Abstract
Introduction
The presence of antibiotic allergy labels can have harmful impacts on clinical outcomes, particularly among immunosuppressed patients, in whom there have been associations with increased complications, readmission rates, and mortality. We explore the effects of a sulfonamide allergy label (SAL) on clinical outcomes in adult patients with Pneumocystis jirovecii pneumonia (PJP).
Methods
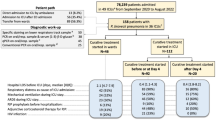
In this retrospective matched cohort study, we utilized TriNetX, a multicenter national database, to match 535 adult patients with PJP and SAL to an equal number of controls. We identified cases indexed between 01/01/2010 and 01/01/2023 utilizing ICD-10 codes for PJP and allergy status to sulfonamides and through detection of P. jirovecii antigen with immunofluorescence or PCR. Propensity score matching was performed in a 1:1 fashion for demographics and comorbidities, and our analysis included clinical outcomes that occurred within 30 days after the occurrence of the index event.
Results
While hospitalization risk tended to be lower among patients with SAL as compared to controls (RR: 0.90; 95% CI 0.81–1.01), there were no major differences in the risk of respiratory failure (RR: 0.94; 95% CI 0.84–1.05), prednisone use (RR: 1; 95% CI 0.91–1.10), intensive level of care requirement (RR: 0.85; 95% CI 0.69–1.06), intubation (RR: 0.85; 95% CI 0.61–1.19), or mortality (RR: 0.98; 95% CI 0.68–1.42). The presence of SAL did however impact antibiotic prescription patterns, with an underutilization of trimethoprim (RR: 0.50; 95% CI 0.43–0.59) and sulfamethoxazole (RR, 0.47; 95% CI 0.40–0.56) and overuse of alternative agents by patients with SAL as compared to controls. Yet, there was no difference in the occurrence of adverse outcomes such as hepatotoxicity (RR: 1.09; 95% CI 0.49–2.45) or acute kidney injury (RR: 0.94; 95% CI 0.78–1.14) between patients with SAL and controls.
Conclusions
The presence of SAL alters antibiotic prescription patterns among adults with Pneumocystis infection but has no clinically significant impact on outcomes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Penicillin allergy label has been associated with various negative clinical outcomes, but the impact of sulfonamide allergy label (SAL), the second most common allergy label, remains unclear. |
We hypothesized that SAL would be associated with negative clinical outcomes in patients with Pneumocystis jirovecii pneumonia such as increased risk of respiratory failure, intensive level of care requirement, or mortality. |
While SAL influenced antibiotic prescription practices with underutilization of sulfamethoxazole-trimethoprim in favor of alternative agents, no impact on clinical outcomes was appreciated. |
Further research to explore the impact of SAL in other clinical settings is needed to guide recommendations regarding de-labeling strategies. |
Introduction
Pneumocystis jirovecii, an opportunistic fungus that causes Pneumocystis jirovecii pneumonia (PJP), is a significant cause of morbidity and mortality in immunocompromised patients [1]. First-line treatment of PJP is trimethoprim-sulfamethoxazole (TMP-SMX), though alternative agents are often required for patients with sulfonamide allergy [2]. In the United States, sulfonamide or sulfa allergy is the second most common drug allergy label, with a reported prevalence of 2–10% [3]. Most sulfa allergy labels (SAL) are attributable to TMP-SMX [4]. In one study that explored the impact of SAL on prescription practices in 94 hospitalized patients, SAL was mostly used to denote an allergy to TMP-SMX (45% of patients) or else was related to an unknown medication (45% of patients) [4]. Sulfonamide allergies can present with various manifestations, but dermatologic complaints are the most frequently reported [5]. Notably, in patients with human immunodeficiency virus (HIV), who are disproportionately affected by PJP, skin eruptions occur 10 to 20 times more frequently than in immunocompetent patients, likely due to a disruption in normal immune regulatory mechanisms [5].
