Abstract
Coral reefs are one of the most productive and biodiverse ecosystems in the world. Humans rely on these coral reef ecosystems to provide significant ecological and economic resources; however, coral reefs are threatened by numerous local and global anthropogenic factors that cause significant environmental change. The interactions of these local and global human impacts may increase the rate of coral reef degradation. For example, there are many local influences (i.e., sedimentation and submarine groundwater discharge) that may exacerbate coral bleaching and mortality. Therefore, researchers and resource managers cannot limit their narratives and actions to mitigating a sole stressor. With the continued increase in greenhouse gas emissions, management strategies and restoration techniques need to account for the scale at which environmental change occurs. This review aims to outline the various local and global anthropogenic stressors threatening reef resiliency and address the recent disagreements surrounding present-day conservation practices. Unfortunately, there is no one solution to preserve and restore all coral reefs. Each coral reef region is challenged by numerous interactive stressors that affect its ecosystem response, recovery, and services in various ways. This review discusses, while global reef degradation occurs, local solutions should be implemented to efficiently protect the coral reef ecosystem services that are valuable to marine and terrestrial environments.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Status of coral reefs
Reef-building corals have existed for over 200 million years, preserving through few challenges in the Holocene, but are now facing new, human-induced challenges in Anthropocene [1,2,3]. In the Holocene (11,000 years ago), reefs were resilient, experiencing rapid recovery and prevalence of acroporids; however, since transitioning into the Anthropocene, reef systems have lost resilience as disturbances increased in frequency and duration [4,5,6]. The importance of the diverse scleractinian family Acroporidae in providing extensive structurally complex habitat across the Indo-Pacific and their dramatic loss observed across the Caribbean makes them a sensitive but important taxon to study under global climate change patterns [7]. Reefs in the Anthropocene have shifted to more dynamic and patchier reef systems where stressors are not purely additive but are interacting in more complex ways [8].
Over the last 3 decades, living coral cover has declined roughly 53% in the Western Atlantic, 40% in the Indo-Pacific, and 50% on the Great Barrier Reef (GBR) [9, 10]. These declines in live coral cover are accompanied by a loss in structural complexity resulting in alterations in trophic structure and reductions in ecosystem services [11]. Globally, 39% of reefs are classified as low risk, and 52% of those reefs are found in the Pacific [12]. Overall, coral reefs in the Central Pacific experience lower rates of decline than those in the Atlantic, Indian, and Southeast-Asian Pacific oceans [12]. Therefore, understanding these drivers' relative influence, local and global, is vital in assessing mechanisms for managing and protecting coral reef systems and reducing secondary stressors during repeated marine heatwaves that cause coral bleaching [13].
While it is estimated that 6% of reefs across the globe will not be affected by either local or global stressors, 11% of reefs will be threatened solely by global factors alone, 22% solely from local factors, and 61% from the combined effects of local and global drivers of environmental change [14]. Globally, ocean warming and acidification are compromising carbonate accretion of coral reefs, resulting in less diverse reef communities [15, 16]. Highly complex coral architecture is relied upon by a diverse array of marine organisms; therefore, reduction in complexity of reefs has severe consequences for biodiversity, ecosystem functioning, and environmental services [17]. Locally, human impacts (i.e., pollution, coastal development, dredging, tourism, etc.) are causing dramatic phase shifts from coral-dominated to algal-dominated systems [18,19,20,21]. Some of these factors of environmental changes are co-occurring, potentially amplifying coral reef decline, and creating cascading effects for coral reef organisms and human populations who rely on the reefs for many ecosystem services [22].
The ongoing, rapid transformation of coral reefs creates challenges for conservation and management strategies due to a growing spatial mismatch between the scale of threats and planned responses. Additionally, coral reefs are not solely challenged by a single stressor. The various local, anthropogenic factors degrading reefs exacerbate the global effects of warming and acidification [8]. Global climate change has profound implications on reef health; however, the potential synergies between climate, human pressure, and biogeochemical factors must be alleviated for successful restoration and rehabilitation. The interactions between multiple stressors can be defined as synergistic (the combined effects exceed their individual effects), additive (the combined effect is equal to the sum of their individual effects), or antagonistic (the combined effect is less than the additive) [8, 23]. The ongoing increase of local and global threats on coral reefs highlights a critical gap in our knowledge of how these stressors may interact and shape future coral reefs [24]. Therefore, there is a need to reform management and conservation strategies that combat both local and global drivers of environmental change.
Coral reef status and resilience can be location-specific due to interactions of localized environmental stressors. For example, in Hawaiʻi, it has been suggested that regional management of multiple factors will benefit fish biomass and coral resistance to elevated temperatures [25,26,27,28]. In the Philippines, reefs have experienced a continued decline in hard coral cover over the past three decades, not solely due to the third global bleaching event (2014–2017) [29]. In Malaysia, coral reefs that contained high levels of live coral cover (> 25 colonies) were geographically isolated and experienced less coastal development and human activity [30], highlighting the importance of land use management practices. Across the Western Caribbean, four decades of coastal development has significantly reduced coral cover (15–20% in most regions). In turn, these reefs experienced increased fleshy macroalgae and herbivorous fish abundance, particularly between 2006 and 2016 [31]. In the Florida Keys, a 30-year study suggests that local nutrient enrichment and discharge from the Everglades contributes to eutrophication that has exacerbated coral stress and decline [32]. Results of this long-term monitoring suggest a balanced nitrogen to phosphorus (N:P) stoichiometry ratio would reduce the risk of coral bleaching disease and mortality in the future [32]. The disease occurrence on key coral species in the 1970s (Acropora spp.) and 2004 (Orbicella spp.) was linked to an increase of climate change impacts, including the intensity and frequency of hurricanes within the same regions. While climate stressors play a role, reef status and coral cover decline trends were significantly impacted at sites with increased coastal development and human-induced pollution [33]. Globally, there have been documented synergies between different environmental changes (i.e., anthropogenic stress, bleaching events, disease outbreaks, and hurricane damage) driving coral decline [4].
2 Global impacts
Projected increases in carbon dioxide (CO2) over the next 50 years will exceed the conditions coral reefs have survived over the past half-million years [15]. The industrial revolution has led to rapid increases in atmospheric greenhouse gas emissions, which have caused dramatic shifts in environmental conditions [34]. In particular, global changes in ocean chemistry and sea surface temperatures have promoted significant ecological decline in coral reef ecosystems [35,36,37]. In response, the 2015 Paris Agreement was developed to globally manage greenhouse gas emissions, which is vital for coral reefs' persistence [38]. Without radically reducing carbon emissions, the ocean is predicted to be 1–3 °C warmer, 0.2 pH units more acidic, and up to 1 m higher by 2050 [39].
2.1 Ocean warming
Corals worldwide live between 1 and 2 °C below their maximum summer temperature [40]. As the ocean continues to warm, corals are being pushed into their upper lethal temperature tolerances [41]. The resulting coral stress response includes the breakdown in the symbiosis between the coral host and its algal symbiont, commonly referred to as coral bleaching [36]. Other extreme changes in environmental conditions (e.g., irradiance, salinity sedimentation) can also cause coral bleaching; however, there has been an increase in the frequency and severity of mass coral bleaching events that are linked to increases in ocean temperatures [4, 36]. Additionally, coral bleaching events can be episodic and can coincide with ocean–atmosphere phenomena, such as El Niño-Southern Oscillation (ENSO) events [42]. To date, there have been three global bleaching events from 2014 to 2017 [43]. The number of areas (~ 6%) that have previously escaped bleaching will decrease each year as the potential for bleaching and mortality increases with or without ENSO events [42]. Examining recovery and adaptation rates of various reef systems over time has proved that bleaching events are likely to become chronic stress in the coming decades. Consequently, many coral communities will not recover quickly enough to maintain stable, coral-covered communities [44].
Before the third global coral bleaching event (2014–2017), many “low risk reefs” were situated in Australian waters [12, 43]. One year following the 2016 bleaching event, these reefs experienced large reductions (51%) in live coral cover, but recovery varied by region due to functional changes in the coral and fish community compositions [45]. The reefs in the northern half of the GBR were severely impacted (40%) [12, 45]. Community-wide trophic restructuring, specifically fish that scrape algae from reef surfaces, reduces competition between stressed corals and algae overgrowth and is critical for reef-scale recovery from bleaching [45].
