Abstract
It is well accepted that the phenology of insects whose life activities are closely related to temperature is changing in response to global climate warming. To investigate the impacts of climate warming on the phenology of Bactrocera dorsalis (Hendel) across large temporal and spatial scales, this study collected historical data on the occurrence and population dynamic of this pest in China, and systematically explored its phenological responses. The results showed a delayed trend for the dates of first occurrence, end occurrence, population initial growth, and population peak of B. dorsalis in China during 40 years, and the changes of the latter two phenological parameters were significant. The mean temperature in spring and summer were the key climatic factors affecting the occurrence and population growth of B. dorsalis in China, respectively. Moreover, the B. dorsalis data in eastern, southern, central, and southwestern China showed spatial heterogeneity of phenological responses to climate warming at a regional scale. B. dorsalis phenology and their changing patterns with climate warming varied by geographical location. This study provides valuable information for future monitoring, prediction, and prevention of the oriental fruit fly in the context of climate warming.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Climate change, the most debated issue of time, threatens many organisms. The Intergovernmental Panel on Climate Change (IPCC) reported that the average global temperature had increased by 0.72 °C with a mean increase rate of 0.012 °C per year from 1880 to 2012 (IPCC 2014). The global temperature rise significantly impacts individual development, population dynamics, phenology, and the geographical range of organisms, including insects, which has recently attracted much attention (Wu et al. 2020). Insects, a large group of poikilotherms, are highly sensitive to climate warming. The life activities of insects, including growth and development, survival, reproduction, and mobility, are closely associated with ambient temperature, thus, are inevitably influenced by climate warming (Meglitsch 1972; Bale et al. 2002; Meineke et al. 2014). Thus, understanding the proximate mechanisms resulting in these shifts becomes increasingly urgent now that accumulating evidence exhibits that climate change can significantly impact individual survival and the population development of species.
Many studies have demonstrated that climate warming accelerates insects’ growth and development rates, resulting in a shorter life cycle and earlier occurrences (Harrington et al. 2001; Robinet and Roques 2010; Raza et al. 2014). It has been demonstrated that increasing temperature could accelerate the reproductive cycle of insects, such as butterflies, dragonflies, damselflies, flies, aphids, bees, and beetles, to produce more generations (Gordo and Sanz 2005; Harrington et al. 2001). For instance, rising temperatures halved Dendroctonus rufipennis Kirby’s breeding time in northwestern North America (Berg et al. 2006). Based on the data of the British Butterfly Monitoring Schemes (BMS), from 1976 to 1998, the first occurrence of most British butterflies advanced and the period of their mean flight was prolonged due to the increasing temperatures in spring and summer in central England. Similar results were observed for the butterflies in Spain (Stefanescu et al. 2010) and California, USA (Forister and Shapiro 2003), and 37 Odonata species in the Netherlands (Dingemanse and Kalkman 2008). In China, three aphid pests, Myzus persicae Sulzer, Aphis gossypii Glover, and Sitobion avenae Fabricius are occurring earlier, and their migration seasons have become longer due to climate warming (Wu et al. 2020).
Bactrocera dorsalis (Hendel), commonly known as the oriental fruit fly, is a highly polyphagous species, which attacks more than 300 host plants of commercially grown vegetables and fruits, and is thus regarded as a major pest worldwide (Clarke et al. 2005; Vargas et al. 2009). First recorded in Kaohsiung of Taiwan, China, in 1912 (Drew and Hancock 1994), B. dorsalis spread beyond tropical Asia over the following years, due to their adaptability to diverse climatic conditions (Clarke et al. 2005; Aketarawong et al. 2007). This pest has recently caused serious economic losses in more than 10 Chinese provinces. These losses in southern China amount to around three billion dollars annually (Ji et al. 2016).
Severe crop damage caused by B. dorsalis prompted research on this pest's biology and population ecology in sub-tropical and tropical regions (Uchida et al. 2007; Piñero et al. 2009; Vargas et al. 2008a, b). The phenology and population dynamics of this pest have been extensively investigated in eastern, southern, southwestern, and central China, such as Fujian (Zheng 2013), Jiangxi (Li et al. 2019), Guangdong (Lv et al. 2008), Yunnan (Chen and Ye 2007; Ye and Liu 2005), and Hubei Provinces (Han et al. 2011). These researches demonstrated that the seasonal occurrence of B. dorsalis was determined by factors, such as temperature, rainfall, and host plant availability. Particularly, the temperature was considered as a crucial factor affecting the occurrence of this pest (Vargas et al. 1996; Michel et al. 2021; Manrakhan et al. 2022). Bactrocera dorsalis development and reproduction permitting temperature range is 15–34 °C, and the optimal temperature range for development is 20–28 °C. The threshold temperature ranges for eggs, larvae, and pupae are 11–12 °C, 9–11 °C, and 9–11 °C, respectively. Many adults and larvae die when the temperature is > 34 °C or < 15 °C (Chen and Ye 2007). Moreover, the low winter temperatures restrict the expansion and establishment of B. dorsalis in the newly invaded regions (Stephens et al. 2007). Han et al. (2011) overwintering experiments in Wuhan, Hubei province, showed that only a small proportion of B. dorsalis pupae might survive the cold winter and bridge the gap between winter and spring, giving a small initial number of viable adults early in the season. In the context of climate change, it is not yet known how climate warm affects population dynamics and the phenology of fruit flies. It is important to investigate the effects of climate warming on the occurrence of fruit flies over a long historical period, which can provide valuable information for forecasting and comprehensive control of this pest.
