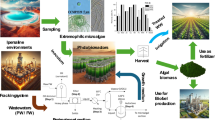
Abstract
The environmental consequences of desalination concentrate disposal have limited the practical adoption of desalination systems for inland brackish water. Desalination concentrate, which is generated by desalination facilities, has the ability to offer water and nutrients for microalgal growth. A useful application for concentrate from desalination systems is required to boost the feasibility of installing desalination procedures for both inland brackish and seawater plants. Several research has been conducted to investigate the use of desalination concentrate as a medium for microalgal culture. This paper reviews the impact of desalination concentrate on microalgal productivity by describing instances of microalgae cultivated in desalination concentrate. Based on the research results, it was found that Chlorella vulgaris, Scendesmus quadricauda, S. platensis, Nannochloropsis oculata and Dunaliella tertiolecta can be cultivated on desalination brine. Also, the paper reviews the different applications of these types which may contribute to adding revenue that will reduce the cost of desalinated water.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
With the increase in human population and water scarcity, the desalination of seawater became the only choice for new water resource. However, desalination operations depend on high power consumption that raises the cost of production to 3–7 times than other water resources (Pistocchi et al. 2020; Dhakal et al. 2022). In recent years, the cultivation of microalgae has been a common trend among several countries as it serves different types of benefits and can be used in different forms. Microalgae can grow in either saline or freshwater under different conditions hence; showing the adaptability of this type of plant-like organism (Sathasivam et al. 2017). Different types of microalgae that can accommodate saline conditions, such as Chlorella and Scenedesmus are the most reported active types cultivated at high salinities. There are plenty of usages for microalgae such as pond stabilization since they can tolerate high amounts of salts (Rahman et al. 2020; Araújo et al. 2021).
Growth conditions for microalgae in brine water
The salinity of brine water used to cultivate microalgae ranges from brackish water to high salinity water, and oil content up to 100 ppm (Sahle-Demessie et al. 2019) as presented in Tables 1 and 2. However, there are specific conditions regarding the cultivation of microalgae, comprising the microalgae constant need for aeration and small quantities of nutrients (Sandeep et al. 2013). Aside from the salinity level that varies from different species to another, pH should be at an alkaline level.
There are several species of microalgae that can adapt to reside in highly saline waters which contribute to biomass production. One of the main species is the cyanobacterium species such as Spirulina platensis, Spirulina maxima and Spirulina argentina (Sánchez et al. 2015). Spirulina or the blue-green microalgae can be cultivated in different types of media including brine medium. It has several benefits and antioxidant factors that can help in human health and can serve as a nutritional factor (Ku et al. 2013; Wells et al. 2017). Mexico is the biggest country that produces Spirulina with over 3000 tons per year. WHO announced theat Spirulina is known as ‘superfood’ meaning that it consists of high nutrient levels (Sandeep et al. 2013).
However, several studies investigate salt stress focusing on four types of microalgae species which are Scenedesmus, Chlorella, Dunaliella and Nannochloropsis (Dolganyuk et al. 2020; Arash et al. 2021 and Udayan et al. 2022). Furthermore, Scenedesmus is a type of freshwater microalgae that can adapt to severe marine environments (Gigante 2013). After different studies on Scenedesmus; it was found that this genus is successful in adaptation to brine water, in addition, it can highly absorb nitrogen, potassium, calcium, sodium, and phosphorus (El-Sayed et al. 2010; El-Sheekh et al. 2018). Scenedesmus is rich in protein and has been seen as a biofuel production tool due to its lipid accumulation capability (Ho et al. 2010; Valdez-Ojeda et al. 2015 and Pushpakumari-Kudahettige et al. 2018). Scenedesmus are highly affected by the pH and light intensity, these two factors have a direct relation to the growth of Scenedesmus and its mass production. Scenedesmus prefers pH of 6.5 as the pH increases the growth rate of the micro microalgae decreases (Fettah et al. 2022; Difusa et al. 2015).