Ocean warming has devastating effects on reef systems across the globe, regardless of protection or isolation. No reef is safe from the unprecedented rate of warming. Hawaiʻi suffered in 2014 and 2015 with 90% bleaching and 50% mortality due to elevated sea surface temperatures. The documented mortality from the warm water events was greater than documented mortality due to visitor trampling [46]. Even the most remote reef ecosystems, such as the Papahānaumokuākea Marine National Monument (PMNM), have suffered significant mortality levels from elevated sea surface temperatures [47]. Although the PMNM has a higher latitude and is far from human pollution and overfishing, historical satellite data confirmed the 2014 bleaching event exposed corals to heat stress that has increased significantly since 1982, confirming the continued, growing threat of climate change [47]. Similar widespread bleaching event scenarios have occurred on reef systems in the Coral Sea and the Gulf of Mexico that were previously known to be isolated and protected [48, 49]. A Coral Sea study showed that after the third global bleaching event, bleaching was less severe compared to other reefs across the globe, indicating that isolation cannot provide refuge from bleaching, but low nutrient levels, high wave energy, and proximity to deeper cooler water can allow reefs to be more resistant to mortality [48].
Alongside bleaching, thermal anomalies cause shifts in the coral microbiome by increasing viral production, which in return increases the carbon and nitrogen flux in the water column and benthos, and enhances rates of coral disease and mortality [50]. This viral production triggers a positive feedback loop that enhances coral decline. White syndrome, a common disease affecting Pacific reef-building corals, is exacerbated by thermal anomalies on reefs with > 50% coral cover [51]. This same disease was recorded dramatically reducing the abundance of Acropora palmata and Acropora cervicornis in the Caribbean. Moreover, data from the GBR supports the detrimental temperature-disease hypothesis while also implying that disease transmission is rapid on healthy reefs where coral cover is high, putting healthy reefs at high risk as warm temperature anomalies increase [51, 52]. Stony Coral Tissue Loss Disease (SCTLD), first occurring at high levels in 2014 along the Florida reef tract and coinciding with summer bleaching events, has since rapidly spread through Caribbean reefs causing unprecedented declines [53]. Using long-term data, there was no SCTLD prevalence in the Mexican Caribbean region before 2018, concluding that the disease was able to spread through the entire region within a few months, severely changing reef community structure [54]. While these diseases are decimating reefs at rapid rates, research has shown reefs containing high densities of coral feeding chaetodontid butterflyfish, from white band disease in the Philippines' to SCTLD in the Florida Keys, has positive correlations with disease prevalence [55, 56]. This suggests that stable, healthy reefs with functionally diverse fish abundances may ameliorate the impact of coral disease [55, 56].
2.2 Ocean acidification
Nearly 30% of the atmospheric CO2 emissions dissolve into the ocean. As CO2 dissociates, it releases hydrogen ions and increases the acidity of the seawater. This increase in the acidity of the ocean, ocean acidification, has several impacts on the marine environment [34, 57, 58]. Corals secrete calcium carbonate skeletons to maintain the ecologically important three-dimensional reef matrix. A change in ocean chemistry affects the shallow, sunlit, alkaline waters corals need to build and sustain their reef structure that provides habitat for many organisms and protects shorelines from bioerosion and storm damage [35, 59].
While ongoing changes in ocean chemistry directly influences coral physiology and accretion, it is difficult to quantify the rate at which ocean acidification is degrading reefs due to a lack of data at large spatial, temporal, and biogeochemical scales. Most acidification studies are conducted on single species, therefore, results are difficult to forecast across ecologically relevant scales [58]. Additionally, there are many assumptions and large discrepancies in coral reef response to ocean acidification [58]. For example, biogeochemical feedback in the open ocean could buffer some effects of acidification; therefore, we must understand this potential feedback and discrepancies to accurately predict the impact of acidification on coral reefs [60]. Also, reef-associated structure, location, hydrodynamics, and biogeochemical processes may vary across reefs and should be considered when calculating net community calcification rates [61].
2.3 Sea level rise
As atmospheric CO2 concentrations increase and our planet continues to warm, ice sheets are melting, causing sea level fluctuations worldwide. In the twenty-first century alone, global sea surface levels are predicted to rise 2 m, creating considerable alterations in coastal shoreline morphology with larger implications (20%) in tropical and subtropical habitats [62]. Low-lying coral atolls are of immediate concern; as the climate continues to change, atolls will be subject to wave-driven flooding and fluctuations in freshwater availability as the reef platforms change [63, 64]. Since the last glacial maximum (30,000 years ago), geologic records from the GBR suggest that the reef has been more resilient to sea-level rise and warming temperatures but was highly affected by increased sediment input [65]. While historical sea-level rise forces a landward migration of shallow-reef habitats, the GBR transitioned from a fringing to a barrier reef system [65]. Although there is evidence that coral reefs can transition as sea level rises, the uncertainty comes from the additive effects of a continued increase in carbon emissions and fishing pressures that prevent reefs from keeping up with rises in sea level expected by 2100 [66].
The structural complexity and integrity of coral reefs supports millions of people worldwide through coastal protection from storm damage and flooding. Coral reefs serve as natural barriers that protect nearly 200 million people worldwide from coastal flooding hazards and associated flooding risk costs (~ $0.8 million km−1 of reef); however, it is predicted that coastal communities are at greater risk if reef structural complexity is not maintained than if sea level continues to rise [64]. The vertical accretion and variation in topographic complexity, along with local rates of sea-level rise, will determine wave height and sediment transport damage. Nonetheless, the wave energy dissipation is determinant on complexity rather than sea level [3]. Future projections indicate that coral reef erosion rates will exceed accretion rates due to unprecedented global climate change, thereby increasing the risk and associated costs of coastal flooding [16, 67]. Current water depths have increased past predicted levels for the year 2100, and regional-scale degradation of coral reefs due to sea-level rise puts many coastal communities in danger [68]. In conclusion, there is no singular solution to combatting global climate change and the negative impacts it has on coral reef ecosystems and the communities that rely on them. As humans continue to amplify greenhouse gas emissions and atmospheric carbon dioxide concentrations, coral reefs will become increasingly vulnerable [69].
3 Local impacts
While global stressors independently affect 11% of reefs worldwide, twice as many (22%) coral reefs are impacted by local impacts. Local disturbances (i.e., water quality, sedimentation, human use, fishing pressure) may potentially influence coral reef responses to and recovery from climatic threats [32]. However, the potential of local action to offset global consequences on coral reefs is relatively unknown because it is difficult to assess across ecological scales in an experimental setting. Therefore, it becomes increasingly important to implement management and monitoring strategies that document the interaction of local and global drivers of environmental change to identify potential mitigation strategies [14]. The primary sources of human-induced impacts result from land-use change and terrestrial runoff, which have increased sedimentation and eutrophication on nearshore coral reefs [70]. Documentation dating back to the 1970s shows many areas are impacted by multiple local impacts and experiencing extensive coral mortality [70, 71]. The reduction of resilience in each area is highly location specific and depends on the type, duration, and magnitude of that impact, local environmental conditions, and overall ecosystem resiliency [72,73,74].
3.1 Human use: tourism and coastal development
Local anthropogenic stressors increase in number, severity, and frequency simultaneously with increasing human population densities [21]. Excess tourism and urban development such as land-use change, sedimentation, untreated sewage discharge, physical damage, and pollution are significant environmental stress sources for corals and coral reefs. Although physical space for development in coastal areas is limited, state governments and commercial companies continue to support urban development, increasing environmental stress on corals [75]. Although coral reef-based tourism is crucial for economic benefits, if degradation continues, socioeconomic services will decline and threaten many reef-dependent sectors of that society [76].
Coral reef-related tourism generates revenues in over 100 countries across the globe [77]. In the United States, the National Ocean and Atmospheric Administration (NOAA) estimated the total economic value of coral reef services to be over USD 3.4 billion [78]. In the Coral Triangle, marine tourism contributes to 36% of the overall tourism market; however, 85% of reefs in the region are threatened by local human activity [79]. Coral reef ecosystems in Hawaiʻi are estimated at USD 360 million a year, acting as an asset that supports many goods and services [80]. Many people rely on coral reefs for food, identity, and wealth in the Dominican Republic [81]. However, alongside the benefits of coral reef-related tourism comes significant impacts on the adjacent coral reefs. A health assessment of local reefs in the Dominican Republic has revealed significant nutrient-based pollution, human-related structural damage, and overfishing that increases coral bleaching, mortality, and disease abundance. Therefore, the Dominican Republic has implemented sustainable, land-based, and marine management practices through ecotourism to sustain economic growth [81]. Likewise, urgent action is necessary to maintain coral reefs on the GBR. Tourism operators have begun acting as stewards to engage their guests on climate change threats and how they can take action to protect the GBR [82]. While Australia and other countries attempt to educate their tourists on environmentally conscious actions, the stress from increasing human impact has decoupled biophysical relationships in coral reefs, causing dominant species to shift due to human-induced selective pressure [83, 84].
