Abstract
Background
Patients with chronic or acute/postoperative pain frequently use opioids. However, opioids may cause considerable adverse reactions (ARs), such as respiratory depression, which could be lethal. Unfortunately, only 5% of drug-related ARs (including those to opioids) are reported to health authorities. Therefore, little is known regarding the occurrence of opioid-related ARs at the population level.
Objective
The aim of this study was to investigate how the rates of reported opioid-related ARs have changed in Canada since 1965.
Methods
Our retrospective study examined trends of reported opioid-related ARs occurring in hospitalized and outpatients. Data on opioid-related ARs and mortality between 1965 and 2019 were obtained from the Canada Vigilance and Statistics Canada databases. Descriptive and Joinpoint regression analyses were performed.
Results
Oxycodone and normethadone were the most and least involved opioid agents, respectively, among the 18,407 reported ARs. The highest rate of reported opioid ARs (3.8 per 100,000 person-years) was recorded in 2012, whereas the lowest was recorded in 1965 (0.1 per 100,000 person-years). Between 1965 and 2019, annual rates climbed by 4.2% (95% confidence interval [CI] 3.1–5.2), and many fluctuations were observed: 1965–1974: +22.3% (95% CI 12.0–33.6); 1974–2000: − 4.1% (95% CI − 5.3 to − 2.9); 2000–2008: +30.3% (95% CI 22.6–38.4); 2008–2014: +4.1% (95% CI − 1.5 to 10.1); 2014–2017: −26.0% (95% CI − 44.7 to − 0.9); and, finally, 2017–2019: +35.4% (95% CI 3.8–76.7).
Conclusion
Reported opioid-related ARs have increased since 1965, although fluctuations were observed in recent decades. The absolute number of opioid-related ARs might be seriously underestimated. Future studies should look into how to close this gap.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Using a descriptive analysis spanning > 50 years, opioid-related adverse reactions occurring in hospitalized and outpatients and reported to the Canada Vigilance database were extracted (n = 18,407) and analyzed |
An increase in reports of opioid-related adverse reactions was noted over time, even though fluctuations were observed in the last 20 years. |
These results show the extensive potential of using the Canada Vigilance database to study trends of adverse reactions in Canada. |
1 Introduction
Prescription opioid use has been steadily decreasing between 2012 and 2017 in Canada. Nevertheless, as many as 21.3 million prescriptions were administered in Canada in 2017 [23]. Opioids have analgesic and sedative effects and are commonly used for the management of pain despite advancements in pain management over the years [12]. McMaster University's most recent guidelines for opioid therapy and management of chronic pain not connected to cancer pain, published in 2017, recommended that nonopioid pharmacotherapy and nonpharmacologic therapy be optimized first, rather than an opioid trial. If this does not work, they recommended starting an opioid trial instead of continuing therapy without opioids. This recommendation shows that opioids are still widely used in pain management [12]. Canada stands among the largest users of opioids in the world [7, 25]. In 2017, 11.8% of the population were claimed to have consumed opioids in the last year [35]; however, this drug class is associated with considerable adverse reactions (ARs).
Indeed, the continuum of ARs related to opioids may vary from constipation to itching and may be lethal when involving decreased central respiratory drive, rate, and tidal volume [11]. ARs resulting from taking an opioid as prescribed represent 41% of opioid-related hospital admissions [23]. Hospital admission, along with death, are serious ARs, according to health authorities. Indeed, “A serious adverse reaction is defined in the Regulations as a noxious and unintended response to a drug or natural health product that occurs at any dose and that requires in-patient hospitalization or prolongation of existing hospitalization, causes congenital malformation, results in persistent or significant disability or incapacity, is life-threatening or results in death” [21]. Unfortunately, even in the safest and most rigorous drug licensing program, only 5% (95% confidence interval [CI] 2–18) of drug-related ARs would be reported for investigation to health authorities [20].
One of the Canadian government drug programs is specifically focused on drug safety: the Canada Vigilance Program (managed by Health Canada) [36]. This program has been collecting declarations of drug-related ARs since 1965 [37]. Healthcare professionals, patients, hospital staff, and drug manufacturers are asked to report promptly via MedEffect any AR that they are made aware of [36]. ARs collected in MedEffect are treated, investigated for causality, and then disclosed publicly in the Canada Vigilance online database [36, 37]. This important information regarding drug-related ARs occurring in Canadian patients is used to better monitor the risk–benefit ratio of commercialized drugs.