While numerous studies have explored the impact of having a penicillin allergy label on clinical outcomes across various clinical settings including in patients with pneumonia; to our knowledge, no studies to date have explored the impact of having SAL on clinical outcomes [6, 7]. Characterizing the impact of SAL on clinical outcomes is especially important given the wide applications of TMP-SMX antibiotic in clinical practice, including in the treatment of PJP, and the high prevalence of SAL [3]. Previous studies demonstrated that avoiding TMP-SMX is associated with an increased risk of nocardiosis in solid organ transplant recipients and that the use of atovaquone, an alternative prophylactic agent against PJP, has been associated with toxoplasmosis reactivation in stem cell transplantation [8,9,10]. These studies indirectly suggest that having a SAL might similarly have negative clinical outcomes by modulating antibiotic prescription practices. While sulfonamide antibiotics have been associated with severe cutaneous allergic reactions, most patients only experience benign cutaneous reactions and are able to tolerate TMP-SMX upon further evaluation [11, 12]. Thus, understanding the impact of SAL can further guide future recommendations regarding the optimal timing for TMP-SMX de-labeling and antibiotic stewardship efforts.
In this study, we leveraged a large multicenter US electronic health record database (TriNetX US Collaborative Network) to explore the impact of having SAL on antibiotic prescription practices and clinical outcomes of PJP and to help guide management decisions in patients with sulfa allergies. We hypothesize that SAL would lead to worse clinical outcomes similar to that observed in penicillin allergy [7].
Methods
Data Source
This retrospective matched cohort study utilized the TriNetX US Collaborative Network database (TriNetX LLC, Cambridge, MA, USA), which was accessible through an institutional agreement, that provides access to ~100 million patients from 60 US healthcare organizations and is de-identified per §164.514(a) of the Health Insurance Portability and Accountability Act (HIPAA).
Study Cohorts and Matching
We utilized the International Classification of Diseases, Tenth Revision (ICD-10) codes and Logical Observation Identifiers Names and Codes (LOINC) to identify adult patients (≥ 18 years old) with PJP [defined by an ICD-10 for pneumocystosis (B59) or detection of P. jirovecii antigen by immunofluorescence or detection of DNA (either positive or > 1450 copies/µL) by polymerase chain reaction (PCR) in sputum, lower respiratory tract specimen, or bronchoalveolar lavage] indexed between 01/01/2010 and 01/01/2023 (Table 1). The sulfa allergy-labeled (SAL) group was denoted by the presence of an ICD-10 code for allergy status to sulfonamides (Z88.2) preceding the indexed pneumonia by ≥ 1 day. The SAL and control cohorts were propensity score-matched in a 1:1 fashion for demographics and comorbidities. These were captured using ICD-10 and Current Procedural Terminology (CPT) codes within 1 year pre-index event (Table 2).
Study Outcomes
After matching, we assessed the 30-day risk of hospitalization [defined by TriNetX-specific term, inpatient encounter], acute respiratory failure [acute respiratory failure, J96.0; OR respiratory failure, unspecified, J96.9; OR acute and chronic respiratory failure, J96.2; OR hypoxemia, R09.02; OR acute respiratory distress, R06.03; OR ventilation assist and management, initiation of pressure volume preset ventilators for assisted or controlled breathing, CPT:94002], need for intensive level of care [initial and continuing intensive care services, CPT:1019140; OR critical care services, CPT:1013729], intubation [insertion of endotracheal airway into trachea, via natural or artificial opening, Procedure Coding System (PCS):0BH17EZ; OR intubation, endotracheal, emergency procedure, CPT:31500; OR insertion of endotracheal tube, Systemized Nomenclature of Medicine (SNOMED):112798008; OR insertion of endotracheal airway into trachea, percutaneous, PCS:0BH13EZ; OR insertion of endotracheal airway into trachea via natural or artificial opening endoscopic, PCS:0BH18EZ], prednisone utilization (RxNorm codes for prednisone or methylprednisolone), acute kidney injury (AKI) [acute kidney failure, N17], hepatotoxicity [toxic liver disease with hepatic necrosis, K71.1; OR toxic liver disease with acute hepatitis, K71.2; OR toxic liver disease with hepatitis, not elsewhere classified, K71.6; OR toxic liver disease, unspecified, K71.9; OR acute and subacute hepatic failure, K72.0), and mortality [TriNetX-specific term: deceased]. We further explored antibiotic prescription patterns using RxNorm medications codes.