Land alteration and coastal development influence several cascading effects that cause perturbations and environmental change within the nearshore coral reef environment. Land-based sediment smothers corals, inhibiting them from receiving adequate light for photosynthesis, and triggers physiological stress that hinders coral recruitment, growth, and other ecosystem services [85,86,87]. Severe sedimentation due to harbor construction caused dramatic ecological declines in Pelekane Bay, Hawaiʻi [87]. Nonetheless, recovery and stabilization of coral cover was attributed to reduced sedimentation from watershed restoration [87]. While recovery is possible, certain taxonomic groups of corals have different thresholds of sediment exposure, including concentration, duration, and frequency; therefore, coral community composition may be determined by species-specific sediment tolerance thresholds [88]. Alongside coastal development, sediment exposure is amplified from dredging activity and has both synergistic and antagonistic effects with thermal stress from global climate change [89]. For example, a long-term coral health monitoring survey was conducted before, during, and after a 530-day dredging project in Barrow Island in Western Australia. This dredging project coincided with a warm water coral bleaching event. The data revealed that suspended sediment had both positive and negative effects on corals during the period of warm water. Under low sediment loads, the cumulative impact of sediment load and thermal stress was antagonistic, but the combined stressors were synergistic [89, 90]. As uncontrolled human pressure continues to have a detrimental effect on coral cover, land to sea management is essential to enhance reef protection and resiliency [89, 91]. The interplay between multiple local and global stressors is not well understood, thereby creating management responses to individual stressors at the community level challenging [73]. In many regions, the cumulative stressors from increased human populations do not receive the attention they require [73]. It is suggested that cumulative impact assessments are the best way to plan conservation strategies to mitigate the effects of coastal development and global climate change [31, 73].
Since early settlement, human use and disturbance have disrupted the unique physical, chemical, and biological features of coral reef ecosystems. In Kāneʻohe Bay, Hawaiʻi, a well-documented coral reef ecosystem, anthropogenic disturbance began with the first European settlers in 1778. By the 1960s, the coastal reefs were devastated by extensive dredging, increased sewage discharge, and excess sedimentation, leading to a loss in coral cover [26]. A reconstruction of the social–ecological relationships in Hawaiian coral reef systems over the past 700 years showed that reefs were able to recover from human impacts when the stressors were reduced over long periods (decades+) and over large spatial scales (> entire island systems or regions) [92]. To this day, across the Hawaiian Archipelago, there is a strong gradient of human impact on fish assemblages and hard coral cover with a few remote areas with small human populations acting as a refuge for fisheries production and biodiversity functioning [93]. A land to sea management technique in West Maui improved coastal water quality through reducing sediment runoff and cesspool effluent, overall enhancing snorkeling experience [94, 95]. Similarly, in Brazil, strategic management actions helped protect refugee areas from stressors (i.e., fishing intensity, land-based activities, coastal development) [73]. These joint integrated land to sea management efforts are the interplay of multiple stressors.
3.2 Pollution
Nearly 25% of coral reefs around the world are threatened by agricultural pollutants [96]. Agriculture is a highly erosive process that passes sediment, inorganic and organic nutrients, and other human contaminants to waterways leading to the ocean and ultimately vulnerable reefs [96,97,98]. Poor watershed management and land-based runoff have been found to cause eutrophication, defined as the excessive richness of nutrients, which creates a dense growth of plant life and an overall decline of oxygen [96, 99]. On coral reefs, eutrophication can cause excessive growth of algae and the suffocation of coral reefs due to a lack of oxygen and being outcompeted for resources by the macroalgae whose growth is being supported by the excessive rich nutrient source [75, 96].
Along with agricultural pollution, sewage effluent is another major source of human pollution, posing significant risks to coral reefs [96]. Sewage pollution sources include domestic and industrial wastewater and urban development from tourism and residential activities. Many coastal communities with fringing coral reef ecosystems, such as Puakō in the Hawaiʻi, rely on cesspools, septic tanks, or aerobic/anaerobic sewage treatments that contribute significantly to the decline in coral reefs [100]. Septic tank location and corresponding sewage pollution aggregates along the coastline, through the surface and benthic waters, increasing fecal indicator bacteria (FIB) and nutrient concentrations that contribute to harmful levels of eutrophication [100, 101]. A study on the effects of increased, wastewater-derived nutrients found that increases in sewage nutrient enrichment from hotel seepage pits could be responsible for lowering coral reef resilience leading to higher degradation rates at heavily affected sites [75]. Human derived sewage, primarily from increased tourism, creates inshore water enrichment from urban wastewater nutrient sources and has led to a reduction in benthic coral reef health and rugosity [59, 75]. It is believed that nutrient-enriched waters from sewage-derived sources are a major local stressor responsible for threatening coral reef ecosystems by reducing their resilience and stability [75].
With the global decline of coral reef ecosystems and a global increase in human populations along coastal areas, there have been management pushes to support wastewater treatment that will protect coral reefs in high-risk areas. Sewage and nitrogen pollution in Hawaiʻi not only led to decreased coral calcification and reproduction but was also a dominant driver in reef fish biomass changes, altering ecosystem function [102]. In the Florida Keys, the local and regional variability of nitrogen enrichment increased the risk of coral bleaching, mortality, and disease under temperature stress [32]. Numerous studies confirm that appropriate wastewater treatment would mitigate local eutrophication and, in response, increase reef refugia areas by 28% across the globe, giving reefs time while climate change is addressed at a global scale [14, 25].
3.3 Fishing pressure
Fishing, combined with other localized human drivers, leads to changes in fish biomass and is used as a direct indicator of reef health status and human disturbance level [103]. Pollution, fishing, and habitat drivers led to an overall decrease in 45% of total fish biomass over 10 years (2008–2018) on the west coast of Hawaiʻi [28]; however, regional management of these multiple factors could benefit fish functional groups. For example, a ban on scuba spearfishing and fishing nets increased grazer populations after a year of management. This confirmed that both recreational and commercial fishing are negatively affecting fish populations, but consistent management will aid in recovery [28, 66, 104]. Additionally, several areas across the Main Hawaiian Islands still support high standing fish stocks, which can be refugia for maintaining fisheries resources [93]. A decline in fish stocks can be attributed to numerous interactive factors, such as nutrient pollution and overfishing [104]. Therefore, management strategies should be implemented (e.g., quantifying fish biomass) to sustain the reefs and the livelihoods of the communities that rely on them [105]. In conclusion, the global decline of reefs is caused by two categories of localized human threats: overfishing and coastal development [106]. Managers must re-examine these threats and create risk assessments to effectively allocate resources and protect coral reefs for generations to come.
4 Actionable science advancements
There is a critical knowledge gap regarding how to deal with these multiple stressors that are causing various environmental changes and a growing need for them to be managed at multiple scales across different regulatory agencies. The increase in the severity and frequency of marine heatwaves challenges conventional management strategies [59, 107]. Therefore, there is a critical need to re-assess restoration and management strategies to sustain coral reef ecosystems under these continuing challenging conditions [108]. The global coral reef degradation that has occurred over the last 30–40 years has shifted coral reef resilience and therefore requires focused science and management efforts to prevent the loss of valuable social, ecological, and cultural resources [109].
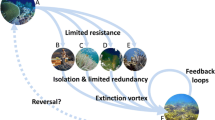
This section discusses (1) advances in science through assisted evolution, (2) creating risk assessments through collaboration with local communities, scientists, managers, and policymakers, and (3) focused management strategies that can be technically and economically achieved (Fig. 1). We aim to synthesize actionable, science-based solutions and collaborations that could benefit the future of coral reefs and the communities that rely on them.