Many therapeutic efforts have been made in recent years to improve the clinical management of patients who use opioids, in addition to the opioid crisis (e.g., naloxone [15]). However, little is known about the progression of opioid-related ARs reported to health officials. The aim of this study was to investigate how AR declarations related to opioids have changed in Canada since 1965. We expected that the rate of opioid-related ARs reported to Health Canada would rise over time, in line with rising cancer rates [5], surgical procedures [6], increased opioid treatment choices, and increased knowledge of opioid-related adverse effects [24].
2 Methods
2.1 Study Design and Period
This was a descriptive retrospective study design using data from 1 January 1965 to 31 October 2019.
2.2 Source of Data
The source of data was the publicly accessible Canada Vigilance database [17, 19] and the Statistics Canada database [41]. We extracted data from each database and treated them independently as described in Sect. 2.6. In the Canada Vigilance database, ARs are coded according to the preferred terminology of the Medical Dictionary for Regulatory Activities (MedDRA v. 22.1) [13]. At the point of data extraction (January 2020), data were available from 1 January 1965 to 31 October 2019. The method for using this source of data for applied pharmacovigilance studies has been previously published [1]. The Statistics Canada database provides the annual Canadian population since 1965, but only since 1971 specifically for female and male Canadians [40]. We used the total number of Canadians available on 1 July of each year as a denominator to calculate the annual rate.
2.3 Drug Class Selection and Eligibility
The therapeutic class of opioids was selected for this study because of its prevalence of use worldwide and its high risk profile [33]. The list of international nonproprietary names marketed in Canada was produced based on the Compendium of Pharmaceuticals and Specialties [4] and validated by two authors (ML and JL) using the Health Canada Drug Product Database [17]. In the Canada Vigilance database, opioids are coded textually by international nonproprietary names. To be eligible for data extraction, ARs declared in the database had to be associated with the use of an opioid drug. No other exclusion criteria were applied.
2.4 Outcomes
Outcomes were opioid-related ARs declared to Health Canada independent of whether or not seriousness criteria were met (death, disability, congenital anomaly, life threatening, hospitalization, other medically important conditions [18]).
2.5 Data Extraction Process
AR-related data were pulled from the Canada Vigilance database in batches of 1000 (the system’s limit) and downloaded into Excel files. Following that, each of the 1000 AR files was merged into a single file. Then only the suspect or concomitant medications were retained [those used to cure the real AR were deleted (Fig. 1)]. Finally, duplicates were deleted from the database to tidy it up and from the Excel database using the Adverse Reaction Report number variable. Our group had already published this strategy [1].
2.6 Data Analyses
We summarized the properties of extracted ARs using descriptive statistics, which served as a proxy for data quality (namely, proportion of missing data by variable). The annual number of ARs per opioid agent was then computed. Then, rates of AR were calculated, with the annual number of ARs (from the Canada Vigilance database) as the numerator and the total Canadian population as the denominator (from the Statistics Canada database) [40]. Rates were calculated for 100,000 person-years. Trends of ARs (either rates or absolute numbers) were stratified according to characteristics associated with the given AR, such as sex, age group, type of drug used, drug class, type of outcome related to the adverse event report as reported to Canada Vigilance (e.g., death vs. recovered), type of reporter source of report (classes: hospital, manufacturer, clinical trial, community or other), type of declaration (spontaneous, study, published, or other), causality assessment, etc. Of note, the causality assessment of the AR (suspect or concomitant) had already been evaluated by Health Canada prior to data extraction. Health authorities consider “suspect” a product if the health product caused the AR and “concomitant” if the health product is not suspected but the patient was taking it at the time of the AR. We used the Joinpoint Regression Program to perform log-linear Joinpoint regression analysis (version 4.8.0.1; April 2020; Statistical Methodology and Applications Branch, Surveillance Research Program, National Cancer Institute). This software calculates the best-fit model, the number of segments in a given trend, and the annual percentage change (APC) in trends, as well as the related 95% CI.
