Abstract
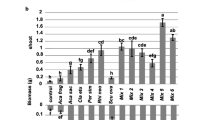
Arbuscular mycorrhizal fungi (AMF) are obligate symbionts that affect plant growth. However, the magnitude and direction of the influences of AMF are variable, and it is yet unclear about the factors contributing to the variability. In this quest, the study evaluated whether the differences are, in part, due to variation among AMF in terms of their phylogeny. In a pot-based experiment, two genotypes of Ocimum tenuiflorum (holy basil) were inoculated with four AMF having different phylogenetic relatedness, namely—Acaulospora mellea, Entrophospora etunicata, Diversispora spurca, and Rhizoglomus intraradices. Effects of the AMF on morphological, physiological, and yield-related parameters of holy basil were evaluated. Though their overall effects were positive, difference in the magnitude of their influences was evident. To elucidate the difference in the influences, hierarchical cluster analysis of the plant responses was carried out. Two distinct clusters were formed, wherein responses induced by members of Diversisporales (A. mellea and D. spurca) formed one cluster, while that of R. intraradices and E. etunicata constituted the second cluster. Similarity or differences in the effect of AMF on holy basil, observed in the cluster analysis, can be based on their phylogeny as related AMF (A. mellea and D. spurca) had similar effect on holy basil as compared with their distant relatives. E. etunicata and R. intraradices resulted in better performance of holy basil, over Diversisporales in both genotypes and this could be related with their percent colonization and higher nutrient uptake capacity. The results suggest functional conservatism among the four AM fungal species based on their influence on the morphological, physiological, and yield-related parameters of holy basil.




Similar content being viewed by others
Abbreviations
- AM:
-
Arbuscular mycorrhiza
- AMF:
-
Arbuscular mycorrhizal fungi
- ANOVA:
-
Analysis of variance
- ATP:
-
Adenine triphosphate
- Chl:
-
Chlorophyll
- Ci:
-
Intercellular CO2 concentration;
- DMSO:
-
Dimethyl sulfoxide
- E:
-
Transpiration rate
- EO:
-
Essential oil
- GC–MS:
-
Gas chromatography–mass spectrometry
- Gs:
-
Stomatal conductance
- IARI:
-
Indian Agricultural Research Institute
- ITS:
-
Internal transcribed region
- M:
-
Mycorrhizal
- MANOVA:
-
Multivariate analysis of variance
- MR:
-
Mycorrhizal responsiveness
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- NBPGR:
-
National Bureau of Plant and Genetic Resources
- NCBI:
-
National Center for Biotechnology Information
- NM:
-
Non-mycorrhizal
- AL:
-
Phenylalanine ammonia lyase
- Pn:
-
Photosynthetic rate
- RDW:
-
Root dry weight
- RFW:
-
Root fresh weight
- RL:
-
Root length
- SDW:
-
Shoot dry weight
- SFW:
-
Shoot fresh weight
- SL:
-
Shoot length
References
Adams RP (2007) Identification of essential oil components by gas chromatography/mass spectrometry. Allured Publishing Corporation, Carol Stream, USA
Adeyemi NO, Atayese MO, Sakariyawo OS, Azeez JO, Ridwan M (2021) Arbuscular mycorrhizal fungi species differentially regulate plant growth, phosphorus uptake and stress tolerance of soybean in lead contaminated soil. J Plant Nutr 44:1633–1648. https://doi.org/10.1080/01904167.2021.1871748
Aguilera P, Ortiz N, Becerra N, Turrini A, Gaínza-Cortés F, Silva-Flores P et al (2022) Application of arbuscular mycorrhizal fungi in vineyards: water and biotic stress under a climate change scenario: new challenge for Chilean grapevine crop. Front Microbiol 13:826571–826583. https://doi.org/10.3389/fmicb.2022.826571