4.1 Advances in science
Science advancements have shifted to promote coral reef resilience through an adaptation-focused intervention that will account for a suite of multiple stressors [57, 108]. Assisted evolution, the acceleration of evolutionary processes through the enhancement of specific traits, includes four mechanisms to increase environmental stress tolerance: (1) epigenetic programming and exposing adult colonies to high levels of environmental stress, (2) manipulation of the microbes associated with the coral holobiont, (3) culturing coral-associated endosymbiotic algae (Symbiodinium spp.) under future environmental conditions to increase bleaching resistance, or (4) selective breeding of the coral host itself (Fig. 1) [38, 110].
Adaptive actions and restoration techniques could address various environmental change drivers, but all stressor interactions and environmental alterations must be considered when analyzing the emergent ecological effects [57]. New interventions, such as genetic engineering, could be added to the coral reef restoration toolbox, but there are challenges in managing risks and uncertainties (Fig. 1) [108]. Therefore, while advances in science are important to consider when making a restoration plan, it is imperative to create a risk assessment outlining all possible options, consequences, uncertainties, and trade-offs to fully understand the goals and objectives in the short and long-term [111].
4.2 Creating risk assessments
The structured decision-making framework, created by Anthony et al., 2020, can help develop a management plan for high-risk ecosystems or watersheds [108]. This framework allows for the creation of a risk-assessment that integrates both local and global management efforts (Fig. 1). For example, the GBR has had approximately 40% coral cover decline on inshore reefs adjacent to human influence and requires a management plan that addresses local and global anthropogenic impacts [21]. Of the funding allocated to threats harming the reefs, some are used inadequately and can be why change or mitigation is not happening quickly (Fig. 1) [106]. It is essential to continue to work towards mitigating local factors because local threats to corals add to the effects seen by global threats and may inhibit recovery [32]. Creating a strategic plan for long-term conservation allows for clear guidelines and strategic investing (Fig. 1) [39]. A risk assessment can also be useful in less vulnerable, more protected reefs, which can be a conservation investment and help to repopulate degraded reefs in the climate becomes stabilized [39, 112].
The carbon emissions crisis has been globally recognized in the Paris Climate agreement; however, the goals are ambitious, and if achieved, coral reefs are still predicted to decline by 70–90% across the globe [112]. However, not all reefs, or species of coral, are at equal risk of global climate change. Coral reefs vary in resilience, defined as the net effect of resistance and recovery following a disturbance [113]. Predicting tipping points to environmental change is common in complex environmental systems, like coral reefs, when there are changes in climate, land-use, biodiversity, and biogeochemical cycles (Fig. 1) [114]. Therefore, strategic management of local factors through risk-sensitivity planning can improve the long-term conservation and persistence of coral reefs under climate pressure [39]. Studies from Kāneʻohe Bay, Hawaiʻi, previously under immense anthropogenic pressure from the 1930s to the 1970s, showed that different species have different pH, temperature, and sediment tolerances [115,116,117]. On the GBR, spatial resilience was determined by water quality levels and could inform strategic planning for future conservation goals [113]. A similar risk assessment was conducted in the Maldives to assess resilience by depth and confirmed bleaching events were too frequent and delayed reef recovery. Therefore, reducing local pressure was suggested to be the only effective way to improve resilience to thermal stress [118]. Resilience can vary region–region [19], reef–reef [113], and species–species [1]. It is imperative to understand multiple responses to both natural and anthropogenic change when conducting assessing management responses (Fig. 1) [119].
Modeling thermal thresholds and environmental influence is an effective way to understand past and future responses to human impact across larger spatial gradients (Fig. 1). In Japan, coral bleaching events were recorded from 2004 to 2016 and revealed multiple factors contributing to bleaching, including a selection of thermal indices and multiple environmental influences [120]. The coral mortality and bleaching output (COMBO) model was created as a tool to calculate the impact of increased greenhouse gas emissions and sea surface temperature on local and regional scales [121]. In the past decade, advancements are now developing more complex ways in which human impacts disrupt and degrade coral reef ecosystem function [83]. Quantifying the types of human impacts and their severity across our gradient would likely improve future interpretation of the spatial patterns on the benthic cover [83]. However, more information about the types of human impacts occurring across our large spatial gradient is required to predict “social–ecological macroecology” accurately [122]. Simulation modeling in the Philippines concluded that increased local water quality management and less management designated to fishing would significantly impact future reef state. The stressors examined interacted antagonistically; therefore, highlighting the importance of combining multiple stressors in a simulation model for supporting management (Fig. 1) [119]. In Karimunjawa National Park, Indonesia, a multivariate statistical model was applied to examine community composition changes and concluded that water quality management across the park is critical to improving resiliency [123]. Lastly, statistical models were used to investigate various coral taxa's responses to local stressors and climate variability in the Red Sea. This model concluded that fishing pressure and eutrophication's synergistic interactions exacerbated the impact of climate change [22]. While policymakers rely on models to predict regional climatic changes, they are non-linear in nature and often contain a high level of uncertainty [124]. Therefore, reliable multivariate models must have a well-monitored design and detailed calibration method [124, 125]. Modeling and incorporating multiple environmental variables that are both natural and human-induced environmental change allows for management and policy to be focused and science-based, improving resiliency of reef communities to future climatic events (Fig. 1).
4.3 Focused management strategies
There are several benefits to implementing focused management strategies that can assess benthic composition status and trends to mitigate local factors [126]. It is essential to consider local factors in management strategies because they play a role in resiliency to global climate change (Fig. 1) [21]. For example, when synthesizing multiple stressor interactions, it was concluded that managing sedimentation and nutrient loading could reduce coral bleaching [8]. To reduce regional threats, managers and other governance authorities should consider a holistic approach [119]. Management efforts should not focus on restoring historical baseline assemblages but should instead adapt to support natural recovery processes and embrace new and evolving conditions on a reef (Fig. 1) [127]. Ecosystem governance needs to shift to a new paradigm that embraces rapid change due to the unprecedented global heatwaves from 2014 to 2017 and continued anthropogenic pressures [128].
The suggested themes to improve governance in complex ecosystems are to address proximal and distal drivers, reduce those drivers' levels, and weaken positive feedback responses contributing to degradation [128]. It is also suggested for coral reefs to prioritize areas that demonstrate fewer bleaching signs than predicted by degree heating weeks recorded in that area [129]. Over the long term, those are the reefs that may be more resilient to thermal stress [129]. Spatial prioritization is also important when implementing marine protected areas (MPAs) (Fig. 1) [130]. Along with providing refuge to corals from human impact (i.e., tourism, fishing, etc.), the systematic conservation planning of an MPA allows for larval connectivity that self-sustains coral and reef fish recruitment (Fig. 1) [71, 131]. For example, zoning was implemented in the GBR protected areas to regulate the overexploitation of valuable resources [130, 132]. There needs to be a fundamental restructuring of institutional governance towards planetary stewardship to navigate the Anthropocene and capitalize on sustainable ecosystem management across the globe. Sustainable development and collaboration needs to be accelerated to mitigate and adapt to the planet's current and future transformations [133].
Recent developments of resilience-based management (RBM) plans aim to sustain the natural reef processes that support valuable ecological and social systems in a localized area, rather than focusing on global climate disturbances such as bleaching events (Fig. 1). RBM highlights reducing local pollution sources, protecting diversity, and maintaining connectivity pathways on reefs to adapt to change while simultaneously supporting resilience [127]. Apart from ecological disturbances, various socioeconomic factors can transform coral reef communities [134]. Participatory planning should be promoted to increase social acceptance and strengthen conservation strategies by considering the community's social and cultural characteristics [134, 135]. Management strategies that foster social adaptive capacity are better fit to address the complex changes in coastal marine socio-ecological systems, such as coral reefs, and should be accompanied by cost–benefit analysis to secure incomes, livelihoods and food security benefits for coastal communities (Fig. 1) [136, 137].
These collaborative, proactive management strategies are crucial for coral reef persistence because prevention is more effective and efficient than repair after the damage is done [138]. Even the GBR, one of the most well-managed systems, has lost a significant percentage of live coral color [9]. Therefore, it is crucial to pursue ecosystem-based management that is adaptive to fully understand the ecological processes that maintain coral reefs at large scales (Fig. 1) [12]. Ecosystem-based management can combat climate change by reducing local anthropogenic stressors and highlighting human-assisted evolution for change-ready MPAs [139]. With climate-induced coral bleaching being the main threat to coral reefs, it is important that management focus on how reefs respond to and recover from these devastating events. Predicting these regime shifts and identifying critical thresholds will guide managers through reef-specific management and adaptation tools (Fig. 1) [140]. Alongside predicting stress tolerances and thresholds, management of coral reef ecosystems will benefit from integrated land to sea models and scenario planning that includes human-induced change (Fig. 1). Planning strategies need to ensure the local human impacts will be minimized to assist in coral reef recovery under the future projected climate change impacts. Awareness of natural resources has led many local communities to implement place-based management enclosures and sustainable practices [141]. This place-based management strategy aims to reduce human impacts on coral reefs in a changing climate (Fig. 1) [141]. Overall, while managers and decision-makers attempt to create management strategies to mitigate global environmental change and its impact on reefs, there is still a research gap surrounding the assessment of the different strategies being utilized to understand the trade-offs and synergies [69]. Collaboration and cooperation across countries to re-organize the scientific research surrounding coral reef management is crucial to ensure science-based solutions lead to coral reef protection moving forward through the Anthropocene.
