Abstract
Background
A high degree of adherence to direct oral anticoagulants is essential for reducing the risk of ischaemic stroke and systemic embolism in patients with atrial fibrillation, owing to the rapid decline in anticoagulation activity when doses are omitted (i.e. rebound effect).
Objective
We aimed to assess the relationship between non-adherence and non-persistence with direct oral anticoagulants and the incidence of ischaemic stroke in patients with atrial fibrillation.
Methods
A nested case–control study was conducted in the Netherlands, Italy and Germany among patients with atrial fibrillation starting direct oral anticoagulants between the drug approval date and the end of database availability. Patients with an ischaemic stroke during the follow-up were selected as cases and compared with matched controls (matched on age ± 5 years, sex, year of cohort entry date and CHA2DS2-VASc-score at cohort entry date). The cohort entry date was the first dispensing date. Study patients were those aged ≥ 45 years, with ≥ 1 year database history, ≥ 1 year follow-up and at least two direct oral anticoagulant dispensings after the cohort entry date. Adherence and persistence to direct oral anticoagulant treatment were defined as the proportion of days covered ≥ 80% or direct oral anticoagulant continuous use between the cohort entry date and the index date (i.e. date of ischaemic stroke), respectively.
Results
In The Netherlands, Italy and Germany, 105 cases and 395 controls, 1580 cases and 6248 controls, and 900 cases and 3570 controls were included, respectively. Odds ratios (ORs) for stroke among current users who were non-adherent compared to adherent users were 0.43 (95% confidence interval [CI] 0.09–1.96) in The Netherlands, 1.11 (95% CI 0.98–1.26) in Italy and 1.21 (95% CI 1.01–1.45) in Germany. The risk of stroke was significantly higher among non-persistent users compared with persistent users in all three databases [OR 1.56 (95% CI 1.00–2.44), OR 1.48 (1.32–1.65) and OR 1.91 (95% CI 1.64–2.22), respectively]. In The Netherlands and Germany, the risk of stroke was higher the longer a patient had stopped using direct oral anticoagulants.
Conclusions
Both non-adherence (in Germany) and non-persistence increased the risk of stroke, either using a once-daily or twice-daily regime.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Direct oral anticoagulants are effective in reducing the risk of stroke but continuous use is essential. |
Non-adherence and non-persistence of direct oral anticoagulants and the relationship with the risk of stroke were assessed in patients with atrial fibrillation. |
Non-adherence (in Germany) and non-persistence, either with once-daily or twice-daily direct oral anticoagulants, increase the risk of ischaemic stroke. |
1 Introduction
Non-valvular atrial fibrillation (AF) is a common cardiac rhythm disorder and has long been identified as a significant risk factor for disabling or fatal ischaemic stroke and systemic embolism. It has been estimated that around 37 million people worldwide have this condition (0.51% of the worldwide population) and that its prevalence has increased by 33% during the last 20 years [1].
Overall, AF increases the stroke risk five-fold; the attributable risk of ischaemic stroke related to AF ranges from 4.6% for the ages of 50–59 years to 20.2% for the ages of 80–89 years [2]. The risk of stroke in AF is reduced by antithrombotic therapy, specifically vitamin K antagonists (VKAs, e.g. warfarin) or direct oral anticoagulants (DOACs) [3, 4].
Based on the results of multiple randomised controlled trials, DOACs are now frequently used to reduce stroke risk in patients with AF [5,6,7]. Favorable risk-benefit profiles compared with warfarin have been published with significant risk reductions in stroke, intracranial haemorrhage (a serious side effect of oral anticoagulants) and mortality [8]. However, despite their increasing popularity because of a reduced need for routine coagulation testing, improved patient convenience, greater predictability, faster onset of anticoagulation effect, and a lower potential for food and drug interactions, concerns have been raised regarding the heightened consequences of poor adherence to DOACs [9, 10].
A high degree of adherence to DOACs is in fact essential for reducing the risk of ischaemic stroke and systemic embolism in patients with AF, owing to the rapid decline in anticoagulation activity when doses are omitted (i.e. rebound effect) [11]. Poor medication adherence and persistence in the real-world setting may therefore affect the efficacy and safety estimates derived from randomised controlled trials, translating into poor health outcomes and increased healthcare costs [12, 13].
Therapy-related factors have been recognised as one of the most important factors positively or negatively affecting medication adherence. Among them, dosing frequency may play a key role. For example, several studies have shown that once-daily dosing (vs more frequent dosing) [e.g. rivaroxaban vs dabigatran or apixaban] significantly increases the likelihood of keeping patients under treatment [14, 15].