Moreover, Chlorella which is a freshwater alga that consists of proteins and B-complex vitamins that can be extracted for medical purposes, adapted to live in saline conditions (Wells et al. 2017). Moreover, Chlorella been used to remove the actual nutrients from wastewater (Ismail et al. 2017; Chamberlin et al. 2018; Znad et al. 2018). Znad et al. (2018), experimented nutrient removal capability of Chlorella from primary wastewater, secondary wastewater, and petroleum effluent. It was found that Chlorella could remove 100 and 82 of macro-nutrients (N and P) in primary wastewater. Matos et al. (2017), cultivated Chlorella in different brine concentration levels, while Wang et al. (2016) conclude that Chlorella was able to survive in conditions of 50 g L−1.
Dunaliella, another type of microalgae that can withstand high variances of pH, temperature, and salinity (Rodríguez-DeLaNuez et al. 2012; Oren 2014), it is also a common resident in salt works (Oren 2014). Since Dunaliella can adapt to different environmental conditions, its effect on nutrient uptake due to salt stress has been presented in a few studies. Moreover, other species like Stichococcus, Dunaliella salina and Stephanoptera gracilis are species that can tolerate high temperatures and saline water (Van and Glaser 2022; Javor 1989). Not only that but microalgae such as N. pseudostigmata (UTEX 1249) and N. conjuncta (CCAP 254/1), had the highest concentrate tolerance (Hiibel et al. 2015).
Microalgal species respond to different stressors like hypersalinity, for example, D. salina responds to the high osmotic pressure of the hypersaline environment by increasing the production of glycerol. This occurs via starch degradation and shifting the use of carbon flow towards glycerol biosynthesis which acts as an osmotic element. The outcome is the ability of the cells to survive in the hypersaline environment (Sedjati et al. 2019).
Growing microalgae with desalination brine water has many economic and environmental advantages. This system has lower cost, smoother construction, no energy demands and it valorizes a waste product (El Sergany et al. 2014).
Potential of microalgae mass production in desalination brine
Several features and conditions influence microalgae growth and biomass output. Generally, microalgae need both light and nutrients at a certain temperature to grow properly. However, too much light or Oxygen might have a detrimental impact on the development of microalgae, the type of reliance is inextricably linked to the specific microalgae species. Some of them grow well at low temperatures and light intensities, while others require more light. Other considerations, such as the balance between operating parameters (levels of Oxygen and CO2), pH, and product quality, must be considered while selecting the best species concentration or water consumption.
Different pilot plants were done in open-air under environmental conditions for microalgae cultivation, this type of cultivation consists of storage tanks, aerators, and manual microalgae separator units (El Sergany et al. 2014). Basins are usually used to place different total dissolved solids (TDS) concentrations and apply different concentrations of artificial saline water. For example, Scenedesmus microalgae were added with a rate of 0.4 lit/basin/run to treat the saline water for up to 7 retention days (El Sergany et al. 2014). Another study grew Dunaliella in a growth chamber with an 18/6 h light/dark cycle at 26/20 °C and 140 μmol m−2 s−1 light intensity that contained fluorescent and incandescent lamps, within an aquarium where atmospheric air was pumped through a 2-L flask with sterile water (Hiibel et al. 2015). A pilot plant was created for Scenedesmus where it was operated under climatic conditions such as temperature, sunlight, and humidity with three storage tanks. Such tanks were divided into three parallel equal parts (El Sergany et al. 2014). Another cultivation medium contains artificial seawater (33 g L−1 natural salt) with 2.5 g L−1 NaHCO3 that took place in 720 mL glass photobioreactors to cultivate the cyanobacterium Arthrospira platensis (Markou et al. 2020).
Spirulina is usually produced by either culture pond or open pond methods, which is a rectangular pond that is opened and exposed to sunlight, this shape and form helps the water to flow in a circulation that prevents any stagnation point (Shimamatsu 2004). While in a large-scale cultivation process, Spirulina can be produced in open ponds and open raceways ponds, ponds can be up to 605 m2 and a volume of 193 m3. The operating cost per unit area for Spirulina production can vary according to the materials used in the construction of ponds, volume, space, and concentrate optimization (Bioeng 2020).
