Abstract
The native savanna ecosystem of the Orinoquia region is the habitat of 50% of the wild ungulate species reported for Colombia. Over the last 20 years, this high species diversity has been strongly threatened by the human transformation of the natural land cover causing connectivity loss of the habitats. The Orinoquia region lacks a biological connectivity analysis with a multi-species approach involving species groups that are representative of the savanna ecosystem such as the ungulates. Understanding the spatial distributions of suitable areas and the main habitats that act as primary habitats for ungulate species in these landscapes is fundamental for the design of conservation strategies. We use an occurrence dataset for lowland tapir (Tapirus terrestris), white-tailed deer (Odocoileus virginianus), and white-lipped peccary (Tayassu pecari) in the development of species’ potential distribution models, binarization process, and morphological spatial pattern analysis. This information was used for the modeling of dispersal corridors connecting the core habitats of the ungulate focal species using the randomized shortest path algorithm and quantifying the weighted global connectivity metrics. Our results suggest an integral corridor with potential least-cost dispersal routes between the native savanna landscape on the middle basins of the savanna rivers and the Meta River. These areas associated with the fluvial dendritic systems are connected, while the core habitats in the eastern part of the savanna landscape are disconnected. We discuss how the application of such knowledge on the spatial ecology of ungulate focal species might improve the management of the metapopulations in the Orinoquia region.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Human activities have drastically shaped the earth’s surface, causing fluctuations in the connectivity of the landscape that lower species diversity (Palmara et al. 2023), besides negatively impacting the ecological, genotypes, species, and functional characteristics of landscape units (de Chazal and Rounsevell 2009; Haddad et al. 2015; IPBES 2019). On a global scale, loss of landscape connectivity generates an increase in the costs of dispersal of mammals, limiting, in turn, the large movements by increasing the distances between habitat patches (Bolger et al. 2008; Bonte et al. 2012; Seidler et al. 2015). Dispersing individuals in unconnected landscapes must travel longer distances through unsuitable parts of the landscape as the matrix, requiring time and energy with the additional risk of an unsuccessful dispersal (Baguette et al. 2013; Bonte et al. 2012). The risk is greater for species from biomes with low levels of connectivity, like savanna landscapes. Savanna is the biome with the lowest connectivity of the 14 world’s biomes assessed (Saura et al. 2017; Herrera et al. 2017). In this context, connectivity of wildlife populations that inhabit the Neotropical savannas is a critical factor in their long-term survival, particularly for large mammals facing multiple threats such as habitat loss and hunting pressure (Bogoni et al. 2020; 2022). Nowadays, wildlife is increasingly confined in ever more smaller and sparse habitat patches (Fahrig 2003; Boitani et al. 2007); and low habitat connectivity impedes ecological processes like daily foraging, seasonal migration, successful reproduction, and juvenile dispersal (Rayfield et al. 2011; Braaker et al. 2014). In the long term, poor connectivity increases extinction risk and decreases genetic diversity because of population isolation, inbreeding depression, demography stochasticity, and limitation of their evolutionary potential and response to fluctuations in landscape connectivity (Hayward and Kerley 2009; Dirzo et al. 2014; Koen et al. 2014; Correa Ayram et al. 2016). These effects are mainly seen in the large mammal species with broad-scale spatial requirements (Bogoni et al. 2023). These are the wild ungulates species that require large areas (Crooks et al. 2017). It should be noted some ungulates are reported as threated species because of habitat loss and degradation of their habitats (Lino et al. 2019).
The Perissodactyla represent 3 families and a total of 16 species, and the Artiodactyla is represented by 10 families and 380 species (Fennessy et al. 2016; Wilson and Mittermeier 2011). These ungulates are distributed mainly in savannas, grasslands, and forests of Africa, America, Asia, and Europe; and they also appear in Australia as introduced species (Wilson and Mittermeier 2011). The Neotropics contain about 30% of the total world mammalian diversity (Carrillo et al. 2015). In the northern South American savannas, ungulates are particularly vulnerable to different anthropogenic threats (Gallina-Tessaro 2019). In Colombia, 12 species of wild ungulates are known, represented by 3 species of tapir, 2 peccary species, and 7 species of deer. Of this diversity 50% of the species are reported in the native savannas of the Orinoco River basin, containing one tapir species, two peccary species, and three deer species (Montenegro et al. 2019). The ungulates are typically herbivorous and inhabit a wide range of diverse habitats (Wilson and Mittermeier 2011), and generally show high dispersal capacity, allowing individuals to find suitable habitat. Ecological conditions indicate some degree of connectivity of these terrestrial landscapes, converting these wild ungulate to “focal species” allowing the creation of connectivity networks, habitat modeling, selection of conservation areas, and the assessment of functional connectivity across large landscapes such as the savanna ecosystems (Riggio and Caro 2017; Riggio et al. 2022). In this context, it is crucial to assess connectivity patterns and identify key areas for implementation of conservation actions for the metapopulations of these mammals (Saranholi et al. 2022). Such patterns must satisfy the ecological requirements of a wide range of organisms in biodiverse countries like Colombia where human transformation has affected nearly 50% of the ecosystems and more than 1500 species of fauna and flora are classified in some category of threat (Andrade-Correa 2011; Etter et al. 2017).
At present, landscape connectivity assessments and corridor designs are being conducted in savanna ecosystems to maintain a suite of ecological processes only supported if multiple species can move across it. These assessments have been explored in a multi-species context (e.g., Brodie et al. 2016; Riggio and Caro 2017; Niyogi et al. 2021; Crego et al. 2021; Riggio et al. 2022). Evaluating landscape connectivity and identifying corridors could be done using different approaches (Littlefield et al. 2019). One of the most used is least-cost modeling or circuit theory (McClure et al. 2016; Jackson et al. 2016). This involves (i) the collection of focal species occurrences or movement data, (ii) assembling spatial variables that are hypothesized to be associated with species occurrence or movement, (iii) fitting appropriate habitat models that allow the prediction of how spatially variables promote or impede species movement or presence in the landscape, and (iv) estimating corridors based on the spatial arrangement of the resistance surface (Bond et al. 2017). Landscape metric indices have been used to estimate and quantify the connectivity of patches (Saura and Pascual-Hortal 2007; Saura and Torné 2009; Niyogi et al. 2021). These indices are based on estimations of the distances between habitats that allow the calculation of the Euclidean distance as the shortest straight line between habitats (Jordan et al. 2007; Andersson & Bodin 2009; Norman and Boesch 2009).
This paper presents a comprehensive assessment of connectivity and corridor designs for Tapirus terrestris, Odocoileus virginianus, and Tayassu pecari in the high plains native savanna located in Vichada department, Colombia. Our approach involved four key steps (i) modeling suitable areas of occurrence for lowland tapir, white-tailed deer, and white-lipped peccary, (ii) classifying these areas into either suitable or unsuitable habitats, as well as identifying core patches within those that were deemed viable, (iii) designing dispersal corridors to connect these essential habitat cores, and finally (iv) quantifying weighted global connectivity metrics to measure the overall connectedness across this expansive region. Our research aims to provide valuable scientific information useful for prioritizing conservation management efforts within eastern Colombia's native savanna ecosystems. Given the increasing vulnerability of the region to climate change and other global factors affecting wildlife populations, our findings can support decision-making processes concerning effective measures toward ensuring the long-term sustainability of both native savanna ecosystems and associated wild ungulate metapopulations.