5 The path forward
Multiple environmental stressors significantly impact coral reefs. There is a global disparity in reef resilience across the globe; therefore, their persistence depends on conservation and management strategies specific to impacts experienced within that ecosystem [8, 19]. Therefore, local and regional managers should work together to mitigate the effects of climate change based on local conditions and responses [22, 121]. For example, large declines in reef populations across the Hawaiian Archipelago raised concerns of dangerous levels of overfishing and led to a local, watershed-based framework for resource management to protect key species [93]. Since global factors require numerous countries' assistance, it will be difficult and timely to get a proper collaboration or agreement that is agreed upon by all parties. In contrast, local factors can be dealt with immediately and directly. For example, appropriate wastewater treatments can mitigate local eutrophication and increase temporary refugia areas to 28%, allowing coral reefs to be relieved of the local stressor while international agreements are found to abate global stressors [14]. It is suggested that managers spend time identifying coral reef locations that, in the absence of other impacts, are most likely to have a heightened chance of surviving projected climate changes relative to other reefs and protect those reefs (Fig. 1) [39]. Multiple environmental variables are simultaneously interacting on coral reefs causing widespread degradation and understanding climate change impacts on reefs starts with managing local stressors that will, in return, help mitigate the global drivers of change.
The advancements outlined in this review have created a debate between scientists and resource managers about whether focusing efforts on mitigating local impacts will increase the resiliency of coral reef ecosystems to global climate change. This lack of clarity surrounding the recent disagreements on coral reef conservation practices has created many challenges in implementing actionable, science-based solutions that will benefit the communities that rely on them (Fig. 1). For example, some researchers argue that global warming is the universal threat to coral reef integrity and function [12, 37, 142]. In contrast, others believe local activities and land-based sources of pollution are the most critical threats [83, 96, 109]. Lastly, some believe local and global threats act in combination [66, 104]. To plan, mitigate, and restore the future of coral reefs, it is essential to acknowledge that 61% of reefs are simultaneously affected by local and global stressors. It cannot be a ‘one size fits all’ solution for reef protection [14]. Our review recommends actionable science-based advancements based on a local risk assessment that are unique to each region or reef system.
References
Putnam HM, Barott KL, Ainsworth TD, Gates RD (2017) The vulnerability and resilience of reef-building corals. Curr Biol 27:R528–R540. https://doi.org/10.1016/j.cub.2017.04.047
Woodroffe CD, Webster JM (2014) Coral reefs and sea-level change. Mar Geol 352:248–267
Harris DL, Rovere A, Casella E, et al (2018) Coral reef structural complexity provides important coastal protection from waves under rising sea levels. Sci Adv 4:eaao4350. https://doi.org/https://doi.org/10.1126/sciadv.aao4350
Hughes TP, Barnes ML, Bellwood DR et al (2017) Coral reefs in the Anthropocene. Nature 546:82–90
Roff G (2020) Reef accretion and coral growth rates are decoupled in Holocene reef frameworks. Mar Geol 419:106065. https://doi.org/10.1016/j.margeo.2019.106065
Waters CN, Zalasiewicz J, Summerhayes C, et al (2016) The Anthropocene is functionally and stratigraphically distinct from the Holocene. Science 351:aad2622–aad2622
Wallace CC (2012) Acroporidae of the Caribbean
Ban SS, Graham NAJ, Connolly SR (2014) Evidence for multiple stressor interactions and effects on coral reefs. Glob Chang Biol 20:681–697. https://doi.org/10.1111/gcb.12453
De’Ath G, Fabricius KE, Sweatman H, Puotinen M (2012) The 27-year decline of coral cover on the Great Barrier Reef and its causes. Proc Natl Acad Sci U S A 109:17995–17999. https://doi.org/10.1073/pnas.1208909109
Gardner TA, Côté IM, Gill JA et al (2003) Long-term region-wide declines in Caribbean corals. Science 301:958–960. https://doi.org/10.1126/science.1086050
Birkeland C (2015) Coral reefs in the anthropocene. Coral Reefs Anthr. https://doi.org/10.1007/978-94-017-7249-5
Bellwood DR, Pratchett MS, Morrison TH et al (2019) Coral reef conservation in the Anthropocene: confronting spatial mismatches and prioritizing functions. Biol Conserv 236:604–615. https://doi.org/10.1016/j.biocon.2019.05.056
Kramer KL, Cotton SP, Lamson MR, Walsh WJ (2016) Bleaching and catastrophic mortality of reef-building corals along west Hawai‘i island: findings and future directions. In: Proceedings of 13th international coral reef symposium Honolulu, pp 219–230
Guan Y, Hohn S, Wild C, Merico A (2020) Vulnerability of global coral reef habitat suitability to ocean warming, acidification and eutrophication. Glob Chang Biol 26:5646–5660. https://doi.org/10.1111/gcb.15293
Hughes TP, Baird AH, Bellwood DR et al (2003) Climate change, human impacts, and the resilience of coral reefs. Science 301:929–933. https://doi.org/10.1126/science.1085046
Hoegh-Guldberg O, Mumby PJ, Hooten AJ, Steneck RS, Greenfield P, Gomez E, Harvell CD, Sale PF, Edwards AJ, Caldeira K, Knowlton N, Eakin CM, Iglesias-Prieto R, Muthiga N, Bradbury RH, Dubi MEH (2007) Coral reefs under rapid climate change and ocean acidification. Science 1737:1737–1743. https://doi.org/10.1126/science.1152509
Alvarez-Filip L, Dulvy NK, Gill JA et al (2009) Flattening of Caribbean coral reefs: Region-wide declines in architectural complexity. Proc R Soc B Biol Sci 276:3019–3025. https://doi.org/10.1098/rspb.2009.0339
Hughes TP, Graham NAJ, Jackson JBC et al (2010) Rising to the challenge of sustaining coral reef resilience. Trends Ecol Evol 25:633–642. https://doi.org/10.1016/j.tree.2010.07.011
Roff G, Mumby PJ (2012) Global disparity in the resilience of coral reefs. Trends Ecol Evol 27:404–413. https://doi.org/10.1016/j.tree.2012.04.007
Bellwood DR, Hughes TP, Folke C, Nyström M (2004) Confronting the coral reef crisis. Nature 429:827–833. https://doi.org/10.1038/nature02691
Smith JE, Brainard R, Carter A et al (2016) Re-evaluating the health of coral reef communities: Baselines and evidence for human impacts across the central pacific. Proc R Soc B Biol Sci 283:1–9. https://doi.org/10.1098/rspb.2015.1985
Ellis JI, Jamil T, Anlauf H et al (2019) Multiple stressor effects on coral reef ecosystems. Glob Chang Biol 25:4131–4146. https://doi.org/10.1111/gcb.14819
Brown CJ, Saunders MI, Possingham HP, Richardson AJ (2013) Managing for interactions between local and global stressors of ecosystems. PLoS ONE. https://doi.org/10.1371/journal.pone.0065765
Abelson A (2020) Are we sacrificing the future of coral reefs on the altar of the “climate change” narrative? ICES J Mar Sci 77:40–45. https://doi.org/10.1093/icesjms/fsz226
Coles SL, Bahr KD, Rodgers KS et al (2018) Evidence of acclimatization or adaptation in Hawaiian corals to higher ocean temperatures. PeerJ 2018:1–24. https://doi.org/10.7717/peerj.5347
Bahr KD, Jokiel PL, Toonen RJ (2015) The unnatural history of Kāne’ohe bay: coral reef resilience in the face of centuries of anthropogenic impacts. PeerJ. https://doi.org/10.7717/peerj.950