As unforeseen and as yet unresolvable technical issues were encountered with the Canada Vigilance database (some data were not extractable; listed in Table S1 in the electronic supplementary material [ESM]), we performed three post-hoc sensitivity analyses to validate the initial findings. Our first sensitivity analysis strategy was radical: we removed the trend analysis (1965–2019) for the whole opioids group with which we encountered some problems during data extraction, independently of their seriousness criteria and their source of report (Table S1 in the ESM). We recognized that this would definitely result in a significant underestimation of the true situation, so we conducted a second, more conservative sensitive analysis, excluding only opioids that were difficult to extract for a specific study period (e.g., codeine, hydromorphone, fentanyl, and oxycodone; Table S1 in the ESM). We took an even more conservative approach for the last sensitivity analysis, removing opioids that were hard to extract for a specific study period and report source (e.g., ARs associated with fentanyl and reported by manufacturers were removed from the trend analysis; Table S1 in the ESM). Trends were then re-evaluated in all cases, as described in the main analysis.
2.7 Ethics and Data Protection
This study was conducted at the Université du Québec à Trois-Rivières' Department of Nursing and the Centre de recherche de l'Institut universitaire de cardiologie et de pneumologie de Québec-Université Laval. Because the data were publicly available, no review ethic board certificate was required. Data were designated and protected by the Privacy Act. It was not necessary to obtain informed consent from the patients.
3 Results
3.1 Description of the Database
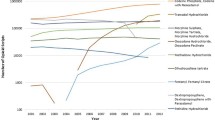
The Canada Vigilance database had 41 variables that could be extracted, and the majority of the key variables had no missing data (Table S2 in the ESM). Figure S1 in the ESM depicts the data extraction flowchart. In total, 689,323 ARs were extracted from the database at first; 189,528 ARs remained after deleting the non-opioid-related ARs. Then, only the cases (n = 189,001) in which the opioid was associated with the AR (but not used to treat it) were kept. After removing duplicates, our final database for this analysis contained 18,407 opioid-related ARs. Between 1965 and 2019, there were 18,407 reports of ARs linked to opioids in Canada (Fig. S1 in the ESM). The numbers of opioid-related ARs for each agent are summarized in Table 1 and illustrated in Fig. S1 in the ESM. The largest overall proportion of ARs was for oxycodone (n = 4053 [22.0%]), and the lowest was for normethadone (n = 14 [0.1%]). The causality assessment found that, of the total number of opioid-related ARs (n = 18,407) that were reported, 16,445 were likely caused by the opioid (labeled as “suspect” in the causality assessment). The other opioids mentioned in AR reports were only labeled as “concomitant” in 1962. No other medicine was shown to be the cause of the AR in the latter study. More than half of the AR cases were classified as serious (n = 10,105), with the remaining 8285 classified as non-serious. Healthcare professionals (physicians, pharmacists, and other health professionals) reported 5639 serious ARs (Fig. 1). Furthermore, fewer ARs (serious or non-serious) were reported by physicians (n = 2746) and pharmacists (n = 2963) than by consumers or non-healthcare professionals (n = 4642) (Fig. 1). Since 1965, manufacturers have been the greatest source of reporters (n = 8908), as expected.
3.2 Description of Trends
3.2.1 Absolute Numbers of Opioid-Related ARs
Since 1965, the total number of opioid-related ARs reported to Health Canada has risen steadily, from 19 in 1965 to 1002 in 2019 (Fig. S3 in the ESM). More non-serious ARs were recorded than serious ARs until 2001 (Fig. S4 in the ESM), at which point the trend reversed. The maximum number of serious ARs (n = 1097) was reported in 2012, and the lowest number (n = 1) was reported in 1965, the first year of this study. The largest total of non-serious ARs was recorded in 2014 (n = 468), and the lowest was recorded in 1965 (n = 18).
Over the years, the trends regarding AR report sources have changed (Fig. S5 in the ESM). Indeed, between 1965 and 1997, most reports originated from hospitals. However, since 1998, reports from hospitals declined and reports from manufacturers increased. Interestingly, since 1999, more reports have come from the community. The source of the report is considered as “other” if it does not come from one of the following sources: manufacturer, clinical trials, community (e.g., pharmacy in community), or hospital.
Opioids were suspected as causative agents in ARs throughout the study period, which is unsurprising (Fig. S6 in the ESM). Since 1965, the majority of AR cases have been recorded spontaneously (n = 16,548; Fig. S7 in the ESM). Finally, Health Canada generally did not know the outcome for the patient experiencing the AR throughout the study period.