Methods
Study area
The Colombian Orinoquia represents 33% of the Orinoco River basin and 34% of the national territory (Romero-Ruíz et al. 2011). In this region, there are three main types of native savannas (i) floodplain savanna, (ii) piedmont savanna, and (iii) high plains savanna; and each contains different landforms, soil, savanna formations, and vegetation patterns that belong to a macrosystem of global importance (the Neotropical savannas; Rippstein et al. 2001). The high plains native savanna are represented by the flat and dissected highlands and rocky outcrops associated with the Guiana Shield made up of Tertiary sedimentary rocks, overlying crystalline Precambrian rocks and sediments of the Paleozoic Era and Cretaceous Period of the Mesozoic Era that stand out from the savanna landscape matrix (Huber 2006; SGC 2020). We conducted the study, encompassing 36.841,4 km2 of the high plains native savanna associated with the Meta, Bita, Tomo, Tuparrito, and Tuparro Rivers in the Vichada department (4° 38′ N, 69° 14′ W; 65 m altitude), in the Orinoco River basin, within and in the area of influence of the El Tuparro Biosphere Reserve, east of the Colombian Andes (Rippstein et al. 2001, Fig. 1). The Orinoquia region is biogeographically located east of the Andes with Amazonia to the south, the Caribbean to the northwest, and the Guiana Shield to the east. The region has a tropical climate with an average annual temperature and precipitation of 30 °C and 1115 mm year−1 with a unimodal pattern in the temporal distribution of precipitation during the annual rainy season cycle from April to October and with a dry season from November to March and is largely associated with activity generated by the southern migration of the Intertropical Convergence Zone (Pimentel et al. 2021). The Vichada department’s landscapes are made up of a mosaic of extensive native savanna ecosystems with riparian forests associated with the dendritic pattern of the complex hydric network (Veneklaas et al. 2005; Romero-Ruíz et al. 2011; Pimentel et al. 2021). This landcover functions as a biological corridor connecting fauna from the west to the east and south to north (Mosquera-Guerra et al. 2018).
Focal species
We focus on three of the six species of threatened ungulate species reported for the Orinoquia region (i) lowland tapir (Tapirus terrestris) categorized as Vulnerable (VU) by the International Union for Conservation of Nature—IUCN, (ii) white-tailed deer (Odocoileus virginianus) Least Concern (LC), and (iii) white-lipped peccary (Tayassu pecari) Vulnerable (VU—Montenegro et al. 2019). The three other species of ungulates, the collared peccary (Dicotyles tajacu), red brocket deer (Mazama americana), and the Amazonian brown brocket deer (Mazama nemorivaga), have a limited number of records of occurrence for the native savanna ecosystems of Vichada. The ungulate focal species are morphologically and behaviorally heterogeneous and have different habitat and foraging preferences in the native savanna ecosystem. The lowland tapir is forest-dependent species where are the prime seed dispersers for the palm (Mauritia flexuosa), indicating the close evolutionary relationship between both species (Varela et al. 2019; González et al. 2021; Álviz et al. 2023). The white-lipped peccary is primarily frugivorous, but will eat other plant resources, invertebrates, fungi and even occasional fish, and prefer natural forests (Keuroghlian et al. 2008, 2012, 2013). The white-tailed deer consume leaves, shoots, fruits, seeds, and grasses, a generalist condition that explains their wide distribution in savanna ecosystems (Gallina and López-Arévalo 2016; Roden-Reynolds et al. 2022). The white-lipped peccary has an average weight of less than 40 kg (Dubost et al. 2003), while the white-tailed deer is over 80 kg (Weber and Hidalgo 1999) while the lowland tapir average weight is 200 kg (Medici 2010).
Wild ungulate species occurrence data
Our analyses used a set of occurrence data of the ungulate focal species from different sources such as camera-trap surveys, databases, direct observations, and reference collections reported from 2016 to 2022 for the savanna ecosystem of the Vichada department. Camera-trap campaigns were carried out by the Grupo de Ecología del Paisaje y Modelación de Ecosistemas—ECOLMOD, Fundación Omacha, Wildlife Conservation Society—WCS Colombia, Parque Nacional Natural El Tuparro, and the Instituto de Investigaciones de Recursos Biológicos Alexander von Humboldt—IavH; direct observations reported in the social media iNaturalist, and specimens deposited in the mammals’ reference collection of the IAvH—M.
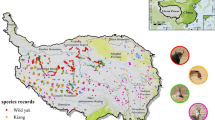
We consolidated a matrix with 985 occurrence registers of lowland tapir, n = 338 (34%); white-tailed deer, n = 205 (21%); and white-lipped peccary, n = 442 (45%, Fig. 1). Subsequently, we created a presence–absence matrix by removing duplicate records taking the coordinates of each record, and establishing a value of 1 for each place when present and 0 otherwise. The data used in the analyses are free to access information consolidated in the Global Biodiversity Information Facility (GBIF www.gbif.org, Accessed Jan 2023), and used in the construction of species distribution models. Geographic coordinates of occurrence records were reported in decimal degrees based on WGS 84 data.
Species distribution model and suitable habitats
We modeled the ungulate focal species using the MaxEnt algorithm version 3.4.3. Habitat suitability was then used to derive the resistance surface and the core habitat patches (Niyogi et al. 2021). We evaluated the performance of the MaxEnt models by calculating commission and omission errors (Anderson et al. 2003), the area under the curve (AUC) of the receiver operating characteristic (ROC) curve (Elith et al. 2006, 2011), as well as by testing partial ROC curves (Lobo et al. 2008). The construction of the suitability models for the species considered 12 predictors: climatic predictors were temperature seasonality (BIO 04), temperature annual range (BIO 07), precipitation of driest month (BIO 14), precipitation of coldest quarter (BIO 19), and isothermality (BIO 03). Habitat quality predictors were (i) normalized difference vegetation index—NDVI for the dry season (October to March), period when the riparian forest is not inundated by the flood pulse of the rivers and calculated for the last 10 years, (ii) distance to rivers and forests, (iii) elevation, (iv) distance to roads and urban centers, and (v) global human modification dataset (gHM). Collinearity between variables was evaluated using the variance inflation factor (VIF, Babak et al. 2014; Akinwande et al. 2015) analysis in the open-source software R, version 4.0.3 (R Core Team 2023). The spatial resolution of the bioclimatic predictors was 500 m; and they were obtained from the WorldClim dataset (Hijmans et al. 2005), habitat quality predictors (Farr et al. 2007; Didan 2015; IDEAM 2021; IGAC 2021), and the global human modification dataset provides a cumulative measure of human modification of terrestrial lands globally at 1 km2 resolution (Kennedy et al. 2019). The gHM values range from 0 to 1 and were calculated by estimating the proportion of a given location (pixel) that is modified, the estimated intensity of modification associated with a given type of human modification or “stressor”. Five major anthropogenic stressors are: (i) human settlement, (ii) agriculture, (iii) transportation, (iv) mining and energy production, and (v) electrical infrastructure.
Binarization process and morphological spatial pattern analysis (MSPA)
We applied the specificity–sensitivity threshold using the R package Presence–Absence test (Freeman and Moisen 2008; Freeman 2012), and binarization converted the continuous gradient maps into binary distributions of suitable and unsuitable classes (Niyogi et al. 2021). Subsequently, we performed the morphological spatial pattern analysis (MSPA) on the suitable areas using the Guidos Toolbox (Hiratra 1996; Meijster et al. 2000; Soille and Vogt 2009; Vogt and Riitters 2017) to identify core habitat patches (Niyogi et al. 2021). In addition, the MSPA classifies the image into seven categories (i) core, (ii) islet, (iii) loop, (iv) bridge, (v) perforation, (vi) edge, and (vii) branch (Soille and Vogt 2009), based on the shape and physical appearance of its features. MSPAs are useful for mapping core habitats; however, their use for mapping corridors is not recommended due to the non-inclusion of spatial metrics such as dispersal capabilities (maximum dispersal distance) of species (Niyogi et al. 2021). In this context, our approach to corridor modeling considered the dispersal capabilities of ungulate focal species reported in the scientific literature (see below).
Modeling dispersal corridors
We modeled dispersal corridors connecting core habitats of species using the randomized shortest path algorithm (RSPA) from the R package “gdistance” (Van Etten et al. 2017). The conceptual approach considered was that of circuit theory (setting θ equal to 0 in RSPA). This method is reported by Zeller et al. (2018) as the most appropriate for modeling dispersal corridors when the starting data are locations of species presence (θ equal to 1 would simulate the least-cost path) (Niyogi et al. 2021). RSPA estimates density values for each grid cell in the landscape, where higher values represent a higher probability of a random walker passing through the cell (McRae and Beier 2007). The data used for this analysis were (i) core habitat patches, (ii) resistance surface, and (iii) maximum dispersal distances of ungulate focal species (Niyogi et al. 2021). The resistance surface represents the degree to which some landscape feature impedes or facilitates some movement process of the species (Adriaensen et al. 2003) and generated using a negative exponential transformation of the habitat suitability gradient maps of the focal species (Trainor et al. 2013; Keeley et al. 2016). For dispersal ability, we employ the “dispersal range” concept which refers to the distance a species can move from an existing population or the parent organism (Clobert et al. 2001). The analyses incorporated the data of maximum dispersal range obtained from satellite telemetry surveys reported in wild individuals by Medici (2010) for the lowland tapir 6.112 m in 20 months of data collection; Roden-Reynolds et al. (2022) for the white-tailed deer 2.114 m in 24 months of tracked, and Hofman et al. (2016) for the white-lipped peccary 3.800 m in 4 month data set.