Allen SE (1989) Chemical analysis of ecological materials. Blackwell Scientific, Oxford
Amiri R, Nikbakht A, Rahimmalek M, Hosseini H (2017) Variation in the essential oil composition, antioxidant capacity, and physiological characteristics of Pelargonium graveolens L. inoculated with two species of mycorrhizal fungi under water deficit conditions. J Plant Growth Regul 36:502–515. https://doi.org/10.1007/s00344-016-9659-1
Arnon DI (1949) Copper enzymes in isolated polyphenol oxidase in Beta vulgaris. Plant Physiol 24:1–15. https://doi.org/10.1104/pp.24.1.1
Arpanahi AA, Feizian M, Mehdipourian G, Khojasteh DN (2020) Arbuscular mycorrhizal fungi inoculation improve essential oil and physiological parameters and nutritional values of Thymus daenensis Celak and Thymus vulgaris L. under normal and drought stress conditions. Eur J Soil Biol 100:103–217. https://doi.org/10.1016/j.ejsobi.2020.103217
Baon JB, Smith SE, Alston AM (1993) Mycorrhizal responses of barley cultivars differing in P efficiency. Plant Soil 157:97–105. https://doi.org/10.1007/BF00038752
Bennett AE, Groten K (2022) The costs and benefits of plant–arbuscular mycorrhizal fungal interactions. Ann Rev Plant Biol 73:649–672. https://doi.org/10.1146/annurev-arplant-102820-124504
Błaszkowski J, Sanchez-Garcia M, Niezgoda P, Zubek S, Fernandez F, Vila A et al (2022) A new order, Entrophosporales, and three new Entrophospora species in Glomeromycota. Front Microbiol 13:4615–4645. https://doi.org/10.3389/fmicb.2022.962856
Bouskout M, Bourhia M, Al Feddy MN, Dounas H, Salamatullah AM, Soufan W et al (2022) Mycorrhizal fungi inoculation improves Capparis spinosa’s yield, nutrient uptake and photosynthetic efficiency under water deficit. Agronomy 12:149–160. https://doi.org/10.3390/agronomy12010149
Brito I, Goss MJ, Alho L, Brígido C, van Tuinen D, Félix MR, Carvalho M (2019) Agronomic management of AMF functional diversity to overcome biotic and abiotic stresses-the role of plant sequence and intact extraradical mycelium. Fungal Ecol 40:72–81. https://doi.org/10.1016/j.funeco.2018.06.001
Bulgarelli RG, Marcos FCC, Ribeiro RV, de Andrade SAL (2017) Mycorrhizae enhance nitrogen fixation and photosynthesis in phosphorus-starved soybean (Glycine max L. Merrill). Environ Exp Bot 140:26–33. https://doi.org/10.1016/j.envexpbot.2017.05.015
Burducea M, Zheljazkov VD, Dincheva I, Lobiuc A, Teliban GC, Stoleru V et al (2018) Fertilization modifies the essential oil and physiology of basil varieties. Ind Crops Prod 121:282–293. https://doi.org/10.1016/j.indcrop.2018.05.021
Cesaro P, Massa N, Cantamessa S, Todeschini V, Bona E, Berta G et al (2020) Tomato responses to Funneliformis mosseae during the early stages of arbuscular mycorrhizal symbiosis. Mycorrhiza 30:601–610. https://doi.org/10.1007/s00572-020-00973-9
Chandrasekaran M (2022) Arbuscular mycorrhizal fungi mediated enhanced biomass, root morphological traits and nutrient uptake under drought stress: a meta-analysis. J Fungi 8:660–672. https://doi.org/10.3390/jof8070660
Crossay T, Majorel C, Redecker D, Gensous S, Medevielle V, Durrieu G et al (2019) Is a mixture of arbuscular mycorrhizal fungi better for plant growth than single-species inoculants? Mycorrhiza 29:325–339. https://doi.org/10.1007/s00572-019-00898-y
de Souza TAF, de Andrade LA, Freitas H, da Silva SA (2018) Biological invasion influences the outcome of plant-soil feedback in the invasive plant species from the Brazilian semi-arid. Microb Ecol 76:102–112. https://doi.org/10.1007/s00248-017-0999-6
