Abstract
Purpose
To investigate the feasibility of a municipality-based 16-week group-based HIFT-program (e.g. CrossFit) as a part of the physical rehabilitation of cancer survivors at different stages of cancer treatment.
Methods
Non-randomised clinical feasibility study. Younger adult patients (age 18–44 years) diagnosed with cancer who were referred to rehabilitation between August 2019 to December 2019 were eligible for inclusion. The group-based HIFT intervention was designed as a 16-week program with two sessions weekly (1.25 h each). The intervention program was not developed with pre-defined progression in terms of gradually added resistance, intensity, or volume during the 16 weeks period but the physiotherapist leading the sessions was trained in scalability. Feasibility was evaluated as retention, adherence, and accrual rates. Data on quality of life and cancer-related fatigue were measured EORTC QLQ-C-30 and evaluated using paired t-tests or Wilcoxon signed-rank test.
Results
Eighty-three percent of the eligible patients were included and initiated the HIFT program. However, 25% of the patients were not adherent to the intervention and only 34% of the patients were still adherent to the intervention after 4 months. Nonetheless, a significant improvement in cancer specific HRQoL was found from baseline [Mean = 53.4, 95%CI (47.6, 59.1)] to the end of the intervention [Mean = 66.3, 95%CI (60.8, 71.9)].
Conclusion
It is possible to recruit patients diagnosed with cancer to a municipality-based HIFT rehabilitation program, however, adherence to the intervention is found to be difficult for the majority of the patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
High Intensity Functional training (HIFT) is defined as a form of combined aerobic and resistance training that incorporates functional, multimodal movements, performed at relatively high intensity (relative to an individual’s ability), and designed to improve parameters of general physical fitness and performance [12]. In the last two decades HIFT has gained increasing attention in the fitness industry and in recent years also in research [40], especially due to the increased popularity of the HIFT program and worldwide fitness brand CrossFit© [12]. HIFT facilitates strong sense of community, continued participation, exercise enjoyment, satisfaction and intrinsic motivation among healthy participants [5, 15, 20, 32, 42]. HIFT is also associated with numerous physiological benefits in healthy adults including improvements in muscle flexibility and endurance [9], maximal strength [9, 11, 13, 33], physical work capacity [11], anaerobic capacity [11], aerobic capacity [27, 33], reduced resting heart rate [4], body composition and bone health [4, 13, 27, 33].
Systematic reviews highlight HIFT as a safe type of training with low injury rates that are similar to other types of aerobic and resistance training modalities [2, 16]. Recently, the HIFT methodology has drawn research attention for individuals with chronic conditions including cancer patients [3, 14, 29].Combined aerobic and resistance training is recommended for both healthy individuals and for patients diagnosed with cancer in the latest guidelines by the World Health Organization and the American College of Sports Medicine [7, 13]. Patients diagnosed with cancer experience a variety of ongoing physical and psychological symptoms associated with the treatment of cancer [7, 31]. Additionally, survivors of cancer have an elevated risk of comorbid conditions such as cardiovascular disease and diabetes [31]. Consequently, there is a growing emphasis on rehabilitation strategies to optimise health-related quality of life (HRQoL) and cancer-related fatigue (CRF) during and after cancer treatment [34]. Studies show that exercise interventions are safe and effective for patients diagnosed with cancer [21], improving cardiovascular fitness and strength [41]. Interventions designed as high-intensity interval training (HIIT) and moderate-intensity training interventions seems to produce similar positive effects on physical fitness and health-related outcomes [22, 26]. Thus, clinical guidelines worldwide recommend both aerobic and resistance training as an integral part of the rehabilitation of patients diagnosed with cancer [7, 18, 35].
However, despite the established recommendations and demonstrated effectiveness, reported adherence rates to exercise interventions and physical activity guidelines among cancer patients is often as low as a third of patients [25, 28, 30]. Barriers to adherence include symptoms of pain and fatigue as well as lack of enjoyment and motivation to exercise [28].