Statistical Analysis
Statistical analyses were performed using the TriNetX built-in analytical feature. We used composite outcomes to account for rare events, as TriNetX obfuscates counts ≤ 10 to safeguard patients’ confidentiality. Significance was assessed using the Z-test to compare proportions. For propensity score matching, TriNetX generates a propensity score for each patient and uses a “greedy nearest-neighbor matching” with a caliper of 0.1 pooled standard deviations to identify matched subsets. The relative risk (RR) and 95% confidence interval (CI) characterized the direction and strength of associations between study outcomes and SAL.
Ethical Approval
Ethical approval was not required for this study. This retrospective study is exempt from informed consent. The data reviewed is a secondary analysis of existing data, does not involve intervention or interaction with human subjects, and is de-identified per the de-identification standard defined in §164.514(a) of the HIPAA Privacy Rule. The process by which the data is de-identified is attested to through a formal determination by a qualified expert as defined in §164.514(b)(1) of the HIPAA Privacy Rule. This formal determination by a qualified expert refreshed on December 2020.
Results
We successfully matched 535 patients with PJP and SAL to an equal number of controls (patients with PJP but without SAL). Baseline demographics and comorbidities of the matched cohorts were well balanced (standardized mean difference < 1 across all variables) (Table 2). Within 30 days of the indexed PJP, there was a trend toward decreased hospitalization in the SAL group compared to controls [273 (51%) SAL versus 303 (56.6%); RR: 0.90; 95% CI, 0.81–1.01; P = 0.066] (Fig. 1) (Table 3). There was no significant difference in prednisone use [322 (60.2%) versus 322 (60.2%) controls; RR: 1; 95% CI 0.91–1.10; P = 1.00], acute respiratory failure [275 (51.4%) versus 294 (55) controls; RR: 0.94; 95% CI 0.84–1.05; P = 0.24], the need for intensive level of care [122 (22.8%) patients with SAL versus 143 (26.7%) controls; RR: 0.85; 95% CI 0.69–1.06; P = 0.14], or intubation [56 (10.5%) versus 66 (12.3%) controls; RR: 0.85; 95% CI 0.61–1.19; P = 0.34] (Fig. 1) (Table 3). There was also no difference in the risk of acute kidney injury [146 (27.3%) versus 155 (29%) controls; RR: 0.94; 95% CI, 0.78–1.14; P = 0.54], or hepatotoxicity [12 (2.2%) versus 11 (2.1%) controls; RR: 1.09; 95% CI, 0.49–2.45; P = 0.83) (Fig. 1) (Table 3). Lastly, there was no mortality difference [51 (9.5%) versus 52 (9.7%) controls; RR: 0.98; 95% CI 0.68–1.42; P = 0.92] (Fig. 1) (Table 3).
Sulfonamide allergy label (SAL) was not associated with worse clinical outcomes in patients with Pneumocystis jirovecii pneumonia (PJP). Shown are the % of cohorts with the different study outcomes, along with respective relative risk (RR) and 95% confidence interval (CI) for having each outcome among patients with SAL relative to controls. Results are after propensity score matching (PSM) for demographics and comorbidities
SAL altered antibiotic prescription patterns among patients with PJP (Fig. 2) (Table 3). Relative to controls, trimethoprim and sulfamethoxazole were underutilized in the SAL group (RR: 0.50; 95% CI, 0.43–0.59; P < 0.001 and RR, 0.47; 95% CI 0.40–0.56; P < 0.001, respectively) (Fig. 2) (Table 3). There was over-utilization of alternative anti-PJP agents including dapsone (RR: 3.35; 95% CI 2.20–5.10; P < 0.001), pentamidine (RR: 2; 95% CI 1.31–3.05; P = 0.001), atovaquone (RR: 2.35; 95% CI 1.85–2.97; P < 0.001), primaquine (RR: 2.65; 95% CI, 1.91–3.69; P < 0.001) and clindamycin (RR: 2.37; 95% CI 1.77–3.18; P < 0.001) in the SAL cohort (Fig. 2) (Table 3).