Bahr KD, Rodgers KS, Jokiel PL (2017) Impact of three bleaching events on the reef resiliency of Kāne’ohe Bay. Hawai’i Front Mar Sci. https://doi.org/10.3389/fmars.2017.00398
Foo SA, Walsh WJ, Lecky J et al (2020) Impacts of pollution, fishing pressure, and reef rugosity on resource fish biomass in West Hawaii. Ecol Appl. https://doi.org/10.1002/eap.2213
Licuanan WY, Robles R, Reyes M (2019) Status and recent trends in coral reefs of the Philippines. Mar Pollut Bull 142:544–550. https://doi.org/10.1016/j.marpolbul.2019.04.013
Shahbudin S, Fikri Akmal K, Faris S et al (2017) Current status of coral reefs in tioman island, Peninsular Malaysia. Turk J Zool 41:294–305. https://doi.org/10.3906/zoo-1511-42
Schmitter-Soto JJ, Aguilar-Perera A, Cruz-Martínez A et al (2018) Interdecadal trends in composition, density, size, and mean trophic level of fish species and guilds before and after coastal development in the Mexican Caribbean. Biodivers Conserv 27:459–474. https://doi.org/10.1007/s10531-017-1446-1
Lapointe BE, Brewton RA, Herren LW et al (2019) Nitrogen enrichment, altered stoichiometry, and coral reef decline at Looe Key, Florida Keys, USA: a 3-decade study. Springer, Berlin Heidelberg
Rioja-Nieto R, Álvarez-Filip L (2019) Coral reef systems of the Mexican Caribbean: status, recent trends and conservation. Mar Pollut Bull 140:616–625. https://doi.org/10.1016/j.marpolbul.2018.07.005
Hönisch B, Ridgwell A, Schmidt DN et al (2012) The geological record of ocean acidification. Science 335:1058–1063. https://doi.org/10.1126/science.1208277
Albright R (2018) Ocean acidification and coral bleaching. Springer, Cham, pp 295–323
Brown BE (1997) Coral bleaching: causes and consequences. Coral Reefs 16:S129–S138. https://doi.org/10.1007/s003380050249
Hughes TP, Kerry JT, Baird AH et al (2018) Global warming transforms coral reef assemblages. Nature 556:492–496. https://doi.org/10.1038/s41586-018-0041-2
van Oppen MJH, Gates RD, Blackall LL, et al (2017) Shifting paradigms in restoration of the world’s coral reefs. Glob Chang Biol
Beyer HL, Kennedy E V., Beger M, et al (2018) Risk-sensitive planning for conserving coral reefs under rapid climate change. Conserv Lett 11
Coles SL, Jokiel PL (1977) Effects of temperature on photosynthesis and respiration in hermatypic corals. Mar Biol 43:209–216. https://doi.org/10.1007/BF00402313
Jokiel PL, Coles SL (1990) Response of Hawaiian and other Indo-Pacific reef corals to elevated temperature. Coral Reefs 8:155–162. https://doi.org/10.1007/BF00265006
Hughes TP, Anderson KD, Connolly SR et al (2018) Spatial and temporal patterns of mass bleaching of corals in the Anthropocene. Science 359:80–83. https://doi.org/10.1126/science.aan8048
Eakin CM, Sweatman HPA, Brainard RE (2019) The 2014–2017 global-scale coral bleaching event: insights and impacts. Coral Reefs 38:539–545. https://doi.org/10.1007/s00338-019-01844-2
Baker AC, Glynn PW, Riegl B (2008) Climate change and coral reef bleaching: an ecological assessment of long-term impacts, recovery trends and future outlook. Estuar Coast Shelf Sci 80:435–471. https://doi.org/10.1016/j.ecss.2008.09.003
Stuart-Smith RD, Brown CJ, Ceccarelli DM, Edgar GJ (2018) Ecosystem restructuring along the Great Barrier Reef following mass coral bleaching. Nature 560:92–96. https://doi.org/10.1038/s41586-018-0359-9
Rodgers KS, Bahr KD, Jokiel PL, Donà AR (2017) Patterns of bleaching and mortality following widespread warming events in 2014 and 2015 at the Hanauma Bay Nature Preserve. Hawai’i PeerJ. https://doi.org/10.7717/peerj.3355
Couch CS, Burns JHR, Liu G et al (2017) Mass coral bleaching due to unprecedented marine heatwave in Papahānaumokuākea Marine National Monument (Northwestern Hawaiian Islands). PLoS ONE 12:e0185121. https://doi.org/10.1371/journal.pone.0185121
Harrison HB, Álvarez-Noriega M, Baird AH et al (2019) Back-to-back coral bleaching events on isolated atolls in the Coral Sea. Coral Reefs 38:713–719. https://doi.org/10.1007/s00338-018-01749-6
Wright RM, Correa AMS, Quigley LA, et al (2019) Gene expression of endangered coral (Orbicella spp.) in flower garden banks national marine sanctuary after Hurricane Harvey. Front Mar Sci https://doi.org/https://doi.org/10.3389/fmars.2019.00672
Thurber RV, Payet JP, Thurber AR, Correa AMS (2017) Virus–host interactions and their roles in coral reef health and disease. Nat Rev Microbiol 15:205–216
Bruno JF, Selig ER, Casey KS et al (2007) Thermal stress and coral cover as drivers of coral disease outbreaks. PLoS Biol 5:1220–1227. https://doi.org/10.1371/journal.pbio.0050124
Aronson RB, Precht WF (2001) White-band disease and the changing face of Caribbean coral reefs. The ecology and etiology of newly emerging marine diseases. Springer, Netherlands, pp 25–38
Walton CJ, Hayes NK, Gilliam DS (2018) Impacts of a regional, multi-year, multi-species coral disease outbreak in Southeast Florida. Front Mar Sci 5:323. https://doi.org/10.3389/fmars.2018.00323
Alvarez-Filip L, Estrada-Saldívar N, Pérez-Cervantes E et al (2019) A rapid spread of the stony coral tissue loss disease outbreak in the Mexican Caribbean. PeerJ 2019:e8069. https://doi.org/10.7717/peerj.8069
Raymundo L, Halford AR, Maypa AP, Kerr AM (2009) Functionally diverse reef-fish communities ameliorate coral disease. Proc Natl Acad Sci USA 106:17067–17070. https://doi.org/10.1073/pnas.0913116107
Noonan KR, Childress MJ (2020) Association of butterflyfishes and stony coral tissue loss disease in the Florida Keys. Coral Reefs. https://doi.org/10.1007/s00338-020-01986-8
Doney SC, Busch DS, Cooley SR, Kroeker KJ (2020) The impacts of ocean acidification on marine ecosystems and reliant human communities. Annu Rev Environ Resour 45:83–112. https://doi.org/10.1146/annurev-environ-012320-083019
Jokiel PL (2016) Predicting the impact of ocean acidification on coral reefs: evaluating the assumptions involved. ICES J Mar Sci 73:550–557. https://doi.org/10.1093/icesjms/fsv091
Hoegh-Guldberg O, Poloczanska ES, Skirving W, Dove S (2017) Coral reef ecosystems under climate change and ocean acidification. Front Mar Sci 4
Takeshita Y (2017) Understanding feedbacks between ocean acidification and coral reef metabolism. J Geophys Res Ocean 122:1639–1642
Nakamura T, Nadaoka K, Watanabe A et al (2018) Reef-scale modeling of coral calcification responses to ocean acidification and sea-level rise. Coral Reefs 37:37–53. https://doi.org/10.1007/s00338-017-1632-3
Slangen ABA, Carson M, Katsman CA et al (2014) Projecting twenty-first century regional sea-level changes. Clim Change 124:317–332. https://doi.org/10.1007/s10584-014-1080-9
Webb AP, Kench PS (2010) The dynamic response of reef islands to sea-level rise: Evidence from multi-decadal analysis of island change in the Central Pacific. Glob Planet Change 72:234–246. https://doi.org/10.1016/j.gloplacha.2010.05.003
Storlazzi CD, Gingerich SB, Van Dongeren A, et al (2018) Most atolls will be uninhabitable by the mid-21st century because of sea-level rise exacerbating wave-driven flooding. Sci Adv 4:eaap9741. https://doi.org/https://doi.org/10.1126/sciadv.aap9741
Webster JM, Braga JC, Humblet M et al (2018) Response of the Great Barrier Reef to sea-level and environmental changes over the past 30,000 years. Nat Geosci 11:426–432. https://doi.org/10.1038/s41561-018-0127-3