3.2.2 Rates of Opioid-Related ARs per 100,000 Person-Years
The rate of declared ARs has fluctuated over time, from 0.1 per 100,000 person-years in 1965 to 2.7 per 100,000 person-years in 2019 (Fig. 2). The highest rate of AR declarations was recorded in 2012 (3.8 per 100,000 person-years). Using a log-linear regression model, we calculated that rates grew by 4.2% each year (95% CI 3.1–5.2) across the study period. The best fitted Joinpoint regression model, in particular, allowed for identification of six segments of the trend. Between 1965 and 1974, the yearly percentage change in AR declaration rate per 100,000 person-years increased by 22.3% (95% CI 12.0–33.6). The second period spanned 1974 to 2000, with an estimated drop of 4.1% (95% CI 2.9–5.3). The third segment was predicted to be +30.3% (95% CI 22.6–38.4) between 2000 and 2008, and the fourth section was expected to be +4.1% (95% CI −1.5–10.1) between 2008 and 2014. Fifth, between 2014 and 2017, there was a 26.0% drop (95% CI 0.9–44.7) in opioid-related AR declarations, with a final increase of 35.4% (95% CI 3.8–76.7) between 2017 and 2019.
Stratification analysis revealed that, for the most part, females had greater rates of declared ARs than males during the study period, with the exception of 2008–2013 (Fig. 3). During the study period, the annual rate of declared ARs for females increased by 3.3% (95% CI 2.4–4.3). Males saw a similar increase of 4.3% (95% CI 3.0–5.7). Throughout the study period, rates of declared ARs increased in both the group aged 0–64 years (3.4% [95% CI 2.2–4.6]) and those aged 65 years (1.9% [95% CI 0.9–2.9]) (Fig. 4).
Throughout the study period, the rates of serious and non-serious ARs reported to Health Canada fluctuated widely (Fig. S8 in the ESM). During the study period, the annual rate of non-serious ARs grew by 1.5% (95% CI 0.7–2.3), and the rate of serious ARs increased by 7.3% (95% CI 5.4–9.2).
3.3 Post-Hoc Sensitivity Analysis
Figures S9 (radical), S10 (moderate), and S11 (conservative) in the ESM show the results of the post-hoc sensitivity analyses (even more conservative). In the first, the log-linear model predicted a yearly increase of 0.5% (95% CI −0.5–1.4) that was non-statistically significant over the study period (Fig. S9 in the ESM). The second sensitivity analysis showed a 1.4% total increase (95% CI 0.6–2.2) (Fig. S10 in the ESM). Finally, a 1.7% rise (95% CI 1.0–2.5%) was reported in the third sensitivity analysis (Fig. S11 in the ESM). Even though the 95% CI did not match those in our initial study (95% CI 3.1–5.2), the conservative sensitivity analyses all showed rising and statistically significant trends.
4 Discussion
4.1 Major Findings
We demonstrate in this study that, notwithstanding changes in the last 20 years, trends for declared opioid-related ARs have increased since 1965. This retrospective population-wide investigation of opioid-related ARs is, to our knowledge, the first of its kind and demonstrates the vast potential of using the Canada Vigilance database to examine AR patterns in Canada.
In recent years, manufacturers have been the most prominent reporting body. This is consistent with the Canadian homologation procedure [14], which requires manufacturers to disclose any ARs they become aware of, especially in the first few years after a new product is commercialized. However, it was not mandatory for non-manufacturers to disclose ARs during the period covered by this study.
4.2 Increasing and Fluctuating Trends of Opioid-Related AR Declarations
As expected, we saw an upward trend in opioid-related AR declarations over time (4.2% [95% CI 3.1–5.2]). This is consistent with our hypotheses, including the effects of the increasing incidence of cancer in Canada [5], along with the aging of the population [40].
The rise in opioid-related AR declarations is also consistent with a report released by Health Canada in 2018 [35], which revealed the total number of AR declarations (not rates) for any type of substance (including opioids) from 2008 to 2017. However, in our study, we were able to fine tune the methodology to produce rates rather than just absolute numbers. Furthermore, rather than a 10-year span, we looked at trends over the last 50 years.
In the last 20 years, reporting rates have fluctuated. Many other factors were changing at the same time, which could have led to some of the fluctuations. These could be explained by, first, the increased availability of various surgical procedures for the general public [6]; the availability of long-acting, but no less strong, opioid therapy choices over time (Table 1) [24]; and the growth of the medical world [26]. Perhaps it is the increased knowledge and other factors involving individuals that have changed over time, such as patient features, comorbidities, and so on.