The resistance values of each grid cell in the high plains native savanna connecting the core habitats and the maximum dispersal capacity of the species were used to derive the probabilistic dispersal area, where each cell was assigned a corridor value (range 0 to 100). The corridor values, together with core habitats, were used to create an integrated conservation area for the three ungulate focal species that highlighted regions with high probability corridors and core habitats (Niyogi et al. 2021).
Quantifying functional connectivity in the savanna landscapes
The weighted global connectivity metrics used in the process of quantifying the connectivity of savanna native ecosystem for ungulate focal species were as follows (i) flux (F), (ii) equivalent probability (EC), (iii) probability of connectivity (PC), and (iv) integral index of connectivity (IIC), calculated using Graphab version 2.4 software (Clauzel et al. 2019—Supplementary Information). Input data were (i) core habitats, (ii) resistance surfaces, and (iii) maximum dispersal costs (threshold) for each species (Niyogi et al. 2021).
Statistical analysis
Sample size was limited and influenced the type of statistical analysis of the results carried out. A Kruskal–Wallis test was performed to determine whether or not there was a statistically significant difference between the areas suitable and core habitats for the ungulate focal species. Ninety-five percent confidence intervals (CI 95%) of area sizes of suitable habitats, core habitats, and dispersal distance were calculated for the ungulate focal species and integrated across species. The tests reported in this paper were calculated using the open-source software R, version 4.0.3 (R Core Team 2023) for the graphical representation using the R software package ggplot2 (Wickham 2016).
Results
Modeling suitable habitats, binarization process, and morphological spatial pattern analysis (MSPA)
The variance inflation factor analysis made it possible to exclude four highly correlated climatic variables with the variables initially considered: mean annual temperature (BIO 01), mean diurnal range (BIO 02), annual precipitation (BIO 12), and precipitation of the driest quarter (BIO 17). The models obtained using the Maxent’s algorithm gave AUC values > 0.9 (lowland tapir = 0.94, white-tailed deer = 0.95, white-lipped peccary = 0.96 and integrated = 0.94; Fig. 2). Habitat suitability ensemble output maps, binarized suitability maps (Fig. 2), and MSPA output maps of focal species and integrated across species showed the spatial distribution of suitable and core habitats (Fig. 3).
The areas values of suitable and core habitats reported for the focal species analyzed varied significantly between land-use categories: (i) unprotected areas, (ii) protected areas (PAs), (iii) forests, and (iv) savanna (suitable habitats p = 0.002, and core habitats p = 0.004; Figs. 4 and 5). The integrated cross-species areas reported for suitable habitats were 4694 km2 and core habitats 1905 km2, respectively.
Modeling dispersal corridors
The dispersal corridor maps for focal species and the integrated cross-species showed patches of riparian forest associated with the fluvial dendritic network that offers low resistance for dispersal in the high plains native savanna (Fig. 6). The patterns are relatively similar across the species and the integrated area considers the minimal requirement for allowing dispersal across all the wild ungulate species in the savanna ecosystem of Vichada.
Quantifying potential functional connectivity
The reported values of global connectivity metrics showed that there will be much lower global connectivity in the high plains native savanna if habitats outside the protected areas were not conserved (see Figs. 7 and 8—Supplementary Information).
Mapping of ungulate focal species and integrated core habitats and links: a lowland tapir (LT), b white-tailed deer (WTD), c white-lipped peccary (WLP), and d ungulate focal species (UFS). Core habitat patches are shown as nodes, while the connections between the nodes are shown as edges. The size of the nodes is proportional to the area of the different habitat patches in the high plain native savanna landscape
Discussion
Multispecies connectivity analyses that include wild ungulate species provide elements at species and community level for conservation and conservation decision-making and have been used successfully in savanna landscapes of Asia (e.g., Liang et al. 2021; Niyogi et al. 2021; Malakoutikhah et al. 2020), Africa (e.g., Riggio and Caro 2017; Riggio et al. 2022), and South America (e.g., Herrera et al. 2017; Sancha et al. 2021; Saranholi et al. 2022). Our study is the first to use a multi-species approach to evaluate biological connectivity in the high plains native savanna in eastern Colombia, besides presenting detailed analyses that identify suitable habitats and potential corridors for three vulnerable ungulate species. The information here generated is of utility for the conservation interventions focused on ungulate species based on their dispersal and geographical distributions for the Orinoquia region.
The distribution of the suitable habitats and the core habitats suggests that large areas are outside the protected areas and act as primary habitats for T. terrestris, O. virginianus, and T. pecari. The maps show that most of the core habitats are located in the middle and lower-middle basins of the Bita, Tomo, Tuparrito, and Tuparro savanna rivers and, to a lesser extent, the lower basin of the Meta River. These areas are located in the riparian forests of the connected dendritic systems, while the core habitats in the eastern part of the savanna rivers are largely disconnected. Our results agree with that reported by Ang’ila et al. (2023) on the influences of (i) plant communities, (ii) topography, and (iii) soil for the distributional patterns of large mammals in the savanna landscapes. In recent decades, the increases in the high plains native savanna areas transformed by livestock and agro-industrial models and the increases in the frequency and extent of forest fires are considered degradation factors for riparian forests in Vichada department, due to recurrent burnings of the surrounding savanna landscapes. These conditions could affect the connectivity on the regional scale (Armenteras et al. 2005; Huidobro et al. 2021; Jiménez et al. 2022; Ramírez-Restrepo et al. 2023). In this context, maintaining or improving the quality of the larger patches associated with the fluvial dendritic network will contribute to more successful use of these patches that can function as “stepping stones” by focal species (Saura and Rubio 2010; Herrera et al. 2017). Fundamental areas that facilitate the movement across savanna matrix for these species can traverse relatively long distances, e.g., tapirs greater than 5000 m (Medici 2010) or intermediate distances > 4000 for peccary and deer (Hofman et al. 2016; Roden-Reynolds et al. 2022). For example, during the rainy season, the riparian forests of the savanna rivers are flooded, causing wildlife to move to higher elevations where the M. flexuosa formations are located (Mosquera-Guerra et al. 2018). Throughout this climatic season, the “savanna” matrix plays a fundamental role in connecting the riparian forest and the palm formations where wildlife can feed the fruit of this palm, the main resource for some large mammal species including the wild ungulates (González et al. 2021; Álviz et al. 2023). Thus, within the high plains native savanna and for the ungulate focal species, different elements of landscape structure may play different roles depending on their topological position and intrinsic habitat characteristics. One of these is passage patches used as intermediate points to subsequently reach other habitat patches in a dispersal process that encompasses multiple movements as steps through the dendritic network of the savanna landscape (Herrera et al. 2017; Riggio and Caro 2017).
The intra- and inter-specific variations in the dispersal of ungulate species demonstrate the difficulty of implementing suitable corridors and stepping stones to provide functional ecological networks (Baguette et al. 2013). Connectivity models have proven useful for designing biological corridors (Beier et al. 2008; Freeman et al. 2019). There is a growing recognition that wild mammal corridors should be planned, designed, and implemented for multiple species so that, ideally, the full range of crucial ecosystem processes can be maintained across large spatial scales (Brodie et al. 2015; Lawler et al. 2020; Crego et al. 2021; Riggio et al. 2022). The circuit theory is a useful tool for modeling species dispersal patterns (McRae et al. 2008; Liang et al. 2021). When interpreting the maps resulting from circuit models, the individual connectivity analyses for the three ungulate focal species yielded “traffic hubs” similar to connecting paths through the middle and lower basins of the Meta and savanna rivers. Thus, the integral corridor largely confirmed potential least-cost dispersal routes within and between the high plains native savanna. This outcome was not unexpected since these threatened ungulate species share habitat affinities. The three species positively select riparian forests associated with rivers and streams of the savanna landscape in search of shelter (predators and high temperatures), water, and food in addition to avoiding human proximity and the open conditions of savannas (Mosquera-Guerra et al. 2018).