Cacciapaglia CW, van Woesik R (2020) Reduced carbon emissions and fishing pressure are both necessary for equatorial coral reefs to keep up with rising seas. Ecography (Cop) 43:789–800. https://doi.org/10.1111/ecog.04949
Quataert E, Storlazzi C, Van Rooijen A et al (2015) The influence of coral reefs and climate change on wave-driven flooding of tropical coastlines. Geophys Res Lett 42:6407–6415. https://doi.org/10.1002/2015GL064861
Yates KK, Zawada DG, Smiley NA, Tiling-Range G (2017) Divergence of seafloor elevation and sea level rise in coral reef ecosystems. Biogeosciences 14:1739–1772. https://doi.org/10.5194/bg-14-1739-2017
Comte A, Pendleton LH (2018) Management strategies for coral reefs and people under global environmental change: 25 years of scientific research. J Environ Manag 209:462–474. https://doi.org/10.1016/j.jenvman.2017.12.051
Johannes RE (1975) Chapter 2. Pollution and degradation of coral reef communities. Elsevier Oceanogr Ser 12:13–51. https://doi.org/https://doi.org/10.1016/S0422-9894(08)71107-3
Mumby PJ, Steneck RS (2008) Coral reef management and conservation in light of rapidly evolving ecological paradigms. Trends Ecol Evol 23:555–563. https://doi.org/10.1016/j.tree.2008.06.011
Cinner JE, Maire E, Huchery C et al (2018) Gravity of human impacts mediates coral reef conservation gains. Proc Natl Acad Sci USA 115:E6116–E6125. https://doi.org/10.1073/pnas.1708001115
Magris RA, Grech A, Pressey RL (2018) Cumulative human impacts on coral reefs: assessing risk and management implications for brazilian coral reefs. Diversity 10:1–14. https://doi.org/10.3390/d10020026
Ferrigno F, Bianchi CN, Lasagna R et al (2016) Corals in high diversity reefs resist human impact. Ecol Indic 70:106–113. https://doi.org/10.1016/j.ecolind.2016.05.050
Lachs L, Johari NAM, Le DQ et al (2019) Effects of tourism-derived sewage on coral reefs: isotopic assessments identify effective bioindicators. Mar Pollut Bull 148:85–96. https://doi.org/10.1016/j.marpolbul.2019.07.059
Pendleton L, Hoegh-Guldberg O, Albright R et al (2019) The great barrier reef: vulnerabilities and solutions in the face of ocean acidification. Reg Stud Mar Sci 31:100729
Spalding M, Burke L, Wood SA et al (2017) Mapping the global value and distribution of coral reef tourism. Mar Policy 82:104–113. https://doi.org/10.1016/j.marpol.2017.05.014
Edwards P (2013) Summary report: the economic value of U.S. Coral Reefs
Huang Y, Coelho VR (2017) Sustainability performance assessment focusing on coral reef protection by the tourism industry in the Coral Triangle region. Tour Manag 59:510–527. https://doi.org/10.1016/j.tourman.2016.09.008
Cesar HSJ, van Beukering PJH (2004) Economic valuation of the coral reefs of Hawai’i. Pac Sci 58:231–242. https://doi.org/10.1353/psc.2004.0014
Eastwood EK, Clary DG, Melnick DJ (2017) Coral reef health and management on the verge of a tourism boom: a case study from Miches, Dominican Republic. Ocean Coast Manag 138:192–204. https://doi.org/10.1016/j.ocecoaman.2017.01.023
Goldberg J, Birtles A, Marshall N et al (2018) The role of Great Barrier Reef tourism operators in addressing climate change through strategic communication and direct action. J Sustain Tour 26:238–256. https://doi.org/10.1080/09669582.2017.1343339
Williams GJ, Gove JM, Eynaud Y et al (2015) Local human impacts decouple natural biophysical relationships on Pacific coral reefs. Ecography (Cop) 38:751–761. https://doi.org/10.1111/ecog.01353
Piggott-McKellar AE, McNamara KE (2017) Last chance tourism and the Great Barrier Reef. J Sustain Tour 25:397–415. https://doi.org/10.1080/09669582.2016.1213849
Rogers CS (1990) Responses of coral reefs and reef organisms to sedimentation. Mar Ecol Prog Ser 62:185–202. https://doi.org/10.1109/acssc.2005.1599849
Richmond RH (1993) Coral reefs: Present problems and future concerns resulting from anthropogenic disturbance. Integr Comp Biol 33:524–536. https://doi.org/10.1093/icb/33.6.524
Stender Y, Jokiel PL, Rodgers KS (2014) Thirty years of coral reef change in relation to coastal construction and increased sedimentation at Pelekane Bay. Hawai’i PeerJ 2014:e300. https://doi.org/10.7717/peerj.300
Tuttle LJ, Johnson C, Kolinski S et al (2020) How does sediment exposure affect corals? A systematic review protocol. Environ Evid 9:17
Fisher R, Bessell-Browne P, Jones R (2019) Synergistic and antagonistic impacts of suspended sediments and thermal stress on corals. Nat Commun. https://doi.org/10.1038/s41467-019-10288-9
Moore JAY, Bellchambers LM, Depczynski MR et al (2012) Unprecedented Mass Bleaching and Loss of Coral across 12° of Latitude in Western Australia in 2010–11. PLoS ONE 7:e51807. https://doi.org/10.1371/journal.pone.0051807
Suchley A, Alvarez-Filip L (2018) Local human activities limit marine protection efficacy on Caribbean coral reefs. Conserv Lett 11
Kittinger JN, Pandolfi JM, Blodgett JH et al (2011) Historical reconstruction reveals recovery in Hawaiian coral reefs. PLoS ONE 6:e25460. https://doi.org/10.1371/journal.pone.0025460
Friedlander AM, Donovan MK, Stamoulis KA et al (2017) Human-induced gradients of reef fish declines in the Hawaiian Archipelago viewed through the lens of traditional management boundaries. Aquat Conserv Mar Freshw Ecosyst 28:146–157. https://doi.org/10.1002/aqc.2832
Oleson KLL, Bagstad KJ, Fezzi C et al (2020) Linking land and sea through an ecological-economic model of coral reef recreation. Ecol Econ 177:106788. https://doi.org/10.1016/j.ecolecon.2020.106788
Rodgers KS, Jokiel PL, Brown EK et al (2015) Over a decade of change in spatial and temporal dynamics of Hawaiian coral reef communities. Pacific Sci 69:1–13. https://doi.org/10.2984/69.1.1
Carlson RR, Foo SA, Asner GP (2019) Land use impacts on coral reef health: a ridge-to-reef perspective. Front Mar Sci 6:562
Kroon FJ, Schaffelke B, Bartley R (2014) Informing policy to protect coastal coral reefs: Insight from a global review of reducing agricultural pollution to coastal ecosystems. Mar Pollut Bull 85:33–41. https://doi.org/10.1016/j.marpolbul.2014.06.003
Dadhich AP, Nadaoka K, Motomura Y, Watanabe A (2017) Potential impacts of land use change dynamics and submarine groundwater discharge on fringing reefs of Kuroshima Island, Japan. J Coast Conserv 21:245–254. https://doi.org/10.1007/s11852-017-0495-7
Risk MJ (2014) Assessing the effects of sediments and nutrients on coral reefs. Curr Opin Environ Sustain 7:108–117
Abaya LM, Wiegner TN, Beets JP et al (2018) Spatial distribution of sewage pollution on a Hawaiian coral reef. Mar Pollut Bull 130:335–347. https://doi.org/10.1016/j.marpolbul.2018.03.028
Morrison RJ, Denton GRW, Bale Tamata U, Grignon J (2013) Anthropogenic biogeochemical impacts on coral reefs in the Pacific Islands—an overview. Deep Sea Res Part II Top Stud Oceanogr. https://doi.org/10.1016/j.dsr2.2013.02.014
Barnes MD, Goodell W, Whittier R et al (2019) Decision analysis to support wastewater management in coral reef priority area. Mar Pollut Bull 148:16–29. https://doi.org/10.1016/j.marpolbul.2019.07.045
McClanahan TR (2011) Human and coral reef use interactions: from impacts to solutions? J Exp Mar Bio Ecol 408:3–10. https://doi.org/10.1016/j.jembe.2011.07.021
Zaneveld JR, Burkepile DE, Shantz AA et al (2016) Overfishing and nutrient pollution interact with temperature to disrupt coral reefs down to microbial scales. Nat Commun. https://doi.org/10.1038/ncomms11833