Consequently, the development of exercise interventions and strategies improving adherence remains a critical challenge in cancer rehabilitation settings. Effective interventions aimed at increasing motivation and enjoyment as well as treating symptoms hold the potential to improve adherence to exercise and decrease the risk of comorbid conditions including cardiovascular disease and diabetes [25]. The preliminary evidence showing that HIFT is safe [20] and associated with physiological and psychosocial benefits as well as continued participation [6, 15, 19, 32, 42], suggests its potential utility within an outpatient cancer rehabilitation setting. The findings from one study indeed indicate that a HIFT intervention is feasible for patients diagnosed with cancer as 80% of the eligible participants initiated participation, and of these 75% adhered to 5 weeks intervention [20]. However, the findings should be interpreted with caution as the study only included eight participants. Hence, the feasibility of HIFT interventions for cancer patients remains unknown.
Therefore, the primary aim of this study was to investigate the feasibility of a municipality-based 16-week group-based HIFT-program as a part of the physical rehabilitation of younger cancer survivors (18–44 years) at different stages of cancer treatment. Additionally, we investigated changes in HRQoL and CRF following the 16-week exercise intervention. Finally, we investigated the continuation of participation in any HIFT program, 3 months following completion of the exercise intervention.
Materials and Methods
Study Design
This study was a non-randomised clinical feasibility study. The manuscript is reported as recommended in the CONSORT checklist for reporting pilot or feasibility trials.
Setting
The Copenhagen Centre for Cancer and Health (CCCH) a municipality-based rehabilitation centre functioned as the non-hospital clinical facility site. Referral for rehabilitation at CCCH could come from various hospitals and discharging departments within the Capital Region of Denmark. Regulated by law, patients can only be referred to rehabilitation in the municipality if there is a medically justified need for rehabilitation, and furthermore, by the referring doctor is responsible for the content of the rehabilitation plan including any possible restrictions for rehabilitation (e.g. cancer-specific exercise contraindications).
Patients
Patients referred to municipality-based rehabilitation are upon referal divided into two groups defined by age younger adults (age 18–44 years) and adults (age 45 + years). In this study only the younger adults referred to CCCH between August 2019 to December 2019 were eligible for inclusion. All referred patients had an individual assessment with a physiotherapist upon referral that included goal setting with aim to increase long-term vision and motivation. All young adults were in the study period given the choice to participate in HIFT or the standard, group-based physical rehabilitation intervention. All cancer diagnoses and stages were included, as long as the patients were enrolled in rehabilitation at the CCCH. Hence, patients undergoing active cancer treatment, and patients who had completed active treatment as well as cancer survivors were considered eligible for inclusion. Patients were excluded if they did not have an email (to answer surveys) and if they were not able to read and understand Danish. Eligible patients received oral and written information about the study by a physiotherapist, and if willing to participate in the study, the patient signed an informed consent. Patients could without any reason given decline to participate in the study without further consequences for the present or future treatment and were offered usual care rehabilitation which includes an individualised assessment of rehabilitation needs, and as standard, a group-based physical rehabilitation intervention.
Intervention
The HIFT intervention, as defined by Feito et al. as “a training style or program that incorporates functional, multimodal movements, performed at a relatively high intensity, and designed to improve parameters of general physical fitness and performance” [13]. The designed intervention was based on the principles of CrossFit®. CrossFit is described as a strength and conditioning program that focuses on “constantly varying functional movements, performed at a relatively high intensity” [17]. This type of HIFT includes a variety of elements from gymnastics (e.g., floor, bar, or ring exercises), weightlifting (e.g., squats, cleans, snatches and presses with a barbell, dumbbell, or kettlebell), and cardiovascular training (e.g., running or rowing) [8]. Hence, this intervention is a pragmatic intervention where flexibility in the individual training response is accepted. Hence, no load control or ongoing measure of biochemical and physiological optics were registered.
Depending on the number of patients one or two (if more than 12 participants) physiotherapists trained to deliver HIFT lead the supervised group-based sessions. All sessions were conducted in the clinical setting at CCCH. As the intervention was designed to reflect the clinical setting patients were consecutively included in the open-ended cohort. The intervention was designed with 2 weekly exercise sessions for 16 weeks (supplementary file 1), and each HIFT session lasted for one hour and 15 min and included a warmup, a strength-focused workout, and an aerobic-focused workout. The exercise program was not developed with pre-defined progression in terms of gradually added resistance, intensity, or volume over the cause of 16 weeks. However, the physiotherapists leading the sessions was trained in scalability, and hence the difficulty of the exercises could be scaled (e.g. complexity, intensity, volume, load) to fit the individual skill and physical condition with the aim to ensure high intensity.
