Abstract
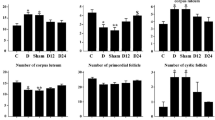
Diabetes mellitus is a common chronic metabolic disorder. This study aimed to investigate the effects of co-treatment with l-carnitine (LC) and zinc oxide nanoparticles (ZnONPs) on serum levels of sex hormones, oxidative stress, and ovarian histopathology in streptozotocin (STZ)-induced diabetic rats. Female Wistar rats (n = 56, 180–220 g) received a single intraperitoneal (IP) injection of STZ (65 mg/kg). They were randomly assigned into the following groups: diabetic group (Dia), Dia+Met group (100 mg metformin/kg/day), Dia+LC group (200 mg/kg/day), Dia+ZnONPs group (10 mg/kg/day), and Dia+LC+ZnONPs group (200 mg LC/kg/day and 10 mg ZnONPs/kg/day). Control group (Ctl) received the same volume of STZ solvent. After 21 days of treatment, blood serum was centrifuged for sex hormone assays. The right ovary was used for biochemical analysis, and the left ovary was fixed in 10% neutral buffered formalin for histological assessment. The levels of estradiol, progesterone, FSH, and LH significantly increased in the Dia+ZnONPs+LC group (P < 0.001) compared with the Dia group. Co-treatment with LC and ZnONPs reduced malondialdehyde and carbonyl protein and increased glutathione, catalase, and superoxide dismutase activities in ovarian tissue compared with the Dia group (P < 0.05). Moreover, the number of all ovarian follicles significantly increased in this group compared with the Dia group (P < 0.05). The results of this study indicated that co-treatment with LC and ZnONPs could preserve ovarian function by increasing sex hormones levels and antioxidant activity and decreasing lipid peroxidation in diabetic rats. Therefore, this compound supplementation may improve ovulation and fertility in people with diabetes mellitus.


Similar content being viewed by others
References
Shah A, Afzal M. Prevalence of diabetes and hypertension and association with various risk factors among different Muslim populations of Manipur, India. J Diabetes Metab Disord. 2013;12(1):52.
Oueslati N, Charradi K, Bedhiafi T, Limam F, Aouani E. Protective effect of grape seed and skin extract against diabetes-induced oxidative stress and renal dysfunction in virgin and pregnant rat. Biomed Pharmacother. 2016;83:584–92.
Rashid K, Sil PC. Curcumin ameliorates testicular damage in diabetic rats by suppressing cellular stress-mediated mitochondria and endoplasmic reticulum-dependent apoptotic death. Biochim Biophys Acta. 2015;1852(1):70–82.
Tok EC, Ertunc D, Evruke C, Dilek S. The androgenic profile of women with non-insulin-dependent diabetes mellitus. J Reprod Med. 2004;49(9):746–52.
Yeshaya A, Orvieto R, Dicker D, Karp M, Ben-Rafael Z. Menstrual characteristics of women suffering from insulin-dependent diabetes mellitus. Int J Fertil Menopausal Stud. 1995;40(5):269–73.
Dorman JS, Steenkiste AR, Foley TP, Strotmeyer ES, Burke JP, Kuller LH, et al. Menopause in type 1 diabetic women: is it premature? Diabetes. 2001;50(8):1857–62.
Garris DR. Effects of progressive hyperglycemia on ovarian structure and function in the spontaneously diabetic Chinese hamster. Anat Rec. 1984;210(3):485–9.
Cox N, Meurer K, Carlton C, Tubbs R, Mannis D. Effect of diabetes mellitus during the luteal phase of the oestrous cycle on preovulatory follicular function, ovulation and gonadotrophins in gilts. J Reprod Fertil. 1994;101(1):77–86.
Cheng P-P, Xia J-J, Wang H-L, Chen J-B, Wang F-Y, Zhang Y, et al. Islet transplantation reverses the effects of maternal diabetes on mouse oocytes. Reproduction. 2011;141(4):417–24.
Sanchez-Rangel E, Inzucchi SE. Metformin: clinical use in type 2 diabetes. Diabetologia. 2017;60(9):1586–93.
Di Pietro M, Parborell F, Irusta G, Pascuali N, Bas D, Bianchi MS, et al. Metformin regulates ovarian angiogenesis and follicular development in a female polycystic ovary syndrome rat model. Endocrinology. 2015;156(4):1453–63.
Duan X, Li Y. Physicochemical characteristics of nanoparticles affect circulation, biodistribution, cellular internalization, and trafficking. Small. 2013;9(9–10):1521–32.
