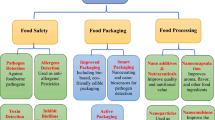
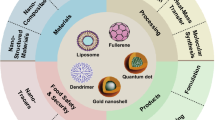
Abstract
Packaging films help to prevent damage, by providing an extended shelf life. From the post-harvest period to final delivery to the consumer is the most crucial time span for maintaining the shelf life of many fruits and vegetables. The gasotransmitters play a vital role in the delayed ripening and senescence of many fruits and vegetables, during the post-harvest period. Apart from their application in maintaining the shelf life of fruits and vegetables, these gaseous signaling molecules are also used for different therapeutic applications. Handling, storage, and transport of these gas molecules are the main problems when to be directly used in an open environment. To avoid these issues, porous organic/inorganic scaffolds are generally used to immobilize them. Recently nanostructures act as an encapsulation system and carrier of these molecules. This system not only avoids the burst release and toxicity that is associated with the burst release but also helps to release them in a controllable manner at a required site under induced stimuli. These nanocarriers on forming a composite with packaging materials provide several new properties to the packaging film. In this paper, the interaction of gasotransmitters with different nanostructures, their controlled release from nanostructures, and further use for advanced packaging film are reviewed.
Graphical Abstract




Similar content being viewed by others
References
Xue L et al (2017) Missing food, missing data? A critical review of global food losses and food waste data. Environ Sci Technol 51(12):6618–6633. https://doi.org/10.1021/acs.est.7b00401
Joardder MUH, Masud MH (2019) Food preservation in developing countries: challenges and solutions. Food Preserv Dev Ctries Chall Solut. https://doi.org/10.1007/978-3-030-11530-2
Sheldon RA, Norton M (2020) Green chemistry and the plastic pollution challenge: towards a circular economy. Green Chem 22(19):6310–6322. https://doi.org/10.1039/d0gc02630a
Chaudhary P, Fatima F, Kumar A (2020) Relevance of nanomaterials in food packaging and its advanced future prospects. J Inorg Organomet Polym Mater 30(12):5180–5192. https://doi.org/10.1007/s10904-020-01674-8
Tiwari K, Singh R, Negi P, Dani R, Rawat A (2021) Application of nanomaterials in food packaging industry: a review. Mater Today Proc 46:10652–10655. https://doi.org/10.1016/j.matpr.2021.01.385
Kuswandi B (2017) Environmental friendly food nano-packaging. Environ Chem Lett 15(2):205–221. https://doi.org/10.1007/s10311-017-0613-7
Youssef AM, El-Sayed SM (2018) Bionanocomposites materials for food packaging applications: Concepts and future outlook. Carbohydr Polym 193:19–27. https://doi.org/10.1016/j.carbpol.2018.03.088
Gong T, Li C, Bian B, Wu Y, Dawuda MM, Liao W (2018) Advances in application of small molecule compounds for extending the shelf life of perishable horticultural products: a review. Sci Hortic (Amsterdam) 230(2017):25–34. https://doi.org/10.1016/j.scienta.2017.11.013
Jin Z, Sun L, Yang G, Pei Y (2018) Hydrogen sulfide regulates energy production to delay leaf senescence induced by drought stress in arabidopsis. Front Plant Sci 871:1–11. https://doi.org/10.3389/fpls.2018.01722
Dugbartey GJ, Juriasingani S, Zhang MY, Sener A (2021) H2S donor molecules against cold ischemia-reperfusion injury in preclinical models of solid organ transplantation. Pharmacol Res 172:105842. https://doi.org/10.1016/j.phrs.2021.105842
Wang L, Xie X, Ke B, Huang W, Jiang X, He G (2021) Recent advances on endogenous gasotransmitters in inflammatory dermatological disorders. J Adv Res. https://doi.org/10.1016/j.jare.2021.08.012