The present study aims to confirm the hypothesis that in the real-world setting DOACs reduce the risk of stroke, requiring continuous use for optimal effectiveness. We therefore assessed among patients with AF the association between non-adherence and non-persistence with DOAC treatment and the risk of ischaemic stroke in three European countries: The Netherlands, Italy and Germany.
2 Methods
2.1 Data Sources
Three different databases were used from three European countries: healthcare records from the PHARMO Database Network in The Netherlands (7 million residents), the Italian Agenzia Regionale di Sanità della Toscana database [ARS] (3.7 million residents) in Italy and the German InGef database (8 million insured persons; an anonymised sample of approximately 4 million insured persons representative of the age and sex structure of the total German population was used for this study) in Germany. The databases are described in detail elsewhere [16,17,18]. ARS and InGef created common input files, whereafter common programming was performed by PHARMO. Thereafter, validation between SAS and R programming for ARS and InGef was carried out and aggregated output files were created.
2.2 Study Design and Patient Selection
All patients were selected if they met the inclusion criteria and received at least DOACs from the date of the positive Committee for Medicinal Products for Human Use opinion on the AF indication (14 April, 2011 for dabigatran, 22 September, 2011 for rivaroxaban, 20 September, 2012 for apixaban and 23 April, 2015 for edoxaban) until the end of data availability for each database (31 December, 2017 in PHARMO, 31 December, 2019 in ARS and 31 December, 2018 in InGef).
During this period, the date of the first dispensing of a DOAC was defined as the cohort entry date (CED). The study cohort was restricted new users (i.e. no previous dispensing of DOACs in the year before CED, no multiple different DOACs on the CED and no VKA dispensing on the CED), aged 45 years or older at CED, and with at least 1 year of database history before CED. Patients were also required to have at least 1 year of follow-up after CED and at least two DOAC dispensings after the CED.
To limit the study cohort to patients with AF, DOAC users were required to meet at least one of the following criteria: have a recorded (primary or secondary) discharge diagnosis of AF (ICD-9 code 427.31, 427.32 or ICD-10 code I48) any time before or on the CED in hospital data and/or have a coded diagnosis indicating AF any time before the CED or in the same quarter as the CED in out-patient data (InGef only). Patients were excluded from the study if they had a diagnosis for different labeled indications such as valvular heart disease, heart valve replacement, venous thromboembolism or pericarditis any time before CED. They were also excluded if they had a procedure code for a hip or knee replacement within 6 weeks prior to the CED. Patients were followed up from CED until the end of data collection (e.g. patient moved out of the catchment area/ended insurance), death or the end of database availability, whichever occurred first.
2.3 Case–Control Definition and Matching
A nested case–control design was used for this study. Cases were selected from the study cohort, based on the following criteria: (a) a recorded (primary or secondary) discharge diagnosis for an ischaemic stroke during the follow-up (ICD-9-CM codes 433 and 434, or the ICD-10 code I63) and (b) at least two DOAC dispensings and no recorded discharge diagnosis for an ischaemic stroke before CED, or between CED and the second DOAC dispensing. The date of the first hospitalisation for ischaemic stroke was considered as the index date. For each selected case, up to four controls were assigned by matching on age ± 5 years, sex, year of cohort entry and CHA2DS2-VASc-score at CED. Controls were assigned the index date of the corresponding case. Controls were required to have at least two DOAC dispensings between CED and index date. Once a patient had been assigned as a case, the patient could not be assigned as a control anymore.
2.4 Patient Characteristics
Sex, age and time since the first DOAC dispensing were determined at the index date for all patients. The following characteristics were determined at CED: year of CED, prior VKA treatment (i.e. VKA dispensing in the year before CED), polypharmacy (i.e. the number of all different pharmacological subgroups [Anatomical Therapeutic Chemical third level] dispensed in the 3 months before or on CED), the systemic use of lipid-modifying agents, antihypertensive drugs, glucose-lowering drugs, antiarrhythmic drugs, antiplatelet drugs and corticosteroids (i.e. dispensing in the 3 months before or on CED), CHA2DS2-VASc-score and the diagnosis of haemorrhagic stroke, renal disease, moderate/severe liver disease and COPD in the year before or on CED [19].
2.5 Adherence and Persistence with DOAC Use
The period between the index date and CED was defined as the exposure period. For each patient, the number of DOAC dispensings within the exposure period was assessed as well as the DOAC type(s) dispensed. Furthermore, all DOAC dispensings in the exposure period were converted into treatment episodes of uninterrupted DOAC use. The duration of each dispensing was calculated by dividing the number of tablets dispensed by the number of tablets to be used per day according to the dosing instructions (or the number of defined daily dose for InGef). If a dispensing preceded the end date of the former dispensing, the date of the latest dispensing was shifted forward to account for possible stockpiling. In InGef and PHARMO, in case of an interruption between DOAC dispensings, use of DOACs was considered uninterrupted if the duration of this gap was less than half the duration of the preceding dispensing with a minimum of 7 days [20]. In ARS, use of DOACs was considered uninterrupted if the duration of the gap was 90 days or less, as in a previous study it was concluded that this method best fitted the ARS data [21]. Patients could have several treatment episodes after CED.