Siddiqui SA, Or Rashid MM, Uddin MG, Robel FN, Hossain MS, Haque MA, et al. Biological efficacy of zinc oxide nanoparticles against diabetes: a preliminary study conducted in mice. Biosci Rep. 2020;40(4):BSR20193972.
Akpolat M, Gulle K, Topcu-Tarladacalisir Y, Safi Oz Z, Bakkal BH, Arasli M, et al. Protection by L-carnitine against radiation-induced ileal mucosal injury in the rat: pattern of oxidative stress, apoptosis and cytokines. Int J Radiat Biol. 2013;89(9):732–40.
Turker Y, Naziroglu M, Gumral N, Celik O, Saygin M, Comlekci S, et al. Selenium and L-carnitine reduce oxidative stress in the heart of rat induced by 2.45-GHz radiation from wireless devices. Biol Trace Elem Res. 2011;143(3):1640–50.
Celik F, Kose M, Yilmazer M, Koken GN, Arioz DT, Kanat PM. Plasma L-carnitine levels of obese and non-obese polycystic ovary syndrome patients. J Obstet Gynaecol. 2017;37(4):476–9.
Mendez-Cuesta LA, Marquez-Valadez B, Perez-De la Cruz V, Maldonado PD, Santana RA, Escobar-Briones C, et al. Early changes in oxidative stress markers in a rat model of acute stress: effect of l-carnitine on the striatum. Basic Clin Pharmacol Toxicol. 2011;109(2):123–9.
Bene J, Hadzsiev K, Melegh B. Role of carnitine and its derivatives in the development and management of type 2 diabetes. Nutr Diabetes. 2018;8(1):8.
Marcondes F, Bianchi F, Tanno A. Determination of the estrous cycle phases of rats: some helpful considerations. Braz J Biol. 2002;62(4A):609–14.
Mohammadi M, Zare Z. Effects of treadmill exercise on cognitive functions and anxiety-related behaviors in ovariectomized diabetic rats. Physiol Behav. 2020;224:113021.
Rezaei N, Mardanshahi T, Shafaroudi MM, Abedian S, Mohammadi H, Zare Z. Effects of L-carnitine on the follicle-stimulating hormone, luteinizing hormone, testosterone, and testicular tissue oxidative stress levels in streptozotocin-induced diabetic rats. J Evid Based Integr Med. 2018;23:2515690x18796053.
Beutler E, Duron O, Kelly B. Improvement method for the determination of glutathione in tissue homogenate. J Lab Clin Med. 1963;61:882–8.
Kei S. Serum lipid peroxide in cerebrovascular disorders determined by a new colorimetric method. Clin Chim Acta. 1978;90(1):37–43.
Levine RL, Garland D, Oliver CN, Amici A, Climent I, Lenz AG, et al. Determination of carbonyl content in oxidatively modified proteins. Methods Enzymol. 1990;186:464–78.
Khedr NF. Fish oil and wheat-germ oil supplementation restores ovarian function in streptozotocin-diabetic rats. Reprod Fertil Dev. 2017;29(9):1689–98.
Ranasinghe P, Pigera S, Galappatthy P, Katulanda P, Constantine GR. Zinc and diabetes mellitus: understanding molecular mechanisms and clinical implications. Daru. 2015;23:44.
Rani AJ, Mythili SV. Study on total antioxidant status in relation to oxidative stress in type 2 diabetes mellitus. J Clin Diagn Res. 2014;8(3):108–10.
Garris DR, Garris BL. Cytolipotoxicity-induced involution of the female reproductive tract following expression of obese (ob/ob) and diabetes (db/db) genotype mutations: progressive, hyperlipidemic transformation into adipocytic tissues. Reprod Toxicol. 2004;18(1):81–91.
Wu J, Yan LJ. Streptozotocin-induced type 1 diabetes in rodents as a model for studying mitochondrial mechanisms of diabetic β cell glucotoxicity. Diabetes Metab Syndr Obes. 2015;8:181–8.
Vella S, Buetow L, Royle P, Livingstone S, Colhoun HM, Petrie JR. The use of metformin in type 1 diabetes: a systematic review of efficacy. Diabetologia. 2010;53(5):809–20.
Livingstone R, Boyle JG, Petrie JR. A new perspective on metformin therapy in type 1 diabetes. Diabetologia. 2017;60(9):1594–600.