Sulfonamide allergy label (SAL) altered antibiotic prescription practices for patients with Pneumocystis jirovecii pneumonia (PJP). There was underutilization of trimethoprim and sulfamethoxazole and overutilization of alternative anti-PJP agents. Results are after propensity score matching (PSM) for demographics and comorbidities
Discussion
This is the first multicenter study to investigate the impact of SAL on clinical outcomes in patients with PJP. Several studies have explored the effects of antibiotic allergy labels on clinical outcomes in immunocompromised patients demonstrating associations with increased medical complications, readmission rates, and mortality [13, 14]. In this study, we anticipated similar findings, yet observed no impact of SAL on acute respiratory failure, need for intensive care, or mortality in patients with PJP. In fact, there was a slight tendency toward lower hospitalization rates among the SAL group. There was also no difference in prednisone utilization, an intervention reserved for severe PJP. SAL did, however, influence the choice of antibiotics with underutilization of trimethoprim and overutilization of alternative agents.
In this study, patients with HIV accounted for ~40% of the study cohorts, and, after matching the study cohorts for HIV status, we similarly did not observe altered clinical outcomes driven by SAL albeit altered antibiotic prescription practices. Despite a decreasing incidence of PJP among patients with HIV due to recommendations for earlier diagnosis and initiation of antiretroviral therapy, PJP remains one of the most common opportunistic infections in HIV-infected individuals [15]. It is well known that patients with HIV experience a higher rate of adverse drug reactions, particularly to TMP-SMX [5], which complicates the management of PJP and begs the question of which patients should be treated through hypersensitivity reactions. The findings of this study suggest that the use of alternative agents in the presence of SAL might have no impact on clinical outcomes and may serve to inform clinicians’ decision regarding sulfa allergy de-labeling or desensitization.
The lack of association with worse outcomes may be explained by the effectiveness of alternative antibiotic regimens. In a literature review by McDonald and colleagues, alternative agents including dapsone-trimethoprim, and pentamidine were shown to have point estimates favoring their use versus TMP-SMX with respect to overall mortality [16]. In another study, approximately 90% patients with PJP who failed conventional therapy with TMP-SMX had a good clinical response to the combination of clindamycin and primaquine [17]. Alternatively, patients with allergic phenotypes may mount an altered immune response to PJP [18]. Under normal circumstances, Pneumocystis is recognized by antigen-presenting cells and presented to CD4+ T-lymphocytes and B-lymphocytes in bronchus-associated lymphoid tissue. The combined activity of CD4+ T-lymphocytes and B-lymphocytes activates macrophages with a preference for M2 polarization, leading to clearance of the fungus with little to no inflammation [18]. Whereby in immunocompromised hosts, the population that accounts for most cases of PJP, an improper T-helper 1 (Th1) predominant response leads to the activation of M1 macrophages with infiltration of CD8 T cells and neutrophils that is toxic to lung tissue [18]. In allergic patients with a T-helper 2 predisposition, we propose that this altered Th1 predominant immune response may be ameliorated. Furthermore, this may provide an explanation for the slight tendency toward lower hospitalization among the SAL group.
As SAL is often used to indicate allergy to TMP-SMX, [4] both trimethoprim and sulfamethoxazole were underutilized in our SAL cohort in favor for alternative agents (50% less likely). Yet, despite such altered prescription patterns, we observed no increased side effects such as AKI or hepatotoxicity as was previously observed with penicillin allergy-labeled individuals with pneumonia [6]. The lack of association may otherwise be explained by both trimethoprim and alternative agents carrying the risk of these side effects [19, 20] or else attributed to insufficient sample size.