The three ungulate focal species are considered landscape species because they have extensive area requirements, low population densities, and importance for local human communities. As such, their conservation contributes to maintaining ecological integrity and ecosystem services provided by savanna landscapes (Álviz and Pérez 2015; Mosquera-Guerra et al. 2018; Montenegro et al. 2019; Khosravi and Hemami 2019). Consequently, it is not surprising that the main corridors identified in the present study overlapped with important areas for biodiversity conservation with different approaches as follow: “Bita Corredor para la Vida” (Bita Life Corredor) in the framework of designation of wetlands of international importance, Ramsar site (Mosquera-Guerra et al. 2018), “Proyecto Vida Silvestre” (Wildlife Project) (Álviz and Pérez 2015), Natural National Park El Tuparro, and the El Tuparro Biosphere Reserve designated recently as other effective area-based conservation measures—OECMs (Protected Planet 2022). Our results suggest that optimizing ungulate focal species corridors using either a multi-species or a focal species approach can be a cost-effective method for securing savanna landscape permeability for a large mammal assemblage associated with these ecosystems (e.g., predators of wild ungulates such as Panthera onca, Puma concolor, and Speothos venaticus). Therefore, the riparian forests that give rise to the dendritic network of the middle and lower river basins of the savanna rivers and Meta River are crucial for maintaining connectivity for all three focal species and deserve to be granted conservation priority, because once that is denied, the dispersal and gene flow of all three species will be inhibited with serious consequences for the biodiversity of the high plains native savanna (Mosquera-Guerra et al. 2018).
Functional connectivity estimates are mainly used to design viable metapopulations of ungulate focal species (Saranholi et al. 2022). The spatial dynamics of these metapopulations are often described as continuous source-sink dynamics, whereby upstream populations serve as sources and downstream populations serve as sinks (Baguette et al. 2013). Global connectivity metrics and corridor structure for the three focal species reported for the high plains native savanna of the Colombian Orinoquia support the view that some species are better connected in these landscapes, for example the lowland tapir. This may be a function of their home range size and dispersal ability, so while lowland tapir is widely distributed with largely contiguous habitats, white-tailed deer and white-lipped peccary remain limited to certain patches, and their core habitats are connected only when they are in proximity. Therefore, some species are more prone to geographic isolation and population decline compared to others within the same landscape (Poethke et al. 2003; Reinhardt et al. 2005; Niyogi et al. 2021). Connectivity per se also affects the dispersal of organisms in dendritic networks (Carrara et al. 2012). Headwaters are often distinct evolutionary populations, whereas confluence patches are a genetic mix of several headwater patches and hence reservoirs for genetic diversity (Baguette et al. 2013). Confluence patches are thought to be more stable; however, headwater patches are the sources of diversity in a basin and therefore very important from a conservation viewpoint (Campbell Grant et al. 2007; Baguette et al. 2013). Our results show high disconnection of the patches of riparian forest that forms the dendritic network associated with sectors of the upper basin of the rivers evaluated. Similar results are reported in the Serra do Mar Biodiversity Corridor designed in the Brazilian largest Atlantic Forest to promote the conservation of lowland tapir and white-lipped peccary where anthropogenic barriers can influence the corridor effectiveness for promoting gene flow at some scales (e.g., within 30 km). This corridor may not be fully effective over its entire extension (Saranholi et al. 2022). The situation is possibly similar to what is happening with the populations of ungulate focal species in our study area.
Consequently, the high plains native savanna are underrepresented within the national system of protected areas. However, the region has a network of private nature reserves that contribute to the conservation of the biodiversity associated with those ecosystems located in the area of influence and inside the Tuparro Biosphere Reserve (López-Arévalo et al. 2021). These private nature reserves are generally small and isolated from each other (RUNAP 2023). This condition translates into different levels of connectivity for ungulate focal species ranging from networks with few large areas and distant from each other, to networks consisting of many small areas where connectivity depends mainly on the dispersal capacity of mammal species (Santini et al. 2016). Limitations in the dispersal capacity of individuals between patches of the protected areas reduce the opportunities for adaptation in response to global changes in subpopulations (Riggio and Caro 2017). In this context, it is a priority to plan the conservation and restoration actions of the riparian forest in the savanna ecosystems at a landscape scale focused on facilitating dispersal between subpopulations to ensure long-term survival as a metapopulation (Hanski and Ovaskainen 2000; Blazquez-Cabrera et al. 2014). It is also necessary to integrate connectivity analyses with management plans for vulnerable ungulate species (e.g., Álviz and Pérez 2015). In addition, high plains native savanna are listed among the ecosystems most vulnerable to degradation processes driven by climate change in Colombia, a situation that could increase the levels of land cover transformation in the region (Etter et al. 2017). This condition makes necessary the construction and integration of adaptation strategies against climate change in the planning of conservation actions and the design of new protected areas (Heller and Zavalenta 2009; Hannah 2011; Magris et al. 2014; Armenteras and Barrera 2023). It is also necessary to consider factors such as variation in habitat connectivity levels of vulnerable ungulate species as individuals move within their areas of occurrence, as well as the uncertainties of connectivity changes driven by current and future climate variability (Albert et al. 2017).
We suggest that our results at species and community levels should be considered in the national program for the conservation of Tapirus in Colombia (Montenegro 2005) and other strategic planners engaged in the conservation of ungulate species and large mammals in the Colombian savanna ecosystem. These actions will be in line with a recent claim to conserve biodiversity conservation under the Convention on Biological Diversity—CDB, and the Aichi Target 11 (Hoban et al. 2020; Saranholi et al. 2022). We also suggest that in the El Tuparro Biosphere Reserve and the Bita River Ramsar site, considered the conservation figures with the greatest extension of savanna landscape in Colombia, that policies for the conservation of the wild ungulate species should include reinforcement (or perhaps restoration) of population connectivity and reduction of illegal hunting pressures (Saranholi et al. 2022), including subsequent protection or restoration of wildlife corridors—i.e., patches of land that connect two or more protected areas or seasonal ranges of focal species (Jones et al. 2012; Beale et al. 2013; Bond et al. 2017)—because of the contribution that ungulates make in carbon exchange and accumulation in the Orinoco high plains native savanna (Morales-Rincon et al. 2021). This is essential in the current global climate crisis. Finally, we recommend (i) exploring the combined effects of climate, land cover, and anthropogenic influences such as wildfire on ungulate focal species in the climate change scenarios, (ii) incorporating more detailed species information such as dispersal limitation, biotic interactions, and population density into the connectivity analysis, and (iii) conducting fine-scale monitoring with camera traps and GPS collars to verify the proposed connectivity priority.
Availability of data and material
The data sets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
The code supporting the current study is available from the corresponding author on request.
