Abstract
Purpose
At present, autologous platelet-derived therapies such as platelet-rich plasma is widely used in the clinic, but allogeneic and xenogeneic therapies are currently in the research stage, mainly due to their biocompatibility in vivo. Platelet lysates depleted of antigens such as blood cells are a potential solution for allogeneic or xenogeneic applications. Platelet lysates can successfully promote the growth and differentiation of xenogeneic cells in vitro, but in vivo data are not yet available. This study aims to evaluate whether porcine platelet lysate can effectively avoid inflammatory reaction in rabbit knee joint, and then treat cartilage defect and arthritis.
Methods
We developed porcine platelet lysates containing undetectable antigens such as blood cells and complement. For xenogeneic application, platelet lysate was injected into rabbit knee joints to observe joint responses. To examine cartilage repair, osteochondral defects were created in rabbit knee joints. Rabbits were sacrificed three months after treatment with platelet lysate to observe cartilage regeneration and arthritis.
Results
The tissue sections in the rabbit knee joints showed no inflammatory reaction. Furthermore, the injection of platelet lysate was found to effectively inhibit the formation of cartilage arthritis in rabbit knee joints.
Conclusion
Our experimental results show that xenogeneic platelet lysate is a safe and effective method in the treatment of arthritis, which can be used as a research basis for future medical applications. The use of xenogeneic platelet lysate for regenerative therapy in vivo is feasible.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Regenerative medicine is recognized as one of the most promising osteoarthritis treatments and has been extensively studied over the past few decades [1, 2], and platelet-derived therapy is one of its most popular applications [3,4,5,6,7]. Growth factors released by α-granules contained in platelets are a group of bioactive polypeptides with anabolic and anti-catabolic effects that can promote cell production, regulate new cartilage formation and chondrocyte metabolism and differentiation, and promote tissue and cartilage repair [8, 9]
At present, autologous platelet-rich plasma (PRP) is widely used in clinical practice. The advantage of autologous PRP is that there is no rejection problem. After centrifuging the autologous blood, the platelet dense layer is extracted and then injected into the affected area. However, inconveniences remain in clinical use as it is time-consuming and the final PRP growth factor concentration can vary. Many studies have been carried out to ameliorate this disadvantage. The production and extraction of recombinant human growth factors is one of the sources [10], but it provides a single growth factor and carries concerns regarding carcinogenicity [11]. The development of allogeneic platelet-derived preparations has also been discussed. But the allogeneic PRP still has the problem of certain unremoved antigens, which reduces the tissue regeneration capability of the growth factors [12]. Therefore, platelet lysate (PL) is a potential application method that further removes antigens. PL is using physical methods to destroy the membrane of the platelets in PRP. Filter large proteins and platelet fragments and then heat to remove complements, so that possible antigens can be removed and obtained growth factors from platelets. Platelet lysate, as well as other autologous platelet-derived preparations as a source of growth factors and cytokines, have been successfully used in the regenerative engineering of bone and cartilage [13,14,15]. Platelet lysate has been widely used in cell culturing as a replacement for fetal bovine serum to obtain a better cell culturing effect. Autologous PL has been a replacement for PRP in clinical practice. Clinical evidence and products of autologous PL have been launched but allogeneic and xenogeneic PL still needs more research to confirm its effectiveness. Aldén et al. used porcine PL to culture monkey and hamster cells and obtained good differentiation and value-added effects [16]. There is still no paper published on the safety and efficacy of xenogeneic PL in vivo. We designed an experiment to conduct an in vivo study of xenogeneic PL to confirm its safety and efficacy. In the future, it can be developed for the treatment of cartilage damage and arthritis in animals, and further applied to the regenerative treatment of humans and other tissues with xenogeneic PL. Herein, we developed porcine PL containing undetectable antigens such as blood cells and complement. The porcine PL was injected into the rabbits’ joints to observe whether it caused an inflammatory reaction. Then, osteochondral defects were created in rabbits’ knee joints and they were injected with porcine PL to observe the repair of osteochondral defects and the development of knee joint arthritis.