To limit B. dorsalis damage, a series of area-wide Integrated Pest Management (IPM) were implemented in recent years. This IPM program is based on biological and sustainable technologies, including (1) trapping adults using methyl eugenol (ME) and other lures (Uchida et al. 2007; Vargas et al. 2008b; Gu et al. 2018; Lin et al. 2022), (2) protein bait sprays (Wang et al. 2021), (3) promptly removal of infested fruits, (4) elimination of overwintering pupae by turning soil, (4) bagging fruit to prevent fly infestation (Mau et al. 2007; Vargas et al. 2008a), (5) application of parasitoids (Cai et al. 2017, 2020, 2022; Yang et al. 2018), (6) mass release of sterile insect (Cai et al. 2018; Lin et al. 2020; Zhang et al. 2021). However, whether climate warming would influence the timeline of when and how to perform these effective control methods across different geographical scales remained unknown. Therefore, exploring this issue with this notorious pest from different geographical regions is urgently needed.
With vast territory, varied topography, and various climate types, China is also significantly influenced by global warming, with a temperature growth rate of 0.026 ℃/a over the past 70 years, which is significantly higher than that in the world or the northern hemisphere (Climate Change Center of China Meteorological Administration 2022). However, the long-term impacts of climate warming on fruit fly pests remained largely unknown due to the absence of long-term population monitoring data. To address this research need, historical data extracted from the literature may provide insight into this issue (Tian et al. 2010; Hu et al. 2019; Matsuda et al. 2018). Thus, our research thoroughly collected historical data on the seasonal occurrence and population dynamics of B. dorsalis, rated as the TOP 10 invasive pests in China (Wan et al. 2017). Based on the collected historical data, the effects of long-term climate change on B. dorsalis were determined by analyzing changes in several phenological parameters.
Materials & methods
Phenological data
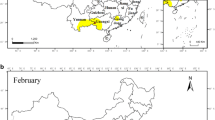
Bactrocera dorsalis phenological data were extracted and compiled from historical literature, most of which originated from the CNKI database (http://www.cnki.net), the most extensive and comprehensive database of Chinese periodicals (Tu et al. 2017), and Web of Science (http://www.webofknowledge.com/). Firstly, the common and Latin names of this fruit fly specie were used as subject words and searched by subject word retrieval. Afterward, the related literature from January 1980 to March 2022 recording the occurrences, geographical distributions, and population dynamics of B. dorsalis in diverse areas of China was consulted. The specific time and geographic information on life cycle parameters were extracted and a database was constructed. All data collection sites documented in the B. dorsalis collected literature were georeferenced into geographical maps utilizing ArcGIS 10.2 (ESRI, Inc., Redlands, CA, USA).
The collected data were organized based on the four most frequently recorded life cycle parameters, the first occurrence date, the end occurrence date, the population peak date, and the population initial growth date. In this study, the first occurrence date was defined as the time when adult flies were first detected in the fields, the end occurrence date was the time when no adult flies were detected in the field, the population peak date as the time when the trapping amount of adult flies reached the highest levels in the field, and the time when the population of adult flies began to grow rapidly as the population initially growth data. For each parameter, “change of days” was quantified by calculating the differences (number of days) between the dates of first occurrence, end occurrence, population peak or population increase records in our dataset, and January first of that year. In some literature, time descriptions of these parameters were vague, such as “the beginning of the month”, “the middle of the month”, “the end of the month”, “the first (middle or last) ten days”. Thus, such time information without specific dates was specifically approximated. For instance, the description about the beginning of a month was set as the first day of that month while the end of a month was set as the last day of that month, and the first, middle, and last ten days of a month were set as 5th, 15th, and 25th of that month, respectively.
Meteorological data
The annual and seasonal mean temperatures of each province in China from 1980 to 2020 were obtained. Temperature records were downloaded from Chinese meteorological websites (http://data.cma.cn/). According to the collected literature, the annual mean temperature of southern China, eastern China, central China, southwestern China, and whole China where had phenological records of B. dorsalis were calculated and analyzed the overall change rates by a simple linear regression method to clarify the trend and temperature change about the past 40 years, respectively.
Statistical analysis
All analyses were conducted using SPSS for Windows version 20.0 (SPSS Inc., Chicago, IL, USA). The phenological responses of B. dorsalis were analyzed by plotting changes of the phenological parameters described above (the first occurrence, the end occurrence, the population increase, and the population peak). The change in days for each parameter was taken as the Y‐axis, and the occurrence year was taken as the X‐axis. Linear regression analysis was used to construct regression equations and reveal the trends of the four parameters in the time series. A normal test for all estimated data sets was performed, showing that all data sets were under normal distributions. Pearson correlation analysis was performed to verify the correlation between the phenological parameters of B. dorsalis and seasonal mean temperature.
