 2021;16:251–63. https://doi.org/10.51220/jmr.v16i3.26.
Khuraijam JS, Huidrom E. Medicinal and nutritive value of Ficus auriculata-the wonder fig. Indian J Tropical Biodivers. 2014;22(1):70–3.
Hussain SZ, Naseer B, Qadri T, Fatima T, Bhat TA. Fig (Ficus carica)—Morphology, taxonomy, composition and health benefits. In: Hussain SZ, Naseer B, Qadri T, Fatima T, Bhat TA, editors. Fruits grown in highland regions of the Himalayas. Berlin: Springer; 2021. p. 77–90. https://doi.org/10.1007/978-3-030-75502-7_6.
Teruel-Andreu C, Andreu-Coll L, López-Lluch D, Sendra E, Hernández F, Cano-Lamadrid M. Ficus carica fruits, by-products and based products as potential sources of bioactive compounds: a review. Agronomy. 2021;11(9):1834. https://doi.org/10.3390/AGRONOMY11091834.
Rasool IFU, Aziz A, Khalid W, Koraqi H, Siddiqui SA, Al-Farga A, Ali A. Industrial application and health prospective of fig (Ficus carica) by-products. Molecules. 2023;28(3):960. https://doi.org/10.3390/molecules28030960.
Gaire BP, Lamichhane R, Sunar CB, Shilpakar A, Neupane S, Panta S. Phytochemical screening and analysis of antibacterial and antioxidant activity of Ficus auriculata (Lour.) stem bark. Pharmacogn J. 2011;3(21):49–55. https://doi.org/10.5530/pj.2011.21.8.
George M, Joseph L, Pau NM. Ficus auriculata: a pharmacological update. Int J Curr Res Acad Rev. 2016;4(7):26–31. https://doi.org/10.20546/ijcrar.2016.407.003.
Nuri ZN, Uddin MS. A review on nutritional values and pharmacological importance of Ficus carica. J Curr Res Food Sci. 2021;2(1):07–11.
Flowers of India. https://www.flowersofindia.net/catalog/slides/Elephant%20Ear%20Fig.html. Accessed 27 Aug 2024.
Thingbaijam R, Dutta BK, Paul SB. In vitro antioxidant capacity, estimation of total phenolic and flavonoid content of Ficus auriculata lour. Int J Pharm Pharm Sci. 2012;4(4):518–21.
Saklani S, Chandra S. In vitro antimicrobial activity, nutritional profile and phytochemical screening of wild edible fruit of Garhwal Himalaya (Ficus auriculata). Int J Pharm Sci Rev Res. 2012;12(2):61–4.
Walia A, Kumar N, Singh R, Kumar H, Kumar V, Kaushik R, Kumar AP. Bioactive compounds in Ficus fruits, their bioactivities, and associated health benefits: a review. J Food Qual. 2022;2022:1–19. https://doi.org/10.1155/2022/6597092.
Anjum N, Tripathi YC. Assessment of polyphenolics in Ficus auriculata and F. palmata fruits and their antioxidants activity. University. 2020
Shah AF. Pharmacological evaluation of Murraya koenigii and Ficus carica in management of diabetic complications diabetic nephropathy and diabetic neuropathy. 2018.
El-Fishawy A, Zayed R, Afifi S. Phytochemical and pharmacological studies of Ficus auriculata Lour. J Nat Prod. 2011;4:184–95.
Shao TM. Chemical constituents from stems of Ficus auriculata. Chin Tradit Herb Drugs. 2013. https://doi.org/10.7501/J.ISSN.0253-2670.2013.16.003.
Shao TM, Zheng CJ, Han CR, Chen GY, Dai CY, Song XP, Chen WH. Lactones from Ficus auriculata and their effects on the proliferation function of primary mouse osteoblasts in vitro. Bioorg Med Chem Lett. 2014;24(16):3952–5. https://doi.org/10.1016/J.BMCL.2014.06.035.
Olaoluwa O, Taiwo O, Nahar L, Sarker SD. Ethnopharmacology, phytochemistry and biological activities of the African species of the genus Ficus L. Trends Phytochem Res. 2022;6(1):46–69. https://doi.org/10.30495/tpr.2022.1939285.1219.
Dueñas M, Pérez-Alonso JJ, Santos-Buelga C, Escribano-Bailón T. Anthocyanin composition in fig (Ficus carica L.). J Food Compos Anal. 2008;21(2):107–15. https://doi.org/10.1016/j.jfca.2007.09.002.
El-Shobaki FA, El-Bahay AM, Esmail RSA, El-Megeid AAA, Esmail NS. Effect of figs fruit (Ficus carica L.) and its leaves on hyperglycemia in alloxan diabetic rats. World J Dairy Food Sci. 2010;5(1):47–57.
Oliveira AP, Valentão P, Pereira JA, Silva BM, Tavares F, Andrade PB. Ficus carica L.: metabolic and biological screening. Food Chem Toxicol. 2009;47(11):2841–6. https://doi.org/10.1016/j.fct.2009.09.004.
