Abstract
Improvement of coking properties of sub-bituminous coal (A) and bituminous coal (B) was done using blended organic solvents, namely, n-methyl-2-pyrrolidinone (NMP) and ethylenediamine (EDA). Various solvent blends were employed for the coal extraction under the total reflux condition. A low-cost ceramic membrane was fabricated using industrial waste iron ore slime of M/s TATA steel R&D, Jamshedpur (India) to separate out the dissolved coking fraction from the solvent-coal mixture. Membrane separations were carried out in a batch cell, and around 75 % recovered NMP was reused. The fractionated coal properties were determined using proximate and ultimate analyses. In the case of bituminous coal, the ash and sulfur contents were decreased by 99.3 % and 79.2 %, respectively, whereas, the carbon content was increased by 23.9 % in the separated coal fraction. Three different cleaning agents, namely deionized water, sodium dodecyl sulphate and NMP were used to regain the original membrane permeability for the reusing.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Incomplete coal combustion results in carbon monoxide formation along with carbon dioxide. Coal contains mainly combustible hydrocarbons and about 10 % inorganic impurities. These impurities enhance the deposition of uncontrolled ash on the boiler tube wall, and the boiler efficiency is deteriorated (Couch 1994). There are several methods employed for extraction of coal, such as soxhlet, supercritical fluid and microwave-assisted extractions, and chemical leaching with alkali and acid solutions. The ultra-clean coal (ash <1 %) could be produced by these techniques. The yield of low-rank coals can be enhanced by the extraction with inorganic acids, such as HCl, at the elevated temperature. The extraction yield also depends on acid strength, temperature of extraction and time (Kashimura et al. 2006). A single solvent is not usually effective for the removal of impurities to obtain the desired level of purification (Mishra and Sharma 1999). The blended solvents such as CS2 and NMP have also been proven useful in coal purification (Iino et al. 1989). These, are in particular, effective up to 65 %–85 % organic fraction extraction (Shui et al. 2006). Ethylenediamine (EDA) gives better extraction efficiency under the mild conditions (60–80 °C and 1 atm). The solvent is also cheaper compared to NMP but recovery is difficult (Chawla and Davis 1989). The ceramic membrane processes especially offer effective recovery of solvent from the coal-solvent mixture (Singh et al. 2012). Iron ore slime (IOS) is one of the waste materials formed in the steel production unit. It contains around 35 %–40 % iron. IOS is not normally suitable for steel production for extremely fine particle size <50 µm containing a significant amount of Fe along with SiO2 and Al2O3. IOS has good thermal stability and could be a suitable raw material for low-cost ceramic membrane fabrication upon appropriate blending with other components.
Therefore, the objectives are (1) utilization of waste IOS for the fabrication of ceramic membrane which is a step forward for the minimization of solid waste, (2) characterization of the fabricated membrane and (3) its application in solvent-coke separation for the improvement of coke properties, namely, ash and sulphur content reduction and increment of carbon content. This work focuses on the improvement of the coal extraction yield using the blended solvents of NMP and EDA.
2 Experimental
2.1 Materials
Both the coals (coal A: sub-bituminous and coal B: bituminous) and IOS samples were supplied by M/s TATA Steel R&D, Jamshedpur, India. Other raw materials utilized for the ceramic membrane fabrication, namely, kaolin (99.5 %), quartz (99.5 %), boric acid (99.5 %), sodium carbonate (99.9 %), calcium carbonate (99 %), and sodium metasilicate (99.5 %) were purchased from Merck India Ltd. Quartz powder was provided by Research-Lab Fine Chem Industry, Mumbai, India. The solvents, n-methyl-2-pyrrolidane (NMP) (99.5 %) and ethylenediamine (EDA) (99 %) were obtained from Merck India Ltd. All the chemicals were analytical grade and used without further purification. Quartz was added to provide thermal and mechanical strength (Jana et al. 2010).
2.2 Physical and chemical properties of IOS
Iron ore slime is consisting of particle size <50 µm with a density of 5.1 g/cm3. It is rich in iron oxides with rusty red color appearance. Energy dispersive X-ray (EDX) analysis of IOS showed the elemental composition (wt%) as follows: oxygen (41.80), iron (35.63), silicon (10.79), aluminium (10.47) and thallium (1.29). Iron in IOS acts as a binder for ceramic membrane fabrication providing strength and reduces the cost of the fabricated membrane.