For the adherence analysis, DOAC exposure at the index date was assessed and patients were classified into one of the following exposure categories: current users (when the index date fell between the start and end date of a treatment episode or the last treatment episode ended within 30 days prior to the index date), recent users (when treatment episode ended within 31 and 365 days before the index date) and past users (when the last treatment episode ended more than 365 days before the index date).
Among the current users, we assessed the effect of adherence to DOAC treatment. Adherence to DOAC treatment was defined as the proportion of days covered (PDC) during the exposure period [22]. The PDC was calculated as the total days of supply divided by the number of days in the exposure period. The PDC values ranged from 0 to 1, with higher values suggesting higher adherence. Mean ± standard deviation PDC are presented. Furthermore, in line with previous studies, patients were classified into the following adherence levels: adherent (PDC ≥ 0.80) and non-adherent (PDC < 0.80) [23, 24].
For the persistence analysis, discontinuation of DOAC treatment during the exposure period was assessed based on DOAC treatment episodes. Patients were classified as persistent users (i.e. those with continuous DOAC use from CED to the index date) or non-persistent users (i.e. those who discontinued DOAC use between CED and the index date). As non-persistent users may have restarted treatment, we also assessed the effect of treatment discontinuation based on the above-mentioned exposure categories at the index date (i.e. current, recent and past users). For the assessment of both adherence and persistence, switches between types of DOACs were allowed, i.e. patients switching from for example dabigatran to edoxaban were still considered as adherent to and persistent with DOAC treatment.
2.6 Statistical Analysis
Odds ratios (ORs) with 95% confidence intervals (CIs) for ischaemic stroke were determined in logistic regression models. These models were stratified for each database as well as for patients using only once-daily (QD) DOACs or only twice-daily (BID) DOACs during the exposure period for the adherence analysis. Among current users at the index date, non-adherent patients were compared to adherent patients. For the persistence analysis, non-persistent users were compared to persistent users, and recent and past users at the index date were compared to current users. The non-adherence and non-persistence analyses were also stratified by age (0–64, 65–74 and ≥ 75 years) and CHA2DS2-VASc score (0–3, 4–5 and ≥ 6).
Odds ratio adjustment was considered for the following risk factors: age, VKA treatment prior to DOAC initiation, polypharmacy, co-morbidities (haemorrhagic stroke, renal disease, moderate/severe liver disease, COPD) and CHA2DS2-VASc score at CED. At PHARMO, all data were analysed using SAS programs organised within SAS Enterprise Guide version 8.2 (SAS Institute Inc., Cary, NC, USA) and conducted under Windows using SAS version 9.4. At ARS and InGef, R version 4.03 and version 3.5 were used correspondingly for data processing and analyses. A p value less than 0.05 was considered statistically significant.
3 Results
In The Netherlands, a total of 8421 patients with AF were included for the selection of cases and controls. After matching, 105 (1%) cases and 395 (5%) matched controls were selected for the study. In Italy, 34,713 patients with AF were included, and 1580 (5%) cases and 6248 (18%) matched controls were selected. In Germany, 26,438 patients with AF were included and 900 (3%) cases and 3570 (14%) matched controls were assigned (Fig. 1).
Selection of the study cohort, cases and controls in PHARMO (The Netherlands [NL]), Agenzia Regionale di Sanità della Toscana [ARS] (Italy [IT]) and InGef (Germany [DE]). *Patients aged ≥ 45 years receiving direct oral anticoagulants (DOACs) during the selection period (selection period: from the date of a positive Committee for Medicinal Products for Human Use opinion on the atrial fibrillation [AF] indication for each drug). **A diagnosis for valvular heart disease, heart valve replacement, venous thromboembolism or pericarditis prior to cohort entry date (CED), or a procedure for a hip or knee replacement within 6 weeks prior to CED. VKA vitamin K antagonists
The percentage of male individuals in The Netherlands, Italy and Germany was respectively: 59, 52 and 49%. Furthermore, in The Netherlands, the mean age was 75 years among the cases and 78 years among the controls, and in Italy and Germany, the mean age among both cases and controls was 81 and 80 years, respectively (Table 1). In all databases, among both cases and controls, antihypertensive drugs were used the most (90–93%). In both The Netherlands and Germany, lipid-modifying agents were the second most common co-medication in cases as well as controls, in 52 and 40% of the cases and 47 and 40% of the controls, respectively. In Italy, the second most common co-medication was glucose-lowering drugs among both cases and controls, 22 and 19%, respectively. Polypharmacy was most frequently observed in The Netherlands and Germany. To illustrate, in The Netherlands and Germany, 30 and 34%, respectively of the cases and 32% of the controls used eight or more drugs, while this was only the case in 9% of the cases and 7% of the controls in Italy. The mean CHA2DS2-VASc-score in The Netherlands and Italy was 4 and in Germany this mean score was 6. Furthermore, COPD was the most common comorbidity in The Netherlands and in Italy (19–21% and 15–16% of patients, respectively) and was present in 18% of the patients in Germany. Renal disease was the most common comorbidity in Germany (31–33% of the patients) and was present in about 5% of patients in The Netherlands and 1% of patients in Italy.