Results
Phenological records of B. dorsalis in China
In March 2022, 150 pieces of literature that documented the phenological records of B. dorsalis in China were found in the CNKI and Web of Science databases (see supplemental files Table S1), involving 4 regions namely, southern China (Guangdong, Guangxi, and Hainan provinces), southwestern China (Sichuan province, Guizhou province, and Yunnan province), central China (Hubei, Hunan, and Henan provinces), eastern China (Shanghai city, and the Jiangsu, Jiangxi Fujian, Anhui, Zhejiang, and Shandong provinces) (Fig. 1). The collected data on B. dorsalis were mainly concentrated in southwestern, southern, and eastern China. These provide data support for investigating the impacts of climate warming to B. dorsalis at different geographical scales. The most phenological records of B. dorsalis have been reported from Yunnan province, followed by Guangxi and Fujian provinces, as they are the major fruit-producing areas where climatic conditions are also favorable for the survival, growth, and reproduction of this pest (Table 1).
Temperature changes in B. dorsalis-infested areas in China over time
In the past 40 years, the annual mean temperature (AMT) in eastern (Jiangsu, Jiangxi, Fujian, Anhui, Shanghai, Zhejiang, and Shandong), southern (Guangdong, Guangxi, and Hainan), central (Hubei, Hunan, and Henan), and southwestern (Sichuan, Guizhou, and Yunnan) China exhibited a significant upward trend with fluctuations, with the temperature rising rates of 0.0485 ± SE 0.0044 ℃ year−1, 0.0254 ± SE 0.0050 ℃ year−1, 0.0384 ± SE 0.0044 ℃ year−1 and 0.0364 ± SE 0.0040 ℃ year−1, as revealed by linear regression calculations (all p < 0.0001, Fig. 2).
Between 1980 and 2020, the spring (March–May, SPMT), summer (June–August, SUMT), autumn (September–November, AUMT), and winter (December-February, WMT) mean temperatures in the areas infested by B. dorsalis in China had also increased over the years (Fig. 3). The linear regression analysis indicated that SPMT, SUMT, AUMT, and WMT in eastern China increased about 0.0677 ± SE0.0082 ℃ year−1, 0.0383 ± SE0.0062 ℃ year−1, 0.0449 ± SE0.0082 ℃ year−1, 0.0453 ± SE0.0122 ℃ year−1, respectively (all p < 0.001). Similar increasing trends of seasonal temperature parameters could be found in southern, central, and southwestern China. Still, the WMT in southern China did not show a significant rising trend (p = 0.0629).
The temporal trend of occurrence
Based on the collected long-term historical data, the first appearance of B. dorsalis in southern and central China followed an insignificantly downward tendency over the years, indicating that the first occurrence times of oriental fruit flies moved earlier by -1.74 ± SE1.1 days year−1 (p = 0.1198) and -1.822 ± 1.977 days year−1 (p = 0.3684), respectively (Fig. 4bc). However, the first occurrence date of B. dorsalis in eastern China and southwestern China were significantly delayed by 3.73 ± SE1.062 days year−1 (p = 0.007) and 2.138 ± SE0.7702 days year−1(p = 0.0066), respectively (Fig. 4ad). For China as a whole, the scatter plots of the first occurrence change of B. dorsalis showed delayed tendency in the time series by 0.8197 ± SE0.5087 days per year (p = 0.1083, Fig. 4e).
For the end occurrence, the scatter plots of B. dorsalis in eastern and southern China showed a climbing trend over time, predicting that the end occurrence date was slightly delayed by 1.088 ± SE0.965 days year−1 (p = 0.2629, Fig. 5a) and 0.666 ± SE0.5151 days year−1 (p = 0.2029, Fig. 5b) respectively. However, this parameter of oriental fruit flies in central and southwestern China advanced by 4.984 ± SE2.006 days year−1 (p = 0.0274, Fig. 5c) and 0.3162 ± SE0.383 days year−1 (p = 0.4123, Fig. 5d), respectively. For China as a whole, the last occurrence times of B. dorsalis showed a slight upward shift, with a change rate of 0.4612 ± SE0.3657 days each year (p = 0.2087, Fig. 5e).
The temporal trend of population initial growth and peak
The current data collection revealed that the changes in population initial growth and peak of oriental fruit flies in central China exhibited significantly advanced tendencies by 13.47 ± SE2.707 days year−1 (p = 0.0025, Fig. 6c) and 3.563 ± SE1.599 days year−1 (p = 0.0370, Fig. 7c). In contrast, B. dorsalis population dynamics parameters in eastern, southern, and southwestern China were delayed. The population time change rates initially increased in the field were 8.52 ± SE1.152 days year−1 (p < 0.0001, Fig. 6a), 1.816 ± SE0.5468 days year−1 (p = 0.0013, Fig. 6b) and 0.3516 ± SE0.5528 days year−1(p = 0.5261, Fig. 6d), respectively. While the date of their population reached the maximum in the regions delayed by 1.356 ± SE0.6899 days year−1 (p = 0.0509, Fig. 7a), 1.033 ± SE0.3647 days year−1 (p = 0.0051, Fig. 7b) and 2.833 ± SE0.4133 days year−1 (p < 0.0001, Fig. 7d), respectively. For China as a whole, the initial increase date and peak times of the B. dorsalis population were both significantly delayed over the years and with the change rates of 1.755 ± SE0.3923 days each year (p < 0.0001, Fig. 6e) and 1.618 ± SE0.2525 days each year (p < 0.0001, Fig. 7e), respectively.
