Abstract
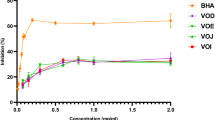
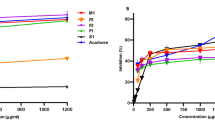
The 70% ethanolic extracts from eight neglected fruits; Muntingia calabura, Leucaena leucocephala, Spondias dulcis, Syzygium jambos, Mangifera caesia, Ardisia elliptica, Cynometra cauliflora and Ficus auriculata were evaluated for their 2,2-diphenyl-1-picrylhydrazyl (DPPH) free radical scavenging, α-glucosidase inhibitory activities as well as total phenolic content. The results of this study revealed that M. caesia fruit extract demonstrated the most potent radical scavenging activity. Among the fruits examined for α-glucosidase inhibitory activity, M. calabura and F. auriculata exhibited strong activity with no significant difference. The Pearson correlation indicated that the activities of M. caesia and F. auriculata contributed by phenolic compounds. A total of 65 metabolites were tentatively identified by using ultra-high-performance liquid chromatography tandem mass spectrometry (UHLPC-MS/MS). These findings suggested that the possible application of M. caesia and F. auriculata as a functional food with antioxidant and α-glucosidase inhibitory properties.


Similar content being viewed by others
References
Abd Ghafar SZ, Mediani A, Maulidiani, Ramli AS, Abas F. Antioxidant, α-glucosidase and nitric oxide inhibitory activities of Phyllanthus acidus and LC-MS/MS profile of the active extract. Food Biosci. 25: 134-140 (2018)
Abu-Reidah IM, Ali-Shtayeh MS, Jamous RM, Arraez-Roman D, Swgura-Carretero A. HPLC-DAD-ESI-MS/MS screening of bioactive components from Rhus coriaria L. (Suman) fruits. Food Chem. 166: 179-191 (2015)
Ahmad A, Misra LN. Isolation of herniarin and other constituents from Matricaria chamomilla flowers. Int. J. Pharmacog. Phytochem. 35: 121-125 (1997)
Akhtar N, Jafri L, Green BD, Kalsoom S, Mirza B. A multi-mode bioactive agent isolated from Ficus microcarpa L. Fill. with therapeutic potential for type 2 diabetes mellitus. Front. Pharmacol. 9: 1376 (2018)
Anjum N. Tripathi YC. In vitro alpha-amylase and alpha-glucosidase inhibitory activities of fruits of Ficus auriculata Lour. Int. J. Pharm. Bio. Sci. 10: 134-141 (2019)
Nor-Azman NII, Hashim N, Ahmad R. In-vitro α-Glucosidase inhibitory activities of Muntingia calabura Linn. Int. J. Eng. Tech. 7: 183-185 (2018)
Barros A, Girones-Vilaplana A, Teixeira A, Collado-Gonzalez J, Moreno DA, Gil-Izqueierdo A, Rosa E, Dominguez-Perles R. Evaluation of grape (Vitis vinifera L.) stems from Portuguese varieties as a resource of (poly)phenolic compounds: A comparative study. Food Res. Int. 65: 375-384 (2014)
Berardini N, Schieber A, Klaiber I, Beifuss U, Carle R, Conrad J. 7-O-methylcyanidin 3-O-β-D-galactopyranoside, a novel anthocyanin from Mango (Mangifera indica L. cv. ‘Tommy Atkins’) peels. Z. Naturforsch. 60: 801-804 (2005)
Chew YL, Chan WWL, Tan PL, Stanslas J, Goh JK. Assessment of phytochemical content, polyphenolic composition, antioxidant and antibacterial activities of Leguminosae medical plants in Peninsular Malaysia. BMC Complement. Altern. Med. 11: 2-10 (2011)
Dias T, Bronze MR, Houghton PJ, Mota-Filipe H, Paulo A. The flavonoid-rich fraction of Coreopsis tinctoria promotes glucose tolerance regain through pancreatic function recovery in streptozotocin-induced glucose-intolerant rats. J. Ethnopharmacol. 132: 483-490 (2010)