In The Netherlands, dabigatran BID (the first approved DOAC) was most frequently used (42–43% of patients) followed by rivaroxaban QD (24% of cases and 32% of controls). In Italy, rivaroxaban QD, apixaban BID and dabigatran BID were used equally often. In Germany, apixaban BID was most frequently used (60–61% of patients). In the overall study population in The Netherlands, the median (interquartile range) exposure period was 12 (6–21) months for cases and 13 (5–22) months for controls. This was 12 (5–23) months in both cases and controls in Germany and 15 (7–29) months among cases and 13 (5–27) months among controls in Italy. The median exposure period among users of only QD or only BID DOACs was comparable to the exposure period in the overall population.
At the index date, 75% of the cases and 89% of the controls in The Netherlands were current DOAC users (Table 2). In Italy, this was 77% of the cases and 81% of the controls and in Germany, this was 69% of the cases and 83% of the controls. The proportion of current users among QD DOAC users and BID DOAC users was also similar in all countries. Mean PDC among current users was 0.96–0.98 in The Netherlands, 0.73–0.74 in Italy and 0.70–0.71 in Germany. In The Netherlands and Italy, the mean PDC among current users did not significantly differ between cases and controls in the overall population, nor in the QD or BID users. In Germany, only among BID users, the mean PDC was significantly lower among cases compared with controls, 0.73 versus 0.77 (p < 0.05). In The Netherlands, the largest part of the study population had a PDC ≥ 0.80, 97% among the cases and 95% among the controls. In Italy and Germany, most patients had a PDC < 0.80, 56% of cases and 52–53% of the controls.
In The Netherlands, 51% of the cases and 42% of the controls (p = 0.093) were non-persistent DOAC users. In Italy, this was 58% of the cases and 48% of the controls (p < 0.05) and in Germany 49 and 34%, respectively (p < 0.05). Once-daily and BID users showed similar results, some results were statistically significant among users in Italy and Germany (data not shown here).
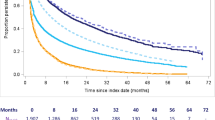
Figure 2 illustrates the association of non-adherence and non-persistence with ischaemic stroke. In Italy, the risk of ischaemic stroke in non-adherent versus adherent patients was not statistically significant [1.11 (95% CI 0.98–1.26)]. In Germany, the risk of ischaemic stroke was 1.21 higher among non-adherent patients compared with adherent patients (95% CI 1.01–1.45). In The Netherlands, where more than 95% of the current users had a PDC ≥ 0.80, no significant relationship between adherence to DOAC treatment and incidence of ischaemic stroke was found. Among QD and BID users in The Netherlands and Italy, ORs were non-significant. In Germany, non-adherent BID users had a significantly higher risk for ischaemic stroke compared with adherent users [1.42 (95% CI 1.15–1.75)]. In all countries, the risk of ischaemic stroke was (borderline) statistically significantly higher among non-persistent users, OR 1.56 (95% CI 1.00–2.44) in The Netherlands, OR 1.48 (95% CI 1.32–1.65) in Italy and OR 1.91 (95% CI 1.64–2.22) in Germany. In The Netherlands and Germany, the comparison between past and recent users at the index date to current users demonstrated that the risk of ischaemic stroke became higher the longer a patient had stopped using DOACs. In The Netherlands, the OR was statistically significant among past DOAC users [4.19 (95% CI 1.99–8.81] and in Germany among both recent and past users [OR 2.07 (95% CI 1.72–2.48) and 2.45 (95% CI 1.76–3.41) respectively]. In Italy, recent users had a significantly higher risk compared with current users [OR 1.47 (95% CI 1.26–1.70)] (Fig. 2).