Purdue Pharma’s oxycodone, for example, was taken off the market in Canada in 2012 [22]. The onset of this withdrawal appears to coincide with the peak in opioid-related AR rates in 2012 (Fig. 2). Perhaps the heightened awareness and vigilance in the community and health professionals increased their willingness to report opioid-related ARs. Additionally, between 2011 (n = 454) and 2012 (n = 698), oxycodone was the only opioid for which there was a significant rise in AR declarations.
The averted opioid crisis of the mid-2000s could possibly explain, at least in part, the fluctuations in the trends (Fig. 2). In fact, the community of Stand Off in Alberta, Canada, raised the alarm for the first time in 2015, when family physicians on the Blood Tribe reserve were treating two to three fentanyl-related overdoses per work shift [22]. This was discovered to be just the tip of the iceberg, whether for prescription or illegal opioids. As a result, the government of Canada introduced steps that may have aided in raising awareness of severe opioid-related ARs.
Indeed, the Minister of Health convened a meeting of health partners to commit to a collaborative action aimed at minimizing the detrimental effects of opioids in Canada [16], as it is globally [43]. Similarly, Health Canada dropped the medical prescription requirement for obtaining naloxone nasal spray in 2016, making it a more accessible treatment for opioid-related overdoses [15]. With all of the media attention given to the opioid problem and naloxone availability, we believe that public awareness of the need to report opioid-related ARs to Health Canada has grown.
The unexpected decrease in the rates of reported opioid-related ARs in 2016 is intriguing. According to our data, hydromorphone (83 ARs in 2015; 18 ARs in 2016), codeine (144 in 2015; 73 in 2016), and buprenorphine (186 in 2015; 105 in 2016) were all declared lower in 2016. This decline could not, to our knowledge, be explained by any removal from the market of these compounds, let alone shortages. Notably, since 2016, Health Canada has mandated that medication makers disclose actual and predicted drug shortages, as well as discontinuations, to Drug Shortages Canada [34]. It is not unlikely that this drop could be caused by the data extraction problems we encountered. For instance, we were unable to retrieve data on fentanyl (Table S1 in the ESM) after November 2015. Fortunately, we believe this information bias does not render the entire trend study incorrect, as the findings of our conservative sensitivity tests were positive (rates increased over time).
4.3 Sex and Gender Differences in Trends of Opioid-Related AR Declarations
Although the overall increases in AR rates for both sexes were statistically similar, female patients had overall higher rates than male patients. This is in line with research that claims ARs have a greater impact on women. Furthermore, the Canadian Tobacco, Alcohol and Drugs survey revealed in 2015 [39] and 2017 [40] that females consumed more prescribed opioids than did males (13.9 vs. 12.1%) [38]. Male trends, on the other hand, were higher between 2008 and 2013. Unfortunately, stratified analyses by gender were not possible because of a lack of this information in our database (we only had access to the biological sex, not the social construct of it). In future studies, sex and gender disparities in opioid-related ARs should be explored and comprehended more thoroughly.
4.4 Strengths and Limitations
Although various descriptive studies on ARs have previously been published [2, 3, 9, 29,30,31, 35], a key strength of our work was the use of national datasets that encompass practically the whole population and opioid-related AR rates since 1965. As a result, selection bias was reduced and generalizability improved. The data for this study were publicly available through the Canada Vigilance database, and we share how to extract and analyze it in a way that is both sustainable and democratic. In terms of transparency, our final dataset is available to the public. Our group has previously worked on validating the methodologies for using Canada Vigilance data in research, boosting confidence in the quality of the information [1]. Despite these strengths, some limitations were unavoidable. First, the reported trends in declared opioid-related ARs should be viewed as an underestimation of the true situation. This underestimation is because of a potential information bias, as some molecules were not extractable throughout the entire study period/reporter types, despite multiple interactions with the database owner (Table S2 in the ESM). We were able to conduct post-hoc sensitivity analyses, which yielded reassuring results. According to a meta-analysis of international data, the underestimating of self-reported ARs could be as high as 95% [20]. Second, despite all efforts to avoid residual information bias [8], given the secondary data analysis nature of this vast database and the small number of non-essential missing data, we cannot rule out the chance of mistakes. Obviously, some external variables over the last 50 years may have influenced opioid-related AR reporting. For example, increased awareness of Purdue Pharma’s oxycontin when it was taken off the market in 2012 could have influenced the number of reports. Finally, we were unable to obtain statistics on the population’s drug prescriptions and usage over time. This would have aided in refining the AR rates on a smaller denominator rather than the entire population. Nonetheless, this field of study is rapidly growing, and we believe our work contributes to pharmacovigilance improvement for better patient care.