Dou J, Lee VSY, Tzen JTC, Lee M-C. Identification and comparison of phenolic compounds in the preparation of Oolong tea manufactured by semifermentation and drying processes. J. Agric. Food Chem. 55: 7462-7468 (2007)
El-Fishawy A, Zayed R, Afifi S. Phytochemical and pharmacological studies of Ficus auriculata Lour. J. Nat. Prod. 4: 184-195 (2011)
Ertas A, Yilmaz MA, Firat M. Chemical profile by LC-MS/MS, GC-MS and antioxidant activities of the essential oils and crude extracts of two Euphorbia species. Nat. Prod. Res. 29: 529-534 (2014)
Fathalla N, Bishr M, Singab AN, Salama O. GC-MS and LC-MS identification of the phenolic compounds present in the ethyl acetate fraction obtained from Senna tora, L. Roxb. seeds. Nat. Prod. Res. 16: 1-4 (2018)
Gomathi R, Anusuya N, Manian S. A dietary antioxidant supplementation of Jamaican cherries (Muntingia calabura L.). attenuates inflammatory related disorders. Food Sci. Biotechnol. 22: 787–794 (2013)
Hainida E, Ikram K, Hock K, Maleyki A, Jalil M. Antioxidant capacity and total phenolic content of Malaysian underutilized fruits. J. Food Compost. Anal. 22: 388–393 (2009)
Hofmann T, Nebehaj E, Albert L. Antioxidant properties and detailed polyphenol profiling of European hornbeam (Carpinus betulus L.) leaves by multiple antioxidant capacity assays and high-performance liquid chromatography/multistage electrospray mass spectrometry. Ind. Crops Prod. 87: 340-349 (2016)
Jin MJ, Kim IS, Rehman SU, Dong M-S, Na C-S, Yoo HH. A liquid chromatograph-tandem mass spectrometry method for simultaneous quantitation of 10 bioactive components in Rhus verniciflua extracts. J. Chromatogr. Sci. 4: 390-396 (2015)
Kajdzanoska M, Gjamovski V, Stefova M. HPLC-DAD-ESI-MSn identification of phenolic compounds in cultivated strawberries from Macedonia. Maced. J. Chem. Chem. En. 29: 181-194 (2010)
Kumar S, Chandra P, Bajpai V, Singh A. Rapid qualitative and quantitative analysis of bioactive compounds from Phyllanthus amarus using LC/MS/MS techniques. Ind. Crops Prod. 69: 143-152 (2015)
Lawal U, Leong SW, Shaari K, Ismail IS, Khatib A, Abas F. α-glucosidase inhibitory and antioxidant activities of different Ipomoea aquatica cultivars and LCMS/MS profiling of the active cultivar. J. Food Biochem. 41(2): 1-8 (2017)
Li Y, Guo H, Wu Y, Geng Q, Dong D, Wu H, Li E. A sensitive and selective method for determination of Aesculin in Cortex fraxini by liquid chromatography quadrupole time-off light tandem mass spectrometry and application in pharmacokinetic study. J. Anal. Methods Chem. 2013: 1-6 (2013)
Lin L-Z, He X-G, Lindenmaier M, Yang J, Cleary M, Qiu S-X, Cordell GA. LC-ESI-MS study of the flavonoid glycoside malonates of red clover (Trifolium pratense). J. Agric. Food Chem. 48: 354-365 (2000)
Maity S, Chatterjee S, Variyar PS, Sharma A, Adhikari S, Mazumder S. Evaluation of antioxidant activity and characterization of phenolic constituents of Phyllantus amarus root. J. Agric. Food Chem. 61: 3443-3450 (2013)
McCune LM, Kubota C, Stendell-Hollis NR, Thomson CA. Cherries and Health: A review. Crit. Rev. Food Sci. Nutr. 51: 1-12 (2011)
Mediani A, Abas F, Tan CP, Khatib A. Effects of different drying methods and storage time on free radical scavenging activity and total phenolic content of Cosmos caudatus. Antioxidants 3: 358–370 (2014)