Association of non-adherence and non-persistence with ischaemic stroke, pooled estimates stratified by database. Analyses were adjusted for age, co-morbidities, prior vitamin K antagonist treatment and CHA2DS2-VASc score. PHARMO (The Netherlands), Agenzia Regionale di Sanità della Toscana [ARS] (Italy), InGef (Germany). BID twice daily (apixaban and dabigatran), CI confidence interval, QD once daily (edoxanban and rivaroxaban)
The relationship between non-adherence and non-persistence with ischaemic stroke was also assessed for different age categories and the CHA2DS2-VASc score categories. The risk of ischaemic stroke due to non-persistence was highest in the oldest age group (≥ 75 years) in The Netherlands and in Italy, 2.49 (95% CI 1.30–4.74) and 1.54 (95% CI 1.35–1.75), respectively. In Germany, this risk was highest in the 65–74 years age group, 2.31 (1.63–3.26) [Table S1 of the Electronic Supplementary Material (ESM)]. Furthermore, the risk of ischaemic stroke among non-persistent users was highest in patients with the highest CHA2DS2-VASc-score (≥ 6) in The Netherlands and Italy, 3.09 (1.34–7.11) and 1.61 (1.26–2.07), respectively. In Germany, this was the case for patients with a CHA2DS2-VASc score of 0–3, 2.35 (1.38–4.00) [Table S2 of the ESM]. For the risk of ischaemic stroke due to non-adherence, no trend among different age or CHA2DS2-VASc score groups was observed (Tables S1 and S2 of the ESM). In The Netherlands, this could not be assessed because of the low number of cases.
4 Discussion
In this study, we assessed among patients with nonvalvular AF the association between non-adherence and non-persistence with DOAC treatment and the incidence of ischaemic stroke in three European countries: The Netherlands, Italy and Germany. To our best knowledge, this is the first study to estimate this relationship in a multi-country study using a validated common data mode.
In The Netherlands, there was no relationship between adherence to DOAC treatment and the incidence of ischaemic stroke. However, because of the low number of cases in The Netherlands, this result has to be interpreted with caution. In Italy and Germany, patients who were non-adherent had a 1.1–1.2 times higher risk of ischaemic stroke compared with adherents. However, results for Italy were not significant. In all countries, non-persistent DOAC users had a higher risk of ischaemic stroke compared with persistent users. The OR (95% CI) ranged from 1.48 in Italy to 1.91 in Germany. These results are in line with previous studies that found that suboptimal adherence to DOACs was associated with an increased hazard of ischaemic stroke [11, 22, 25,26,27,28,29]. Results of the analyses in the different age and CHA2DS2-VASc score groups showed that the risk of ischaemic stroke due to non-persistence or non-adherence remained high in all countries.
Among the three databases, similar age and sex distributions were found. Mean age ranged from 75 to 81 years and the proportion of male individuals varied from 49 to 59%. Concerning non-persistence with DOAC use during the exposure period, 49–58% of the cases and 34–51% of the controls were non-persistent. As non-persistent users may have restarted treatment, we also classified patients as current, recent or past users at the index date. In The Netherlands and Germany, the comparison of past/recent users to current users at the index date showed that the OR increased the longer a patient had stopped using DOACs.
In Italy, the OR showed no trends of increase and only recent users had a significantly higher risk compared with current users. Because of the low proportions of patients in the recent and past user groups in all countries, a post-hoc analysis was performed where recent/past users were combined into one previous user category. This analysis showed that previous users had a significantly higher risk of ischaemic stroke compared with current users in all countries. These results are in line with an increase in coagulation activity when DOACs are discontinued.
Results of the separate once-daily and twice daily regime analyses were mostly in line with the overall study results, showing that when using either a QD or BID regime ensuring continuous use is essential. Our study results are in line with previous studies that indicated that continuing DOAC treatment with adequate adherence is crucial for the beneficial effects on stroke [11, 22, 25,26,27,28,29]. Previous studies of the effect of non-persistence on the risk of stroke are limited. Komen et al. did study persistence as well as adherence and their association with stroke risk [26]. Compared with our study, the maximum number of spillover days, i.e. gap days, differed slightly. Regarding adherence, the researchers used a medication possession rate and perfect adherence was defined as a medication possession rate > 95%, while we defined adherent as a PDC ≥ 80% [24]. Definitions regarding current, recent and past users were comparable between the two studies. Komen et al. found that persistence rates decreased slowly over time, nonetheless, in persistent patients, high adherence was observed. In line with our study results, poor adherence and non-persistence were both associated with an increased stroke risk [26]. The study of Banerjee et al. focused on long-term persistence and adherence and its general relationship with DOACs [30]. The researchers stress the importance of these two concepts with regard to DOAC use as these are usually life-long therapies; however, without focusing on the corresponding risk of stroke when doses are omitted [30]. Regarding non-adherence, results showed a significant risk of ischaemic stroke only in Germany. A previous study showed that patients with a CHA2DS2-VASc score ≥ 4 were at an increased risk of stroke when they were not taking DOAC for ≥ 1 month [29]. It could be that the high CHA2DS2-VASc score of patients in the German sample affected this result. We would also expect to find this result in the stratified analyses [CHA2DS2-VASc score (0–3, 4–5, ≥ 6)]; however, the numbers of cases was too low to interpret these results.