Oliveira AP, Silva LR, Andrade PB, Valentao P, Silva BM, Goncalves RF, Guedes de Pinho P. Further insight into the latex metabolite profile of Ficus carica. J Agric Food Chem. 2010;58(20):10855–63. https://doi.org/10.1021/jf1031185.
Rubnov S, Kashman Y, Rabinowitz R, Schlesinger M, Mechoulam R. Suppressors of cancer cell proliferation from fig (Ficus carica) resin: isolation and structure elucidation. J Nat Prod. 2001;64(7):993–6. https://doi.org/10.1021/np000592z.
Solomon A, Golubowicz S, Yablowicz Z, Grossman S, Bergman M, Gottlieb HE, Flaishman MA. Antioxidant activities and anthocyanin content of fresh fruits of common fig (Ficus carica L.). J Agric Food Chem. 2006;54(20):7717–23. https://doi.org/10.1021/jf060497h.
Vallejo F, Marín JG, Tomás-Barberán FA. Phenolic compound content of fresh and dried figs (Ficus carica L.). Food Chem. 2012;130(3):485–92. https://doi.org/10.1016/j.foodchem.2011.07.032.
Vaya J, Mahmood S. Flavonoid content in leaf extracts of the fig (Ficus carica L.), carob (Ceratonia siliqua L.) and pistachio (Pistacia lentiscus L.). BioFactors. 2006;28(3–4):169–75. https://doi.org/10.1002/biof.5520280303.
Yemiş O, Bakkalbaşı E, Artık N. Changes in pigment profile and surface colour of fig (Ficus carica L.) during drying. Int J Food Sci Technol. 2012;47(8):1710–9. https://doi.org/10.1111/j.1365-2621.2012.03025.x.
El-Kholy IE-S, Shaban MAM. Constituents of the leaves of Ficus carica, L. Part II. Isolation of a ψ-taraxasteryl ester, rutin, and a new steroid sapogenin. J Chem Soc C. 1966. https://doi.org/10.1039/j39660001140.
Oliveira AP, Silva LR, de Pinho PG, Gil-Izquierdo A, Valentão P, Silva BM, Andrade PB. Volatile profiling of Ficus carica varieties by HS-SPME and GC–IT-MS. Food Chem. 2010;123(2):548–57. https://doi.org/10.1016/j.foodchem.2010.04.064.
Oliveira AP, Silva LR, Ferreres F, Guedes de Pinho P, Valentao P, Silva BM, Andrade PB. Chemical assessment and in vitro antioxidant capacity of Ficus carica latex. J Agric Food Chem. 2010;58(6):3393–8. https://doi.org/10.1021/jf9039759.
Shao TM, Liao HX, Li XB, Chen GY, Song XP, Han CR. A new isoflavone from the fruits of Ficus auriculata and its antibacterial activity. Nat Prod Res. 2022;36(5):1191–6. https://doi.org/10.1080/14786419.2020.1864368.
Khatun MJM, Rahman MM, Rahim MA, Jakariya M, Mirdah MH. Study on the ethnobotany and nutritional status of three edible Ficus species in hill district of Bangladesh. Int J Minor Fruits Med Aromat Plants. 2016;2(1):35–40.
U.S. Department of Agriculture, Agricultural Research Service, Beltsville, Human Nutrition Research Center. FoodData Central. https://fdc.nal.usda.gov/fdc-app.html#/food-details/173021/nutrients. Retrieved Jun 11 2023.
Idrus RBH, Sainik NQAV, Ansari AS, Zulfarina MS, Razali RA, Nordin A, Saim A. Bin, Naina-Mohamed I. Ficus carica and bone health: a systematic review. Sains Malays. 2018;47:2741–55. https://doi.org/10.17576/jsm-2018-4711-17.
Pant S, Samant SS, Arya SC. Diversity and indigenous household remedies of the inhabitants surrounding Mornaula reserve forest in West Himalaya. Indian J Tradit Knowl. 2009;8(4):606–10.
Sirisha N, Sreenivasulu M, Sangeeta K, Chetty CM. Antioxidant properties of Ficus species-a review. Int J PharmTech Res. 2010;2:2174–82.
Jagadeesan G, Muniyandi K, Lydia MA, Nataraj G, Thamburaj S, Sathyanarayanan S, Thangaraj P. Analysis of discrete and combined effect of solvent, extraction time, and extraction temperature on polyphenol compounds extraction from roxburgh fig (Ficus auriculata Lour.) fruit using response surface methodology. Phytomedicine. 2020. https://doi.org/10.1201/9781003014898-5.
Li Z, Yang Y, Liu M, Zhang C, Shao J, Hou X, Tian J, Cui Q. A comprehensive review on phytochemistry, bioactivities, toxicity studies, and clinical studies on Ficus carica Linn. leaves. Biomed Pharmacother. 2021;137:111393. https://doi.org/10.1016/j.biopha.2021.111393.
Tamta G, Mehra N, Tandon S, Nand V, Biswas S. In vitro antimicrobial assay of leaves, bark and fruits of Ficus auriculata collected from two different regions of Uttarakhand. Int J Chem Stud. 2021;9(1):930–5. https://doi.org/10.22271/chemi.2021.v9.i1m.11347.