Diagne N, Ngom M, Djighaly PI, Fall D, Hocher V, Svistoonoff S (2020) Roles of arbuscular mycorrhizal fungi on plant growth and performance: importance in biotic and abiotic stressed regulation. Diversity 12:370–395. https://doi.org/10.3390/d12100370
Dodd JC, Boddington CL, Rodriguez A, Gonzalez-Chavez C, Mansur I (2000) Mycelium of arbuscular mycorrhizal fungi (AMF) from different genera: form, function and detection. Plant Soil 226:131–151. https://doi.org/10.1023/A:1026574828169
Ferrol N, Azcón-Aguilar C, Pérez-Tienda J (2019) Arbuscular mycorrhizas as key players in sustainable plant phosphorus acquisition: an overview on the mechanisms involved. Plant Sci 280:441–447. https://doi.org/10.1016/j.plantsci.2018.11.011
Giovannetti M, Mosse B (1980) An evaluation of techniques for measuring vesicular arbuscular mycorrhizal infection in roots. New Phytol 84:489–500
Golubkina N, Logvinenko L, Novitsky M, Zamana S, Sokolov S, Molchanova A et al (2020) Yield, essential oil and quality performances of Artemisia dracunculus, Hyssopus officinalis and Lavandula angustifolia as affected by arbuscular mycorrhizal fungi under organic management. Plants 9:375–387. https://doi.org/10.3390/plants9030375
Gupta S, Thokchom SD, Kapoor R (2021) Arbuscular mycorrhiza improves photosynthesis and restores alteration in sugar metabolism in Triticum aestivum L. grown in arsenic contaminated Soil. Front Plant Sci 12:334–347. https://doi.org/10.3389/fpls.2021.640379
Gupta S, Thokchom SD, Koul M, Kapoor R (2022) Arbuscular Mycorrhiza mediated mineral biofortification and arsenic toxicity mitigation in Triticum aestivum L. Plant Stress 5:100086–100102. https://doi.org/10.1016/j.stress.2022.100086
Hart MM, Reader RJ (2002) Does percent root length colonization and soil hyphal length reflect the extent of colonization for all AMF? Mycorrhiza 12:297–301. https://doi.org/10.1007/s00572-002-0186-5
Heydari S, Pirzad A (2021) Efficiency of Funneliformis mosseae and Thiobacillus sp. on the secondary metabolites (essential oil, seed oil and mucilage) of Lallemantia iberica under salinity stress. J Hortic Sci Biotechnol 96:249–259. https://doi.org/10.1080/14620316.2020.1833764
Hiscox JD, Israelstam GF (1979) A method for the extraction of chlorophyll from leaf tissue without maceration. Canad J Bot 57:1332–1334. https://doi.org/10.1139/b79-163
Huang GM, Zou YN, Wu QS, Xu YJ, Kuča K (2020) Mycorrhizal roles in plant growth, gas exchange, root morphology, and nutrient uptake of walnuts. Plant Soil Environ 66:295–302. https://doi.org/10.17221/240/2020-PSE
Jakobsen I, Abbott LK, Robson AD (1992) External hyphae of vesicular-arbuscular mycorrhizal fungi associated with Trifolium subterraneum L. 1. Spread of hyphae and phosphorus inflow into roots. New Phytol 120:371–380. https://doi.org/10.1111/j.1469-8137.1992.tb01077.x
Jerbi M, Labidi S, Laruelle F, Tisserant B, Dalpé Y, Lounès-Hadj Sahraoui A et al (2022) Contribution of native and exotic arbuscular mycorrhizal fungi in improving the physiological and biochemical response of hulless barley (Hordeum vulgare ssp. nudum L.) to drought. J Soil Sci Plant Nutr 22:2187–2204. https://doi.org/10.1007/s42729-022-00802-2
Kaur S, Suseela V (2020) Unraveling arbuscular mycorrhiza-induced changes in plant primary and secondary metabolome. Metabolites 10:335–365. https://doi.org/10.3390/metabo10080335
Khalediyan N, Weisany W, Schenk PM (2021) Arbuscular mycorrhizae and rhizobacteria improve growth, nutritional status and essential oil production in Ocimum basilicum and Satureja hortensis. Ind Crops Prod 160:113–163. https://doi.org/10.1016/j.indcrop.2020.113163