This study is subject to some limitations. First, in The Netherlands, the number of cases (n = 105) was low and nearly all current users had a PDC ≥0.80 therefore, the statistical power for this cohort was rather low. The latter limited the analyses of non-adherence in The Netherlands. The low proportion of patients with ischaemic stroke could be explained by the end of data availability of the Dutch database, 31 December, 2017, while in Germany and Italy this was later, i.e. 31 December, 2018 and 31 December, 2019, respectively. The high adherence rates in The Netherlands might be elucidated by the fact that patients in The Netherlands are regularly seen by their healthcare provider, which likely has a positive effect on adherence to treatment. Second, a well-known limitation of calculating adherence and persistence with dispensing data is that one never knows the real intake of a medicine of a patient; however, this is not specific to our study but accounts for all studies addressing this topic and using prescription or dispensing data. For example, one can assume that patients started using the drug at the dispensed day; however, one does not know whether a gap of several days means that a patient did not take the drug at separate days or over a period of several days. However, we based our calculations on consecutive dispensing dates from the outpatient pharmacies. Here, we assumed that patients continued their medication intake. However, using real-word data, this study was not susceptible to social desirability nor recall bias. Third, in this study, we reduced selection bias by matching on the observable variables: age ± 5 years, sex, year of cohort entry and CHA2DS2-VASc score at CED. However, for a future study, baseline adherence to hypertensive medications (as almost all patients were taking these) should also be taken into account to further reduce selection bias.
Our study also has several strengths. First, data from unselected populations from three different countries (The Netherlands, Italy, Germany) were used, yielding, as much as possible, generalisable results. Second, because of matching, we limited potential differences between the cases and controls. In addition to matching, ORs were also adjusted for potential confounders. Third, this study distinguished between adherence and persistence. Adherence focuses on medication use during a certain study period, i.e. when a patient stops using medication for a certain time and restarts afterwards, this is taken into account in the analyses. In the case of persistence, the focus is on when a patient stops using medication irrespective whether this stop is temporary or not. Both methods are included, the results of this study are very robust and well established. Fourth, in this study, a common data model was used that delivered consequent results across the three different databases, which is to our best knowledge, rather unique. We also used validated software for the analysis, discrepancies in the results across the databases can, for the majority, be explained by differences in the healthcare systems of the countries.
Future research could focus on the relationship between non-adherence and non-persistence and the risk of stroke for individual DOACs as there might be differences between dabigatran and apixaban (BID) and between rivaroxaban and edoxaban (QD) [22, 31]. As a result of findings from the previous literature and our study, we want to stress the importance of closely monitoring patients taking DOACs as the risk of ischaemic stroke is increased when doses are omitted.
5 Conclusions
The study suggests that, in the real-world setting, DOACs are effective in reducing the risk of stroke but continuous use is essential because of the rapid decline in anticoagulation activity when doses are omitted. Regardless of age and the baseline risk of stroke, both non-adherence (in DE) and non-persistence were found to increase the risk of ischaemic stroke, either using a QD regime or a BID regime. The results of this study are in line with previous research and emphasise the importance of optimal adherence and persistence to achieve ideal care and reduce costs, especially since these treatment regimes are usually life-long.
References
Lippi G, Sanchis-Gomar F, Cervellin G. Global epidemiology of atrial fibrillation: an increasing epidemic and public health challenge. Int J Stroke. 2021;16(2):217–21. https://doi.org/10.1177/1747493019897870.
Björck S, Palaszewski B, Friberg L, et al. Atrial fibrillation, stroke risk, and warfarin therapy revisited: a population-based study. Stroke. 2013;44(11):3103–8. https://doi.org/10.1161/strokeaha.113.002329.
Camm AJ, Lip GY, De Caterina R, et al. 2012 focused update of the ESC guidelines for the management of atrial fibrillation: an update of the 2010 ESC guidelines for the management of atrial fibrillation. Developed with the special contribution of the European Heart Rhythm Association. Eur Heart J. 2012;33(21):2719–47. https://doi.org/10.1093/eurheartj/ehs253.
January CT, Wann LS, Alpert JS, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2014;64(21):e1-76. https://doi.org/10.1016/j.jacc.2014.03.022.
Connolly SJ, Ezekowitz MD, Eikelboom YS, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009;361(12):1139–51. https://doi.org/10.1056/NEJMoa0905561.