References
Adriaensen F, Chardon JP, De Blust G, Swinnen E, Villalba S, Gulinck H, Matthysen E (2003) The application of “least-cost” modelling as a functional landscape model. Landsc Urban Plan 64:233–247. https://doi.org/10.1016/S0169-2046(02)00242-6
Akinwande MO, Hussaini GD, Agboola S (2015) Variance inflation factor: as a condition for the inclusion of suppressor variable(s) in regression analysis. J Stat 5:754–767. https://doi.org/10.4236/ojs.2015.57075
Albert CH, Rayfield B, Dumitru M, Gonzalez A (2017) Applying network theory to prioritize multispecies habitat networks that are robust to climate and land-use change. Conserv Biol 31:1383–1396. https://doi.org/10.1111/cobi.12943
Álviz A, Pérez AK (2015) Plan para la conservación de la danta de tierras bajas (Tapirus terrestris) en los departamentos de Casanare, Arauca y Vichada. Corporinoquia-Fundación Orinoquia Biodiversa, Yopal
Álviz A, González-González P, Pérez-Torres J (2023) Scientific and traditional knowledge meet: diet of the lowland tapir Tapirus terrestris in the Orinoquia region of Colombia. Anim Biodivers Conserv 46:87–97. https://doi.org/10.32800/abc.2023.46.0087
Anderson R, Lew D, Peterson A (2003) Evaluating predictive models of species’ distributions: criteria for selecting optimal models. Ecol Modell 162:211–232. https://doi.org/10.1016/S0304-3800(02)00349-6
Andersson E, Bodin O (2009) Practical tool for landscape planning? an empirical investigation of network based models of habitat fragmentation. Ecography 32:123–132. https://doi.org/10.1111/j.1600-0587.2008.05435.x
Andrade-Correa MG (2011) Estado del conocimiento de la biodiversidad en Colombia y sus amenazas. Consideraciones para fortalecer la interacción ciencia-política. ACCEFYN 35:491–508
Ang’ila RO, Kimuyu DM, Wambugu GM, Kenfack D, Musili PM, Kartzinel TR, (2023) Fine-scale variation in soil and topography influences herbaceous vegetation and the distribution of large mammalian herbivores. Afr J Ecol 61:1–11. https://doi.org/10.1111/aje.13166
Armenteras D, de la Barrera F (2023) Landscape management is urgently needed to address the rise of megafires in South America. Comm Earth Environ 4:305–309. https://doi.org/10.1038/s43247-023-00964-6
Armenteras D, Romero M, Galindo G (2005) Vegetation fire in the savannas of the Llanos Orientales of Colombia. World Resour Rev 17:531–543
Babak N, Hamm NAS, Groen TA, Skidmore AK, Toxopeus AG (2014) Where is positional uncertainty a problem for species distribution modelling? Ecography 37:191–203. https://doi.org/10.1111/j.1600-0587.2013.00205.x
Baguette M, Blanchet S, Legrand D, Stevens VM, Turlure C (2013) Individual dispersal, landscape connectivity and ecological networks. Biol Rev 88:310–326. https://doi.org/10.1111/brv.12000
Beale CM, van Rensberg S, Bond WJ, Coughenour M, Fynn RWS, Gaylard A, Grant R, Harris B, Jones T, Mduma S, Owen-Smith N, Sinclair ARE (2013) Ten lessons for the conservation of African savannah ecosystems. Biol Conserv 167:224–232. https://doi.org/10.1016/j.biocon.2013.08.025
Beier P, Majka DR, Spencer WD (2008) Forks in the road: choices in procedures for designing wildland linkages. Conserv Biol 22:836–851. https://doi.org/10.1111/j.1523-1739.2008.00942.x
Blazquez-Cabrera S, Bodin O, Saura S (2014) Indicators of the impacts of habitat loss on connectivity and related conservation priorities: do they change when habitat patches are defined at different scales? Ecol Indic 45:704–716. https://doi.org/10.1016/j.ecolind.2014.05.028
Bogoni J, Peres C, Ferraz K (2020) Extent, intensity and drivers of mammal defaunation: a continental-scale analysis across the Neotropics. Sci Rep 10:1–16. https://doi.org/10.1038/s41598-020-72010-w
Bogoni JA, Ferraz KMPMB, Peres CA (2022) Continental-scale local extinctions in mammal assemblages are synergistically induced by habitat loss and hunting pressure. Biol Conserv 272:109635. https://doi.org/10.1016/j.biocon.2022.109635
Bogoni JA, Boron V, Peres CA, Coelho MEMS, Morato RG, Oliveira-da-Costa M (2023) Impeding anthropogenic threats and protected area prioritization for jaguars in the Brazil. Comm Biol 6:132–143. https://doi.org/10.1038/s42003-023-04490-1
Boitani L, Falcucci A, Maiorano L, Rondinini C (2007) Ecological networks as conceptual frameworks or operational tools in conservation. Conserv Biol 21:1414–1422. https://doi.org/10.1111/j.1523-1739.2007.00828.x
Bolger DT, Newmark WD, Morrison TA, Doak DF (2008) The need for integrative approaches to understand and conserve migratory ungulates. Ecol Lett 11:63–77. https://doi.org/10.1111/j.1461-0248.2007.01109.x
Bond ML, Bradley CM, Kiffner C, Morrison TA, Lee DE (2017) A multi-method approach to delineate and validate migratory corridors. Landsc Ecol 32:1705–1721. https://doi.org/10.1007/s10980-017-0537-4
Bonte D, Van Dyck H, Bullock JM, Coulon A, Delgado M, Gibbs M, Lehouck V, Matthysen E, Mustin K, Saastamoinen M, Schtickzelle N, Stevens V, Vandewoestijne S, Baguette M, Barton K, Benton TG, Chaput-Bardy A, Clobert J, Dytham C, Hovestadt T, Meier CM, Palmer S, Turlure C, Travis JMJ (2012) Costs of dispersal. Biol Rev 87:290–312. https://doi.org/10.1111/j.1469-185X.2011.00201.x
Braaker S, Moretti M, Boesch R, Ghazoul J, Obrist MK, Bontadina F (2014) Assessing habitat connectivity for ground-dwelling animals in an urban environment. Ecol Appl 24:1583–1595. https://doi.org/10.1890/13-1088.1
Brodie JF, Giordano AJ, Dickson BG, Hebblewhite M, Bernard H, Mohd-Azlan J, Anderson J, Ambu L (2015) Evaluating multispecies landscape connectivity in a threatened tropical mammal community. Conserv Biol 29:122–132. https://doi.org/10.1111/cobi.12337
Brodie JF, Mohd-Azlan J, Schnell JK (2016) How individual links affect network stability in a large-scale, heterogeneous metacommunity. Ecology 97:1658–1667. https://doi.org/10.1890/15-1613.1
Campbell Grant EH, Lowe WH, Fagan WF (2007) Living in the branches: population dynamics and ecological processes in dendritic networks. Ecol Lett 10:165–175. https://doi.org/10.1111/j.1461-0248.2006.01007.x
Carrara F, Altermatt F, Rodriguez-Iturbe I, Rinaldo A (2012) Dendritic connectivity controls biodiversity patterns in experimental metacommunities. PNAS 109:5761–5766. https://doi.org/10.1073/pnas.1119651109
Carrillo JD, Forasiepi A, Jaramillo C, Sánchez-Villagra MR (2015) Neotropical mammal diversity and the Great American Biotic Interchange: spatial and temporal variation in South America’s fossil record. Front Genet 5:1–11. https://doi.org/10.3389/fgene.2014.00451
Clauzel C, Foltête JC, Girardet X, Vuidel G (2019) User manual. Graphab. Version 2.4. https://sourcesup.renater.fr/www/graphab/download/manual-2.4-en.pdf. Accessed Feb 2023
Clobert J, Danchin E, Dhondt AA, Nichols JD (2001) Dispersal. Oxford University Press, Oxford
Correa Ayram CA, Mendoza ME, Etter A, Perez Salicrup DR (2016) Habitat connectivity in biodiversity conservation: a review of recent studies and applications. Prog Phys Geogr 40:7–37. https://doi.org/10.1177/0309133315598713
Crego RD, Wells HBM, Ndung’u KS, Evans L, Njeri Nduguta R, Chege MA, Brown MB, Ogutu JO, Ojwang GO, Fennessy J, O’Connor D, Stacy-Dawes J, Rubenstein DI, Martins DJ, Leimgruber P, Stabach JA (2021) Moving through the mosaic: identifying critical linkage zones for large herbivores across a multiple-use African landscape. Landsc Ecol. https://doi.org/10.1007/s10980-021-01232-8
Crooks KR, Burdett CL, Theobald DM, King SRB, Di Marco M, Rondinini C, Boitani L (2017) Quantification of habitat fragmentation reveals extinction risk in terrestrial mammals. PNAS 114:7635–7640. https://doi.org/10.1073/pnas.1705769114
de Chazal J, Rounsevell MDA (2009) Land-use and climate change within assessments of biodiversity change: a review. Glob Environ Change 19:306–315. https://doi.org/10.1016/j.gloenvcha.2008.09.007
Didan K (2015) MOD13A1 MODIS/Terra vegetation indices 16-day L3 global 500m SIN grid V006 . NASA EOSDIS land processes DAAC. https://lpdaac.usgs.gov/products/mod13a1v006/. Accessed Feb 2023
Dirzo R, Young HS, Galetti M, Ceballos G, Isaac NJB, Collen B (2014) Defaunation in the anthropocene. Science 345:401–406. https://doi.org/10.1126/science.1251817
Dubost G, Dutertre C, Henry O (2003) Body weight increase in the two peccary species of the genus Tayassu (Tayassuidae, Artiodactyla). Mammalia 67:5–645. https://doi.org/10.1515/mamm.2003.67.1.55
Elith J, Graham CH, Anderson RP, Dudik M, Ferrier S, Guisan A, Hijmans RJ, Huettmann F, Leathwick JR, Lehmann A, Jin L, Lohmann LG, Loiselle BA, Craig Mortiz GM, Nakamura M, Nakazawa Y, Overton JMCCM, Townsend Peterson A, Phillips SJ, Richardson K, Scachetti-Pereira R, Schapire RE, Soberón J, Williams S, Wisz MS, Zimmermann NE (2006) Novel methods improve prediction of species’ distributions from occurrence data. Ecography 29:129–151. https://doi.org/10.1111/j.2006.0906-7590.04596
Elith J, Phillips SJ, Hastie T, Dudík M, Chee YE, Yates CJ (2011) A statistical explanation of MaxEnt for ecologists. Divers Distrib 17:43–57. https://doi.org/10.1111/j.1472-4642.2010.00725.x
Etter A, Andrade A, Saavedra K, Amaya P, Arévalo P (2017) Estado de los ecosistemas colombianos una aplicación de la metodología de la lista roja de ecosistemas. Version 2.0. Pontificia Universidad Javeriana y Conservación Internacional, Bogotá, D.C.