Cinner JE, Zamborain-Mason J, Gurney GG et al (2020) Meeting fisheries, ecosystem function, and biodiversity goals in a human-dominated world. Science 368:307–311. https://doi.org/10.1126/science.aax9412
Wear SL (2016) Missing the boat: Critical threats to coral reefs are neglected at global scale. Mar Policy 74:153–157. https://doi.org/10.1016/j.marpol.2016.09.009
Van Hooidonk R, Maynard J, Tamelander J et al (2016) Local-scale projections of coral reef futures and implications of the Paris Agreement. Sci Rep 6:1–8. https://doi.org/10.1038/srep39666
Anthony KRN, Helmstedt KJ, Bay LK et al (2020) Interventions to help coral reefs under global change—a complex decision challenge. PLoS ONE 15:e0236399. https://doi.org/10.1371/journal.pone.0236399
McLean M, Cuetos-Bueno J, Nedlic O et al (2016) Local stressors, resilience, and shifting baselines on coral reefs. PLoS ONE 11:e0166319. https://doi.org/10.1371/journal.pone.0166319
Muller-Parker G, D’Elia CF, Cook CB (2015) Interactions between corals and their symbiotic algae. Coral Reefs in the Anthropocene. Springer, Netherlands, pp 99–116
Goergen EA, Schopmeyer S, Moulding A, et al (2020) Coral reef restoration monitoring guide: best practices for monitoring coral restorations from local to ecosystem scales
Hoegh-Guldberg O, Kennedy EV, Beyer HL et al (2018) Securing a long-term future for coral reefs. Trends Ecol Evol 33:936–944
Mellin C, Matthews S, Anthony KRN et al (2019) Spatial resilience of the Great Barrier Reef under cumulative disturbance impacts. Glob Chang Biol 25:2431–2445. https://doi.org/10.1111/gcb.14625
Moore JC (2018) Predicting tipping points in complex environmental systems. Proc Natl Acad Sci USA 115:635–636
Jury CP, Toonen RJ (2019) Adaptive responses and local stressor mitigation drive coral resilience in warmer, more acidic oceans. Proc R Soc B Biol Sci 286:20190614. https://doi.org/10.1098/rspb.2019.0614
Bahr KD, Rodgers KS, Jokiel PL et al (2020) Pulse sediment event does not impact the metabolism of a mixed coral reef community. Ocean Coast Manag 184:105007. https://doi.org/10.1016/j.ocecoaman.2019.105007
Bahr KD, Jokiel PL, Rodgers KS (2016) Relative sensitivity of five Hawaiian coral species to high temperature under high-pCO2 conditions. Coral Reefs 35:729–738. https://doi.org/10.1007/s00338-016-1405-4
Montefalcone M, Morri C, Bianchi CN (2020) Influence of local pressures on Maldivian coral reef resilience following repeated bleaching events, and recovery perspectives. Front Mar Sci 7:1–14. https://doi.org/10.3389/fmars.2020.00587
Gurney GG, Melbourne-Thomas J, Geronimo RC et al (2013) Modelling coral reef futures to inform management: can reducing local-scale stressors conserve reefs under climate change? PLoS ONE 8:e80137. https://doi.org/10.1371/journal.pone.0080137
Kumagai NH, Yamano H (2018) High-resolution modeling of thermal thresholds and environmental influences on coral bleaching for local and regional reef management. PeerJ 2018:e4382. https://doi.org/10.7717/peerj.4382
Buddemeier RW, Jokiel PL, Zimmerman KM et al (2008) A modeling tool to evaluate regional coral reef responses to changes in climate and ocean chemistry. Limnol Oceanogr Methods 6:395–411. https://doi.org/10.4319/lom.2008.6.395
Williams GJ, Graham NAJ, Jouffray JB et al (2019) Coral reef ecology in the Anthropocene. Funct Ecol 33:1014–1022. https://doi.org/10.1111/1365-2435.13290
Kennedy EV, Vercelloni J, Neal BP et al (2020) Coral reef community changes in Karimunjawa National Park, Indonesia: assessing the efficacy of management in the face of local and global stressors. J Mar Sci Eng 8:1–27. https://doi.org/10.3390/jmse8100760
Hawkins E, Smith RS, Gregory JM, Stainforth DA (2016) Irreducible uncertainty in near-term climate projections. Clim Dyn 46:3807–3819. https://doi.org/10.1007/s00382-015-2806-8
Ho CK, Stephenson DB, Collins M et al (2012) Calibration strategies a source of additional uncertainty in climate change projections. Bull Am Meteorol Soc 93:21–26. https://doi.org/10.1175/2011BAMS3110.1
Williams ID, Couch C, Beijbom O et al (2019) Leveraging automated image analysis tools to transform our capacity to assess status and trends on coral reefs. Front Mar Sci 6:1–14. https://doi.org/10.3389/fmars.2019.00222
Mcleod E, Anthony KRN, Mumby PJ et al (2019) The future of resilience-based management in coral reef ecosystems. J Environ Manage 233:291–301. https://doi.org/10.1016/j.jenvman.2018.11.034
Morrison TH, Adger N, Barnett J et al (2020) Advancing coral reef governance into the Anthropocene. One Earth 2:64–74. https://doi.org/10.1016/j.oneear.2019.12.014
Heron S, Johnston L, Liu G et al (2016) Validation of reef-scale thermal stress satellite products for coral bleaching monitoring. Remote Sens 8:59. https://doi.org/10.3390/rs8010059
Hock K, Wolff NH, Ortiz JC et al (2017) Connectivity and systemic resilience of the Great Barrier Reef. PLOS Biol 15:e2003355. https://doi.org/10.1371/journal.pbio.2003355
Harrison HB, Williamson DH, Evans RD et al (2012) Larval export from marine reserves and the recruitment benefit for fish and fisheries. Curr Biol 22:1023–1028. https://doi.org/10.1016/j.cub.2012.04.008
Day JC (2002) Zoning—lessons from the great barrier reef marine park. Ocean Coast Manag 45:139–156. https://doi.org/10.1016/S0964-5691(02)00052-2
Biermann F, Abbott K, Andresen S et al (2012) Navigating the anthropocene: improving earth system governance. Science 335:1306–1307
Darling ES, McClanahan TR, Maina J et al (2019) Social–environmental drivers inform strategic management of coral reefs in the Anthropocene. Nat Ecol Evol 3:1341–1350. https://doi.org/10.1038/s41559-019-0953-8
Hein MY, Birtles A, Willis BL et al (2019) Coral restoration: socio-ecological perspectives of benefits and limitations. Biol Conserv 229:14–25. https://doi.org/10.1016/j.biocon.2018.11.014
D’agata S, Darling ES, Gurney GG, et al (2020) Multiscale determinants of social adaptive capacity in small-scale fishing communities. Environ Sci Policy 108:56–66. https://doi.org/10.1016/j.envsci.2020.03.006
Whitney CK, Bennett NJ, Ban NC et al (2017) Adaptive capacity: from assessment to action in coastal social–ecological systems. Ecol Soc. https://doi.org/10.5751/ES-09325-220222
Birkeland C (2018) Global status of coral reefs: in combination, disturbances and stressors become ratchets. In: World seas: an environmental evaluation volume III: ecological issues and environmental impacts. Elsevier, pp 35–56
Harvey BJ, Nash KL, Blanchard JL, Edwards DP (2018) Ecosystem-based management of coral reefs under climate change. Ecol Evol 8:6354–6368
Graham NAJ, Jennings S, MacNeil MA et al (2015) Predicting climate-driven regime shifts versus rebound potential in coral reefs. Nature 518:94–97. https://doi.org/10.1038/nature14140
Delevaux JMS, Stamoulis KA, Whittier R et al (2019) Place-based management can reduce human impacts on coral reefs in a changing climate. Ecol Appl 29:e01891. https://doi.org/10.1002/eap.1891
Hughes TP, Kerry JT, Álvarez-Noriega M et al (2017) Global warming and recurrent mass bleaching of corals. Nature 543:373–377. https://doi.org/10.1038/nature21707
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Good, A.M., Bahr, K.D. The coral conservation crisis: interacting local and global stressors reduce reef resiliency and create challenges for conservation solutions. SN Appl. Sci. 3, 312 (2021). https://doi.org/10.1007/s42452-021-04319-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42452-021-04319-8