The change of initially growth times of B. dorsalis population in eastern China (a), southern China (b), central China (c), southwestern China (d) and whole China (e) for the period 1990–2020. The solid lines represent the trends of the end of occurrence and the dots indicate different phenological records
Field phenology of B. dorsalis in relation to temperature
As a result, the date of the population peak of oriental fruit flies in eastern China was significantly negative correlated with the spring mean temperature (Pearson correlation coefficient = -0.4732, p = 0.0302, Fig. 8a). In southwestern China, there was a significant positive correlation between summer mean temperature and population peak date (Pearson correlation coefficient = 0.4332, p = 0.0240, Fig. 8d). For China as a whole, the date of the end occurrence and initial growth of B. dorsalis population were significantly positive correlated with spring mean temperature (Pearson correlation coefficient = 0.4556, p = 0.0380, Fig. 8e) and summer mean temperature (Pearson correlation coefficient = 0.3917, p = 0.0433, Fig. 8e).
The correlations between the phenological parameters of B. dorsalis and annual and seasonal mean temperatures in eastern China (a), southern China (b), central China (c), southwestern China (d) and whole China (e). The asterisk indicated the Pearson correlations were significant at the levels of p < 0.05
Discussion
The analyses support several studies suggesting that, during four decades (1980–2020), the annual mean temperatures of the four regions infested by B. dorsalis in China have shown rising trends, as well as the seasonal mean temperatures of these regions. The population initial growth date and population peak date of B. dorsalis in China have been significantly delayed due to climate warming. The results of Pearson correlations analysis indicated that the initial growth of the B. dorsalis population had a significantly positive correlation with the summer mean temperature, implying that the initial growth of B. dorsalis population was similar to the summer mean temperature rising. The frequent appearance of extreme heat in the summer season may postpone the emergence of fruit flies to avoid exposure to high temperatures, leading to a delay in the initial growth date. With delayed population initial growth, there was a concurrent deferment of population peak occurrence. It is widely accepted that increased ambient temperature would generally accelerate the completion of the insect reproductive cycle to produce more generations in different insect species, ultimately leading to enhanced potential for insect pest outbreaks (Raza et al. 2014). Thus, B. dorsalis produced more generations due to rising temperature, resulting in more serious generation overlap, possibly explaining the delayed reaching of its maximum population size.
We also found that the first and end occurrence dates of B. dorsalis in China exhibited a slight delay. These findings did not agree with the general prediction that climate warming would lead to the advanced occurrence of insects (Harrington et al. 2001; Robinet and Roques 2010; Raza et al. 2014). Although the environmental temperature was considered as a crucial variable affecting the occurrence of B. dorsalis (Vargas et al. 1996; Su et al. 2020), other factors not included in this study such as atmospheric humility, rainfall, sunlight hours, and host plant availability. may also influence the phenology of fruit flies. For example, in Mengzi city, Yunnan province, the population peak date of B. dorsalis in different orchards was quite different due to differences in the phenology of host fruits. In peach orchards, B. dorsalis first occurred in mid-April, and in mid-March in jujube orchards, while on early-April in loquat orchards (Fang et al. 2017). Moreover, soil moisture, closely related to precipitation and rainfall frequency, was supposed to be an important factor influencing the pupation and emergence of B. dorsalis (Duyck et al. 2010). Previous research found it difficult for adults to emerge when soil moisture is more than 30% (Alyokhin et al. 2001). Thus, the observed changes in phenology appear to be caused neither by a general elevation in the overall temperature over time nor by a general response of fruit flies to temperature, but instead by the combined effects of changes in temperature regimes and other environmental variables that influence larval development and/or adult survival.
Additionally, our data in eastern, southern, central, and southwestern China show spatial heterogeneity of phenological responses to climate warming at a regional scale, as was found in previous research concerning three aphid species in Xinjiang, China (Wu et al. 2020). Firstly, the occurrence times of oriental fruit flies varied in different regions in China due to the existing differences in the annual mean temperature in different regions. For instance, the average first appearance date of fruit flies in southern China was around mid-April, while the average first occurrence date in eastern, central, and southwestern China were around late May, mid-late June, and early May, respectively. The data for the other three phenological parameters in different regions in China also suggested geographical variation at a regional scale. Secondly, the long-term trends of population occurrence and dynamic of B. dorsalis showed different patterns between four regions in China. For example, the first occurrence of B. dorsalis in southern China was slightly ahead of time and their end occurrence date slightly delayed, contrary to that of B. dorsalis in southwest China (Table S2). It is possible that this variation may be a real reflection of spatial heterogeneity of the effect of climate warming.
Considering that the phenology of fruit flies is temperature-dependent, there is a possibility that “noise” such as unusual values for the field phenological parameters may be introduced in a long‐term data set due to weather anomalies in a specific year. For example, in 2008, the first and end occurrence dates of B. dorsalis in central China (data mainly originated from Wuhan city, Hubei province) were notably later than that of other years of this region (Han et al. 2011). According to literature records, low temperatures and heavy snow storms happened at the beginning of 2008 in Wuhan, which may postpone the occurrence of oriental fruit flies. Similarly, frost caused by a strong decreased temperature occurred in the Fujian province of eastern China in 2009. Thus, the first appearance date of B. dorsalis was deferred to mid-late August (Lin 2014).