2 Materials and Methods
2.1 PL Preparation
Porcine platelet lysate was produced following the protocol of Jonsdottir-Buch SM et al. [17]. Porcine whole blood was centrifuged and the supernatant was taken, leukocytes were filtered, platelets were lysed by the freeze–thaw method, heated to remove the complement, and filtered with a 0.22 µm pore size filter. The blood cell count was performed and the growth factor concentration of the lysate was detected by TGF-ß ELISA Kit. Growth factors in porcine blood PL include PDGF, EGF, TGF-α, β, VEGF, and FGF, which exist in fixed proportions. In this study, TGF-β was used as the representative for detection. The measurement of TGF-β concentration in samples was based on the recommended protocols of TGF-β DuoSet ELISA Kit (catalog number DY240-05, R&D Systems, MN, USA) and R&D DuoSet Ancillary Reagent Kit 1 (catalog number DY007, R&D Systems, MN, USA).
Figure 1 shows the TGF-β standard curve obtained by following the aforementioned protocol. Once the concentration of TGF-β was determined for each batch of extraction process of PL, quantitative freeze-dried PL with known TGF-β was weighted and diluted with normal saline for the subsequent animal experiments.
2.2 Inflammation Assessment After PL Injection in Rabbit Knee Joint
A biocompatibility test was designed for the New Zealand rabbit’s hind knee joint after PL injection (IACUC Approval No. 21T10-10, Master Laboratory Co. Ltd., Taiwan). The animals used in this experiment were six male New Zealand rabbits weighing 3.0–3.5 kg (N = 6). This animal experiment followed the guidelines of ISO 10993–6:2016 edition for local effects after implantation issued by International Organization for Standardization (ISO). In this study, one hind knee joint of each rabbit was randomly selected as the experimental group, and the other hind knee joint was selected as the control group (normal saline). On day 1, 2, and 4, the joints in the experimental group were injected with 0.3 mL of PL with a total content of 7.64 ng of TGF-β (three joints) and 0.3 mL of PL with a total content of 3.82 ng of TGF-β (three joints) in the experimental group. The PL was injected into the joint cavity with injection site lateral to the patella tendon. The control group were injected with 0.3 mL of normal saline. During the test, observations were made of whether the injection site was red and swollen, and whether the physiological functions (such as changes in diet and weight) were normal. On day 14 of the experiment, the experimental animals were sacrificed and the tested joints were taken for pathological section and staining to observe the inflammation condition.
Table 1 shows the inflammation scores in ISO10993-6:2006 edition. The total score for the experimental group was subtracted from the total score for the control group to obtain the inflammatory reaction rating.
2.3 Osteochondral Defect Experiment
A New Zealand rabbit hind knee joints osteochondral defect regeneration experiment was designed (IACUC Approval No. 22T10-10, Master Laboratory Co. Ltd., Taiwan). The animals used in this experiment were three male New Zealand rabbits weighing 3.0–3.5 kg (N = 3). This animal experiment followed the guidelines of ISO 10993–6:2016 edition. In this experiment, the experimental group and the control group were randomly selected. The femur trochlea groove of the left and right hind knee joints were used to create osteochondral defects of 4 mm diameter and 4 mm depth. On the day after the operation and the second and fourth weeks after the operation, the experimental group was injected with PL 0.5 mL (the total content of TGF-β was 25 ng). In our previous studies [18, 19], 10 ng of TGF-β can promote cartilage regeneration. The control group was without the injection of TGF-β. The PL was injected into the joint cavity with injection site lateral to the patella tendon. The animals were sacrificed at the 12th week and the appearance of osteochondral regeneration and pathological sections were observed. The repaired cartilage was scored using the ICRS scoring system for macroscopic evaluation [20] and histological assessment [21] (Table 2).
3 Results
3.1 PL Preparation
Porcine whole blood was centrifuged, the supernatant was taken to filter out white blood cells, the platelets were lysed by the freeze–thaw method for five times, and the complement and protein were removed by heating and then filtered through a 0.22 µm filter to complete the PL preparation process. The PL sample was performed the blood count and biochemical analysis. The blood count showed that the white blood cell, red blood cell, and platelet counts were all zero (Table 3). No residual blood cells were noted. Biochemical analysis showed that the contents of total protein and albumin were trace amounts. This method can remove most antigenic components, including blood cells and proteins.