2.3 Membrane fabrication and characterization
2.3.1 Fabrication method
The uniaxial die pressing method was employed to synthesize the membranes. All the membrane materials were heated for 3 h at 100 °C in a hot air oven for moisture removal. The membrane materials of the composition in Table 1 were mixed in a ball mill. It was then compacted in a rigid die, with 55 mm diameter and 5 mm thick, under an applied load of 2 tons in a single axial direction with the help of a rigid piston for 2 min. The compacted membrane weighs about 22 g. The disk was calcined in a programmable muffle furnace (Make: Reico Equipment and Instrument Pvt. Ltd., Kolkata, India) in two sequential stages. At first, the rate of temperature increment was 1 °C per min for 4 h at the starting temperature of 25 °C and, the final temperature was maintained for 20 h (Singh et al. 2012). Initially, the low heating rate was adopted to reduce the crack formation of the membrane. In the second stage, the temperature was increased at a rate of 2 °C per min from 265 °C to desired sintering temperature, such as 700 °C. The membrane was kept for 6 h at the final sintering temperature. After this, the sintered membrane was cooled to the room temperature.
2.3.2 Pore size distribution
The pore size distribution of prepared IOS ceramic membrane was analyzed by SEM/EDX technique (make: Oxford; Model: LEO 1430VP, UK). Small pieces of the prepared IOS ceramic membranes were cut out without distorting the surface geometry. The top image of membrane active layers was recorded and the average pore diameter was determined by using the imageJ software (Version 1.37) (Singh et al. 2012). SEM micrographs were recorded from the different sections of the fabricated IOS membranes. The diameters of 200 pores of each membrane were determined using the image J software. The average pore diameters (d avg) were calculated from Eq. (1) (Singh et al. 2012). Where, n i is the pores numbers, and d i is the diameter (μm) of the ith pore.
2.3.3 Membrane porosity
The membrane porosity was determined by the psychometric method using de-ionized water as the wetting liquid. The membrane was immersed in de-ionized water for two consecutive days, and the porosity was determined by standard equation (Singh et al. 2012).
2.3.4 Hydraulic permeability
Hydraulic permeability was determined from the steady state permeate flux value measured after stabilization at different transmembrane pressure drops (after 35 min of water filtration experiment). The steady state permeate flux value increased linearly with applied transmembrane pressure drops. The membrane hydraulic permeability was determined from the slope of steady state permeates flux with transmembrane pressure drop (Jana et al. 2010).
2.3.5 Chemical stability test
Chemical/corrosion stability test of the membrane was performed both at acidic (pH 4) and alkaline (pH 10) solution. Porosity and EDX analysis were performed in order to explain the chemical attack and elemental dissolution by comparing the results obtained from the acid or alkali treatment (Jana et al. 2010).
2.4 Coal extraction followed by membrane separation
The performance of the fabricated membrane was tested for solvent recovery and coking coal separation followed by solvent extraction.
2.4.1 Extraction experiment
The coal samples (A and B) were dried overnight at 100 °C, grounded and screened through 200 mesh (74 µm). The undersized (<74 µm) was utilized for coal extraction test using the mixed organic solvent of NMP and EDA (9:1 v/v). The extraction experiment was performed with 50 mL mixed solvents and 5 g coal samples in a round bottom flask extractor of 500 mL capacity. The extraction was carried out for 1 h at 180 °C under total reflux with the help of a reflux condenser (Christy et al. 1992). The entire hot extractor content was first filtered using two successive 500 mesh (31 µm) stainless steel sieves to separate out the coking coal fractions remained dissolved in the solvent (extracted fraction) and the residues (un-extracted fraction). The dissolved coking parts and solvents were separated from the hot extracted fraction by filtration using the fabricated ceramic IOS membrane in a batch cell.
2.4.2 Membrane experiment
The experimental setup consisted of a stainless steel cylindrical batch cell of 5 × 10−4 m3 capacity (ϕ 57 mm). The applied transmembrane pressure drop of 203 kPa was maintained using compressed nitrogen gas. A porous stainless steel plate was placed at the bottom of the membrane to provide the extra mechanical support. The flange was bolted to the cell body to make the cell unit leak proof. The dissolved coking parts were accumulated on the membrane surface and the solvents collected from the permeate side. The schematic diagram of the main membrane module and other auxiliaries are shown in Fig. 1.