Besides changing the phenology of B. dorsalis, a warming climate may facilitate range expansion of this pest and help them invade and colonize new territories (de Villiers et al. 2016). Previous research projected B. dorsalis to be capable of invading new territories and establishing persistent populations throughout the tropics and subtropics under predicted future climatic conditions (Stephens et al. 2007; de Villiers et al. 2016). It is worth noting that the time of B. dorsalis damage in China has been changed due to rising temperatures, which may result in a mismatch of phenological synchronicity between fruit flies and host plants or nature enemies (Visser 2008, 2017). If the pest emerges early, but the fruit of host plants does not germinate simultaneously, B. dorsalis females were finding less developed fruits during their oviposition period and this may have influenced their chances of reproduction (Gordo and Sanz 2005). Alternatively, B. dorsalis may shift to new plants for oviposition to maintain the population, expanding the host plant range. Moreover, the shift in fruit flies' occurrence may also influence the original phenological synchronization between pests and parasitoids, helping fruit flies avoid parasitoids' parasitism (Meineke et al. 2014). These are challenges to detecting and suppressing fruit flies in the future.
The long-term effect of climate warming on insects is a pressing issue for science and application. Due to the insufficiency of the long-term dataset, evidence concerning this issue remains limited (Hu et al. 2019; Matsuda et al. 2018). Though collecting a 40 years dataset on B. dorsalis population dynamics, this study reveals the long-term effect of climate warming on this notorious pest. It is worth noting that some uncertainty could happen in quantifying time data due to the vagueness of time information extracted from some literature. Thus, careful and thorough data collection, standardization, and careful analyses are essential to minimize the influence of uncertainty of historical literature data. In the future, a more detailed examination of the long-term effect of climate warming on fruit flies and other pests and the construction of a standard detection network are urgently required in China.
Conclusion
This research uncovered the long‐term impacts of climate warming on the phenology of B. dorsalis in China across large temporal and spatial scales by collecting and analyzing historical data. The results showed that the annual and seasonal mean temperatures of four regions (eastern, southern, central, and southwestern China) infested by B. dorsalis have increasing trends at different change rates over the past 40 years. Under climate warming, the first and end occurrence date of B. dorsalis in China became later, and the initial growth and peak times of the B. dorsalis population were significantly delayed. The phenological date of B. dorsalis in eastern, southern, central, and southwestern China suggests spatial heterogeneity of the effect of climate warming at a regional scale. This research provides practical implications for understanding the effects of climate warming on insect pests, and theoretical guidance for future fruit fly pests' prediction and control.
Data availability
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
References
Aketarawong N, Bonizzoni M, Thanaphum S, Gomulski LM, Gasperi G, Malacrida AR, Gugliemino CR (2007) Inferences on the population structure and colonization process of the invasive oriental fruit fly, Bactrocera dorsalis (Hendel). Mol Ecol 16(17):3522–3532. https://doi.org/10.1111/j.1365-294X.2007.03409.x
Alyokhin AV, Mille C, Messing RH (2001) Selection of pupation habitats by oriental fruit fly larvae in the laboratory. J Insect Behav 14(1):57–67. https://doi.org/10.1023/A:1007849629409
Bale JS, Masters GJ, Hodkinson ID, Awmack C, Bezemer TM, Brown VK, Butterfield J, Buse A, Coulson JC, Farrar J, Good JEG, Harrington R, Hartley S, Jones TH, Lindroth RL, Press MC, Symrnioudis L, Watt AD, Whittaker JB (2002) Herbivory in global climate change research: Direct effect of rising temperature on insect herbivores. Glob Change Biol 8(1):1–16. https://doi.org/10.1046/j.1365-2486.2002.00451.x
Berg EE, Henry JD, Fastie CL, De Volder AD, Matsuoka SM (2006) Spruce beetle outbreaks on the Kenai Peninsula, Alaska, and Kluane National Park and Reserve, Yukon Territory: Relationship to summer temperatures and regional differences in disturbance regimes. For Ecol Manage 227(3):219–232. https://doi.org/10.1016/j.foreco.2006.02.038
Cai PM, Gu XH, Yao MY, Zhang HH, Huang J, Idrees A, Ji QE, Chen JC, Yang JQ (2017) The optimal age and radiation dose for Bactrocera dorsalis (Hendel) (Diptera: Tephritidae) eggs as hosts for mass-reared Fopius arisanus (Sonan) (Hymenoptera: Braconidae). Biol Control 108:89–97. https://doi.org/10.1016/j.biocontrol.2017.02.006
Cai PM, Hong JF, Wang C, Yang YC, Yi CD, Chen JH, Ji QE (2018) Effects of Co-60 radiation on the activities of three main antioxidant enzymes in Bactrocera dorsalis (Hendel) (Diptera: Tephritidae). J Asia-Pac Entomol 21:345–351. https://doi.org/10.1016/j.aspen.2018.01.006