In our study, it was found that the concentration of growth factor varied with the batches of porcine whole blood and the production process. For the growth factor TGF-β, which can induce the chondrogenesis of stem cells, its concentrations in three different productions (three different porcine) results were 9.6 ng/mL, 12.8 ng/mL, and 6.0 ng/mL, respectively, and their average was 9.5 ng/mL.
3.2 Inflammation Assessment After PL Injection in Rabbit Knee Joints
There was no redness, swelling, or infection in the six experimental rabbits’ joints compared to the control group, which indicated that PL obtained by our method did not cause severe joint inflammation.
In the pathological section, no obvious aggregation of inflammatory cells such as neutrophils, eosinophils, basophils, mast cells, lymphocytes or macrophages were found in cartilage, joint synovium, or meniscus (Figs. 2, 3). Only one experimental rabbit joint (rabbit 1002) injected with TGF-β 7.64 ng showed a notable moderate inflammatory reaction with polymorphonuclear and macrophage infiltration in the synovium region compared to the control group (Fig. 3). For the rabbit (1002), the inflammatory reaction ratings of the experimental joint and the control joint were 15 and 2, respectively and indicated a moderate inflammatory reaction.
Hematoxylin & eosin staining of the control group knee joints (The magnification: 40x). The pathological sections of six rabbits 1001–1006 in the control group (injected with normal saline) showed no abnormal aggregated neutrophils, eosinophils, basophils, mast cells, lymphocytes, and macrophages in articular cartilage (a), meniscus (b) and synovium (c)
Hematoxylin & eosin staining of the experimental group knee joint (The magnification: 40x). The pathological sections of six rabbits (1001 ~ 1006) in the experimental group (rabbits 1001–1003 injected with 7.64 ng TGF-β of PL and rabbits 1004–1006 injected with 3.82 ng TGF-β of PL). The experimental knee joint of 1002 showed moderate inflammation reaction compared to control group, aggregated polymorphonuclear, lymphocytes, and macrophages were noted in the synovium (c) (magnified circle) with cell nuclei stained blue-purple. The rest of the pathological sections showed no abnormal aggregated neutrophils, eosinophils, basophils, mast cells, lymphocytes, and macrophages in articular cartilage (a), meniscus (b) and synovium (c)
3.3 Osteochondral Defect Experiment
An osteochondral defect of 4 mm diameter and 4 mm depth was created in the distal femur patellofemoral surface region of the rabbit hind knee joint. PL was injected into the experimental group on the day (1st) after the operation and the second (2nd) and fourth (4th) weeks after the operation. The rabbits were sacrificed after 12 weeks for the evaluation of cartilage regeneration and arthritis. In macroscopic evaluation, the ICRS cartilage repair score of the control group was 11.7 and that for the experimental group was 19.4. No cartilage regeneration was noted at the defect region in rabbit numbered 2 in the control group (Fig. 4). Both the others in control group and all those in the experimental group showed > 75% cartilage regeneration in the defect area. The most obvious difference in the overall joint appearance evaluation was the occurrence of traumatic arthritis at the distal femur patellofemoral surface. The three joints in the control group had obvious traumatic arthritis (Fig. 4), and none of the experimental group subjects showed traumatic arthritis. The results showed that the injection of porcine-derived PL could effectively slow down the occurrence of arthritis.
Macroscopic observation: the yellow arrows are where the osteochondral defects were created and the red arrows are the distal femur patellofemoral surface. a The control group: the osteochondral defects of rabbits 1 and 3 are covered with new cartilage > 75% area (yellow arrows), while rabbit 2 shows no new cartilage formation. All three control groups had obvious traumatic arthritis in the distal femur patellofemoral surface (red arrows), irregular cartilage surface and subchondral bone exposure were noted. b In the experimental group, > 75% of the osteochondral defect had newly-formed cartilage (yellow arrow). No arthritis was observed in the distal femur patellofemoral surface and the articular surface was smooth indicating and no sign of degeneration (red arrow). (Color figure online)
Histological evaluation of osteochondral defects of those in the experimental group scored 9 points, while those in the control group scored 7.8 points. The regeneration of cartilage defects in the experimental group was slightly better than that in the control group. However, the histological results of cartilage cross section showed that most of the regenerated cartilage was fibrocartilage or incompletely differentiated hyaline cartilage (Figs. 5, 6).