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Syst 34:487–515. https://doi.org/10.1146/annurev.ecolsys.34.011802.132419
Farr T, Rosen P, Caro E, Crippen R, Duren R, Hensley S, Kobrick M, Paller M, Rodríguez E, Roth L, Seal D, Shaffer S, Shimada J, Umland J, Werner M, Oskin M, Burbank D, Alsdorf D (2007) The shuttle radar topography mission. Rev Geophys 45:1–33. https://doi.org/10.1029/2005RG000183
Fennessy J, Bidon T, Reuss F, Kumar V, Elkan P, Nilsson MA, Vamberger M, Fritz U, Janke A (2016) Multi-locus analyses reveal four giraffe species instead of one. Curr Biol 26:2543–2549. https://doi.org/10.1016/j.cub.2016.07.036
Freeman E, Moisen G (2008) PresenceAbsence: an R package for presence-absence model analysis. J Stat Sofw 23:1–31. https://doi.org/10.18637/jss.v023.i11
Freeman B, Roehrdanz PR, Peterson AT (2019) Modeling endangered mammal species distributions and forest connectivity across the humid upper Guinea lowland rainforest of west Africa. Biodivers Conserv 28:671–685. https://doi.org/10.1007/s10531-018-01684-6
Freeman E (2012) Package ‘PresenceAbsence’, R Package. Version 1.1.9. https://cran.r-project.org/package=PresenceAbsence. Accessed Mar 2023
Gallina S, López-Arévalo H (2016) Odocoileus virginianus. The IUCN red list of threatened species 2016: e.T42394A22162580. https://doi.org/10.2305/IUCN.UK.2016-2.RLTS.T42394A22162580.en. Accessed May 2023
Gallina-Tessaro S (2019) Ecology and conservation of tropical ungulates in Latin America. Springer, Cham
González TM, González-Trujillo JD, Muñoz A, Armenteras D (2021) Differential effects of fire on the occupancy of small mammals in neotropical savanna-gallery forests. Perspect Ecol Conserv 19:179–188. https://doi.org/10.1016/j.pecon.2021.03.005
Haddad NM, Brudvig LA, Clobert J, Davies KF, González A, Holt RD, Lovejoy TE, Sexton JO, Austin MP, Townshend JR (2015) Habitat fragmentation and its lasting impact on earth’s ecosystems. Sci Adv 1:e1500052. https://doi.org/10.1126/sciadv.1500052
Hannah L (2011) Climate change, connectivity, and conservation success. Conserv Biol 25:1139–1142. https://doi.org/10.1111/j.1523-1739.2011.01788.x
Hanski I, Ovaskainen O (2000) The metapopulation capacity of a fragmented landscape. Nature 404:755–758. https://doi.org/10.1038/35008063
Hayward MW, Kerley GIH (2009) Fencing for conservation: restriction of evolutionary potential or a riposte to threatening processes? Biol Conserv 142:1–13. https://doi.org/10.1016/j.biocon.2008.09.022
Heller NE, Zavalenta ES (2009) Biodiversity management in the face of climate change: a review of 22 years of recommendations. Biol Conserv 142:14–32. https://doi.org/10.1016/j.biocon.2008.10.006
Herrera LP, Sabatino MC, Jaimes FR, Saura S (2017) Landscape connectivity and the role of small habitat patches as stepping stones: an assessment of the grassland biome in South America. Biodivers Conserv 26:3465–3479. https://doi.org/10.1007/s10531-017-1416-7
Hijmans RJ, Cameron SE, Parra JL, Jones PG, Jarvis A (2005) Very high resolution interpolated climate surfaces for global land areas. Int J Climatol 25:1965–1978. https://doi.org/10.1002/joc.1276
Hiratra T (1996) A unified linear-time algorithm for computing distance maps. Inform Process Lett 58:129–133. https://doi.org/10.1016/0020-0190(96)00049-X
Hoban S, Bruford M, D’Urban Jackson J, Lopes-Fernandes M, Heuertz M, Hohenlohe PA, Paz-Vinas I, Sjögren-Gulve P, Segelbacher G, Vernesi C, Aitken S, Bertola LD, Bloomer P, Breed M, Rodríguez-Correa H, Chris Funk W, Grueber CE, Hunter ME, Jaffe R, Liggins L, Mergeay J, Moharrek F, O’Brien D, Ogden R, Palma-Silva C, Pierson J, Ramakrishnan U, Simo-Droissart M, Tani N, Waits L, Laikre L (2020) Genetic diversity targets and indicators in the CBD post-2020 Global Biodiversity Framework must be improved. Biol Conserv 248:108654. https://doi.org/10.1016/j.biocon.2020.108654
Hofman MPG, Signer J, Hayward MW, Balkenhol N (2016) Spatial ecology of a herd of white-lipped peccaries (Tayassu pecari) in Belize using GPS telemetry: challenges and preliminary results. Therya 7:21–37. https://doi.org/10.12933/therya-16-335
Huber O (2006) Herbaceous ecosystems on the Guayana Shield, a regional overview. J Biogeogr 33:464–475. https://doi.org/10.1111/j.1365-2699.2005.01454.x
Huidobro G, Bernal R, Wagner S (2021) Post-fire regeneration of the palm Mauritia flexuosa in Vichada, Orinoco region of Colombia. In: Leal Filho W, Azeiteiro UM, Setti AFF (eds) Sustainability in natural resources management and land planning. Springer, Berlin, pp 445–463
IDEAM-Instituto de Hidrología, Meteorología y Estudios Ambientales (2021) Mapa nacional de coberturas de la tierra 2018. http://www.ideam.gov.co/. Accessed Feb 2023
IGAC-Instituto Geográfico Agustín Codazzi (2021) Cartografía base para Colombia (1:100.000). https://www.igac.gov.co/. Accessed Feb 2023
IPBES-Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services (2019) Global assessment report on biodiversity and ecosystem services of the intergovernmental science-policy platform on biodiversity and ecosystem services. https://ipbes.net/global-assessment. Accessed Feb 2023
Jackson CR, Marnewick K, Lindsey PA, Røskaft E, Robertson MP (2016) Evaluating habitat connectivity methodologies: a case study with endangered African wild dogs in South Africa. Landsc Ecol 31:1433–1447. https://doi.org/10.1007/s10980-016-0342-5
Jiménez R, Ardila AV, Vargas-Burbano AC, Hernandez AJ, León-Velásquez E (2022) Biomass burning-agriculture coupling in the Orinoco savannas—particulate matter emission scenarios. Front Environ Sci 10:689844. https://doi.org/10.3389/fenvs.2022.689844
Jones T, Bamford AJ, Ferrol-Schulte D, Hieronimo P, Mcwilliam N, Rovero F (2012) Vanishing wildlife corridors and options for restoration: a case study from Tanzania. Trop Conserv Sci 5:463–474. https://doi.org/10.1177/194008291200500405
Jordan F, Magura T, Tóthmérész B, Vasas V, Ködöböcz V (2007) Carabids (Coleoptera: Carabidae) in forest patchworks: a connectivity analysis of the Bereg plain landscape graph. Landsc Ecol 22:527–1539. https://doi.org/10.1007/s10980-007-9149-8
Keeley ATH, Beier P, Gagnon JW (2016) Estimating landscape resistance from habitat suitability: effects of data source and nonlinearities. Landsc Ecol 31:2151–2162. https://doi.org/10.1007/s10980-016-0387-5
Kennedy CM, Oakleaf JR, Theobald DM, Baruch-Mordo S, Kiesecker J (2019) Managing the middle: a shift in conservation priorities based on the global human modification gradient. Glob Change Biol 25:811–826. https://doi.org/10.1111/gcb.14549
Keuroghlian A, Donald P, Eaton DP (2008) Fruit availability and peccary frugivory in an isolated Atlantic forest fragment: effects on peccary ranging behavior and habitat use. Biotropica 40:62–70. https://doi.org/10.1111/j.1744-7429.2007.00351.x