Liu T, Mukosera GT, Blood AB (2020) The role of gasotransmitters in neonatal physiology. Nitric Oxide Biol Chem 95:29–44. https://doi.org/10.1016/j.niox.2019.12.002
Garren MR, Ashcraft M, Qian Y, Douglass M, Brisbois EJ, Handa H (2021) Nitric oxide and viral infection: Recent developments in antiviral therapies and platforms. Appl Mater Today 22:100887. https://doi.org/10.1016/j.apmt.2020.100887
Pant J, Mondal A, Manuel J, Singha P, Mancha J, Handa H (2020) H2S-releasing composite: a gasotransmitter platform for potential biomedical applications. ACS Biomater Sci Eng 6(4):2062–2071. https://doi.org/10.1021/acsbiomaterials.0c00146
Pieretti JC, Junho CVC, Carneiro-Ramos MS, Seabra AB (2020) H2S- and NO-releasing gasotransmitter platform: a crosstalk signaling pathway in the treatment of acute kidney injury. Pharmacol Res 161:105121. https://doi.org/10.1016/j.phrs.2020.105121
Corpas FJ, Palma JM (2020) H2S signaling in plants and applications in agriculture. J Adv Res 24:131–137. https://doi.org/10.1016/j.jare.2020.03.011
Yao GF et al (2020) Hydrogen sulfide maintained the good appearance and nutrition in post-harvest tomato fruits by antagonizing the effect of ethylene. Front Plant Sci. https://doi.org/10.3389/fpls.2020.00584
Ziogas V, Molassiotis A, Fotopoulos V, Tanou G (2018) Hydrogen sulfide: a potent tool in postharvest fruit biology and possible mechanism of action. Front Plant Sci. https://doi.org/10.3389/fpls.2018.01375
Li D, Limwachiranon J, Li L, Du R, Luo Z (2016) Involvement of energy metabolism to chilling tolerance induced by hydrogen sulfide in cold-stored banana fruit. Food Chem 208:272–278. https://doi.org/10.1016/j.foodchem.2016.03.113
Aghdam MS, Mahmoudi R, Razavi F, Rabiei V, Soleimani A (2018) Hydrogen sulfide treatment confers chilling tolerance in hawthorn fruit during cold storage by triggering endogenous H2S accumulation, enhancing antioxidant enzymes activity and promoting phenols accumulation. Sci Hortic (Amsterdam) 238:264–271. https://doi.org/10.1016/j.scienta.2018.04.063
Niazi Z, Razavi F, Khademi O, Soleimani M (2021) Scientia Horticulturae Exogenous application of hydrogen sulfide and γ-aminobutyric acid alleviates chilling injury and preserves quality of persimmon fruit (Diospyros kaki, cv. Karaj) during cold storage. Sci Hortic (Amsterdam) 285:110198. https://doi.org/10.1016/j.scienta.2021.110198
Zhu L et al (2019) Scientia horticulturae synergistic effect of nitric oxide with hydrogen sulfide on inhibition of ripening and softening of peach fruits during storage. Sci Hortic (Amsterdam) 256:108591. https://doi.org/10.1016/j.scienta.2019.108591
Siddiqui MW, Deshi V, Homa F, Aftab MA, Aftab T (2021) Inhibitory effects of hydrogen sulfide on oxidative damage and pericarp browning in harvested litchi. J Plant Growth Regul 40(6):2560–2569. https://doi.org/10.1007/s00344-021-10300-x
Al Ubeed HMS, Wills RBH, Bowyer MC, Vuong QV, Golding JB (2017) Interaction of exogenous hydrogen sulphide and ethylene on senescence of green leafy vegetables. Postharvest Biol Technol 133:81–87. https://doi.org/10.1016/j.postharvbio.2017.07.010
Li D et al (2017) Effects of hydrogen sulfide on yellowing and energy metabolism in broccoli. Postharvest Biol Technol 129:136–142. https://doi.org/10.1016/j.postharvbio.2017.03.017
Corpas FJ, González-Gordo S, Palma JM (2020) Nitric oxide: a radical molecule with potential biotechnological applications in fruit ripening. J Biotechnol 324:211–219. https://doi.org/10.1016/j.jbiotec.2020.10.020
Zhang W, Cao J, Fan X, Jiang W (2020) Applications of nitric oxide and melatonin in improving postharvest fruit quality and the separate and crosstalk biochemical mechanisms. Trends Food Sci Technol 99:531–541. https://doi.org/10.1016/j.tifs.2020.03.024