Histological evaluation of the osteochondral defects of knee joints in the control group. Hematoxylin & eosin staining (The magnifications: 40 × and 100x). Red arrows are the osteochondral defect regions. The articular cartilage of rabbit 1 was hypertrophic undifferentiated hyaline cartilage, the defect in rabbit 2 had no cartilage regeneration and the defect in rabbit 3 had a thin layer of fibrocartilage
Histological evaluation of the osteochondral defects of knee joints in the experimental group. Hematoxylin & eosin staining (the magnification: 40 × and 100x). Red arrows are osteochondral defect regions. The articular cartilages for rabbit 1–3 had undifferentiated hyaline cartilage, a thin layer of undifferentiated hyaline cartilage, and fibrocartilage, respectively
4 Discussion
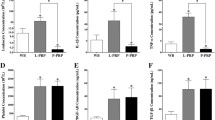
Among inflammatory cytokines, interleukin 1 beta (IL-1β) and tumor necrosis factor-alpha (TNF-α) are considered the major factors in the pathogenesis of osteoarthritis (OA) and leading to chondrocyte death and joint degeneration [22]. Elevated levels of IL-1β and TNF-α have been found in joint fluid affected by OA [23]. Alpha-granules in platelets contain growth factors and proteins have benefits in terms of tissue regeneration [24]. To combat the catabolic milieu of OA-affected joints, PRP is thought to inhibit the catabolic cytokines of IL-1β and TNF-α [25, 26] and to promote the interaction between the cartilage matrix and the paracrine-related factors including fibroblast growth factor, transforming growth factor-beta, vascular endothelial growth factor, and epidermal growth factor [27]. Intra-articular injections of PRP can effectively delay cartilage erosion, but the more effective repair of cartilage defects is to use a composite of PRP and fibrin clots or scaffolds [28].
Platelet lysate contains various growth factors involved in cartilage repair and exerts chondrocyte protection and extracellular matrix modification in a dose-dependent manner. In vitro PL significantly restored TNF-α-inhibited anabolic gene expression (Col2 and aggrecan) and TNF-α increased catabolic gene expression (Col10, Mmp13, Adamts5, and Adamts9) [29] in cartilage repair and arthritis prevention and can hence achieve the same safety and effect as PRP [30].
Camargo Garbin et al. [31] carried out an in vitro cell culture of allogeneic platelet lysate and obtained a good cell proliferation effect. Aldén et al. [16] used porcine PL to culture monkey and hamster cells and obtained good differentiation and value-added effects.
Whether leukocytes should be removed has been discussed in the application of autologous PRP [32]; it is generally believed that high concentrations of leukocytes in PRP preparations can cause inflammation and joint degeneration, and negatively impact the extracellular matrix of cartilage tissue. On allogeneic or xenogeneic platelet-derived preparations, potential antigen removal is very important, especially the removal of leukocytes.
In this study, the preparation process of porcine PL removed leukocytes with a leukocyte filter after centrifugation for removal of erythrocytes. Afterward, a freeze–thaw method was employed to break platelets and to release growth factors, followed by heating the lysates at 56 °C to deactivate the complement. Lastly, a filter of 0.22 μm was used to remove platelet fragments and large proteins. With this process, most antigens including red blood cells, white blood cells, platelets, complement, and large proteins can be effectively removed. As a result, the injection of porcine-derived PL into the knee joint of rabbits did not cause rejection and inflammation reaction. In our osteochondral defect experiment, the injection of porcine-derived PL was significantly effective in preventing the occurrence of arthritis in patellofemoral joints, but for comparison with the control group, the regeneration of the osteochondral defect site had no significant difference, and the treatment of local osteochondral defects with the use of local scaffolds or gels should be able to obtain better therapeutic effects.
The production process of PRP and PL cannot accurately control the amount of platelets or the concentration of growth factors, so each clinical treatment has its own variability. To improve the clinical drawback, freeze-dried PL is one of the solutions. By measuring the concentration of growth factors after PL production, the growth factors required for the injection can be calculated. In addition, freeze-dried PL can maintain good growth factor activity and regeneration ability [29], and can maintain the necessary qualitative and quantitative factors for each injection to achieve a stable therapeutic effect.