Keuroghlian A, Desbiez ALJ, Beisiegel BM, Medici EP, Gatti A, Mendes Pontes AR, Campos CB, Tófoli CF, JrEA M, Azevedo FC, Pinho GM, Cordeiro LP, JrTS S, Morais AA, Mangini PR, Flesher K, Rodrigues LF, Almeida LB (2012) Avaliação do risco de extinção do queixada, Tayassu pecari (Link, 1795), no Brasil [Extinction risk assessment of white-lipped peccaries in Brazil]. Bio Brasil 2:84–102
Keuroghlian A, Desbiez A, Reyna-Hurtado R, Altrichter M, Beck H, Taber A, Fragoso JMV (2013) Tayassu pecari. The IUCN red list of threatened species 2013:e.T41778A44051115. https://doi.org/10.2305/IUCN.UK.2013-1.RLTS.T41778A44051115.en. Accessed May 2023
Khosravi R, Hemami MR (2019) Identifying landscape species for ecological planning. Ecol Ind 99:140–148. https://doi.org/10.1016/j.ecolind.2018.12.010
Koen EL, Bowman J, Sadowski C, Walpole AA, Tatem A (2014) Landscape connectivity for wildlife: development and validation of multispecies linkage maps. Methods Ecol Evol 5:626–633. https://doi.org/10.1111/2041-210X.12197
Lawler JJ, Rinnan DS, Michalak JL, Withey JC, Randels CR, Possingham HP (2020) Planning for climate change through additions to a national protected area network: implications for cost and configuration. Philos Trans R Soc B 375:20190117. https://doi.org/10.1098/rstb.2019.0117
Liang J, Ding Z, Jiang Z, Yang X, Xiao R, Bikram Singh P, Hu Y, Guo K, Zhang Z, Hu H (2021) Climate change, habitat connectivity, and conservation gaps: a case of four ungulate species endemic to the Tibetan plateau. Landsc Ecol 36:1071–1087. https://doi.org/10.1007/s10980-021-01202-0
Lino A, Fonseca C, Rojas D, Fischer E, Ramos Pereira MJ (2019) A meta-analysis of the effects of habitat loss and fragmentation on genetic diversity in mammals. Mamm Biol 94:9–76. https://doi.org/10.1016/j.mambio.2018.09.006
Littlefield CE, Krosby M, Michalak JL, Lawler JJ (2019) Connectivity for species on the move: supporting climate-driven range shifts. Front Ecol Environ 17:270–278. https://doi.org/10.1002/fee.2043
Lobo JM, Jiménez-Valverde A, Real R (2008) AUC: a misleading measure of the performance of predictive distribution models. Glob Ecol Biogeogr 17:145–151. https://doi.org/10.1111/j.1466-8238.2007.00358.x
López-Arévalo HF, Liévano-Latorre LF, Montenegro Díaz OL (2021) The role of small reserves on mammal conservation in Colombia. Caldasia 43:354–365. https://doi.org/10.15446/caldasia.v43n2.85466
Magris RA, Pressey RL, Weeks R, Ban NC (2014) Integrating connectivity and climate change into marine conservation planning. Biol Conserv 170:207–221. https://doi.org/10.1016/j.biocon.2013.12.032
Malakoutikhah S, Fakheran S, Hemami MR, Tarkesh M, Senn J (2020) Assessing future distribution, suitability of corridors and efficiency of protected areas to conserve vulnerable ungulates under climate change. Div Distrib 26:1383–1396. https://doi.org/10.1111/ddi.13117
McClure ML, Hansen AJ, Inman RM (2016) Connecting models to movements: testing connectivity model predictions against empirical migration and dispersal data. Landsc Ecol 31:1419–1432. https://doi.org/10.1007/s10980-016-0347-0
McRae BH, Beier P (2007) Circuit theory predicts gene flow in plant and animal populations. PNAS 104:19885–19890. https://doi.org/10.1073/pnas.0706568104
McRae BH, Dickson BG, Keitt TH, Shah VB (2008) Using circuit theory to model connectivity in ecology, evolution, and conservation. Ecology 89:2712–2724. https://doi.org/10.1890/07-1861.1
Medici EP (2010) Assessing the viability of lowland tapir populations in a fragmented landscape. Ph.D. thesis, Biodiversity Management Durrell Institute of Conservation and Ecology, University of Kent, Canterbury, United Kingdom
Meijster A, Roerdink J, Hesselink W (2000) A general algorithm for computing distance transforms in linear time. In: Goutsias J, Vicent L, Bloomberg D (eds) Mathematical morphology and its applications to image and signal processing. Kluwer Academic Publishers, Dordrecht, pp 331–340
Montenegro OL, López-Arévalo H, Mora-Beltrán C, Lizcano DJ, Serrano H, Mesa E, Bonilla-Sánchez A (2019) Tropical ungulates of Colombia. In: Gallina-Tessaro S (ed) Ecology and conservation of tropical ungulates in Latin America. Springer, Cham, pp 157–195
Montenegro OL (2005) Programa nacional para la conservación del género Tapirus en Colombia. Ministerio de Ambiente, Vivienda y Desarrollo Territorial. Dirección de Ecosistemas, Bogotá D.C., Colombia
Morales-Rincon LA, Hernandez AJ, Rodriguez-Hernandez NS, Jimenez R (2021) Carbon exchange and accumulation in an Orinoco high plains native savanna ecosystem as measured by eddy covariance. Front Environ Sci 9:673932. https://doi.org/10.3389/fenvs.2021.673932
Mosquera-Guerra F, Trujillo F, Díaz-Pulido AP, Mantilla-Meluk H (2018) Diversidad, abundancia relativa y patrones de actividad de los mamíferos medianos y grandes, asociados a los bosques riparios del río Bita, Vichada, Colombia. Biota Colom 19:202–218. https://doi.org/10.21068/c2018.v19n01a13
Niyogi R, Shekhar Sarkar M, Hazra P, Rahman M, Banerjee S, John R (2021) Habitat connectivity for the conservation of small ungulates in a human-dominated landscape. ISPRS Int J Geoinf 10:1–24. https://doi.org/10.3390/ijgi10030180
Norman E, Boesch C (2009) Sophisticated Euclidean maps in forest chimpanzees. Anim Behav 77:1195–1201. https://doi.org/10.1016/j.anbehav.2009.01.025
Palmara GM, Rozenfeld A, de Santana ChN, Klecka J, Riera R, Eguíluz VM, Melián CJ (2023) Biodiversity dynamics in landscapes with fluctuating connectivity. Ecography 2023:e06385. https://doi.org/10.1111/ecog.06385
Pimentel JN, Rogéliz Prada CA, Walschburger T (2021) Hydrological modeling for multifunctional landscape planning in the Orinoquia region of Colombia. Front Environ Sci 9:673215. https://doi.org/10.3389/fenvs.2021.673215
Protected Planet (2022) Discover the world’s protected areas. IOP Publishing Physicsweb. https://www.protectedplanet.net/en. Accessed Mar 2023
Poethke HJ, Hovestadt T, Mitesser O (2003) Local extinction and the evolution of dispersal rates: causes and correlations. Am Nat 161:631–640. https://doi.org/10.1086/368224
R Development Core Team (2023) R: a language and environment for statistical computing. R Foundation for statistical computing. Version 3.1.1. R Foundation for Statistical Computing, Vienna
Ramírez-Restrepo CA, Vera-Infanzón RR, Rao IM (2023) The carbon footprint of young-beef cattle finishing systems in the eastern plains of the Orinoco River basin of Colombia. Front Anim Sci 4:1103826. https://doi.org/10.3389/fanim.2023.1103826
Rayfield B, Fortin M, Fall A (2011) Connectivity for conservation: a framework to classify network measures. Ecology 92:847–858. https://doi.org/10.1890/09-2190.1
Reinhardt K, Köhler G, Maas S, Detzel P (2005) Low dispersal ability and habitat specificity promote extinctions in rare but not in widespread species: the orthoptera of Germany. Ecography 28:593–602. https://doi.org/10.1111/j.2005.0906-7590.04285.x