Cai PM, Peng YW, Song YZ, Lin J, Nie CP, Li YY, Ji QE (2022) Is Fopius arisanus (Hymenoptera: Braconidae) a suitable biological control agent of Bactrocera dorsalis (Diptera: Tephritidae) in multi-crop orchards? Int J Pest Manag (online) 10(1080/09670874):2151055
Cai PM, Song YZ, Huo D, Lin J, Zhang HM, Zhang ZH, Xiao CM, Huang FM, Ji QE (2020) Chemical cues induced from fly-oviposition mediate the host-seeking behaviour of Fopius arisanus (Hymenoptera: Braconidae), an effective egg parasitoid of Bactrocera dorsalis (Diptera: Tephritidae), within a tritrophic context. Insects 11:231. https://doi.org/10.3390/insects11040231
Chen P, Ye H (2007) Population dynamics of Bactrocera dorsalis (Diptera: Tephritidae) and analysis of factors influencing populations in Baoshanba, Yunan, China. Entomol Sci 10:141–147. https://doi.org/10.1111/j.1479-8298.2007.00208.x
Clarke AR, Armstrong KF, Carmichael AE, Milne JR, Raghu S, Roderick GK, Yeates DK (2005) Invasive phytophagous pests arising through a recent tropical evolutionary radiation: the Bactrocera dorsalis complex of tropical fruit flies. Annu Rev Entomol 50(1):293. https://doi.org/10.1146/annurev.ento.50.071803.130428
Climate Change Center of China Meteorological Administration (2022) China Climate Change Blue Book (2022). Beijing: Science Press
de Villiers M, Hattingh V, Kriticos DJ, Brunel S, Vayssières JF, Sinzogan A, Billah MK, Mohamed SA, Mwatawala M, Abdelgader H, Salah FE, De MM (2016) The potential distribution of Bactrocera dorsalis: Considering phenology and irrigation patterns. Bull Entomol Res 106:19–33. https://doi.org/10.1017/S0007485315000693
Dingemanse NJ, Kalkman VJ (2008) Changing temperature regimes have advanced the phenology of Odonata in the Netherlands. Ecol Entomol 33(3):394–402. https://doi.org/10.1111/j.1365-2311.2007.00982.x
Drew R, Hancock D (1994) The Bactrocera dorsalis complex of fruit flies (Diptera: Tephritidae: Dacinae) in Asia. Bull Entomol Res 2(S2):1–68. https://doi.org/10.1017/S1367426900000278
Duyck PF, David P, Quilici S (2010) Climatic niche partitioning following successive invasions by fruit flies in La Réunion. J Anim Ecol 75(2):518–526. https://doi.org/10.1111/j.1365-2656.2006.01072.x
Fang XJ, Yang ZH, Zhang JL, Zhu WL, Zhang WH, Yue R, Jiang XL, Wu H, Chen GH, Tao M (2017) The characteristics of oviposition and population dynamics of Bactrocera dorsalis in different fruit orchards in Mengzi. J Yunnan Agric Univ (Nat Sci) 32(2):212–217. https://doi.org/10.16211/j.issn.1004-390x(n).2017.02.004
Forister ML, Shapiro AM (2003) Climatic trends and advancing spring flight of butterflies in lowland California. Glob Change Biol 9(7):1130–1135. https://doi.org/10.1046/j.1365-2486.2003.00643.x
Gordo O, Sanz JJ (2005) Phenology and climate change: A long-term study in a Mediterranean locality. Oecologia 146(3):484–495. https://doi.org/10.1007/s00442-005-0240-z
Gu XH, Cai PM, Yang YC, Yang QY, Yao MY, Idrees A, Ji QE, Yang JQ, Chen JH (2018) The response of four braconid parasitoid species to methyl eugenol: optimization of a biocontrol tactic to suppress Bactrocera dorsalis. Biol Control 122:101–108. https://doi.org/10.1016/j.biocontrol.2018.04.002
Han P, Xu W, Niu CY, Dong YC, Zhu JQ, Desneux N (2011) Population dynamics, phenology, and overwintering of Bactrocera dorsalis (Diptera: Tephritidae) in Hubei Province, China. J Pest Sci 84(3):289–295. https://doi.org/10.1007/s10340-011-0363-4
Harrington R, Fleming RA, Woiwod IP (2001) Climate change impacts on insect management and conservation in temperate regions: can they be predicted? Agric Entomol 3(4):233–240. https://doi.org/10.1046/j.1461-9555.2001.00120.x
Hu G, Lu MH, Reynolds DR, Wang HK, Xiao C, Liu WC, Zhu F, Wu XW, Xia F, Xie MC (2019) Long-term seasonal forecasting of a major migrant insect pest: The brown planthopper in the Lower Yangtze River Valley. J Pest Sci 92:417–428. https://doi.org/10.1007/s10340-018-1022-9
IPCC. Climate Change (2014) Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Core Writing Team, Pachauri, R.K., Meyer, L.A., Eds.; IPCC: Geneva, Switzerland, 151
Ji QE, Bi K, Chen JH (2016) Response of egg-pupal parasitoid Fopius arisanus (Sonan) to infochemicals from the host eggs’ surface of Bactrocera dorsalis (Hendel). J Asia-Pac Entomol 19:1151–1157. https://doi.org/10.1016/j.aspen.2016.10.005
Li XZ, Yang H, Wang T, Wang J, Wei H (2019) Life history and adult dynamics of Bactrocera dorsalis in the citrus orchard of Nanchang, a subtropical area from China: implications for a control timeline. ScienceAsia 45(3):212. https://doi.org/10.2306/scienceasia1513-1874.2019.45.212
Lin LJ (2014) Test on the dynamics of Bactrocera dorsalis Hendel in Averrhoa carambola L.orchards of Yunxiao County and its control. Subtrop Agric Res 10(1):23–26
Lin J, Yamada H, Lu NF, Ao GF, Yuan WW, Liu XX, Cai PM, Zheng ML, Yang JQ, Ji QE (2020) Quantifification and impact of cold storage and heat exposure on mass rearing program of Bactrocera dorsalis (Diptera:Tephritidae) genetic sexing strain. Insects 11:821. https://doi.org/10.3390/insects11110821