Mass production of Scenedesmus takes place using Raceways bioreactor to have suitable conditions for the cultivation and production of this type of microalgae. The Institute for Agricultural and Fisheries Research and Training (IFAPA) in Almería, Spain, is cultivating Scenedesmus in raceway bioreactors inside a greenhouse to have full control of the optimization of conditions for mass production. The scale of such a cultivation process can be limited by the feasibility of smart greenhouses, yet Scenedesmus can be grown outside in photobioreactors on a large scale to produce biodiesel. The efficiency and growth rate are highly affected by the seasons, the highest growth rates will be found in August, September, and October. Operating cost of such a method is relatively low compared to smart greenhouses, however, its mass production and efficiency is highly dependent on the season and time (Darwish et al. 2015).
Chlorella vulgaris is one of the most used commercial types of microalgae, as it can be cultivated under different conditions. Chlorella can be cultivated for mass production in a short time and without control factors. Chlorella grows faster in fresh water under sunlight, so the best method for its cultivation is open-air ponds, but different pilot bioreactors were established to cultivate Chlorella under salt stress conditions. It turned out that the salt stress condition has repeatedly enhanced some of the fatty acid releases and lipids that are highly valuable (Ali et al. 2020).
Recent applications of cultivating microalgae in desalination brine water
Microalgae production utilizing brine generated by desalination facilities has recently been investigated as presented in Table 3. Dunaliella salina, has gained the most attention due to its exceptional environmental adaption, which includes the production of huge amounts of carotenoids and glycerol (Raja et al. 2007 and Giwa et al. 2017). A pilot scale study was carried out to develop Dunaliella salina in outdoor ponds using brine with salinity ranging from 40,000 to 80,000 ppm. It resulted in brine salinity reductions ranging from 13 to 63%, depending on brine content, duration in ponds, and changes in climatic conditions (El Sergany et al. 2014).
Uses of the cultivated microalgae
There are multiple uses for cultivated microalgae, such as Spirulina, Dunaliella spp., in brine water. They can be utilized in the food industry, cosmetics, as well as pharmaceutical industry where specific nutrients exist such as the antioxidant agent, phycocyanin (Sandeep et al. 2013) and β-carotene which is converted to vitamin A which is essential for human health. Dunaliella sp. is highly used in pharmaceutical industries as it has the capacity to produce two main products which are β-carotene and glycerol. Many countries cultivate Dunaliella sp. for the use of β-carotene and glycerol such as Australia, Kuwait, Chile, and Spain (Oren 2014). Recent studies proved that Nannochloropsis could produce fatty acids and proteins which are essential in food production (Hulatt et al. 2017).
In addition, microalgae are utilized in agricultural production as an organic biofertilizer stimulating the growth of the plants as well as improving the soil properties (Piwowar and Harasym 2020). Moreover, there are several ways in using microalgae in brine aside from using it for agriculture purposes, in which it can be utilized to detoxify minerals and heavy metals (Sánchez et al. 2015). In addition to that, the microalgae can remove calcium and salts from their own growth mediums reducing the overall brine salinity by 20 percent which decreases the environmental impact of brine disposal (Figler et al. 2019).
Furthermore, using microalgae as a source of energy, the biomass could be harvested to produce energy as biofuel. The genus Nannochloropsis has gained a strong position in the biofuel research field. Plenty of studies were tested on several strains which can promote lipid production. Additionally, the ability of microalgae to utilize wastewater nutrients and saltwater for growth not only reduces the production costs by replacing the commercial growth medium but also purifies the wastewater for reuse in irrigation and other commercial applications (Sheets et al. 2014; Mitra and Mishra 2019).
Moreover, green microalgae are used for the remediation of industrial wastewater generated from natural gas productions with a TDS level of up to 25,000 ppm and oil content of up to 100 ppm (El Sergany et al. 2014; Ismail 2021). Another potential application of microalgal biomass cultivated in brine would be pigments’ production and as animal or fish fodders (El Sergany et al. 2014).
The actual cost of phycocyanin is 0.3 USD per mg which makes it very attractive for businesses to invest in such a component (Sandeep et al. 2013). For example, Chile produces about 30 tons per year of microalgae and the estimated production cost of dried Spirulina is 11.4 USD dollars per kg which is highly affordable for businesses (Sánchez et al. 2015).