Granger CB, Lopes RD, Hanna M, et al. Clinical events after transitioning from apixaban versus warfarin to warfarin at the end of the apixaban for reduction in stroke and other thromboembolic events in atrial fibrillation (ARISTOTLE) trial. Am Heart J. 2015;169(1):25–30. https://doi.org/10.1016/j.ahj.2014.09.006.
Patel MR, Mahaffey KW, Garg J, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011;365(10):883–91. https://doi.org/10.1056/NEJMoa1009638.
Ruff CT, Giugliano RP, Braunwald E, et al. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: a meta-analysis of randomised trials. Lancet. 2014;383(9921):955–62. https://doi.org/10.1016/s0140-6736(13)62343-0.
Raparelli V, Proietti M, Cangemi R, et al. Adherence to oral anticoagulant therapy in patients with atrial fibrillation: focus on non-vitamin K antagonist oral anticoagulants. Thromb Haemost. 2017;117(2):209–18. https://doi.org/10.1160/th16-10-0757.
Forslund R, Wettermark B, Hjemdahl P. Comparison of treatment persistence with different oral anticoagulants in patients with atrial fibrillation. Eur J Clin Pharmacol. 2016;72(3):329–38. https://doi.org/10.1007/s00228-015-1983-z.
Alberts MJ, Peacock WF, Fields LE, et al. Association between once- and twice-daily direct oral anticoagulant adherence in nonvalvular atrial fibrillation patients and rates of ischemic stroke. Int J Cardiol. 2016;215:11–3. https://doi.org/10.1016/j.ijcard.2016.03.212.
Deshpande CG, Kogut S, Willey C. Real-world health care costs based on medication adherence and risk of stroke and bleeding in patients treated with novel anticoagulant therapy. J Manag Care Spec Pharm. 2018;24(5):430–9. https://doi.org/10.18553/jmcp.2018.24.5.430.
Jackevicius CA, Tsadok MA, Essebag V, et al. Early non-persistence with dabigatran and rivaroxaban in patients with atrial fibrillation. Heart. 2017;103(17):1331–8. https://doi.org/10.1136/heartjnl-2016-310672.
Ageno W, Beyer-Westendorf J, Rubboli A. Once- versus twice-daily direct oral anticoagulants in non-valvular atrial fibrillation. Expert Opin Pharmacother. 2017;18(13):1325–32. https://doi.org/10.1080/14656566.2017.1361405.
Weeda ER, Coleman CI, McHorney CA, et al. Impact of once- or twice-daily dosing frequency on adherence to chronic cardiovascular disease medications: a meta-regression analysis. Int J Cardiol. 2016;216:104–9. https://doi.org/10.1016/j.ijcard.2016.04.082.
Trifirò G, Gini R, Barone-Adesi F, et al. The role of European healthcare databases for post-marketing drug effectiveness, safety and value evaluation: where does Italy stand? Drug Saf. 2019;42(3):347–63. https://doi.org/10.1007/s40264-018-0732-5.
Kuiper JG, Bakker M, Penning-van Beest FJA, et al. Existing data sources for clinical epidemiology: the PHARMO Database Network. Clin Epidemiol. 2020;12:415–22. https://doi.org/10.2147/clep.S247575.
Andersohn F, Walker J. Characteristics and external validity of the German Health Risk Institute (HRI) Database. Pharmacoepidemiol Drug Saf. 2016;25(1):106–9. https://doi.org/10.1002/pds.3895.
Chen LY, Norby FL, Chamberlain AM, et al. CHA(2)DS(2)-VASc score and stroke prediction in atrial fibrillation in whites, blacks, and Hispanics. Stroke. 2019;50(1):28–33. https://doi.org/10.1161/strokeaha.118.021453.
Catalan VS, LeLorier J. Predictors of long-term persistence on statins in a subsidized clinical population. Value Health. 2000;3(6):417–26. https://doi.org/10.1046/j.1524-4733.2000.36006.x.
Smits E, Andreotti F, Houben E, et al. Adherence and persistence with once-daily vs twice-daily direct oral anticoagulants among patients with atrial fibrillation: real-world analyses from The Netherlands, Italy and Germany. Drugs Real World Outcomes. 2022;9(2):199–209. https://doi.org/10.1007/s40801-021-00289-w
Borne RT, O’Donnell C, Turakhia MP, et al. Adherence and outcomes to direct oral anticoagulants among patients with atrial fibrillation: findings from the Veterans Health Administration. BMC Cardiovasc Disord. 2017;17(1):236. https://doi.org/10.1186/s12872-017-0671-6.
Mazzaglia G, Ambrosioni E, Alacqua M, et al. Adherence to antihypertensive medications and cardiovascular morbidity among newly diagnosed hypertensive patients. Circulation. 2009;120(16):1598–605. https://doi.org/10.1161/circulationaha.108.830299.