Lin J, Hao XX, Yue GQ, Yang DQ, Lu NF, Cai PM, Ao GF, Ji QE (2022) Efficacy of wax-based bait stations for controlling Bactrocera dorsalis (Diptera: Tephritidae). Pest Manag Sci 78(8):3576–3586. https://doi.org/10.1002/ps.7000
Lv X, Han SC, Xu JL, Hang H, Wu H, Ou JF, Sun L (2008) Population dynamics of Bactrocera dorsalis (Hendel) in Guangzhou, Guangdong Province, with analysis of the climate factors. Acta Ecol Sin 28(4):1850–1856. https://doi.org/10.3724/SP.J.1141.2008.00438
Manrakhan A, Daneel JH, Stephen PR, Hattingh V (2022) Cold tolerance of immature stages of Ceratitis capitata and Bactrocera dorsalis (Diptera: Tephritidae). J Econ Entomol toab263. https://doi.org/10.1093/jee/toab263
Matsuda N, Tanaka K, Watari Y, Shintani Y, Goto SG, Nisimura T, Lzumi Y, Numata H (2018) Northward expansion of the bivoltine life cycle of the cricket over the last four decades. Glob Change Biol 24(12):5622–5628. https://doi.org/10.1111/gcb.14436
Mau RFL, Jang EB, Vargas RI (2007) The Hawaii fruit fly area-wide fruit fly pest management programme: influence of partnership and a good education programme. Springer, Netherlands
Meglitsch PA (1972) Invertebrate Zoology, 2nd ed.: Oxford: Oxford University Press
Meineke EK, Dunn RR, Frank SD (2014) Early pest development and loss of biological control are associated with urban warming. Biol Let 10(11):20140586. https://doi.org/10.1098/rsbl.2014.0586
Michel ADK, Fiaboe KKM, Kekeunou S, Nanga SN, Kuate AF, Tonnang HEZ, Gnanvossou D, Hanna R (2021) Temperature-based phenology model to predict the development, survival, and reproduction of the oriental fruit fly Bactrocera dorsalis. J Therm Biol 97:102877. https://doi.org/10.1016/j.jtherbio.2021.102877
Piñero JC, Mau RFL, Vargas RI (2009) Managing oriental fruit fly (Diptera: Tephritidae), with spinosad-based protein bait sprays and sanitation in papaya orchards in Hawaii. J Econ Entomol 3:1123–1132. https://doi.org/10.1603/029.102.0334
Raza MM, Khan MA, Arshad M, Sagheer M, Sattar Z, Shafi J, Haq EU, Ali A, Aslam U, Mushtaq A (2014) Impact of global warming on insects. Arch Phytopathol Plant Prot 48(1):84–94. https://doi.org/10.1080/03235408.2014.882132
Robinet C, Roques A (2010) Direct impacts of recent climate warming on insect populations. Integr Zool 5(2):132–142. https://doi.org/10.1111/j.1749-4877.2010.00196.x
Stefanescu C, Penuelas J, Filella I (2010) Effects of climatic change on the phenology of butterflies in the northwest Mediterranean Basin. Glob Change Biol 9(10):1494–2150. https://doi.org/10.1046/j.1365-2486.2003.00682.x
Stephens AEA, Kriticos DJ, Leriche A (2007) The current and future potential geographical distribution of the oriental fruit fly, Bactrocera dorsalis (Diptera: Tephritidae). Bull Entomol Res 97(4):369–378. https://doi.org/10.1017/S0007485307005044
Su BK, Park JJ, Kim DS (2020) CLIMEX simulated predictions of the potential distribution of Bactrocera dorsalis (Hendel) (Diptera: Tephritidae) considering the northern boundary: With special emphasis on Jeju, Korea. J Asia-Pac Entomol 23(3):797–808. https://doi.org/10.1016/j.aspen.2020.07.006
Tian H, Stige LC, Cazelles B, Kausrud KL, Svarverud R, Stenseth NC, Zhang ZB (2010) Reconstruction of a 1910-y-long locust series reveals consistent associations with climate fluctuations in China. Proc Natl Acad Sci 108(35):14521–14526. https://doi.org/10.1073/pnas.1100189108
Tu HZ, Nie J, Zha WX (2017) An analysis on Pakistan study in China based on the CNKI database. Indian Ocean Econ Polit Rev (03):104–140. https://doi.org/10.16717/j.cnki.53-1227/f.2017.03.007
Uchida GK, Mackey BE, Mclnnis DO, Vargas RI (2007) Attraction of Bactrocera dorsalis (Diptera: Tephritidae) and nontarget insects to methyl eugenol bucket traps with different preservative fluids on Oahu Island, Hawaiian Islands. J Econ Entomol 3:723–729. https://doi.org/10.1603/0022-0493(2007)100[723:AOBDDT]2.0.CO;2
Vargas RI, Walsh WA, Jang EB, Armstrong JW, Kanehisa DT (1996) Survival and development of immature stages of four Hawaiian fruit flies (Diptera: Tephritidae) reared at five constant temperatures. Ann Entomol Soc Am 1:64–69. https://doi.org/10.1093/aesa/89.1.64
Vargas RI, Mau RFL, Jang EB, Faust RM, Wong L (2008) The Hawaii fruit fly area-wide pest management program. In: Koul O, Cuperus GW, Elliott NC (eds) Areawide IPM: theory to implementation. CABI Books, London, pp 300–325
Vargas RI, Stark JD, Hertlein M, Neto AM, Coler R, Piñero JC (2008) Evaluation of SPLAT with spinosad and methyl eugenol or cue-lure for ‘“attract-and-kill”’ of oriental and melon fruit flies (Diptera: Tephritidae) in Hawaii. J Econ Entomol 101(3):759–768. https://doi.org/10.1093/jee/101.3.759
Vargas RI, Piñero JC, Mau RFL, Stark JD, Hertlein M, Mafra-Neto A, Coler R, Getchell A (2009) Attraction and mortality of oriental fruit flies to SPLAT-MAT-methyl engenol with spinosad. Entomol Exp Appl 131(3):286–293. https://doi.org/10.1111/j.1570-7458.2009.00853.x
Visser ME (2008) Keeping up with a warming world; assessing the rate of adaptation to climate change. Proc Royal Soc B: Biol Sci 275(1635):649–659. https://doi.org/10.1098/rspb.2007.0997
Visser ME (2017) Phenological shifts in animals under contemporary climate change. In: Reference Module in Life Sciences. Elsevier: Amsterdam, Netherlands.