Outcomes
Participant characteristics including age, sex, cancer type, treatment status, body mass index, educational level, and smoking and employment status were obtained from the electronical patient journal. Data for measuring adherence, retention, and accrual rate were obtained from the electronical registration system at CCCH.
Adherence
As the intervention were planned as a 16-week program with two sessions weekly, a pragmatic adherence cut-off for the intervention were set to once weekly. The rationale for this cut-off is that muscle training stimuli once a weak is generally considered to maintain muscle mass [36]. Hence, a participant was categorised as adherent to the intervention if they participated in at least 16 sessions throughout the intervention period.
Retention
It was considered that the pragmatic design with a consecutive inclusion over time, holidays might cause smaller periods of non-adherence. Patients were categorised as still being adherent to the intervention after 16 weeks of intervention if they participated in at least one of the last four sessions (week 15 or 16).
Accrual Rate
Accrual rate was determined by the total number patients assessed for eligibility and patients enrolled in the study divided by the number of months recruitment occurred.
A baseline survey was sent to the patients prior to commencing the HIFT sessions. The baseline survey consisted of items regarding sociodemographic and lifestyle and the cancer specific health-related quality of life questionnaire, European Organisation for Research and Treatment of Cancer (EORTC QLQ-C-30). EORTC QLQ-C-30 includes five functional domains (physical, role, cognitive, emotional and social, where higher scores represent greater function or quality of life) and three symptom scales (fatigue, pain and nausea, where lower scores represent greater quality of life/lower symptom severity). EORTC QLQ-C-30 was chosen for its established reliability and validity with specific emphasis on use in cancer populations [1, 24]. Scoring was conducted in accordance with the official user manual [39], and a 10 point difference between assessments were considered as the minimal important difference. After the end of the intervention (16 weeks) the patients were fulfilled the survey again. To minimize non-response and loss to follow-up all patients received a survey reminder by email if the surveys were not fulfilled after 3 days.
Harms
All adverse events occurring after entry into the study were recorded. The participants were asked to report any serious adverse events (requiring hospitalization) or adverse events (all others) before every training session and in the survey at the end of the intervention period. Furthermore, withdrawals due to adverse events were registered.
Sample Size
Due to the exploratory nature of this study, no power calculation was conducted. Data collection methods.
Self-reported baseline data were collected via online survey sent to patients 1 week prior to beginning the supervised HIFT intervention. After the intervention period patients received another survey with the purpose of evaluating the feasibility of the intervention.
Statistical Methods
A statistical analysis plan was developed prior to evaluation of data.
Descriptive statistics were used to summarize patient characteristics including age, sex, cancer diagnosis and type of treatment. Data were summarized as means with standard deviations (SDs) and ranges or frequencies with percentages as appropriate. Feasibility were evaluated in relation to adherence, retention, and accrual rate, respectively and presented as frequencies and percentages. Data on HRQoL and cancer-related fatigue were evaluated using paired t-tests or Wilcoxon signed-rank test respectively of the distribution of data, and missing data were imputed using last observation carried forward. The proportion of patients who continued participation in HIFT at follow-up were presented as frequency and percentage. RStudio version 1.3.1093 was used for all analyses and illustrations. An alpha level of 0.05 was considered statistically significant.
Results
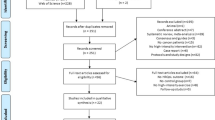
Thirty-six patients were assessed for eligibility. The full study flow diagram is presented in Fig. 1.
Thirty-two patients completed the baseline assessment. Participants were primarily women (78%), with a median age of 33, and 50% were diagnosed with breast cancer, and 60% of the included patients were in active medical treatment with either chemo- or radiation-therapy at the time of inclusion. A full list of patient characteristics is presented in Table 1.
Tables 2 and 3 respectively presents the findings for feasibility and effectiveness of the HIFT intervention. All included participants provided data in relation to feasibility, and 78% fulfilled the patient reported outcomes at the end of the intervention period.
No withdrawals due to adverse events were registered. No serious adverse events were registered. As expected, all participants experienced at least some degree of delayed onset muscle soreness, however, no other adverse events were registered Fig. 2.