The findings of this study must be viewed considering several limitations. Reliance on ICD-10 codes that are subject to reporting bias represents a potential constraint. However, the TriNetX platform employs machine learning to extract codes from clinical documentation, thereby lessening such bias. Additionally, we utilized antigen detection methods, like immunofluorescence and PCR, to confirm the diagnosis of PJP when able. The relatively short follow-up period of 30 days represents another potential limitation, as this may have prevented certain outcomes from becoming more apparent with added monitoring. Lastly, since our data were obtained from large healthcare organizations in the United States, our findings may not apply outside the US due to regional differences in P. jirovecii transmission and antibiotic availability. The lack of prior studies on this topic makes it difficult to predict outcomes and draw conclusions. Therefore, future research is warranted to elucidate any potential associations. Investigators could employ a prospective study design with a longer follow-up period and include data from international populations.
Conclusions
Through leveraging a multicenter database, we found that SAL altered antibiotic prescription practices with underutilization of TMP-SMX, but such altered antibiotic prescription practices had no clinically meaningful impact on outcomes of patients with Pneumocystis jirovecii pneumonia such as progression to acute respiratory failure, the need for intensive care, or mortality. As such, the de-labeling of sulfa allergy may not have an appreciable positive impact on clinical outcomes in the setting of PJP. Further research is needed to elucidate the impact of SAL on clinical outcomes in other clinical settings to better guide future recommendations regarding optimal timing for sulfa allergy de-labeling.
Data Availability
No database was downloaded or downloadable, and the presented analysis represents live analysis of the TriNetX US collaborative Database which was accessible through an institutional agreement.
References
Schmidt JJ, Lueck C, Ziesing S, et al. Clinical course, treatment and outcome of Pneumocystis pneumonia in immunocompromised adults: a retrospective analysis over 17 years. Crit Care. 2018;22(1):307.
Ibrahim A, Chattaraj A, Iqbal Q, et al. Pneumocystis jiroveci Pneumonia: A Review of Management in Human Immunodeficiency Virus (HIV) and Non-HIV Immunocompromised Patients. Avicenna J Med. 2023;13(1):23–34.
Khan DA, Knowles SR, Shear NH. Sulfonamide hypersensitivity: fact and fiction. J Allergy Clin Immunol Pract. 2019;7(7):2116–23.
Hemstreet BA, Page RL 2nd. Sulfonamide allergies and outcomes related to use of potentially cross-reactive drugs in hospitalized patients. Pharmacotherapy. 2006;26(4):551–7.
Giles A, Foushee J, Lantz E, Gumina G. Sulfonamide Allergies. Pharmacy (Basel). 2019;7(3).
Kaminsky LW, Ghahramani A, Hussein R, Al-Shaikhly T. Penicillin allergy label is associated with worse clinical outcomes in bacterial pneumonia. J Allergy Clin Immunol Pract. 2022;10(12):3262–9.
Castells M, Khan DA, Phillips EJ. Penicillin Allergy. N Engl J Med. 2019;381(24):2338–51.
Yetmar ZA, Chesdachai S, Duffy D, et al. Risk factors and prophylaxis for nocardiosis in solid organ transplant recipients: A nested case-control study. Clin Transplant. 2023;37(9): e15016.
Chueng TA, Moroz IV, Anderson AD, Morris MI, Komanduri KV, Camargo JF. Failure of atovaquone prophylaxis for prevention of toxoplasmosis in hematopoietic cell transplant recipients. Transpl Infect Dis. 2020;22(1): e13198.
Adekunle RO, Sherman A, Spicer JO, et al. Clinical characteristics and outcomes of toxoplasmosis among transplant recipients at two US academic medical centers. Transpl Infect Dis. 2021;23(4): e13636.
Khan DA, Banerji A, Blumenthal KG, et al. Drug allergy: a 2022 practice parameter update. J Allergy Clin Immunol. 2022;150(6):1333–93.
Gorsline CA, Afghan AK, Stone CA Jr, Phillips EJ, Satyanarayana G. Safety and value of pretransplant antibiotic allergy delabeling in a quaternary transplant center. Transpl Infect Dis. 2022;24(5): e13885.