Wan F, Jiang M, Zhan A (2017) Biological Invasions and Its Management in China. Springer, Netherlands
Wang B, Huang ZF, Yang DQ, Ji QE, Cai PM (2021) Improvement of protein bait produced from beer yeast waste for controlling Bactrocera dorsalis (Diptera: Tephritidae) in China. J Asia-Pac Entomol 24:573–579. https://doi.org/10.1016/j.aspen.2021.04.017
Wu YX, Li JJ, Liu HH, Qiao GX, Huang XL (2020) Investigating the impact of climate warming on phenology of aphid pests in China using long-term historical data. Insects 11(3):167. https://doi.org/10.3390/insects11030167
Yang JQ, Cai PM, Chen J, Zhang HH, Wang C, Xiang HJ, Wu J, Yang YC, Chen JH, Ji QE, Song DB (2018) Interspecific competition between Fopius arisanus and Psyttalia incisi (Hymenoptera: Braconidae), parasitoids of Bactrocera dorsalis (Diptera: Tephritidae). Biol Control 121:183–189. https://doi.org/10.1016/j.biocontrol.2018.02.003
Ye H, Liu JH (2005) Population dynamics of the oriental fruit fly, Bactrocera dorsalis (Diptera: Tephritidae) in the Kunming area, southwestern China. Insect Sci 12(05):387–392. https://doi.org/10.1111/j.1005-295X.2005.00048.x
Zhang QW, Cai PM, Wang B, Liu XX, Lin J, Hua RH, Zhang HH, Yi CD, Song XS, Ji QE (2021) Manipulation of gut symbionts for improving the sterile insect technique: Quality parameters of Bactrocera dorsalis (Diptera: Tephritidae) genetic sexing strain males after feeding on bacteria-enriched diets. J Econ Entomol 114:560–570. https://doi.org/10.1093/jee/toaa294
Zheng SN (2013) Population dynamics and density of Bactrocera dorsalis (Hendel) in different habitats. Acta Ecol Sin 33(24):7699–7706. https://doi.org/10.5846/stxb201304060610
Acknowledgements
We are very grateful to Mr. Huameng Zhang for providing some literature, Mr. Fanhua Meng for preparing GIS figures, and especially to Prof. Xiaolei Huang from the College of Plant Protection, Fujian Agriculture and Forestry University for guiding us in collecting meteorological data.
Funding
This research was funded by the Special Program for Supporting Industry of Wuyi University (2021XJFWCY01), the Education and Scientific Research Project for Young and Middle-aged Teachers in Fujian Province (JAT220382), Advanced Talents Introduction Project of Wuyi University (YJ201910), Innovation and Entrepreneurship Training Program for College Students at Provincial Level (S202210397072), Fujian Natural Science Foundation Youth Program (2020J05216), Scientific Research Team Jointly Constructed By Teachers and Students of Wuyi University (2020-SSTD-006), Provincial Natural Science Foundation of Fujian (2022J011198), the Central Guidance on Local Science and Technology Development Fund of Fujian Province (2021L3058). The funders had no role in study design, data collection, analysis, the decision to publish, or the preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Pumo Cai conceived and designed the experiments, performed the experiments, analyzed the data, prepared figures and tables, authored or reviewed drafts of the paper, and approved the final draft. Yunzhe Song collected the data, prepared figures and tables, revised the manuscript, and approved the final draft. Litao Meng analyzed the data, prepared figures and tables, and approved the final draft. Jia Lin collected the data, prepared figures and tables, and approved the final draft. Mengting Zhao collected the data, prepared figures and tables, and approved the final draft. Qingfeng Wu collected the data, prepared figures and tables, and approved the final draft. Chuanpeng Nie collected the data, prepared figures and tables, and approved the final draft. Yanyan Li collected the data and approved the final draft. Qinge Ji conceived and designed the experiments, authored or reviewed drafts of the paper, and approved the final draft. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interest
The authors declare no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cai, P., Song, Y., Meng, L. et al. Phenological responses of Bactrocera dorsalis (Hendel) to climate warming in China based on long-term historical data. Int J Trop Insect Sci 43, 881–894 (2023). https://doi.org/10.1007/s42690-023-00996-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-023-00996-7