Kheyri Z, Moghaddam M, Farhadi N (2022) Inoculation efficiency of different mycorrhizal species on growth, nutrient uptake, and antioxidant capacity of Calendula officinalis L.: a Comparative Study. J Soil Sci Plant Nutr 22:1160–1172. https://doi.org/10.1007/s42729-021-00721-8
Krüger M, Krüger C, Walker C, Stockinger H, Schüßler A (2012) Phylogenetic reference data for systematics and phylotaxonomy of arbuscular mycorrhizal fungi from phylum to species level. New Phytol 193:970–984. https://doi.org/10.1111/j.1469-8137.2011.03962.x
Le Pioufle O, Ganoudi M, Calonne-Salmon M, Ben Dhaou F, Declerck S (2019) Rhizophagus irregularis MUCL 41833 improves phosphorus uptake and water use efficiency in maize plants during recovery from drought stress. Front Plant Sci 10:897–910. https://doi.org/10.3389/fpls.2019.00897
Moukarzel R, Ridgway HJ, Waller L, Guerin-Laguette A, Cripps-Guazzone N, Jones EE (2022) Soil arbuscular mycorrhizal fungal communities differentially affect growth and nutrient uptake by grapevine rootstocks. Microb Ecol 1:15–30. https://doi.org/10.1007/s00248-022-02160-z
Ostadi A, Javanmard A, Amani Machiani M, Sadeghpour A, Maggi F, Nouraein M et al (2022) Co-application of TiO2 nanoparticles and arbuscular mycorrhizal fungi improves essential oil quantity and quality of sage (Salvia officinalis L.) in drought stress conditions. Plants 11:1659–1681. https://doi.org/10.3390/plants11131659
Pan J, Huang C, Peng F, Zhang W, Luo J, Ma S, Xue X (2020) Effect of arbuscular mycorrhizal fungi (AMF) and plant growth-promoting bacteria (PGPR) inoculations on Elaeagnus angustifolia L. in saline soil. Appl Sci 10:945–967. https://doi.org/10.3390/app10030945
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res 29:45–45. https://doi.org/10.1093/nar/29.9.e45
Phillips JM, Hayman DS (1970) Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans Br Mycol Soc 55:158-IN18. https://doi.org/10.1016/S0007-1536(70)80110-3
Porter SS, Bantay R, Friel CA, Garoutte A, Gdanetz K, Ibarreta K et al (2020) Beneficial microbes ameliorate abiotic and biotic sources of stress on plants. Funct Ecol 34:2075–2086. https://doi.org/10.1111/1365-2435.13499
Powell JR, Parrent JL, Hart MM, Klironomos JN, Rillig MC, Maherali H (2009) Phylogenetic trait conservatism and the evolution of functional trade-offs in arbuscular mycorrhizal fungi. Proc Royal Soc B 276:4237–4245. https://doi.org/10.1098/rspb.2009.1015
Qu L, Wang M, Biere A (2023) Effects of arbuscular mycorrhizal fungi on plant growth and herbivore infestation depend on availability of soil water and nutrients. Front Plant Sci 14:167–180. https://doi.org/10.3389/fpls.2023.1101932
Rasouli F, Amini T, Asadi M, Hassanpouraghdam MB, Aazami MA, Ercisli S et al (2022) Growth and antioxidant responses of lettuce (Lactuca sativa L.) to arbuscular mycorrhiza inoculation and seaweed extract foliar application. Agronomy 12:401–416. https://doi.org/10.3390/agronomy12020401
Saboor A, Ali MA, Danish S, Ahmed N, Fahad S, Datta R et al (2021) Effect of arbuscular mycorrhizal fungi on the physiological functioning of maize under zinc-deficient soils. Sci Rep 11:18468. https://doi.org/10.1038/s41598-021-97742-1
Säle V, Palenzuela J, Azcón-Aguilar C, Sánchez-Castro I, da Silva GA, Seitz B, Oehl F (2021) Ancient lineages of arbuscular mycorrhizal fungi provide little plant benefit. Mycorrhiza 31:559–576. https://doi.org/10.1007/s00572-021-01042-5
Säle V, Sieverding E, Oehl F (2022) Growth responses of three European weeds on different AMF species during early development. Plants 11:2020–2033. https://doi.org/10.3390/plants11152020