Conclusion
The environmental consequences of concentrate disposal have limited the practical adoption of desalination systems for inland brackish water, diminishing desalination's capacity to solve worldwide water shortages. A useful application for concentrate from desalination systems is required to boost the feasibility of installing desalination procedures for both inland brackish and seawater. The concept of growing microalgae in the concentrate stream to ease desalination concerns while also satisfying energy demands by supplying feedstock for biofuel production. Based on the research results, five main species can be cultivated on desalination brine. These types include Chlorella vulgaris, Scendesmus quadricauda, S. platensis, Nannochloropsis oculata and Dunaliella tertiolecta. They can tolerate brine salinity from 6000 to 80,000 mg L−1. Cultivation of marine microalgal species in brine concentrates from water desalination units might be a potential method of removing contaminants while also creating feedstock for biofuel production.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Ali HEA, El-fayoumy EA, Rasmy WE, Soliman RM, Abdullah MA (2020) Two-stage cultivation of Chlorella vulgaris using light and salt stress conditions for simultaneous production of lipid, carotenoids, and antioxidants. J Appl Phycol 33(1):227–239. https://doi.org/10.1007/s10811-020-02308-9
Almaraz N, Regnery J, Vanzin GF, Riley SM, Ahoor DC, Cath TY (2020) Emergence and fate of volatile iodinated organic compounds during biological treatment of oil and gas produced water. Sci Total Environ 699:134202
Almutairi AW, El-Sayed AEB, Reda MM et al (2021) Evaluation of high salinity adaptation for lipid bio-accumulation in the green microalga Chlorella vulgaris. Saudi J Biol Sci 28(7):3981–3988. https://doi.org/10.1016/j.sjbs.2021.04.007
Ammar SH, Khadim HJ, Mohamed AI (2018) Cultivation of Nannochloropsis oculata and Isochrysis galbana microalgae in produced water for bioremediation and biomass production. Environ Technol Innov 10:132–142. https://doi.org/10.1016/j.eti.2018.02.002
Arash M, Linhua F, Felicity AR (2021) Impact of microalgae species and solution salinity on algal treatment of wastewater reverse osmosis concentrate. Chemosphere 285:131487. https://doi.org/10.1016/j.chemosphere.2021.131487
Araújo R, Vázquez-Calderón F, Sánchez-López J, Azevedo IC, Bruhn A, Fluch S, Garcia-Tasende M, Ghaderiardakani F, Ilmjärv T, Laurans M, Mac-Monagail M, Mangini S, Peteiro C, Rebours C, Stefansson T, Ullmann J (2021) Current status of the microalgae production industry in Europe: an emerging sector of the blue bioeconomy. Front Mar Sci 7:626389. https://doi.org/10.3389/fmars.2020.626389
Bioeng F (2020) Large-scale cultivation of Spirulina for biological CO2 mitigation in open raceway ponds using purified CO2 from a coal chemical flue gas. Frontiers 7:441
Chamberlin J, Harrison K, Zhang W (2018) Impact of nutrient availability on tertiary wastewater treatment by Chlorella vulgaris. Water Environ Res 90(11):2008–2016. https://doi.org/10.2175/106143017X15131012188114
Daliry S, Hallajisani A, Mohammadi Roshandeh J, Nouri H, Golzary A (2017) Investigation of optimal condition for Chlorella vulgaris microalgae growth. Glob J Environ Sci Manage 3(2):217–230. https://doi.org/10.22034/gjesm.2017.03.02.010
Darwish A, Mostafa E, Alamo EB, Ahmad AE (2015) Biodiesel production from Scenedesmus obliquus cultivated in outdoor conditions at large scale. Delta J Sci 201537:190–195
Dhakal N, Salinas-Rodriguez SG, Hamdani J, Abushaban A, Sawalha H, Schippers JC, Kennedy MD (2022) Is desalination a solution to freshwater scarcity in developing countries? Membranes 12:381. https://doi.org/10.3390/membranes12040381