Gergoff Grozeff GE, Alegre ML, Senn ME, Chaves AR, Simontacchi M, Bartoli CG (2017) Combination of nitric oxide and 1-MCP on postharvest life of the blueberry (Vaccinium spp.) fruit. Postharvest Biol Technol 133:72–80. https://doi.org/10.1016/j.postharvbio.2017.06.012
Zhou Y, Li S, Zeng K (2016) Exogenous nitric oxide-induced postharvest disease resistance in citrus fruit to Colletotrichum gloeosporioides. J Sci Food Agric 96(2):505–512. https://doi.org/10.1002/jsfa.7117
Khaliq G, Ullah M, Ahmed S, Arif M, Muhammad A (2020) Exogenous nitric oxide reduces postharvest anthracnose disease and maintains quality of custard apple (Annona squamosa L.) fruit during ripening. J Food Meas Charact. https://doi.org/10.1007/s11694-020-00658-z
Hu M, Zhu Y, Liu G, Gao Z, Li M, Su Z (2019) Scientia horticulturae inhibition on anthracnose and induction of defense response by nitric oxide in pitaya fruit. Sci Hortic (Amsterdam) 245(2018):224–230. https://doi.org/10.1016/j.scienta.2018.10.030
Yan B et al (2019) Scientia horticulturae nitric oxide enhances resistance against black spot disease in muskmelon and the possible mechanisms involved. Sci Hortic (Amsterdam) 256:108650. https://doi.org/10.1016/j.scienta.2019.108650
Li G, Yu Z, Cao J, Peng Y, Shi J (2020) Scientia horticulturae nitric oxide regulates multiple defense signaling pathways in peach fruit response to Monilinia fructicola invasion. Sci Hortic (Amsterdam) 264(2019):109163. https://doi.org/10.1016/j.scienta.2019.109163
Zhu L, Yang R, Sun Y, Zhang F, Du H, Zhang W (2020) Nitric oxide maintains postharvest quality of navel orange fruit by reducing postharvest rotting during cold storage and enhancing antioxidant activity. Physiol Mol Plant Pathol. https://doi.org/10.1016/j.pmpp.2020.101589
Ren Y, He J, Liu H, Liu G, Ren X (2017) Nitric oxide alleviates deterioration and preserves antioxidant properties in ‘Tainong’ mango fruit during ripening. Hortic Environ Biotechnol 58(1):27–37. https://doi.org/10.1007/s13580-017-0001-z
Zheng X, Hu B, Song L, Pan J, Liu M (2017) Scientia horticulturae changes in quality and defense resistance of kiwifruit in response to nitric oxide treatment during storage at room temperature. Sci Hortic (Amsterdam) 222:187–192. https://doi.org/10.1016/j.scienta.2017.05.010
Yang R et al (2021) Scientia horticulturae transcriptome profiling of postharvest kiwifruit in response to exogenous nitric oxide. Sci. Hortic. (Amsterdam) 277(2020):109788. https://doi.org/10.1016/j.scienta.2020.109788
Tian W, Huang D, Geng B, Zhang Q, Feng J, Zhu S (2020) Regulation of the biosynthesis of endogenous nitric oxide and abscisic acid in stored peaches by exogenous nitric oxide and abscisic acid. J Sci Food Agric 100(5):2136–2144. https://doi.org/10.1002/jsfa.10237
Buet A, Steelheart C, Perini MA, Galatro A, Simontacchi M, Gergoff Grozeff GE (2021) Nitric oxide as a key gasotransmitter in fruit postharvest: an overview. J Plant Growth Regul. https://doi.org/10.1007/s00344-021-10428-w
Huang D, Tian W, Feng J, Zhu S (2020) Interaction between nitric oxide and storage temperature on sphingolipid metabolism of postharvest peach fruit. Plant Physiol Biochem 151:60–68. https://doi.org/10.1016/j.plaphy.2020.03.012
Cai H, Han S, Yu M, Ma R, Yu Z (2020) Exogenous nitric oxide fumigation promoted the emission of volatile organic compounds in peach fruit during shelf life after long-term cold storage. Food Res Int 133(2019):109135. https://doi.org/10.1016/j.foodres.2020.109135