Regarding the inflammatory reaction in the rabbit experiment, one rabbit in the experimental group had a moderate inflammatory reaction in the pathological cross section. No redness or swelling was noted in appearance. The other five rabbits in the experimental group showed no inflammatory reaction. The reason may be either that this rabbit was allergic to xenogeneic PL or it sustained injuries due to external forces during the injection process. Future animal experiments can further clarify the problem.
Our experiments provide preliminary evidence for the application of xenogeneic PL in clinical treatment. By removing blood cells, complement, and large proteins in the xenogeneic blood, xenogeneic PL can be safely and effectively used in the treatment of arthritis.
5 Conclusion
The use of xenogeneic PL for regenerative therapy is feasible. Compared with PRP, the PL production process removes possible antigens such as white blood cells, red blood cells, platelets, complement, and large proteins, leaving only the required growth factors to avoid the rejection of xenogeneic sources. Our experimental results show that xenogeneic PL is a safe and effective method in the treatment of arthritis, which can be used as a research basis for future medical applications.
Data Availability Statement
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
References
Filardo, G., Kon, E., Buda, R., Timoncini, A., Di Martino, A., Cenacchi, A., Fornasari, P. M., Giannini, S., & Marcacci, M. (2011). Platelet-rich plasma intra-articular knee injections for the treatment of degenerative cartilage lesions and osteoarthritis. Knee Surgery, Sports Traumatology, Arthroscopy, 19(4), 528–535. https://doi.org/10.1007/s00167-010-1238-6
Ip, H. L., Nath, D. K., Sawleh, S. H., Kabir, M. H., & Jahan, N. (2020). Regenerative medicine for knee osteoarthritis - the efficacy and safety of intra-articular platelet-rich plasma and mesenchymal stem cells injections: A literature review. Cureus, 12(9), e10575. https://doi.org/10.7759/cureus.10575
Carmona, J. U., Argüelles, D., Climent, F., & Prades, M. (2007). Autologous platelet concentrates as a treatment of horses with osteoarthritis: A preliminary pilot clinical study. Journal of Equine Veterinary Science, 27, 167–170. https://doi.org/10.1016/j.jevs.2007.02.007
Sánchez, M., Anitua, E., Azofra, J., Aguirre, J. J., & Andia, I. (2008). Intra-articular injection of an autologous preparation rich in growth factors for the treatment of knee AO: A retrospective cohort study. Clinical and Experimental Rheumatology, 26, 910–913.
Kon, E., Buda, R., Filardo, G., DiMartino, A., Timoncini, A., Cenacchi, A., Fornasari, P. M., Giannini, S., & Marcacci, M. (2010). Platelet-rich plasma: Intra-articular knee injections produced favorable results on degenerative cartilage lesions. Knee Surgery Sports Traumatology Arthroscopy, 18, 472–479. https://doi.org/10.1007/s00167-009-0940-8
Nguyen, R. T., Borg-Stein, J., & McInnis, K. (2011). Applications of platelet-rich plasma in musculoskeletal and sports medicine: An evidence-based approach. PM&, 3, 226–250. https://doi.org/10.1016/j.pmrj.2010.11.007
Textor, J. (2011). Autologous biologic treatment for equine musculoskeletal injuries: Platelet-rich plasma and IL-1 receptor antagonist protein. The Veterinary Clinics of North America. Equine Practice, 27, 275–298. https://doi.org/10.1016/j.cveq.2011.05.001
Van den Berg, W. B., Van Der Kraan, P. M., Scharstuhl, A., & Van Beuningen, H. M. (2001). Growth factors and cartilage repair. Clinical Orthopaedics and Related Research, 391, 244–250. https://doi.org/10.1097/00003086-200110001-00023