Difusa A, Talukdar J, Kalita MC, Mohanty K, Goud VV (2015) Effect of light intensity and pH condition on the growth, biomass and lipid content of microalgae Scenedesmus species. Biofuels 6(1–2):37–44. https://doi.org/10.1080/17597269.2015.1045274
Dolganyuk V, Belova D, Babich O, Prosekov A, Ivanova S, Katserov D, Patyukov N, Sukhikh S (2020) Microalgae: a promising source of valuable bioproducts. Biomolecules 10:1153. https://doi.org/10.3390/biom10081153
El Sergany FAR, El Fadly M, El Nadi MHA (2014) Brine desalination by using microalgae ponds under nature conditions. Am J Environ Eng 4(4):75–79. https://doi.org/10.5923/j.ajee.20140404.02
ElBarmelgy AH, Ismail MM, Sewilam H (2021) Biomass productivity of Nannochloropsis sp. grown in desalination brine culture medium. Desalin Water Treat 216:306–314
El-Sayed AB, El-Fouly MM, Abou-El-Nour EAA (2010) Immobilized microalga Scenedesmus sp. for biological desalination of red sea water: I. Effect Growth Nature Sci 8:9
El-Sheekh M, Abomohra AE-F, Eladel H, Battah M, Mohammed S (2018) Screening of different species of Scenedesmus isolated from Egyptian freshwater habitats for biodiesel production. Renew Energy 129:114–120. https://doi.org/10.1016/j.renene.2018.05.099
Fettah N, Derakhshandeh M, Tezcan-Un U et al (2022) Effect of light on growth of green microalgae Scenedesmus quadricauda: influence of light intensity, light wavelength and photoperiods. Int J Energy Environ Eng 13:703–712. https://doi.org/10.1007/s40095-021-00456-3
Figler A, Béres V, Dobronoki D, Márton K, Nagy SA, Bácsi I (2019) Salt tolerance and desalination abilities of nine common green microalgae isolates. Water 11(12):2527. https://doi.org/10.3390/w11122527
Gigante BM (2013) Saline adaptation of the microalga Scenedesmus dimorphus from fresh water to brackish water [Master's thesis, Cleveland State University]. In: OhioLINK Electronic Theses and Dissertations Center. http://rave.ohiolink.edu/etdc/view?acc_num=csu1382355969
Giwa A, Dufour V, Al Marzooqi F, Al Kaabi M, Hasan SW (2017) Brine management methods: recent innovations and current status. Desalination 407:1–23. https://doi.org/10.1016/j.desal.2016.12.008
Gu N, Lin Q, Li G, Tan Y, Huang L, Lin J (2012) Effect of salinity on growth, biochemical composition, and lipid productivity of Nannochloropsis oculata CS 179. Eng Life Sci 12(6):631–637. https://doi.org/10.1002/elsc.201100204
Hiibel SR, Lemos MS, Kelly BP, Cushman JC (2015) Evaluation of diverse microalgal species as potential biofuel feedstocks grown using municipal wastewater. Front Energy Res 3:893. https://doi.org/10.3389/fenrg.2015.00020
Ho S-H, Chen W-M, Chang J-S (2010) Scenedesmus obliquus CNW-N as a potential candidate for CO2 mitigation and biodiesel production. Bioresour Technol 101(22):8725–8730. https://doi.org/10.1016/j.biortech.2010.06.11
Hulatt CJ, Wijffels RH, Bolla S, Kiron V (2017) Production of fatty acids and protein by Nannochloropsis in flat-plate photobioreactors. PLoS ONE 12(1):e0170440. https://doi.org/10.1371/journal.pone.0170440
Ismail MM (2021) Dual benefits of microalgae in bioremediation, pollutant removal and biomass valorization, a review. In: Bidoia ED, Montagnolli RN (eds) Biodegradation, pollutants and bioremediation principles. CRC Press; Taylor & Francis Group, Florida, pp 174–192. https://doi.org/10.1201/9780429293931-9
Ismail MM, Essam TM, Ragab YM, El-Sayed ABE, Mourad FE (2017) Remediation of a mixture of analgesics in a stirred-tank photobioreactor using microalgal-bacterial consortium coupled with attempt to valorise the harvested biomass. Bioresour Technol 232:364–371. https://doi.org/10.1016/j.biortech.2017.02.062
Javor B (1989) Hypersaline environments. Brock/springer Ser Contemp Biosci. https://doi.org/10.1007/978-3-642-74370-2