Choudhry NK, Shrank WH, Levin RL, et al. Measuring concurrent adherence to multiple related medications. Am J Manag Care. 2009;15(7):457–64.
Kim D, Yang PS, Jang E, et al. The optimal drug adherence to maximize the efficacy and safety of non-vitamin K antagonist oral anticoagulant in real-world atrial fibrillation patients. Europace. 2020;22(4):547–57. https://doi.org/10.1093/europace/euz273.
Komen JJ, Heerdink ER, Klungel OH, et al. Long-term persistence and adherence with non-vitamin K oral anticoagulants in patients with atrial fibrillation and their associations with stroke risk. Eur Heart J Cardiovasc Pharmacother. 2021;7(FI1):f72-80. https://doi.org/10.1093/ehjcvp/pvaa017.
Shore S, Carey EP, Turakhia MP, et al. Adherence to dabigatran therapy and longitudinal patient outcomes: insights from the veterans health administration. Am Heart J. 2014;167(6):810–7. https://doi.org/10.1016/j.ahj.2014.03.023.
Yamashiro K, Kurita N, Tanaka R, et al. Adequate adherence to direct oral anticoagulant is associated with reduced ischemic stroke severity in patients with atrial fibrillation. J Stroke Cerebrovasc Dis. 2019;28(6):1773–80. https://doi.org/10.1016/j.jstrokecerebrovasdis.2018.09.019.
Yao X, Abraham NS, Alexander GC, et al. Effect of adherence to oral anticoagulants on risk of stroke and major bleeding among patients with atrial fibrillation. J Am Heart Assoc. 2016;5(2):e003074. https://doi.org/10.1161/jaha.115.003074.
Banerjee A, Benedetto V, Gichuru P, et al. Adherence and persistence to direct oral anticoagulants in atrial fibrillation: a population-based study. Heart. 2020;106(2):119–26. https://doi.org/10.1136/heartjnl-2019-315307.
Lamberts M, Staerk L, Olesen JB, et al. Major bleeding complications and persistence with oral anticoagulation in non-valvular atrial fibrillation: contemporary findings in real-life Danish patients. J Am Heart Assoc. 2017;6(2):e004517. https://doi.org/10.1161/jaha.116.004517.
Acknowledgements
The authors thank all the healthcare providers contributing information to the PHARMO Database Network, the Italian Agenzia Regionale di Sanità della Toscana database (ARS) and the German InGef database (InGef).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by Daiichi Sankyo Europe GmbH.
Conflict of interest
George Spentzouris was employed at Daiichi Sankyo Europe GmbH. The study was financed by Daiichi Sankyo Europe GmbH. The authors had complete autonomy in the process of establishing the protocol, carrying out the analyses, and interpreting the results and the authors retained the full right to publish the results without limitation. Emily Holthuis, Elisabeth Smits, Fernie Penning-van Beest and Ron Herings are employees of the PHARMO Institute for Drug Outcomes Research. This independent research institute performs financially supported studies for government and related healthcare authorities and pharmaceutical companies. ARS, Giampiero Mazzaglia and InGef were subcontracted by PHARMO. Rosa Gini and Claudia Bartolini are employed by ARS, a public health agency that conducts or participates in pharmacoepidemiology studies compliant with the ENCePP Code of Conduct. The budget of ARS is partially sustained by such studies. Dominik Beier and Dirk Enders are employed by InGef (Institute for Applied Health Research Berlin GmbH), which acted as a subcontractor and received funding from PHARMO for the execution of the study.
Ethics approval
This observational study analysed de-identified data from the PHARMO Database Network; therefore, the study was exempt from ethical review and informed consent was not required. Regarding the ARS database, this study was approved by the ‘Agenzia Regionale di Sanità della Toscana’ Internal Governance Board. All patient-level and provider-level data in the InGef research database are anonymized and are no longer social data in the sense of §67 para. 2 SGB X (Social Security Code X) in combination with Art. 4 Nr. 1 GDPR. Hence, use of the study database for healthcare services research is therefore fully compliant with German federal law and, accordingly, institutional review board or ethical approval for this study was not needed. As this study is based on anonymized claims data, no informed consent of patients was required.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and material
Not applicable.
Code availability
Not applicable.
Author contributions
All authors contributed to the study conception and design. Material preparation and analysis were performed by EH, ES, DE and CB. The first draft of the manuscript was written by EH, ES, DB and GM and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Holthuis, E., Smits, E., Spentzouris, G. et al. Increased Risk of Stroke Due to Non-adherence and Non-persistence with Direct Oral Anticoagulants (DOACs): Real-World Analyses Using a Nested Case–Control Study from The Netherlands, Italy and Germany. Drugs - Real World Outcomes 9, 597–607 (2022). https://doi.org/10.1007/s40801-022-00317-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40801-022-00317-3