2.4.3 Coal extraction and solvent recovery mechanism
NMP is a suitable organic solvent for extraction of bituminous and sub-bituminous coals. The extraction primarily occurs from the outer surface of coal particles due to high viscosity (1.81 × 10−3 Pa s) and low penetration. For this reason, NMP alone has a low extraction yield. The solvent must diffuse inside the coal particles and comes out with soluble coal molecules. During extraction, some amount of the solvent remains in the porous structure of coal resulting in swelling. Hence, the swelling of coal causes a small loss of organic solvents during the extraction process. Organic solvent breaks the intermolecular forces among coal molecules, such as H-bonding, van der Walls forces and other charge transfer π–π interaction and makes bonding with coal molecule resulting in coal extraction (Shui 2005). EDA is commonly blended with NMP to improve the extraction yield as it reduces viscosity, and can easily penetrate the network like coal structure. A 9:1 NMP to EDA ratio could reduce the viscosity to 1.67 × 10−3 from 1.81 × 10−3 Pa s for pure NMP. EDA addition also reduces the cost of solvent (Singh et al. 2012). A mixture of organic solvent and coal particles was collected after the extraction operation as filtrate was permeated through the membrane cell. The organic solvent passed through the membrane pores was recovered as permeate, and the suspended materials (coke) were collected on the membrane surface.
2.5 Sample analysis
2.5.1 Proximate, ultimate and Fourier transform infrared spectroscopy (FTIR) analysis
Proximate and ultimate analysis was performed according to the ASTM D3172-07a and ASTM D3176-89 methods. FTIR spectra of the coals, pure NMP and permeate were recorded using an FTIR spectrometer (Spectrum 65, Perkin Elmer, USA).
2.6 Pore formation mechanism in IOS membrane
Calcium carbonate (CaCO3) played an important role in the formation of pores in IOS ceramic membrane. During calcinations, CaCO3 is decomposed into calcium oxide and carbon dioxide. CO2 dispersion path creates the membrane pores (Jana et al. 2010).
3 Results and discussion
The results and discussions are organized into three sections. Firstly, the membrane fabrication using IOS and their characterizations are provided. The coal extraction test followed by membrane filtration is outlined in the second section. After that, the preliminary cost estimation of the fabricated membrane is discussed.
3.1 Membrane fabrication and characterization
3.1.1 Selection of membrane compositions
Each inorganic material played a different role for fabricating the IOS ceramic membrane. Both IOS and kaolin pose high refractory properties so that the ceramic membrane can withstand higher temperature without degrading the membrane structure. For the admirable thermal and mechanical stability, quartz was added. Boric acid improved the mechanical strength of the membrane. The porous membrane structure was developed by the addition of calcium carbonate and it was dissociated releasing carbon dioxide during sintering. The path followed by the carbon dioxide developed the porous membrane texture. Sodium carbonate and also boric acid improved the dispersion properties and provided a homogeneous membrane structure. To bind all the inorganic materials, sodium metasilicate was added. It made silicate bonds amongst other inorganic elements and gave an extra mechanical strength of the membrane during sintering operation. The inorganic raw materials utilized for the fabrication of IOS ceramic membrane were listed in Table 1. Four different combinations of the compositions designated as I, II, III and IV were used for membrane fabrication. It was one of the reasons that only the concentration of IOS and kaolin were changed keeping the composition of the other materials unchanged. Higher IOS concentration was desirable for low membrane cost. Up to 10 % IOS, dissolution of prepared IOS membrane was around 0.5 %, whereas, it was about 5.5 % with 15 % IOS. The membrane was not good corrosion resistant in acid solutions beyond 10 % IOS. By keeping both the parameters in mind, 10 % IOS composition (II) was selected for fabrication of membrane used in the subsequent experiments.