Discussion
This study aimed to investigate the feasibility of a 16-week group-based HIFT-program for cancer patients at different stages of cancer treatment. Overall, our result shows that it is possible to recruit patients for the intervention and some patients can adhere to a HIFT-program for 16 weeks, however, most of the patients in this study did not adhere to the pre-defined criteria for adherence to intervention and only a third of all patients were still actively participating at the end of the intervention.
In the current study the majority of eligible participants were included. This is in contrast to many studies investigating a HIIT intervention were nearly half of the eligible patients declined to participate, and the primary reason for refusal was a lack of interest in exercise during chemotherapy [23]. However, similar to other studies involving exercise interventions and including cancer patients, adherence to the intervention was low. This is despite using a HIFT methodology to the exercise intervention which has previously in research been shown to facility enjoyment, motivation, sense of community and continued participation. Factors influencing attendance was not investigated in this study, however previous studies have established other barriers to adhering to exercise interventions including time constraints due to medical visits and social support from friends and family and travel distance to rehabilitation centre [30]. Furthermore, some studies have also found adherence to supervised exercise predicted by cancer disease stage and fewer symptoms as well as a higher physical fitness level [10, 28].
It should be noted that 60% of the participants in this present study were in active chemo- or radiation therapy at enrolment. However, it remains unknown whether this or other factors such as lack of interest or motivation towards the exercise intervention are driving forces behind non-adherence.
Although this study was designed with an exploratory aim, and hence not designed to show improvement in a specific outcome, our results showed significant improvements in HRQoL and CRF during the 16-week study period. Despite the low adherence rate in our study, our findings are in line with the body of evidence regarding the effectiveness of rehabilitation intervention on HRQoL and CRF for patients diagnosed with cancer [37].
Methodologically, we aimed for this study to provide an accurate reflection of the everyday clinical setting. This entailed that all referred patients had an individual assessment with a physiotherapist upon referral including functional goal setting with the aim to understand patients rehabilitation needs and motivation. By allowing this, selection bias can be of importance when interpreting the results of this study. Theoretical, one would expect that the individual assessment would lead to a convenience sample of experienced exercise patients who were highly motivated for the intervention. However, only 4 out of 36 (11%) eligible patients declined to participate in the intervention. This is a low non-participant rate when taking the form and extent (16 weeks program) of the HIFT into consideration. Contrary, the low non-participant rate might be a reason for the low adherence rates if the patients, for example, were not adequately informed of the content or the extent of the intervention. On the other hand, it is possible that the low attendance to the exercise intervention partly can be explained by a busy calendar with treatment and medical consultations at the hospital and days with side effects after treatment since 60% of the included patients were treated with either chemo- or radiation-therapy. A study conducted in the same rehabilitation centre found that the primary reasons for cancelling exercise sessions were hospitalization/appointments at the hospital, lack of motivation, or lack of time in a group lung cancer patients [38]. Low participation to exercise sessions in cancer rehabilitation is a condition and found that approximately 50% of the patients had an adherence to supervised group exercise under < 70% [38].
The findings of the present study offer promising implications for both clinical practice and future investigations. Rehabilitation programs may enhance patients' abilities to regain functional movement, promoting faster recovery and improved quality of life and HIFT intervention may be one alternative among others. Moreover, these results beckon further research avenues, encouraging in-depth exploration of HIFT's mechanisms, optimal protocols, and long-term effects.
Strength and Limitations
To our knowledge this is the first study presenting data on a study conducted in municipality-based clinical setting. The intervention was tested in a setting that functions as clinical practice, and hence should be an adequate reflection of the effectiveness of the intervention.
This study aimed to investigate the feasibility of a HIFT-program for a broad group of patients diagnosed with cancer and with few exclusion criteria. This is a strength in terms of generalizability on the other hand the limitation of this study. However, due to the limited sample size it was not possible to conduct meaningful subgroup analysis to test such hypothesis.
A strength of the present study is the acceptable response rate of 78% for the patient reported outcomes, especially taking into consideration the low adherence and retention rates. However, interpretation of the results on effectiveness of the intervention should be performed with caution as no power calculation was conducted due to the exploratory nature of this study. Furthermore, this study recruited a heterogeneous sample of patients with various cancer diagnosis and potentially higher adherence and retention rates in specific patient populations cannot be out-ruled.