Song C, Zhao Y, Li A, Qi S, Lin Q, Duan Y (2021) Plant physiology and biochemistry postharvest nitric oxide treatment induced the alternative oxidase pathway to enhance antioxidant capacity and chilling tolerance in peach fruit. Plant Physiol Biochem 167:113–122. https://doi.org/10.1016/j.plaphy.2021.07.036
Palma F, Carvajal F, Castro-cegrí A, Pulido A, Jamilena M, Jim R (2021) Postharvest biology and technology pre-storage nitric oxide treatment enhances chilling tolerance of zucchini fruit (Cucurbita pepo L.) by S-nitrosylation of proteins and modulation of the antioxidant response. Postharvest Biol Technol. https://doi.org/10.1016/j.postharvbio.2020.111345
Ma Y, Huang D, Chen C, Zhu S, Gao J (2019) Scientia horticulturae regulation of ascorbate-glutathione cycle in peaches via nitric oxide treatment during cold storage. Sci Hortic (Amsterdam) 247(2018):400–406. https://doi.org/10.1016/j.scienta.2018.12.039
Zhao Y, Zhu X, Hou Y, Wang X, Li X (2019) Effects of nitric oxide fumigation treatment on retarding cell wall degradation and delaying softening of winter jujube (Ziziphus jujuba Mill. Cv. Dongzao) fruit during storage. Postharvest Biol Technol 156:110954. https://doi.org/10.1016/j.postharvbio.2019.110954
Gheysarbigi S, Hossein S, Ghasemnezhad M (2020) The inhibitory effect of nitric oxide on enzymatic browning reactions of in-package fresh pistachios (Pistacia vera L.). Postharvest Biol Technol 159(2019):110998. https://doi.org/10.1016/j.postharvbio.2019.110998
Jayarajan S, Sharma RR (2018) “Impact of nitric oxide on shelf life and quality of nectarine (Prunus persica var. nucipersica). Acta Physiol Plant. https://doi.org/10.1007/s11738-018-2779-4
Shi K, Liu Z, Wang J, Zhu S, Huang D (2019) Scientia horticulturae nitric oxide modulates sugar metabolism and maintains the quality of red raspberry during storage. Sci Hortic (Amsterdam) 256:108611. https://doi.org/10.1016/j.scienta.2019.108611
Huang L, Wu G, Zhang S, Kuang F, Chen F (2019) Postharvest biology and technology the identification and functional verification of the cinnamate 4-hydroxylase gene from wax apple fruit and its role in lignin biosynthesis during nitric oxide-delayed postharvest cottony softening. Postharvest Biol Technol 158:110964. https://doi.org/10.1016/j.postharvbio.2019.110964
Soleimani M, Kakavand F, Rabiei V, Zaare-nahandi F (2019) Scientia horticulturae γ-aminobutyric acid and nitric oxide treatments preserve sensory and nutritional quality of cornelian cherry fruits during postharvest cold storage by delaying softening and enhancing phenols accumulation. Sci Hortic (Amsterdam) 246(2018):812–817. https://doi.org/10.1016/j.scienta.2018.11.064
Wasim M, Fozia S, Deep H, Shamsher LM, Surabhi A (2020) Exogenous nitric oxide delays ripening and maintains postharvest quality of pointed gourd during storage. J Plant Growth Regul. https://doi.org/10.1007/s00344-020-10270-6
Sun M, Yang X, Zhu Z, Xu Q, Wu K, Kang Y (2021) Plant physiology and biochemistry comparative transcriptome analysis provides insight into nitric oxide suppressing lignin accumulation of postharvest okra (Abelmoschus esculentus L.) during cold storage. Plant Physiol Biochem 167:49–67. https://doi.org/10.1016/j.plaphy.2021.07.029
Wei F, Fu M, Li J, Yang X, Chen Q, Tian S (2019) Chlorine dioxide delays the reddening of postharvest green peppers by affecting the chlorophyll degradation and carotenoid synthesis pathways. Postharvest Biol Technol 156(2018):110939. https://doi.org/10.1016/j.postharvbio.2019.110939
Yang X, Zhang X, Fu M, Chen Q, Muzammil JM (2018) Chlorine dioxide fumigation generated by a solid releasing agent enhanced the efficiency of 1-MCP treatment on the storage quality of strawberry. J Food Sci Technol 55(6):2003–2010. https://doi.org/10.1007/s13197-018-3114-1