Huang KG, Cluzet V, Hamilton K, Fadugba O. The impact of reported beta-lactam allergy in hospitalized patients with hematologic malignancies requiring antibiotics. Clin Infect Dis. 2018;67(1):27–33.
Waldron JL, Trubiano JA. Antibiotic allergy labels in immunocompromised populations. Transpl Infect Dis. 2022;24(5): e13955.
Buchacz K, Lau B, Jing Y, et al. Incidence of AIDS-defining opportunistic infections in a multicohort analysis of HIV-infected Persons in the United States and Canada, 2000–2010. J Infect Dis. 2016;214(6):862–72.
McDonald EG, Butler-Laporte G, Del Corpo O, et al. On the Treatment of Pneumocystis jirovecii Pneumonia: Current Practice Based on Outdated Evidence. Open Forum Infect Dis. 2021;8(12):ofab545.
Smego RA Jr, Nagar S, Maloba B, Popara M. A meta-analysis of salvage therapy for Pneumocystis carinii pneumonia. Arch Intern Med. 2001;161(12):1529–33.
Charpentier E, Ménard S, Marques C, Berry A, Iriart X. Immune Response in Pneumocystis Infections According to the Host Immune System Status. J Fungi (Basel). 2021;7(8).
Fraser TN, Avellaneda AA, Graviss EA, Musher DM. Acute kidney injury associated with trimethoprim/sulfamethoxazole. J Antimicrob Chemother. 2012;67(5):1271–7.
Briceland LL, Bailie GR. Pentamidine-associated nephrotoxicity and hyperkalemia in patients with AIDS. DICP. 1991;25(11):1171–4.
Mühlethaler K, Bögli-Stuber K, Wasmer S, et al. Quantitative PCR to diagnose Pneumocystis pneumonia in immunocompromised non-HIV patients. Eur Respir J. 2012;39(4):971–8.
Medical Writing, Editorial, and Other Assistance.
None.
Funding
The project was supported by the National Center for Advancing Translational Sciences, National Institutes of Health (NIH), through Grant UL1 TR002014. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. No funding or sponsorship was received for the publication of this article.
Author information
Authors and Affiliations
Contributions
Shane Stone, Maria P. Henao, Timothy J. Craig, and Taha Al-Shaikhly contributed to the study conception and design. Formal analysis and investigation were performed by Taha Al-Shakhily and Shane Stone. The first draft of the manuscript was written by Shane Stone and Taha Al-Shaikhly and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
Shane Stone has nothing to disclose. Paula M Henao has nothing to disclose. Taha Al-Shaikhly has nothing to disclose. Timothy J. Craig is an editorial board member of the Pulmonary Therapy journal. Timothy J. Craig was not involved in the selection of peer reviewers for the manuscript nor any of the subsequent editorial decisions. Timothy J. Craig has received research support from Astria, BioMarin, CSL Behring, GSK, Grfols, Intellia, Ionis, KalVista, Pfizer, Pharvaris, Regeneron, and Takeda; speaking fees from CSL Behring, Grifols, and Takeda; consult- ant fees from Astra, Biocryst, BioMarin, CSL Behring, Intellia, Ionis, Kalvista, and Takeda. He also has center designations from International Hereditary Angioedema Association and Alpha-1 Foundation and is a member of the Medical Advisory Board for the HAE-A.
Ethics/Ethical Approval
Ethical approval was not required for this study. This retrospective study is exempt from informed consent. The data reviewed is a secondary analysis of existing data, does not involve intervention or interaction with human subjects, and is de-identified per the de-identification standard defined in §164.514(a) of the HIPAA Privacy Rule. The process by which the data is de-identified is attested to through a formal determination by a qualified expert as defined in §164.514(b)(1) of the HIPAA Privacy Rule. This formal determination by a qualified expert refreshed on December 2020.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Stone, S., Henao, M.P., Craig, T.J. et al. Impact of Sulfonamide Allergy Label on Clinical Outcomes in Patients with Pneumocystis jirovecii Pneumonia. Pulm Ther (2024). https://doi.org/10.1007/s41030-024-00260-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41030-024-00260-4