3.1.2 Pore size distribution, porosity, hydraulic permeability and corrosion stability
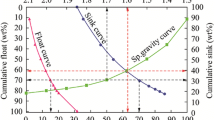
Not only the membrane composition stated above but also the particle size distribution of the main raw materials was selected to obtain the membranes in the microfiltration regime as the particle size of the extracted coal usually comes in the range from 1 to 31 μm. Likewise, the size of the raw materials <10 μm was taken. It was expected that the membrane fabrication protocol used should give the desired membrane in the microfiltration range (Singh et al. 2012). The average particle sizes of IOS, kaolin, calcium carbonate and quartz were 10, 2.37, 4.12 and 8.4 μm, respectively. The average membrane pore diameters were 0.602, 1.053 and 1.103 µm at sintering temperatures of 700, 800 and 900 °C respectively, which come in the microfiltration range. It was observed that as sintering temperature increased, the average pore diameter also increased. The small pores, as well as void spaces, smashed together with big pores and formed uniform packing with the increase in temperature. The membrane porosity was decreased from 0.33 to 0.3 nearly around 9 % when the sintering temperature increased from 700 to 900 °C. The membrane was densified by decreasing the porosity at the higher sintering temperature. Hydraulic permeability was found to increase from 1.58 × 10−12 to 3.81 × 10−12 (m/Pa s) because of larger pore size at the higher temperature. The porosity increased only by 5 %–9 % under chemical/corrosion test at pH 4 and pH 10. It suggests that the membrane fabricated had good chemical stability.
3.1.3 SEM and EDX analysis
The SEM images of prepared IOS ceramic membrane at different sintering temperatures are shown in Fig. 2. It was observed that the small particles merged with large particle during the sintering operation. Highly porous structure was obtained for the substrates sintered at the lower temperature (700 °C) (Fig. 2a). Whereas, the membranes sintered at 800 and 900 °C were more consolidated (Fig. 2b, c).
EDX analysis was performed before (fresh membrane) and after the corrosion test with the membrane sintered at 900 °C (Fig. 3). It was found that the elemental composition of the fabricated membrane couldn’t change even after the corrosion test. The composition of fresh membrane and the same after corrosion test from the EDX analyses is shown in Table S1 of Supplementary data.
3.2 Coal extraction followed by membrane separation
3.2.1 Effect of solvent on coal extraction
The lowest possible extracting solvent requirement to achieve the maximum recovery of solvent is always desirable. Several extraction experiments were performed with both the coal samples under similar test conditions to determine the optimum solvent to coal ratio. In extraction experiment, the yields were 62.8 % and 61 % for coal A and B, respectively, using only NMP as the extracting solvent with the solvent to coal ratio of 10:1 (v/w) (Table 2). The addition of EDA increases the extraction yield by 2.2 % for coal A and 1.8 % for coal B at NMP: EDA: coal = 9:1:1 (v:v:w). It was explained by the synergistic effect of EDA in coal extraction as discussed earlier. The maximum solvent recovery was found to be 75 % with the solvent to coal ratio of 10:1 (v/w) for coal sample A. The same was about 72 % for coal sample B. A slight decrease in both extraction yield and solvent recovery was observed for coal to solvent ratio of 9:1 (v:w) for both the coals.
It is necessary to mention that the loss of the solvent in a laboratory study is significantly higher because a fairly large amount of NMP is retained in the extracted coals. That is why nitrogen contents in extracted coal are rather high as shown in Table 3. However, for a scaled up process for practical application, the typical solvent loss is around 35 % by using thermal distillation at around (204 ± 2) °C (Saha and Mondal 2013).
3.2.2 Proximate and ultimate analysis of raw and extracted coal (coke)
Proximate and ultimate analyses of raw coal and extracted coal (coke) were shown in Table 3. FC content reduced from 51 % to 42.48 % in case of coal A. The reduction was around 3 % for sample B. The ash content reduction in extracted coal sample (ECS) A was about 32 %. It was more than 99 % in ECS B. As a result, VM content was found to increase in both the ECS. Mineral matters in ECS A and B was removed by 26 % and 95.7 %, respectively. The carbon content in ECS increased up to 10 % and 24 % using coals A and B, respectively. The blended solvents effectively reduced sulphur in the ECS by 76 %–79 % in case of both the coal samples. Nitrogen content increased in both ECS due to adsorbed NMP. Gross calorific value (GCV) in ECS increased up to 7 % (coal A) and 17 % (coal B). The results suggest that extracted coal using sample B had better efficiency for iron and steel production.