The present study was designed to evaluating the feasibility of a pragmatic intervention. Hence, interpretation of the results regarding effectiveness of the intervention should be done carefully. The intervention was designed as a pragmatic HIFT intervention where flexibility in the individual training response was accepted using scalability. Hence, it was never designed as a HIIT intervention study with predefined intensity targets and rest periods. The lack of load control or ongoing measures of biochemical and physiological optics is a limitation of the study.
The patients were all residents in the municipality of Copenhagen at the time of referral, however, we did not obtain data in relation to how far away from the rehabilitation center the patients lived. Distance to the rehabilitation center is a known significant predictor for adherence to exercise-based intervention [30]. Therefore, it cannot be ruled out that long transportation time to the rehabilitation center could have been a possible barrier and a reason for the low adherence rates found in this study.
Conclusion
It is possible to recruit patients diagnosed with various cancer diagnoses to a HIFT rehabilitation program delivered in a municipality setting. However, adherence and retention to the intervention in a clinical setting is found to be difficult, and hence, the intervention seems not to be feasible for a heterogenous group of patients diagnosed with cancer in the current context. Though it was possible to find improvements in fatigue and HRQoL in a community-based cancer rehabilitation setting.
Data Availability
By contacting the corresponding author and upon reasonable request, the full anonymized data set can be made available.
References
Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85(5):365–76.
Ángel Rodríguez M, García-Calleja P, Terrados N, Crespo I, Del Valle M, Olmedillas H. Injury in CrossFit®: a systematic review of epidemiology and risk factors. Phys Sportsmed. 2022;50(1):3–10.
Bae M, Kasser SL. High intensity exercise training on functional outcomes in persons with multiple sclerosis: a systematic review. Mult Scler Relat Disord. 2023;75:104748.
Bechke E, Kliszczewicz B, Feito Y, Kelemen H, Nickerson B. Resting cardiac autonomic activity and body composition following a 16-week high-intensity functional training intervention in women: a pilot study. J Hum Sport Exerc. 2017;12(3):480–8. https://doi.org/10.14198/jhse.2017.123.12.
Bycura DK, Feito Y, Prather CC. Motivational factors in CrossFit® training participation. Health Behav Policy Rev. 2017;4:539–50.
Bycura D, Feito Y, Prather CC. Motivational factors in CrossFit® training participation. Health Behav Polic Rev. 2017;4:539.
Campbell KL, Winters-Stone KM, Wiskemann J, May AM, Schwartz AL, Courneya KS. Exercise Guidelines for cancer survivors: consensus statement from international multidisciplinary roundtable. Med Sci Sports Exerc. 2019;51(11):2375–90.
Claudino JG, Gabbett TJ, Bourgeois F, de Souza HS, Miranda RC, Mezêncio B. CrossFit overview: systematic review and meta-analysis. Sports Med—Open. 2018;4(1):11.
Cosgrove SJ, Crawford DA, Heinrich KM. Multiple fitness improvements found after 6-months of high intensity functional training. Sports Basel Switz. 2019;7(9):203.
Courneya KS, Segal RJ, Gelmon K, Reid RD, Mackey JR, Friedenreich CM. Predictors of supervised exercise adherence during breast cancer chemotherapy. Med Sci Sports Exerc. 2008;40(6):1180–7.
Crawford DA, Drake NB, Carper MJ, DeBlauw J, Heinrich KM. Are changes in physical work capacity induced by high-intensity functional training related to changes in associated physiologic measures? Sports Basel Switz. 2018;6(2):26.
Feito Y, Heinrich KM, Butcher SJ, Poston WSC. High-intensity functional training (HIFT): definition and research implications for improved fitness. Sports Basel Switz. 2018;6(3):76.
Feito Y, Hoffstetter W, Serafini P, Mangine G. Changes in body composition, bone metabolism, strength, and skill-specific performance resulting from 16-weeks of HIFT. PLoS ONE. 2018;13(6): e0198324.
Feito Y, Patel P, Sal Redondo A, Heinrich KM. Effects of eight weeks of high intensity functional training on glucose control and body composition among overweight and obese adults. Sports Basel Switz. 2019;7(2):51.