Mirfat AHS, Salma I, Razali M. Natural antioxidant properties of selected wild Mangifera species in Malaysia. J. Trop. Agric. Food Sci. 44: 63-72 (2016)
Muniyandi K, George E, Sathyanarayanan S, George BP, Abrahamse H, Thamburaj S, Thangaraj P. Phenolics, tannins, flavonoids and anthocyanins contents influenced antioxidant and anticancer activities of Rubus fruits from Western Ghats, India. Food Sci Hum Well. 8: 73-81 (2019)
Plazonic A, Bucar F, Males Z, Mornar A, Nigovic B, Kujundzic N. Identification and quantification of flavonoids and phenolic acids in Burr Parsley (Caucalis platycarpos L.), using high-performance liquid chromatography with diode array detection and electrospray ionization mass spectrometry. Molecules 14: 2466-2490 (2009)
Puangpradab R, Suksathan R, Saratan N, Puangsombat P. Antioxidant properties and nutritive values of native figs consumed in northern Thailand. Acta Hortic. 1210: 281-288 (2018)
Putri ND, Nursyamsi KS, Prayogo YH, Sari DR-L, Budiarti E, Batubara I. Exploration of Mango fruits (Mangifera indica) as α-glucosidase inhibitors. Biosaintifika 9: 554-559 (2017)
Saifullah M, Mc-Cullum R, McCluskey A, Vuong Q. Comparison of conventional extraction technique with ultrasound assisted extraction on recovery of phenolic compounds from lemon scented tea tree (Leptospermum petersonii) leaves. Heliyon. 6(4): 1-12 (2020)
Saikia S, Mahnot NK, Lata C. Phytochemical content and antioxidant activities of thirteen fruits of Assam, India. Food Biosci. 13: 15–20 (2016)
Sanchez-Rabaneda F, Jauregui O, Casals I, Andres-Lacueva C, Izqueierdo-Pulido M, Lamuela-Raventos M. Liquid chromatographic/electrospray ionization tandem mass spectrometric study of the phenolic composition of cocoa (Theobroma cacao). J. Mass Spectrom. 38: 35-42 (2003)
Santhirasegaram V, Razali Z, George DS, Somasundram C. Effects of thermal and non-thermal processing on phenolic compounds, antioxidant activity and sensory attributes of Chokonan Mango (Mangifera indica L.) juice. Food Bioproc. Tech. 8: 2256-2267 (2015)
World Health Organization. Diabetes Country Profiles-Malaysia. (2018)
Yao LH, Jiang YM, Shi J, Tomas-Barberan FA, Datta N, Singanusong R, Chen SS. Flavonoids in food and their health benefits. Plant Foods Hum. Nutr. 59: 113-122 (2004)
Ye M, Yang W-Z, Liu K-D, Qiao X, Li B-J, Cheng J, Feng J, Guo D-A, Zhao Y-Y. Characterization of flavonoids in Millettia nitida var. hirsutissima by HPLC/DAD/ESI-MS. J. Pharm. Anal. 2: 35-42 (2012)
Zhou S, Shao Y, Fu J, Xiang L, Zheng Y, Li W. Characterization and quantification of taxifolin related flavonoids in Larix olgensis Henry Var. Koreana Nakai extract analysis and its antioxidant activity assay. Int. J. Pharmacol. 14: 534-545 (2018)
Acknowledgements
This research was supported by grant from Universiti Putra Malaysia (UPM/700/2/1/GPB/2017/9597400) under Putra High Impact Grant Scheme.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mohamed Yunus, S.N., Abas, F., Jaafar, A.H. et al. Antioxidant and α-glucosidase inhibitory activities of eight neglected fruit extracts and UHPLC-MS/MS profile of the active extracts. Food Sci Biotechnol 30, 195–208 (2021). https://doi.org/10.1007/s10068-020-00856-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-020-00856-x