Kim HW, Park S, Rittmann B (2015) Multi-component kinetics for the growth of the cyanobacterium Synechocystis sp. PCC6803. Environ Eng Res 20(4):347–355. https://doi.org/10.4491/eer.2015.033
Ku CS, Yang Y, Park Y, Lee J (2013) Health benefits of blue-green microalgae: prevention of cardiovascular disease and nonalcoholic fatty liver disease. J Med Food 16(2):103–111. https://doi.org/10.1089/jmf.2012.2468
Maeng SK, Khan W, Park JW, Han I, Yang HS, Song KG, Kim H-C (2018) Treatment of highly saline RO concentrate using Scenedesmus quadricauda for enhanced removal of refractory organic matter. Desalination 430:128–135. https://doi.org/10.1016/j.desal.2017.12.056
Markou G, Diamantis A, Arapoglou D, Mitrogiannis D, González-Fernández C, Unc A (2020) Growing Spirulina (Arthrospira platensis) in seawater supplemented with digestate: trade-offs between increased salinity, nutrient and light availability. Biochem Eng J 2020:107815. https://doi.org/10.1016/j.bej.2020.107815
Matos ÂP, MoeckeSant’Anna EHSES (2017) The use of desalination concentrate as a potential substrate for microalgae cultivation in Brazil. Algal Res 24:505–508. https://doi.org/10.1016/j.algal.2016.08.003
Mitra M, Mishra S (2019) A biorefinery from Nannochloropsis spp. utilizing wastewater resources. Appl Microalgae Wastewater Treatment 2019:123–145. https://doi.org/10.1007/978-3-030-13909-4_6
Myint MT, Hussein W, Ghassemi A (2015) Microalgal process for treatment of high conductivity concentrates from inland desalination. Desalin Water Treatment 2015:1–9. https://doi.org/10.1080/19443994.2014.993715
Nascimento-Mata S, de-Souza-Santos T, Guimarães-Cardoso L, Bomfim-Andrade B, Hartwig-Duarte J, Costa JAV, Druzian JI (2020) Spirulina sp. LEB 18 cultivation in a raceway-type bioreactor using wastewater from desalination process: production of carbohydrate-rich biomass. Bioresour Technol 2020:123495. https://doi.org/10.1016/j.biortech.2020.12349
Oren A (2014) The ecology of Dunaliella in high-salt environments. J Biol Res (thessalonike, Greece) 21(1):23. https://doi.org/10.1186/s40709-014-0023-y
Pistocchi A, Bleninger T, Breyer C, Caldera U, Dorati C, Ganora D, Zaragoza G (2020) Can seawater desalination be a win-win fix to our water cycle? Water Res 2020:115906. https://doi.org/10.1016/j.watres.2020.115906
Piwowar A, Joanna H (2020) The Importance and Prospects of the Use of Microalgae in Agribusiness. Sustainability 12(14):5669. https://doi.org/10.3390/su12145669
Pushpakumari-Kudahettige N, Pickova J, Gentili FG (2018) Stressing Microalgae for Biofuel Production: Biomass and Biochemical Composition of Scenedesmus dimorphus and Selenastrum minutum Grown in Municipal Untreated Wastewater. Front Energy Res 2019:6. https://doi.org/10.3389/fenrg.2018.00132
Rahman A, Agrawal S, Nawaz T, Pan S, Selvaratnam T (2020) A review of microalgae-based produced water treatment for biomass and biofuel production. Water 12(9):2351. https://doi.org/10.3390/w12092351
Raja R, Hemaiswarya S, Rengasamy R (2007) Exploitation of Dunaliella for carotene production. Appl Microbiol Biotechnol 74:517–523. https://doi.org/10.1007/s00253-006-0777-8
Rodríguez-DeLaNuez F, Franquiz-Suárez N, Santiago DE, Veza JM, Sadhwani JJ (2012) Reuse and minimization of desalination brines: a review of alternatives. Desalin Water Treat 39(1–3):137–148. https://doi.org/10.1080/19443994.2012.669168
Sahle-Demessie E, Aly Hassan A, El Badawy A (2019) Bio-desalination of brackish and seawater using halophytic microalgae. Desalination 465:104–113. https://doi.org/10.1016/j.desal.2019.05.002
Sánchez AS, Nogueira IBR, Kalid RA (2015) Uses of the reject brine from inland desalination for fish farming, Spirulina cultivation, and irrigation of forage shrub and crops. Desalination 364:96–107. https://doi.org/10.1016/j.desal.2015.01.034