Sun, Y., Feng, Y., Zhang, C. Q., Chen, S. B., & Cheng, X. G. (2010). The regenerative effect of platelet-rich plasma on healing in large osteochondral defects. Int Ortho, 34, 589–597. https://doi.org/10.1007/s00264-009-0793-2
Robson, M. C., Mustoe, T. A., & Hunt, T. K. (1998). The future of recombinant growth factors in wound healing. American Journal of Surgery, 176, 80–82. https://doi.org/10.1016/s0002-9610(98)00186-x
Cianfarani, S. (2019). Risk of cancer in patients treated with recombinant human growth hormone in childhood. Annals of Pediatric Endocrinology & Metabolism, 24(2), 92–98. https://doi.org/10.6065/apem.2019.24.2.92
Akbarzadeh, S., McKenzie, M. B., Rahman, M. M., & Cleland, H. (2021). Allogeneic platelet-rich plasma: Is it safe and effective for wound repair? European Surgical Research, 62, 1–9. https://doi.org/10.1159/000514223
Zaky, S. H., Ottonello, A., Strada, P., Cancedda, R., & Mastrogiacomo, M. (2008). Platelet lysate favours in vitro expansion of human bone marrow stromal cells for bone and cartilage engineering. Journal of Tissue Engineering and Regenerative Medicine, 2, 472–481. https://doi.org/10.1002/term.119
Zhu, Y., Yuan, M., Meng, H. Y., Wang, A. Y., Guo, Q. Y., Wang, Y., & Peng, J. (2013). Basic science and clinical application of platelet-rich plasma for cartilage defects and osteoarthritis: A review. Osteoarthr Cartilage, 21, 1627–1637. https://doi.org/10.1016/j.joca.2013.07.017
Ruggiu, A., Ulivi, V., Sanguineti, F., Cancedda, R., & Descalzi, F. (2013). The effect of platelet lysate on osteoblast proliferation associated with a transient increase of the inflammatory response in bone regeneration. Biomaterials, 34, 9318–9330. https://doi.org/10.1016/j.biomaterials.2013.08.018
Aldén, A., Gonzalez, L., Persson, A., Christensson, K., Holmqvist, O., & Ohlson, S. (2007). Porcine platelet lysate as a supplement for animal cell culture. Cytotechnology, 55, 3–8. https://doi.org/10.1007/s10616-007-9097-9
Jonsdottir-Buch, S. M., Lieder, R., & Sigurjonsson, O. E. (2013). Platelet lysates produced from expired platelet concentrates support growth and osteogenic differentiation of mesenchymal stem cells. PLoS One, 8(7), e68984. https://doi.org/10.1371/journal.pone.0068984
Hsieh, Y. H., Hsieh, M. F., Fang, C. H., Jiang, C. P., Lin, B., & Lee, H. M. (2017). Osteochondral regeneration induced by TGF-β loaded photo cross-linked hyaluronic acid hydrogel infiltrated in fused deposition-manufactured composite scaffold of hydroxyapatite and poly(ethylene glycol)-block-poly(ε-caprolactone). Polymers (Basel), 9(5), 182. https://doi.org/10.3390/polym9050182
Hsieh, Y. H., Shen, B. Y., Wang, Y. H., Lin, B., Lee, H. M., & Hsieh, M. F. (2018). Healing of osteochondral defects implanted with biomimetic scaffolds of poly(ε-caprolactone)/hydroxyapatite and glycidyl-methacrylate-modified hyaluronic acid in a minipig. International Journal of Molecular Sciences, 19(4), 1125. https://doi.org/10.3390/ijms19041125
Van den Borne, M. P., Raijmakers, N. J., Vanlauwe, J., Victor, J., de Jong, S. N., Bellemans, J., & Saris, D. B. (2007). International cartilage repair society (ICRS) and Oswestry macroscopic cartilage evaluation scores validated for use in autologous chondrocyte implantation (ACI) and microfracture. Osteoarthritis and Cartilage, 15, 1397–1402. https://doi.org/10.1016/j.joca.2007.05.005
Mainil-Varlet, P., Aigner, T., Brittberg, M., Bullough, P., Hollander, A., Hunziker, E., Kandel, R., Nehrer, S., Pritzker, K., Roberts, S., et al. (2003). Histological assessment of cartilage repair: A report by the histology endpoint committee of the international cartilage repair society (ICRS). Journal of Bone and Joint Surgery. American Volume, 85, 45–57.