4.5 Implications
Although our findings should be interpreted with caution, they show that, compared with other health professionals, consumers, and other non-health people, pharmacists and physicians appear to declare the fewest opioid-related ARs. Patients take multiple doses of opioids every day, both in hospitals and in the community [23]. In addition to being underreported to health authorities, opioid-related deaths are frequently preventable [10], as evidenced by the loss of many lives, some of whom are quite young [27, 28, 32]. Healthcare professionals provide a solution to increase population safety by reporting ARs to their local health authority as soon as they are discovered. Studying this field may aid clinicians to become more aware of the need to report ARs to health authorities. Developing a partnership of healthcare providers, as recommended by the World Health Organization, will help guarantee that drug safety choices and activities are truly patient centered and result in safe care [42].
5 Conclusion
Even though there have been changes in the previous 20 years, trends of declared opioid-related ARs have increased since 1965. Knowing that the number of opioid-related AR cases is likely to be underestimated, future studies should focus on how to close the gap, as well as the sex and gender variations, in opioid-related AR declarations.
References
Akinola S, da Silveira CG, Corbin S, Lavallée M, Thibault M, Midiani Gonella J, Gimenes FRE, Leclerc J. Using data from the canadian post-marketing spontaneous pharmacovigilance system for drug safety research: a feasibility study. Pharmaceut Med. 2020;34(4):263–9.
Andreaggi CA, Novak EA, Mirabile ME, Sampathkumar S, Gray MP, He M, Kane-Gill SL. Safety concerns reported by consumers, manufacturers and healthcare professionals: a detailed evaluation of opioid-related adverse drug reactions in the FDA database over 15 years. Pharmacoepidemiol Drug Saf. 2020;29(12):1627–35.
Aung AK, Tang MJ, Adler NR, de Menezes SL, Goh MSY, Tee HW, Trubiano JA, Puy R, Zubrinich CM, Graudins LV. Adverse drug reactions reported by healthcare professionals: reaction characteristics and time to reporting. J Clin Pharmacol. 2018;58(10):1332–9.
Association des pharmaciens du Canada, Opioïdes (Monographie de l'APhC), in Compendium des produits et spécialités pharmaceutiques. Ottawa: L'Association des pharmaciens du Canada; 2018.
Canadian Cancer Society, Statistic Canada, Public Health Agency of Canada. Canadian cancer statistics. 2019. https://www.cancer.ca/~/media/cancer.ca/CW/cancer%20information/cancer%20101/Canadian%20cancer%20statistics/Canadian-Cancer-Statistics-2019-EN.pdf?la=en.
Canadian Institute for Health Information. Surgical volumes trends, 2009—within and beyond wait time priority areas. Ottawa: CIHI. 2009. https://secure.cihi.ca/free_products/surgical_volumes_2009_e.pdf.
Centre for Addiction and Mental Health. Prescription opioid policy framework. Toronto: Centre for Addiction and Mental Health. 2016. http://www.camh.ca/-/media/files/pdfs---public-policy-submissions/camhopioidpolicyframework-pdf.pdf.
Dal Pan JG, Lindquist M, Gelperin K. Chapter 7. Postmarketing spontaneous pharmacovigilance reporting systems. In: John Wiley & Sons, editor. Textbook of Pharmacoepidemiology. West Sussex: UK, 2013.
Dang A, Bhandare PN. The profile of voluntary reported adverse drug reactions at a tertiary care hospital: a fifteen month prospective study. J Clin Diagn Res. 2012;6(9):504–1509.
Desrosiers G. Quand l'analgésie par opiacés tue. Perspective infirmière, Vol. 4 (2). Québec: Ordre des infirmiers et infirmières du Québec, 2006. pp. 6–7. https://nanopdf.com/download/quand-lanalgesie-par-opiaces-tue_pdf
Faculty of pain medecine of the Royal College of Anaesthetists. Side effects of opioids. https://www.fpm.ac.uk/opioids-aware-clinical-use-opioids/side-effects-opioids.
Garimella V, Cellini C. Postoperative pain control. Clin Colon Rectal Surg. 2013;26(3):191–6.