3.2.3 Fourier transform infrared spectroscopy (FTIR) analysis
The FTIR spectra of coal sample A, B, pure NMP and permeate (recovered solvent, mainly NMP) is presented in Fig. 4. The broad absorption band at 3440 and 3454 cm−1 in the case of both coal samples and frequencies at 3429, 3435, 1655 and 1661 cm−1 in pure NMP and permeate, were observed due to N–H stretching vibration (Cai and Smart 1993). Two stronger bands 2933 and 2935 cm−1 were observed in pure NMP due to CH3–CH2 vibrations. The band at 2917 cm−1 could be attributed to coals (A and B) due to the presence of C-H group (Krzton et al. 1995). The band at 2136 and 2143 cm−1 for pure NMP and permeate could be assigned to -CN absorption band. It was absent in coal samples. The spectrum at frequency 1641, 1661 and 1655 cm−1 were ascribed in the coal samples, NMP and pure permeate due to the presence of acyclic C–C groups (Bhide and Stern 1991). The spectrum at frequency 1305 cm−1 identified with pure NMP and permeate was resulted from the vibration of C–O groups. The same was not observed in coal samples. The bands 659 and 626 cm−1 were attributed to Si–O vibrations. The distinct peak at 1034 cm−1 was observed in coal samples. The same was absent in NMP and disappeared from permeate because of sulphur removal (Bhide and Stern 1991). The peak at 537 cm−1 appeared in coal sample because of the kaolinite mineral group, and disappeared from permeate. The results indicated that the fabricated membrane was capable of recovering almost pure NMP (permeate) free of dissolved/suspended coal particles. Hence, the recovered solvent must be reused efficaciously for further non-coking to coking coal conversion along with the makeup solvent.
3.3 Membrane cleaning
Membrane fouling caused the permeate flux decline and low solvent recovery on the permeate side. Low solvent recovery was due to the pore blocking and settling of non-coking coal on the membrane surface. The cleaning was necessary to reuse the same membrane for solvent recovery purpose. Fresh IOS ceramic membrane permeability (sintered at 900 °C) was 3.81 × 10−12 m/Pa s. After the experiment, the membrane was fouled and cleaned with three different cleaning agents, such as deionized water, an anionic surfactant (sodium dodecyl sulphate) and an organic solvent (NMP). The cleaning efficiency of the membrane was measured in the terms of recovery of hydraulic permeability. The percentage cleaning efficiency is defined as (Lpc/Lp i ) × 100, where Lpc and Lp i were the membrane hydraulic permeability (m/Pa s) after cleaning and before the fouling experiment. With deionized water, the cleaning efficiency was 48 % and recovered membrane permeability was 1.82 × 10−12 m/Pa s. Hence, deionized water was not sufficient to recover the original permeability. It indicated that irreversible fouling was there on the membrane surface. SDS was used as another cleaning agent for the removal of the irreversible fouling layer. 73 % cleaning efficiency was observed with 5 mM SDS (pH = 11) solution and recovered membrane permeability was 2.78 × 10−12 m/Pa s. With NMP, 90 % cleaning efficiency was observed with membrane permeability of 3.42 × 10−12 m/Pa s. Hence NMP had maximum ability to remove coking coal foulant from membrane surface. A detailed study of fouling and cleaning mechanisms for successive cycles without sacrificing the cleaning efficiency are in progress in our research group.
3.4 Preliminary cost estimation of fabricated membrane
The main drawback of ceramic membranes is its high fabrication cost compared to polymeric membranes. Polymeric membranes are available $50 to 200/m2 and ceramic membranes came in the range from $500 to 2000/m2, of membrane surface area (Koros and Mahajan 2000). The cost of the fabricated IOS ceramic membrane (ϕ 55 mm, thickness 5 mm, membrane II) was calculated from the basis of the analytical grade chemicals (except IOS) used. The cost calculated was then linearly extrapolated to per unit m2 of the membrane surface of the same thickness. Total time of running the muffle furnace (1.8 kW) was 40.42 h. Total fabrication cost including materials and power consumption was $ 53.26/m2 (Table S2 of Supplementary data). For bulk manufacturing including the shipment cost added $ 26.63/m2 in total membrane cost, the estimated total cost reached to $ 79.89/m2 which came near to polymeric membrane production cost. The prepared membrane cost was lower compared to developed low-cost ceramic membrane by (Jana et al. 2010).