Yang X, Yan R, Chen Q, Fu M (2020) Analysis of flavor and taste attributes differences treated by chemical preservatives: a case study in strawberry fruits treated by 1-methylcyclopropene and chlorine dioxide. J Food Sci Technol. https://doi.org/10.1007/s13197-020-04474-7
Sun X et al (2017) Effect of controlled-release chlorine dioxide on the quality and safety of cherry/grape tomatoes. Food Control 82:26–30. https://doi.org/10.1016/j.foodcont.2017.06.021
Wei J, Chen Y, Tiemur A, Wang J, Wu B (2018) Postharvest biology and technology degradation of pesticide residues by gaseous chlorine dioxide on table grapes. Postharvest Biol Technol 137(2017):142–148. https://doi.org/10.1016/j.postharvbio.2017.12.001
Liu X, Jiao W, Du Y, Chen Q, Su Z, Fu M (2020) Chlorine dioxide controls green mold caused by Penicillium digitatum in citrus fruits and the mechanism involved. J Agric Food Chem. https://doi.org/10.1021/acs.jafc.0c05288
Zhang S, Wang Q, Guo Y, Kang L, Yu Y (2020) Carbon monoxide enhances the resistance of jujube fruit against postharvest Alternaria rot. Postharvest Biol Technol 168:111268. https://doi.org/10.1016/j.postharvbio.2020.111268
Hu H, Zhao S, Li P, Shen W (2018) Hydrogen gas prolongs the shelf life of kiwifruit by decreasing ethylene biosynthesis. Postharvest Biol Technol 135:123–130. https://doi.org/10.1016/j.postharvbio.2017.09.008
Zhang Y et al (2019) Nitrite accumulation during storage of tomato fruit as prevented by hydrogen gas. Int J Food Prop 22(01):1425–1438. https://doi.org/10.1080/10942912.2019.1651737
Chang E, Lee J, Kim J (2017) Scientia horticulturae cell wall degrading enzymes activity is altered by high carbon dioxide treatment in postharvest ‘Mihong’ peach fruit. Sci Hortic (Amsterdam) 225:399–407. https://doi.org/10.1016/j.scienta.2017.07.038
Admane N et al (2018) Effect of ozone or carbon dioxide pre-treatment during long-term storage of organic table grapes with modified atmosphere packaging. LWT Food Sci Technol 98:170–178. https://doi.org/10.1016/j.lwt.2018.08.041
Ahn D, Kim I, Lim J, Hee J, Park K, Lee J (2021) The effect of high CO2 treatment on targeted metabolites of ‘Seolhyang’ strawberry (Fragaria × ananassa) fruits during cold storage. LWT Food Sci Technol 143:111156. https://doi.org/10.1016/j.lwt.2021.111156
Li D et al (2020) Delaying the biosynthesis of aromatic secondary metabolites in postharvest strawberry fruit exposed to elevated CO2 atmosphere. Food Chem 306:1–8. https://doi.org/10.1016/j.foodchem.2019.125611
Li D, Zhang X, Li L, Soleimani M, Wei X, Liu J (2019) Elevated CO2 delayed the chlorophyll degradation and anthocyanin accumulation in postharvest strawberry fruit. Food Chem 285:163–170. https://doi.org/10.1016/j.foodchem.2019.01.150
Wang Y, Li W, Chang H, Zhou J, Luo Y, Zhang K (2019) Sweet cherry fruit miRNAs and effect of high-CO2 on the profile associated with ripening. Planta. https://doi.org/10.1007/s00425-019-03110-9
Rodríguez-Ruiz M, Zuccarelli R, Palma JM, Corpas FJ, Freschi L (2019) Biotechnological application of nitric oxide and hydrogen peroxide in plants
Nazir F, Fariduddin Q, Alam T (2020) Chemosphere hydrogen peroxide as a signalling molecule in plants and its crosstalk with other plant growth regulators under heavy metal stress. Chemosphere 252:126486. https://doi.org/10.1016/j.chemosphere.2020.126486
Carné-Sánchez A, Carmona FJ, Kim C, Furukawa S (2020) Porous materials as carriers of gasotransmitters towards gas biology and therapeutic applications. Chem Commun 56(68):9750–9766. https://doi.org/10.1039/d0cc03740k
Chen M, Chen X, Yam K (2020) Encapsulation complex of chlorine dioxide in α-cyclodextrin: Structure characterization and release property. Food Control. https://doi.org/10.1016/j.foodcont.2019.106783