Gouvernement du Canada. Au sujet du Dictionnaire médical des affaires réglementaires: Gouvernement du Canada, 2010. https://www.canada.ca/fr/sante-canada/services/medicaments-produits-sante/medeffet-canada/base-donnees-effets-indesirables/au-sujet-dictionnaire-medical-affaires-reglementaires-base-donnees-ligne-effets-indesirables-canada-vigilance.html.
Gouvernement du Canada. Comment les médicaments sont examinés au Canada, 2015. https://www.canada.ca/fr/sante-canada/services/medicaments-produits-sante/medicaments/feuillets-information/comment-medicaments-sont-examines-canada.html.
Gouvernement du Canada. Foire aux questions : accès à la naloxone au Canada (y compris le Vaporisateur Nasal NARCAN™). 2017. https://www.canada.ca/fr/sante-canada/services/medicaments-produits-sante/medicaments/annonces/vaporisateur-nasal-narcan-foire-questions.html.
Gouvernement du Canada. Mesures du gouvernement du Canada sur les opioïdes: 2016 et 2017. Ottawa: Sa Majesté la Reine du chef du Canada, représentée par la ministre de la Santé. 2017. https://www.canada.ca/fr/sante-canada/services/publications/vie-saine/mesures-opioides-2016-2017.html.
Governement of Canada. Drug product database: Access the database. 2015. https://www.canada.ca/en/health-canada/services/drugs-health-products/drug-products/drug-product-database.html.
Governement of Canada. Glossary of fields in the Canada Vigilance adverse reaction Online Database. 2019. https://www.canada.ca/en/health-canada/services/drugs-health-products/medeffect-canada/adverse-reaction-database/glossary.html.
Government of Canada. Canada Vigilance adverse reaction online databasanada Vigilance adverse reaction online database. https://www.canada.ca/en/health-canada/services/drugs-health-products/medeffect-canada/adverse-reaction-database.html
Hazell L, Shakir S. Under-reporting of adverse drug reactions: a systematic review. Drug Saf. 2006;29(5):385–96.
Health Canada. Reporting adverse reactions to marketed health products. 2018. https://www.canada.ca/content/dam/hc-sc/documents/services/drugs-health-products/reports-publications/medeffect-canada/reporting-adverse-reactions-marketed-health-products-guidance-industry/reporting-adverse-reactions-marketed-health-products-guidance-industry.pdf.
Howlett K. Canada’s opioid crisis. 2017. https://thecanadianencyclopedia.ca/en/article/canadas-opioid-crisis.
Institut canadien d'information sur la santé. Tendances pancanadiennes en matière de prescription d’opioïdes et de benzodiazépines, de 2012 à 2017. Ottawa, 2018.
International Narcotics Control Board. Availability of internationally controlled drugs: ensuring adequate access for medical and scientific purposes: indispensable, adequately available and not unduly restricted. Vienne: United Nations Publications. 2016. https://www.unodc.org/documents/drug-prevention-and-treatment/INCB_Access_Supplement-AR15_availability_English.pdf.
International Narcotics Control Board. Narcotic drugs. 2017. https://www.incb.org/documents/Narcotic-Drugs/Technical-Publications/2017/Narcotic_drugs_technical_publication_2017.pdf
J Esler D. The changes in medicine in the last 30 years. B C Med J. 2019;61(6):234–5.
Larin V. Dose de morphine mortelle: décédé faute d’avoir eu la surveillance requise. Journal de Montréal. Montréal: Journal de Montréal. 2018. https://www.journaldemontreal.com/2018/11/26/mort-faute-davoir-eu-la-surveillance-requise.
LCN. Un médicament prescrit est responsable. TVA Nouvelles. 2006. https://www.tvanouvelles.ca/2006/03/29/un-medicament-prescrit-est-responsable.
Lee JH, Park KH, Moon HJ, Lee YW, Park JW, Hong CS. Spontaneous reporting of adverse drug reactions through electronic submission from regional society healthcare professionals in Korea. Yonsei Med J. 2012;53(5):1022–7.
Lobo MGADA, Pinheiro SMB, Castro JGD, Momenté VG, Pranchevicius MCS. Adverse drug reaction monitoring: support for pharmacovigilance at a tertiary care hospital in Northern Brazil. BMC Pharmacol Toxicol. 2013;14:1–7.
Newbould V, Le Meur S, Goedecke T, Kurz X. Medication errors: a characterisation of spontaneously reported cases in EudraVigilance. Drug Saf. 2017;40(12):1241–8.