Schoch CL, Ciufo S, Domrachev M, Hotton CL, Kannan S, Khovanskaya R et al (2020) NCBI Taxonomy: a comprehensive update on curation, resources and tools. Database 2020:2020–2062. https://doi.org/10.1093/database/baaa062
Shao YD, Zhang DJ, Hu XC, Wu QS, Jiang CJ, Xia TJ et al (2018) Mycorrhiza-induced changes in root growth and nutrient absorption of tea plants. Plant Soil Environ 64:283–289. https://doi.org/10.17221/126/2018-PSE
Thokchom SD, Gupta S, Kapoor R (2020) Arbuscular mycorrhiza augments essential oil composition and antioxidant properties of Ocimum tenuiflorum L.–a popular green tea additive. Ind Crops Prod 153:112418–112430. https://doi.org/10.1016/j.indcrop.2020.112418
Thonar C, Schnepf A, Frossard E, Roose T, Jansa J (2011) Traits related to differences in function among three arbuscular mycorrhizal fungi. Plant Soil 339:231–245. https://doi.org/10.1007/s11104-010-0571-3
Velásquez A, Valenzuela M, Carvajal M, Fiaschi G, Avio L, Giovannetti M et al (2020) The arbuscular mycorrhizal fungus Funneliformis mosseae induces changes and increases the concentration of volatile organic compounds in Vitis vinifera cv Sangiovese leaf tissue. Plant Physiol Biochem 155:437–443. https://doi.org/10.1016/j.plaphy.2020.06.048
Weber SE, Diez JM, Andrews LV, Goulden ML, Aronson EL, Allen MF (2019) Responses of arbuscular mycorrhizal fungi to multiple coinciding global change drivers. Fungal Ecol 40:62–71. https://doi.org/10.1016/j.funeco.2018.11.008
Yan T, Xue J, Zhou Z, Wu Y (2021) Impacts of biochar-based fertilization on soil arbuscular mycorrhizal fungal community structure in a karst mountainous area. Environ Sci Pollut Res 28:66420–66434. https://doi.org/10.1007/s11356-021-15499-6
Acknowledgments
SDT and SG are grateful to Council of Scientific and Industrial Research for providing Senior Research fellowships.
Funding
This work was financially supported by Institution of Eminence (IoE/2022-23/FRP), University of Delhi, India.
Author information
Authors and Affiliations
Contributions
The work was designed and planned by RK and SDT. SDT executed the experiments and analyzed the results. RK, SDT, and SG jointly wrote the manuscript and collectively reviewed the manuscript and approved it.
Corresponding author
Ethics declarations
Competing Interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
Supplementary Fig. 1: A phylogenetic tree showing the positions of the AMF species (Acaulospora mellea, Entrophospora etunicata, Diversispora spurca, Rhizoglomus intraradices), based on Neighbor-Joining of the concatenated nuclear small subunit full (SSUfull) - 5.8S-large subunit (LSU) rDNA strict consensus sequence of some arbuscular mycorrhizal fungal species representative of each family of the Glomeromycota. The sequences used for the construction of the tree were obtained from Krüger et al. (2012). The tree was rooted using the sequence of Paraglomus.
ESM 2
Supplementary Fig. 2: Histochemical staining of the roots colonized by arbuscular mycorrhizal fungi; A. Acaulospora mellea, B. Entrophospora etunicata, C. Diversispora spurca, and D. Rhizoglomus intraradices, showing the mycorrhizal structures (mycorrhizal hyphae and arbuscules).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Thokchom, S.D., Gupta, S. & Kapoor, R. Response of Ocimum tenuiflorum L. (Holy Basil) to Inoculation of Four Arbuscular Mycorrhizal Fungal Species Shows Functional Coherence with Respect to Their Phylogeny. J Soil Sci Plant Nutr 23, 3407–3420 (2023). https://doi.org/10.1007/s42729-023-01259-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42729-023-01259-7