Riggio J, Caro T (2017) Structural connectivity at a national scale: wildlife corridors in Tanzania. PLoS ONE 12:e0187407. https://doi.org/10.1371/journal.pone.0187407
Riggio J, Foreman K, Freedman E, Gottlieb B, Hendler D, Radomille D, Rodriguez R, Yamashita T, Kioko J, Kiffner C (2022) Predicting wildlife corridors for multiple species in an east African ungulate community. PLoS ONE 17:e0265136. https://doi.org/10.1371/journal.pone.0265136
Rippstein G, Amesquita E, Escobar G, Grollier C (2001) Condiciones naturales de las sabanas. In: Rippstein G, Escobar G, Motta F (eds) Agroecología y biodiversidad de las sabanas de los Llanos Orientales. CIAT, Cali, pp 1–21
Roden-Reynolds P, Kent CM, Li AY, Mullinax JM (2022) Patterns of white-tailed deer movements in suburban Maryland: implications for zoonotic disease mitigation. Urban Ecosyst 25:1925–1938. https://doi.org/10.1007/s11252-022-01270-3
Romero-Ruíz MH, Flantua SGA, Tansey K, Berrio JC (2011) Landscape transformations in savannas of northern South America: land use/cover changes since 1987 in the Llanos Orientales of Colombia. Appl Geogr 32:766–776
RUNAP-Registro Único Nacional de Áreas Protegidas (2023) Cartografía de las áreas protegidas para Colombia (1:100.000). IOP Publishing Physicsweb http://runap.parquesnacionales.gov.co/. Accessed Feb 2023
Sancha N, Boyle SA, Mclntryre NE (2021) Identifying structural connectivity priorities in eastern Paraguay’s fragmented Atlantic Forest. Sci Rep 11:16129. https://doi.org/10.1038/s41598-021-95516-3
Santini L, Saura S, Rondinini C (2016) A composite network approach for assessing multi-species connectivity: an application to road defragmentation prioritisation. PLoS ONE 11:e0164794. https://doi.org/10.1371/journal.pone.0164794
Saranholi BH, Sanches A, Moreira-Ramírez JF, da Silva CC, Galetti M, JrPM G (2022) Long-term persistence of the large mammal lowland tapir is at risk in the largest Atlantic forest corridor. Perspect Ecol Conserv 20:263–271. https://doi.org/10.1016/j.pecon.2022.02.002
Saura S, Pascual-Hortal L (2007) A new habitat availability index to integrate connectivity in landscape conservation planning: comparison with existing indices and application to a case study. Landsc Urban Plan 83:91–103. https://doi.org/10.1016/j.landurbplan.2007.03.005
Saura S, Rubio L (2010) A common currency for the different ways in which patches and links can contribute to habitat availability and connectivity in the landscape. Ecography 33:523–537. https://doi.org/10.1111/j.1600-0587.2009.05760.x
Saura S, Torné J (2009) Conefor Sensinode 2.2: a software package for quantifying the importance of habitat patches for landscape connectivity. Environ Model Softw 24:135–139. https://doi.org/10.1016/j.envsoft.2008.05.005
Saura S, Bastin L, Battistella L, Mandrici A, Dubois G (2017) Protected areas in the world’s ecoregions: how well connected are they? Ecol Indic 76:144–158. https://doi.org/10.1016/j.ecolind.2016.12.047
Seidler RG, Long RA, Berger J, Bergen S, Beckmann JP (2015) Identifying impediments to long-distance mammal migrations. Conserv Biol 29:99–109. https://doi.org/10.1111/cobi.12376
SGC—Sistema Geológico Colombiano (2020) Geological map of Colombia. https://www2.sgc.gov.co/MGC/Paginas/agc_500K2020.aspx. Accessed Abr 2023.
Soille P, Vogt P (2009) Morphological segmentation of binary patterns. Pattern Recognit Lett 30:456–459. https://doi.org/10.1016/j.patrec.2008.10.015
Trainor AM, Walters JR, Morris WF, Sexton J, Moody A (2013) Empirical estimation of dispersal resistance surfaces: a case study with red-cockaded woodpeckers. Landsc Ecol 28:755–767. https://doi.org/10.1007/s10980-013-9861-5
Van Etten J (2017) R package gdistance: distances and routes on geographical grids. J Stat Softw 76:1–21. https://doi.org/10.18637/jss.v076.i13
Varela D, Flesher K, Cartes JL, de Bustos S, Chalukian S, Ayala G, Richard-Hansen C (2019) Tapirus terrestris. The IUCN red list of threatened species 2019:e.T21474A45174127. https://doi.org/10.2305/IUCN.UK.2019-1.RLTS.T21474A45174127.en. Accessed May 2023
Veneklaas EJ, Fajardo A, Obregón S, Lozano J (2005) Gallery forest types and their environmental correlates in a Colombian savanna landscape. Ecography 28:236–252. https://doi.org/10.1111/j.0906-7590.2005.03934.x
Vogt P, Riitters K (2017) Guidos toolbox: universal digital image object analysis. Eur J Remote Sens 50:352–361. https://doi.org/10.1080/22797254.2017.1330650
Weber M, Hidalgo R (1999) Morfometría, patrones de crecimiento y ganancia de peso de venados cola blanca (Odocoileus virginianus) en cautiverio en Durango y Toluca, México. Vet Mex 30:183–188
Wickham H (2016) Elegant graphics for data analysis. Springer, New York
Wilson DE, Mittermeier RA (2011) Handbook of the mammals of the world, vol 2. Hoofed mammals. Lynx Editions, Barcelona
Zeller KA, Jennings MK, Vickers TW, Ernest HB, Cushman SA, Boyce WM (2018) Are all data types and connectivity models created equal? Validating common connectivity approaches with dispersal data. Divers Distrib 24:868–879. https://doi.org/10.1111/ddi.12742
Acknowledgements
The authors are grateful to Universidad Nacional de Colombia—UNAL and the Sistema Nacional de Regalías—SNR; in the framework of the project “Diseño participativo de estrategias para la reducción de incendios forestales, la conservación de la biodiversidad y el desarrollo regional en paisajes multifuncionales de Vichada (BPIN 2020000100456)”, and to local communities and government authorities of the department of Vichada, Forest First for all their support in the logistics of the research, the director E. Olaya and the professional D. Echenique from El Tuparro National Natural Park, WCS Colombia, IAvH, IAvH- M, the Asociación Colombiana de Zoología, iNaturalist, and Fundación Omacha and especially its director F. Trujillo for providing the initial raw occurrence dataset information used in the construction of the models of focal ungulate species in the savanna ecosystems of the department of Vichada.
Funding
Open Access funding provided by Colombia Consortium. This study was supported by Universidad Nacional de Colombia—UNAL and the Sistema Nacional de Regalías—SNR.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Ethical approval was granted by Universidad Nacional de Colombia and Natural National Park El Tuparro.
Consent to participate
All authors agreed to participate in this study and all have contributed to its content and current version.
Consent for publication
All authors agreed to submit this manuscript to Mammalian Biology for publication.
Additional information
Handling editor: Vera Rduch.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mosquera-Guerra, F., Barreto, S., Moreno-Niño, N. et al. Habitat connectivity of threatened ungulate species in a native savanna landscape of northern South America. Mamm Biol 104, 259–275 (2024). https://doi.org/10.1007/s42991-024-00404-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42991-024-00404-8