Poulin L, La loi de Vanessa. Journal Le Collectif, Vol. 2019. Sherbrooke: Journal Le Collectif. 2015. http://www.lecollectif.ca/la-loi-de-vanessa/.
Santé Canada. Analgésiques opioides. 2009. https://www.canada.ca/content/dam/hc-sc/migration/hc-sc/hl-vs/alt_formats/pdf/iyh-vsv/med/ana-opioid-med-fra.pdf.
Santé Canada. Drug shortages in Canada. 2017. https://www.canada.ca/en/health-canada/services/drugs-health-products/drug-products/drug-shortages.html.
Santé Canada. Tendances annuelles dans les déclarations de cas d'effets indésirables des produits de santé et des incidents liés aux matériaux médicaux à Santé Canada (2008–2017). Canada. 2018. https://www.canada.ca/content/dam/hc-sc/documents/services/publications/drugs-health-products/tendances-annuelles-declarations-effets-indesirables-produits-sante-incidents-lies-materiels-medicaux-2008-17.pdf.
Santé Canada. Renseignement sur le produit. 2019. https://produits-sante.canada.ca/dpd-bdpp/info.do?lang=fr&code=78104.
Santé Canada. Base de données en ligne Canada Vigilance. 2020. https://www.canada.ca/fr/sante-canada/services/medicaments-produits-sante/medeffet-canada/base-donnees-effets-indesirables.html.
Stabile S, Ruggiero F, Taurasi F, Russo L, Vigano M, Borin F. Gender difference as risk factor for adverse drug reactions: data analysis in salvini hospital. Pharmacologyonline. 2014;2:75–80.
Statistique Canada. Enquête canadienne sur le tabac, l'alcool et les drogues (ECTAD): sommaire de 2013. 2015. https://www.canada.ca/fr/sante-canada/services/enquete-canadienne-tabac-alcool-et-drogues/sommaire-2013.html.
Statistique Canada. Enquête canadienne sur le tabac, l’alcool et les drogues (ECTAD): sommaire des résultats pour 2017: Gouvernement du Canada. 2017. https://www.canada.ca/fr/sante-canada/services/enquete-canadienne-tabac-alcool-et-drogues/sommaire-2017.html.
Statistique Canada. Estimations de la population au 1er juillet, par âge et sexe. 2020. https://www150.statcan.gc.ca/t1/tbl1/fr/tv.action?pid=1710000501.
World Health Organization. Medication without harm. Genève: World Health Organization. 2017. https://www.who.int/publications/i/item/WHO-HIS-SDS-2017.6.
World Health Organization. Opioid overdose, 2020. https://www.who.int/news-room/fact-sheets/detail/opioid-overdose.
Acknowledgements
The authors are grateful to Health Canada and the Canada Vigilance program for the availability of data. Maude Lavallée is also grateful to the Fondation de l’UQTR for the two scholarships received.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No sources of funding were used to conduct this study or prepare this manuscript.
Conflict of interest
Maude Lavallée, Carolina Galli da Silveira, Samuel Akinola, Julie Méthot, Marie-Eve Piché, Anick Bérard, Magalie Thibault, Jennifer Midiani Gonella, Fernanda Raphael Escobar Gimenes, and Jacinthe Leclerc have no conflicts of interest that are directly relevant to the content of this article.
Availability of data and material
The authors will make available the data and program codes used in the analyses to any researcher for the purposes of reproducing the results or replicating the procedure as supplementary material.
Ethics approval
Because the data were publicly available, no review ethic board certificate was required. These data are anonymized and protected under privacy laws; no individual patient may be identified.
Consent
Not applicable.
Author contributions
Maude Lavallée, Carolina Galli da Silveira, Julie Méthot, Marie-Eve Piché, Anick Bérard, Fernanda Raphael Escobar Gimenes, and Jacinthe Leclerc contributed to the study conception and design. Data collection and analysis were performed by Maude Lavallée and Jacinthe Leclerc. The first draft of the manuscript was written by Maude Lavallée and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Lavallée, M., da Silveira, C.G., Akinola, S. et al. Reporting Rates of Opioid-Related Adverse Events Since 1965 in Canada: A Descriptive Retrospective Study. Drugs - Real World Outcomes 9, 153–163 (2022). https://doi.org/10.1007/s40801-021-00275-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40801-021-00275-2