Silva AF, Calhau IB, Gomes AC, Valente AA, Gonçalves IS, Pillinger M (2021) A hafnium-based metal-organic framework for the entrapment of molybdenum hexacarbonyl and the light-responsive release of the gasotransmitter carbon monoxide. Mater Sci Eng C 124:112053. https://doi.org/10.1016/j.msec.2021.112053
Kuai L, Liu F, Sen Chiou B, Avena-Bustillos RJ, McHugh TH, Zhong F (2021) Controlled release of antioxidants from active food packaging: a review. Food Hydrocoll 120:106992. https://doi.org/10.1016/j.foodhyd.2021.106992
Sun X, Wang Y, Wen S, Huang K et al (2021) Novel controlled and targeted releasing hydrogen sulfide system exerts combinational cerebral and myocardial protection after cardiac arrest. J Nanobiotechnol. https://doi.org/10.1186/s12951-021-00784-w
Huang Y et al (2020) Near-infrared photothermal release of hydrogen sulfide from nanocomposite hydrogels for anti-inflammation applications. Chin Chem Lett 31(3):787–791. https://doi.org/10.1016/j.cclet.2019.05.025
Hsu PH et al (2020) Hydrogen sulfide-responsive self-assembled nanogel. ACS Appl Polym Mater 2(9):3756–3760. https://doi.org/10.1021/acsapm.0c00759
Pelegrino MT, Weller RB, Chen X, Bernardes JS, Seabra AB (2017) Chitosan nanoparticles for nitric oxide delivery in human skin. Medchemcomm 8(4):713–719. https://doi.org/10.1039/c6md00502k
Niu X et al (2019) A glutathione responsive nitric oxide release system based on charge-reversal chitosan nanoparticles for enhancing synergistic effect against multidrug resistance tumor. Nanomed Nanotechnol Biol Med 20:102015. https://doi.org/10.1016/j.nano.2019.102015
Ghalei S, Hopkins S, Douglass M, Garren M, Mondal A, Handa H (2021) Nitric oxide releasing halloysite nanotubes for biomedical applications. J Colloid Interface Sci 590:277–289. https://doi.org/10.1016/j.jcis.2021.01.047
Quinn JF, Whittaker MR, Davis TP (2015) Delivering nitric oxide with nanoparticles. J Control Release 205:190–205. https://doi.org/10.1016/j.jconrel.2015.02.007
Yan H et al (2019) Emerging delivery strategies of carbon monoxide for therapeutic applications: from CO gas to CO releasing nanomaterials. Small 15(49):1–22. https://doi.org/10.1002/smll.201904382
Nguyen D, Boyer C (2015) Macromolecular and inorganic nanomaterials scaffolds for carbon monoxide delivery: recent developments and future trends. ACS Biomater Sci Eng 1(10):895–913. https://doi.org/10.1021/acsbiomaterials.5b00230
Chakraborty I, Mascharak PK (2016) Mesoporous silica materials and nanoparticles as carriers for controlled and site-specific delivery of gaseous signaling molecules. Microporous Mesoporous Mater 234:409–419. https://doi.org/10.1016/j.micromeso.2016.07.028
Ho TM, Howes T, Bhandari BR (2014) Encapsulation of gases in powder solid matrices and their applications: a review. Powder Technol 259:87–108. https://doi.org/10.1016/j.powtec.2014.03.054
Chen X, Lee DS, Zhu X, Yam KL (2012) Release kinetics of tocopherol and quercetin from binary antioxidant controlled-release packaging films. J Agric Food Chem 60(13):3492–3497. https://doi.org/10.1021/jf2045813
Funding
This research did not receive any specific grant from any funding agency in the public, commercial or not from profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors have no conflict of interest.
Research Involving Human and Animals Statement
None.
Informed Consent
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wakchoure, D., Azmi, N., Chaskar, J. et al. Application of Gasotransmitters in Nanomaterials-Based Food Packaging. J Package Technol Res 8, 1–13 (2024). https://doi.org/10.1007/s41783-024-00164-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41783-024-00164-3