Fisher J, Sales A, Carlson L, Steele J. A comparison of the motivational factors between CrossFit participants and other resistance exercise modalities: a pilot study. J Sports Med Phys Fitness. 2017;57(9):1227–34.
Gean RP, Martin RD, Cassat M, Mears SC. A systematic review and meta-analysis of injury in Crossfit. J Surg Orthop Adv. 2020;29(1):26–30.
Glassman G. What is CrossFit? Crossfit J. 2004. https://journal.crossfit.com/article/what-is-fitness.
Hayes SC, Spence RR, Galvão DA, Newton RU. Australian association for exercise and sport science position stand: optimising cancer outcomes through exercise. J Sci Med Sport. 2009;12(4):428–34.
Heinrich KM, Patel PM, O’Neal JL, Heinrich BS. High-intensity compared to moderate-intensity training for exercise initiation, enjoyment, adherence, and intentions: an intervention study. BMC Public Health. 2014;14:789.
Heinrich KM, Becker C, Carlisle T, Gilmore K, Hauser J, Frye J. High-intensity functional training improves functional movement and body composition among cancer survivors: a pilot study. Eur J Cancer Care (Engl). 2015;24(6):812–7.
Herranz-Gómez A, Cuenca-Martínez F, Suso-Martí L, Varangot-Reille C, Calatayud J, Blanco-Díaz M. Effectiveness of HIIT in patients with cancer or cancer survivors: an umbrella and mapping review with meta-meta-analysis. Scand J Med Sci Sports. 2022;32(11):1522–49.
Lavín-Pérez AM, Collado-Mateo D, Mayo X, Liguori G, Humphreys L, Copeland RJ. Effects of high-intensity training on the quality of life of cancer patients and survivors: a systematic review with meta-analysis. Sci Rep. 2021;11(1):15089.
Lee K, Kang I, Mack WJ, Mortimer J, Sattler F, Salem G. Feasibility of high intensity interval training in patients with breast cancer undergoing anthracycline chemotherapy: a randomized pilot trial. BMC Cancer. 2019;19(1):653.
Luckett T, King MT, Butow PN, Oguchi M, Rankin N, Price MA. Choosing between the EORTC QLQ-C30 and FACT-G for measuring health-related quality of life in cancer clinical research: issues, evidence and recommendations. Ann Oncol Off J Eur Soc Med Oncol. 2011;22(10):2179–90. https://doi.org/10.1093/annonc/mdq721.
Mazzoni AS, Brooke HL, Berntsen S, Nordin K, Demmelmaier I. Exercise adherence and effect of self-regulatory behavior change techniques in patients undergoing curative cancer treatment: secondary analysis from the phys-can randomized controlled trial. Integr Cancer Ther. 2020;19:1534735420946834.
Mugele H, Freitag N, Wilhelmi J, Yang Y, Cheng S, Bloch W. High-intensity interval training in the therapy and aftercare of cancer patients: a systematic review with meta-analysis. J Cancer Surviv Res Pract. 2019;13(2):205–23.
Murawska-Cialowicz E, Wojna J, Zuwala-Jagiello J. Crossfit training changes brain-derived neurotrophic factor and irisin levels at rest, after wingate and progressive tests, and improves aerobic capacity and body composition of young physically active men and women. J Physiol Pharmacol Off J Pol Physiol Soc. 2015;66(6):811–21.
Ng AH, Ngo-Huang A, Vidal M, Reyes-Garcia A, Liu DD, Williams JL. Exercise barriers and adherence to recommendations in patients with cancer. JCO Oncol Pract. 2021;17(7):e972–81.
Nieuwoudt S, Fealy CE, Foucher JA, Scelsi AR, Malin SK, Pagadala M. Functional high-intensity training improves pancreatic β-cell function in adults with type 2 diabetes. Am J Physiol Endocrinol Metab. 2017;313(3):E314–20.
Ormel HL, van der Schoot GGF, Sluiter WJ, Jalving M, Gietema JA, Walenkamp AME. Predictors of adherence to exercise interventions during and after cancer treatment: a systematic review. Psychooncology. 2018;27(3):713–24.