Faure P, Benhamou P, Perard A, Halimi S, Roussel A. Lipid peroxidation in insulin-dependent diabetic patients with early retina degenerative lesions: effects of an oral zinc supplementation. Eur J Clin Nutr. 1995;49(4):282–8.
Anderson RA, Roussel A-M, Zouari N, Mahjoub S, Matheau J-M, Kerkeni A. Potential antioxidant effects of zinc and chromium supplementation in people with type 2 diabetes mellitus. J Am Coll Nutr. 2001;20(3):212–8.
Faure P, Corticelli P, Richard MJ, Arnaud J, Coudray C, Halimi S, et al. Lipid peroxidation and trace element status in diabetic ketotic patients: influence of insulin therapy. Clin Chem. 1993;39(5):789–93.
Afifi M, Almaghrabi OA, Kadasa NM. Ameliorative effect of zinc oxide nanoparticles on antioxidants and sperm characteristics in streptozotocin-induced diabetic rat testes. Biomed Res Int. 2015;2015:153573.
Wang Q, Ju X, Chen Y, Dong X, Luo S, Liu H, et al. Effects of L-carnitine against H2O2-induced oxidative stress in grass carp ovary cells (Ctenopharyngodon idellus). Fish Physiol Biochem. 2016;42(3):845–57.
Samir SM, Abbas AM, Safwat SM, Elserougy HG. Effect of L-carnitine on diabetes-induced changes of skeletal muscles in rats. J Basic Clin Physiol Pharmacol. 2018;29(1):47–59.
Hino K, Nishikawa M, Sato E, Inoue M. L-carnitine inhibits hypoglycemia-induced brain damage in the rat. Brain Res. 2005;1053(1–2):77–87.
Dupont J, Scaramuzzi RJ. Insulin signalling and glucose transport in the ovary and ovarian function during the ovarian cycle. Biochem J. 2016;473(11):1483–501.
Nasiadek M, Danilewicz M, Sitarek K, Swiatkowska E, Darago A, Stragierowicz J, et al. The effect of repeated cadmium oral exposure on the level of sex hormones, estrous cyclicity, and endometrium morphometry in female rats. Environ Sci Pollut Res Int. 2018;25(28):28025–38.
Yildiz M, Sandikci M. Changes in rat ovary with experimentally induced diabetes and the effects of lycopene on those changes. Romanian J Morphol Embryol. 2016;57(2):703–13.
Pala HG, Pala EE, Artunc Ulkumen B, Aktug H, Yavasoglu A, Korkmaz HA, et al. The protective effect of granulocyte colony-stimulating factor on endometrium and ovary in a rat model of diabetes mellitus. Gynecol Obstet Investig. 2014;78(2):94–100.
Wu Y, Li Y, Liao X, Wang Z, Li R, Zou S, et al. Diabetes induces abnormal ovarian function via triggering apoptosis of granulosa cells and suppressing ovarian angiogenesis. Int J Biol Sci. 2017;13(10):1297–308.
Erfani Majd N, Azizian H, Tabandeh MR, Shahriari A. Effect of Abelmoschus esculentus powder on ovarian histology, expression of apoptotic genes and oxidative stress in diabetic rats fed with high fat diet. Iran J Pharm Res. 2019;18(1):369–82.
Garris DR, Garris BL. Diabetes (db/db) mutation-induced ovarian involution: progressive hypercytolipidemia. Exp Biol Med (Maywood). 2003;228(9):1040–50.
Zare Z, Abouhamzeh B, Masteri Farahani R, Salehi M, Mohammadi M. Supplementation of L-carnitine during in vitro maturation of mouse oocytes affects expression of genes involved in oocyte and embryo competence: an experimental study. Int J Reprod Biomed. 2017;15(12):779–86.
Acknowledgments
The authors thank the Laboratory Animal House of the university (Project number: 97-3038).
Funding
The Immunogenic Research Center (IRC) of Sari Medical Faculty and Chancellor for Research and Technology of Mazandaran University of Medical Sciences, Mazandaran, Iran, financially supported this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Majidi, F.Z., Rezaei, N., Zare, Z. et al. The Protective Effects of l-Carnitine and Zinc Oxide Nanoparticles Against Diabetic Injury on Sex Steroid Hormones Levels, Oxidative Stress, and Ovarian Histopathological Changes in Rat. Reprod. Sci. 28, 888–896 (2021). https://doi.org/10.1007/s43032-020-00317-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-020-00317-0