Sandeep KP, Shukla SP, Harikrishna V, Muralidhar AP, Vennila A, Purushothaman CS, Ratheesh Kumar R (2013) Utilization of inland saline water for Spirulina cultivation. J Water Reuse Desalin 3(4):346–356. https://doi.org/10.2166/wrd.2013.102
Sathasivam R, Radhakrishnan R, Hashem A, Abd-Allah EF (2017) Microalgae metabolites: a rich source for food and medicine. Saudi J Biol Sci. https://doi.org/10.1016/j.sjbs.2017.11.003
Sedjati S, Santosa G, Yudiati E, Supriyantini E, Ridlo A, Kimberly F (2019) Chlorophyll and carotenoid content of Dunaliella salina at various salinity stress and harvesting time. IOP Conf Ser Earth Environ Sci 246:012025. https://doi.org/10.1088/1755-1315/246/1/012025
Sheets JP, Ge X-M, Park SY, Li Y-B (2014) Effect of outdoor conditions on Nannochloropsis salina cultivation in artificial seawater using nutrients from anaerobic digestion effluent. Bioresour Technol 152:154–161. https://doi.org/10.1016/j.biortech.2013.10.115
Shimamatsu H (2004) Mass production of Spirulina, an edible microalga. Hydrobiologia 512(1–3):39–44. https://doi.org/10.1023/b:hydr.0000020364.237
Shirazi SA, Rastegary J, Aghajani M, Ghassemi A (2018) Simultaneous biomass production and water desalination concentrate treatment by using microalgae. Desalin Water Treatment 135:101–107
Talebi AF, Dastgheib SMM, Tirandaz H, Ghafari A, Alaie E, Tabatabaei M (2016) Enhanced algal-based treatment of petroleum produced water and biodiesel production. RSC Adv 6:47001–47009
Udayan A, Pandey AK, Sirohi R, Sreekumar N, Sang BI, Sim SJ, Kim SH, Pandey A (2022) Production of microalgae with high lipid content and their potential as sources of nutraceuticals. In: Phytochemistry reviews: proceedings of the Phytochemical Society of Europe, pp 1–28. https://doi.org/10.1007/s11101-021-09784-y
Valdez-Ojeda R, González-Muñoz M, Us-Vázquez R, Narváez-Zapata J, Chavarria-Hernandez JC, López-Adrián S, Escobedo-Gracia Medrano RM (2015) Characterization of five fresh water microalgae with potential for biodiesel production. Algal Res 7:33–44. https://doi.org/10.1016/j.algal.2014.11.009
Van AT, Glaser K (2022) Pseudostichococcus stands out from its siblings due to high salinity and desiccation tolerance. Phycology 2(1):108–119. https://doi.org/10.3390/phycology2010007
Wang T, Ge H, Liu T, Tian X, Wang Z, Guo M, Chu J, Zhuang Y (2016) Salt stress induced lipid accumulation in heterotrophic culture cells of Chlorella protothecoides: mechanisms based on the multi-levelanalysis of oxidative response, key enzyme activity and biochemicalalteration. J Biotechnol 228:18–27
Wells ML, Potin P, Craigie JS, Raven JA, Merchant SS, Helliwell KE, Smith AG, Camire ME, Brawley SH (2017) Microalgae as nutritional and functional food sources: revisiting our understanding. J Appl Phycol 29(2):949–982. https://doi.org/10.1007/s10811-016-0974-5
Znad H, Al Ketife AM, Judd S, AlMomani F, Vuthaluru HB (2018) Bioremediation and nutrient removal from wastewater by Chlorella vulgaris. Ecol Eng 110:1–7. https://doi.org/10.1016/j.ecoleng.2017.10.008
Acknowledgements
The American University in Cairo's Center for Applied Research on the Environment and Sustainability (CARES), School of Science and Engineering, provided funding for this research.
Funding
Open Access funding enabled and organized by Projekt DEAL. Not applicable.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Al Bazedi, G., Ismail, M.M., Mugwanya, M. et al. Desalination concentrate microalgae cultivation: biomass production and applications. Sustain. Water Resour. Manag. 9, 108 (2023). https://doi.org/10.1007/s40899-023-00887-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40899-023-00887-2