Pachman DR, Barton DL, Swetz KM, Loprinzi CL. Troublesome symptoms in cancer survivors: fatigue, insomnia, neuropathy, and pain. J Clin Oncol. 2012;30(30):3687–96.
Pickett AC, Goldsmith A, Damon Z, Walker M. The influence of sense of community on the perceived value of physical activity: a cross-context analysis. Leis Sci. 2016;38(3):199–214.
Posnakidis G, Aphamis G, Giannaki CD, Mougios V, Aristotelous P, Samoutis G. High-intensity functional training improves cardiorespiratory fitness and neuromuscular performance without inflammation or muscle damage. J Strength Cond Res. 2022;36(3):615–23.
Richards M, Corner J, Maher J. The national cancer survivorship initiative: new and emerging evidence on the ongoing needs of cancer survivors. Br J Cancer. 2011;105(Suppl 1):S1-4.
Rock CL, Doyle C, Demark-Wahnefried W, Meyerhardt J, Courneya KS, Schwartz AL. Nutrition and physical activity guidelines for cancer survivors. CA Cancer J Clin. 2012;62(4):243–74.
Schoenfeld BJ, Ogborn D, Krieger JW. Effects of resistance training frequency on measures of muscle hypertrophy: a systematic review and meta-analysis. Sports Med Auckl NZ. 2016;46(11):1689–97.
Sleight A, Gerber LH, Marshall TF, Livinski A, Alfano CM, Harrington S. Systematic review of functional outcomes in cancer rehabilitation. Arch Phys Med Rehabil. 2022;103(9):1807–26.
Sommer MS, Trier K, Vibe-Petersen J, Missel M, Christensen M, Larsen KR. Perioperative rehabilitation in operable lung cancer patients (PROLUCA): a feasibility study. Integr Cancer Ther. 2016;15(4):455–66.
Sprangers MAG, Bonnetain F. EORTC QLQ-C30. I. In: Michalos AC, Redaktør, editors. Encyclopedia of quality of life and well-being research. Dordrecht: Springer, Netherlands; 2014. pp. 1933–5. https://doi.org/10.1007/978-94-007-0753-5_901.
Thompson WR. Worldwide survey of fitness trends. ACSMs Health Fitness J. 2023;26(1):9–18.
Toohey K, Pumpa K, McKune A, Cooke J, Semple S. High-intensity exercise interventions in cancer survivors: a systematic review exploring the impact on health outcomes. J Cancer Res Clin Oncol. 2018;144(1):1–12.
Whiteman-Sandland J, Hawkins J, Clayton D. The role of social capital and community belongingness for exercise adherence: an exploratory study of the CrossFit gym model. J Health Psychol. 2018;23(12):1545–56. https://doi.org/10.1177/1359105316664132.
Acknowledgements
The authors would like to thank the patients and staff at the CCCH who participated in the study.
Funding
Open access funding provided by National Hospital. This research received no external funding.
Author information
Authors and Affiliations
Contributions
Conceptualization, ALH, and RTL. Methodology, JC, RTL. Formal analysis, JC, ALH, and RTL.; investigation, MSS and RD. Writing original draft preparation, JC, ALH, and RTL Writing review and editing, JC, ALH, MSS, RD and RTL. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflicts of Interest
ALH and RTL are both part time employees at Arca Aps which is a HIFT fitness brand and chain of fitness centres. Arca Aps has supplied some additional exercise equipment for the intervention and offered the five supervising physiotherapists from CCCH to attend on their Trainers Course to develop and improve the physiotherapists HIFT-specific instruction skills. ALH and RTL have not received any funds neither from Arca Aps nor CCCH. Further, Arca Aps and CCCH had no influence on the design of the study; in the collection, analyses or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Ethical Approval
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Regional Scientific Ethics Committee of Capital Region in Denmark [H-19010858]. Handling of data were approved by the Danish Data Protection Agency [514-0306/19-3000]. The study was registered at ClinicalTrials.gov [NCT04001127] before recruitment of the first participant.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Christensen, J., Hessner, A.L., Sommer, M.S. et al. High Intensity Functional Training for Patients Diagnosed with Cancer: A Study Evaluating the Feasibility of a Pragmatic Intervention. J. of SCI. IN SPORT AND EXERCISE (2024). https://doi.org/10.1007/s42978-024-00275-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42978-024-00275-y