Raja R, Kumar N, Duraisamy M. Evaluation of phytochemical screening and antibacterial activity of Ficus auriculata lour. (moraceae)—an traditional medicinal plant. Kongunadu Res J. 2021;8(1):22–9. https://doi.org/10.26524/krj.2021.4.
Shahinuzzaman M, Akhtar P, Amin N, Ahmed Y, Anuar F, Misran H, Akhtaruzzaman M. Extraction process optimization, identification, and profiling of phenolic antioxidant compounds from the fruits of Ficus auriculata. Res Sq. 2020. https://doi.org/10.21203/rs.3.rs-113810/v1.
Akhtar P, Yaakob Z, Ahmed Y, Shahinuzzaman M. Ficus species good sources of natural antioxidant drugs. Turk J Pharm Sci. 2019;2.
Mohamed Yunus SN, Abas F, Jaafar AH, Azizan A, Zolkeflee NKZ, Abd Ghafar SZ. Antioxidant and α-glucosidase inhibitory activities of eight neglected fruit extracts and UHPLC-MS/MS profile of the active extracts. Food Sci Biotechnol. 2021;30:195–208. https://doi.org/10.1007/s10068-020-00856-x.
Anjum N, Tripathi YC. In vitro alpha-amylase and alpha-glucosidase inhibitory activities of fruits of Ficus auriculata Lour. Int J Pharma Bio Sci. 2019;10(4):134–41. https://doi.org/10.22376/ijpbs.2019.10.4.p134-141.
Tiwari Y, Singh A, Kumar B, Kumar A. In vitro evaluation of alpha amylase activity of bark extracts of Ficus auriculata. Int J Innov Sci Res Technol. 2017;12(2):2456.
Doley BK, Kakoti BB. Hepatoprotective potential of Ficus auriculata and Sarcochlamys pulcherrima, two ethnomedicinal plants used by the mishing community of Assam. World J Pharm Pharm Sci. 2018;15(7):913–22. https://doi.org/10.20959/wjpr201815-13033.
Paramanandam V, Jagadeesan G, Muniyandi K, Manoharan AL, Nataraj G, Sathyanarayanan S, Thangaraj P. Comparative and variability analysis of different drying methods on phytochemical, antioxidant and phenolic contents of Ficus auriculata Lour. Fruit Phytomed Plus. 2021;1(3):100075. https://doi.org/10.1016/j.phyplu.2021.100075.
Marvdashti LM, Abdolshahi A, Hedayati S, Sharifi-Rad M, Iriti M, Salehi B, Sharifi-Rad J. Pullulan gum production from low-quality fig syrup using Aureobasidium pullulans. Cell Mol Biol. 2018;64(8):22–6. https://doi.org/10.14715/cmb/2018.64.8.4.
Weibin J, Kai M, Zhifeng L, Yelin W, Lianju W. The production and research of fig (Ficus carica L.) in China. In: II International Symposium on Fig 605. 2001. p. 191–6). https://doi.org/10.17660/ActaHortic.2003.605.28.
Khapre AP, Satwadhar PN, Deshpande HW. Studies on standardization of fig fruit (Ficus carica L.) powder enriched cookies and its composition. Asian J Dairy Food Res. 2015;34(1):71. https://doi.org/10.5958/0976-0563.2015.00015.9.
Shiraishi CSH, Zbiss Y, Roriz CL, Dias MI, Prieto MA, Calhelha RC, Alves MJ, Heleno SA, da Cunha Mendes V, Carocho M, Abreu RMV, Barros L. Fig leaves (Ficus carica L.): source of bioactive ingredients for industrial valorization. Processes. 2023;11(4):1179. https://doi.org/10.3390/pr11041179.
Mathiazhagan M, Chidambara B, Hunashikatti LR, Ravishankar KV. Genomic approaches for improvement of tropical fruits: fruit quality, shelf life and nutrient content. Genes. 2021;12(12):1881. https://doi.org/10.3390/genes12121881.
Belay ZA, James Caleb O. Role of integrated omics in unravelling fruit stress and defence responses during postharvest: a review. Food Chem Mol Sci. 2022;5:100118. https://doi.org/10.1016/j.fochms.2022.100118.
Acknowledgements
Author BN is thankful to the Uttarakhand Council for Science and Technology for providing fellowship to SB.
Funding
None.
Author information
Authors and Affiliations
Contributions
Vijay Kumar and Bindu Naik: conceptualization, analyzed the data, written the original draft, reviewed the manuscript, Saurav Chnadra Bhatt, Arun K.Gupta, Per E. J.Saris, Sarevesh Rustagi, VivekK and Vishal Rajput: analyzed the data, writing the original draft. All the authors read the manuscript and approved its final form.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bhatt, S.C., Kumar, V., Naik, B. et al. Ficus auriculata Lour., an underutilized nonconventional alternative fruit to Ficus carica with nutraceutical potential. Discov Sustain 5, 254 (2024). https://doi.org/10.1007/s43621-024-00480-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s43621-024-00480-3