4 Conclusions
This work summarizes the development of an inorganic ceramic membrane for the purpose of membrane cost reduction and as well as utilization of industrial waste iron ore slimes (IOS) to reduce the environmental consequences.
-
(1)
The average pore diameter of the fabricated IOS ceramic membrane came in the microfiltration range of 0.601–1.103 µm. The IOS membranes showed good corrosion stability both at acidic and alkali conditions.
-
(2)
The fabricated membrane was effective in separation of extraction solvents and dissolved coking coal from the extracted solvent-coal mixture. The efficiency of solvent recovery was dependent on both type of coals and solvent to coal amount. Maximum 75 % and 72 % solvent recovery were obtained at the solvent to coal ratio of 10:1 for coal types A and B, respectively.
-
(3)
NMP had maximum cleaning efficiency (90 %) compared to SDS and deionized water to remove the foulant from the membrane surface.
-
(4)
FTIR spectra confirmed that the recovered solvent was almost free from dissolved coal, could be used further.
-
(5)
The fabricated membrane was more effective for reduction of ash and sulfur content and, enhancement of gross calorific value in case of coal B. The proximate and ultimate analysis showed that the purified coal could be used for industrial application such as steel production.
-
(6)
The cost of prepared IOS ceramic membrane was around $ 79.89/m2 which was less than polymeric membranes.
References
Bhide BD, Stern SA (1991) A new evaluation of membrane processes for the oxygen-enrichment of air. II. Effects of economic parameters and membrane properties. J Membr Sci 62:37–58
Cai MF, Smart RB (1993) Quantitative analysis of N-methyl-2-pyrrolidinone in coal extracts by TGA-FTIR. Energy Fuels 7:52–56
Chawla B, Davis BH (1989) Effect of temperature and solvent on coal extraction under mild conditions. Fuel Process Technol 23:133–148
Christy AA, Liang YZ, Kvalheim OM (1992) On-line pyrolysis of asphaltenes and kerogen studied by diffuse reflectance Fourier transform infrared spectroscopy. Fuel 71:125–127
Couch G (1994) Understanding slagging and fouling during pf combustion. IEACR/72 London: IEA coal research
Iino M, Takanohashi T, Obara S, Tsueta H, Sanokawa Y (1989) Characterization of the extracts and residues from CS2-N-methyl-2-pyrrolidinone mixed solvent extraction. Fuel 68:1588–1593
Jana S, Purkait MK, Mohanty K (2010) Preparation and characterization of low-cost ceramic microfiltration membranes for the removal of chromate from aqueous solutions. Appl Clay Sci 47:317–324
Kashimura N, Takanohashi T, Masaki K, Shishido T, Sato S, Matsumura A, Saito I (2006) Relationship between thermal extraction yield and oxygen-containing functional groups. Energy Fuels 20:2088–2092
Koros WJ, Mahajan R (2000) Pushing the limits on possibilities for large scale gas separation: which strategies? J Membr Sci 175:181–196
Krzton A, Cagniant D, Gruber R, Pajak J, Fortin F, Rouzaud JN (1995) Application of Fourier self-deconvolution to the FT-I.R. characterization of coals and their N-methyl 2-pyrrolidinone extraction products. Fuel 74:217–225
Mishra S, Sharma DK (1999) Solvent extraction and extractive disintegration of coal in anthrancene oil. Fuel 69:1377–1380
Saha B, Mondal C (2013) Removal of ash content from Indian coal by using solvents. Int J Mod Eng Res 3(1):133–138
Shui H (2005) Effect of coal extracted with NMP on its aggregation. Fuel 84:939–941
Shui H, Wang Z, Wang G (2006) Effect of hydrothermal treatment on the extraction of coal in the CS2/NMP mixed solvent. Fuel 85:1798–1802
Singh V, Purkait MK, Chandaliya VK, Biswas PP, Banerjee PK, Das C (2012) Development of membrane based technology for the separation of coal from organic solvent. Desalination 299:123–128
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Singh, V., Meena, N.K., Golder, A.K. et al. Fabrication of cost effective iron ore slime ceramic membrane for the recovery of organic solvent used in coke production. Int J Coal Sci Technol 3, 226–234 (2016). https://doi.org/10.1007/s40789-016-0130-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40789-016-0130-5