Wojdasiewicz, P., Poniatowski, L. A., & Szukiewicz, D. (2014). The role of inflammatory and anti-inflammatory cytokines in the pathogenesis of osteoarthritis. Mediators Inflamm, 2014, 561459. https://doi.org/10.1155/2014/561459
Melchiorri, C., Meliconi, R., Frizziero, L., Silvestri, T., Pulsatelli, L., Mazzetti, I., Borzi, R. M., Uguccioni, M., & Facchini, A. (1998). Enhanced and coordinated in vivo expression of inflammatory cytokines and nitric oxide synthase by chondrocytes from patients with osteoarthritis. Arthritis and Rheumatism, 41, 2165–2174. https://doi.org/10.1002/1529-0131(199812)41:12%3c2165::AID-ART11%3e3.0.CO;2-O
Nurden, A. T. (2011). Platelets, inflammation and tissue regeneration. Thrombosis and Haemostasis, 105(Suppl 1), S13–S33. https://doi.org/10.1160/THS10-11-0720
Daheshia, M., & Yao, J. Q. (2008). The interleukin 1β pathway in the pathogenesis of osteoarthritis. Journal of Rheumatology, 35, 2306–2312. https://doi.org/10.3899/jrheum.080346
Chen, L. X., Lin, L., Wang, H. J., Wei, X. L., Fu, X., Zhang, J. Y., & Yu, C. L. (2008). Suppression of early experimental osteoarthritis by in vivo delivery of the adenoviral vector-mediated NF-kappaBp65-specific siRNA. Osteoarthritis and Cartilage, 16, 174–184. https://doi.org/10.1016/j.joca.2007.06.006
Park, K. H., & Na, K. (2008). Effect of growth factors on chondrogenic differentiation of rabbit mesenchymal cells embedded in injectable hydrogels. Journal of Bioscience and Bioengineering, 106, 74–79. https://doi.org/10.1263/jbb.106.74
Kennedy, M. I., Whitney, K., Evans, T., & LaPrade, R. F. (2018). Platelet-rich plasma and cartilage repair. Current Reviews in Musculoskeletal Medicine, 11(4), 573–582. https://doi.org/10.1007/s12178-018-9516-x
Yan, L., Zhou, L., Xie, D., Du, W., Chen, F., Yuan, Q., Tong, P., Shan, L., & Efferth, T. (2019). Chondroprotective effects of platelet lysate towards monoiodoacetate-induced arthritis by suppression of TNF-α-induced activation of NF-ĸB pathway in chondrocytes. Aging (Albany NY), 11(9), 2797–2811. https://doi.org/10.18632/aging.101952
Al-Ajlouni, J., Awidi, A., Samara, O., et al. (2015). Safety and efficacy of autologous intra-articular platelet lysates in early and intermediate knee osteoarthrosis in humans: A prospective open-label study. Clinical Journal of Sport Medicine, 25(6), 524–528. https://doi.org/10.1097/JSM.0000000000000166
Camargo Garbin, L., McIlwraith, C. W., & Frisbie, D. D. (2019). Evaluation of allogeneic freeze-dried platelet lysate in cartilage exposed to interleukin 1-β in vitro. BMC Veterinary Research, 15, 386. https://doi.org/10.1186/s12917-019-2118-z
Wang, S. Z., Fan, W. M., Jia, J., Ma, L. Y., Yu, J. B., & Wang, C. (2018). Is exclusion of leukocytes from platelet-rich plasma (PRP) a better choice for early intervertebral disc regeneration? Stem Cell Research & Therapy, 9(1), 199. https://doi.org/10.1186/s13287-018-0937-7
Ackenowledgements
The author (Ming-Fa Hsieh) would like to thank National Science and Technology Council of Taiwan for the financial support (grant number: MOST 110–2221-E-033–004-MY2).
Funding
This study was supported by Ministry of Science and Technology, Taiwan, MOST 110-2221-E-033-004-MY2, M.-F. Hsieh
Author information
Authors and Affiliations
Contributions
Y-HH and M-FH contributed to the study conception and design. Material preparation, data collection and analysis were performed by Y-CC, and J-TH. The first draft of the manuscript was written by Y-HH and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hsieh, YH., Chu, YC., Hsiao, JT. et al. Porcine Platelet Lysate Intra-articular Knee Joint Injections for the Treatment of Rabbit Cartilage Lesions and Osteoarthritis. J. Med. Biol. Eng. 43, 102–111 (2023). https://doi.org/10.1007/s40846-023-00776-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40846-023-00776-1