Abstract
Objective
A growing body of research has begun investigating the relationship between hormones and female competitiveness. Many researchers have focused on the effect of the menstrual cycle and hormonal contraceptives. Despite many attempts at understanding hormone-behavior associations, contradictory findings have made it difficult to determine the existence of true effects. The aim of the current research was to use a robust methodological design to investigate the effect of fertility probability on four competitive orientations in naturally cycling women and hormonal contraceptive users.
Methods
Using a longitudinal diary study with over 3,900 observations from 21 countries, we explore the effect of fertility probability on four self-report competitive orientations after controlling for menstruation: self-developmental competition, hyper competitiveness, competition avoidance, and lack of interest toward competition.
Results
Using Bayesian estimation for ordinal mixed models, we found that fertility probability was associated with an increase in self-development competitiveness amongst naturally cycling women but not hormonal contraceptive users. We also found weak evidence that hormonal contraceptive users show reduced interest in competing compared to naturally cycling women. There were no other robust effects of fertility or hormonal contraceptive use.
Conclusions
These results suggest that fertility probability is associated with increased fluctuations in self-development competitive motivation and that hormonal contraceptives interfere with this effect. This research contributes to the growing body of literature suggesting that hormonal contraceptives may influence psychology and behavior by disrupting evolved hormonal mechanisms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
From the simplest unicellular organisms (Baker, 2011), through to complex multicellular species, survival and reproduction are subject to competition (West-Eberhard, 1979). Human competition is generally thought to center around access to material resources, such as shelter, food, safety and mates (Smolla et al., 2015). However, competition for social status is an important social and behavioral outcome that humans use to acquire other goods and services. By competing for status, individuals improve their relative position within a community or group. High-status groups and individuals can then leverage their status in order to access resources that aid survival and reproduction, as evidenced by better physical and mental health amongst high-status individuals (Anderson et al., 2012; Scott et al., 2014). To help understand the social and biological drivers of female competition, researchers have begun to explore within-sex differences in competition and competitive motivation.
One avenue of research suggests that female competitiveness may vary across the menstrual cycle and with the use of hormonal contraceptives (HCs). Hormones originate at different organs within the endocrine system and regulate important bodily functions by sending signals around the body. In women,Footnote 1 gonadal hormones (e.g., estradiol, progesterone) are primarily produced in the ovaries and are vital to sex differentiation, healthy development and reproduction (Burrows, 2013). However, a growing body of research suggests that these same hormones are implicated in a range of other functions, including immunoregulation (Bereshchenko et al., 2018; Bouman et al., 2005), cognitive functioning (Sherwin, 2003, 2012; Toffoletto et al., 2014) and the coordination of adaptive social behavior (Adkins-Regan, 2005; Roney, 2016). Of interest to this paper is the potential for hormones to coordinate adaptive behaviors which enrich competitive outcomes for women.
Across the menstrual cycle gonadal hormones fluctuate to enable healthy reproduction, and many researchers have proposed that certain times of the cycle may be associated with increased competition in naturally cycling (NC) women. One theory – the ovulatory competition hypothesis (Durante et al., 2014; Nikiforidis et al., 2017) – posits that competitiveness may be elevated in women who are approaching ovulation, compared to women in other times in the cycle. Evidence for this theory derives from research revealing a mid-cycle increase in intrasexual competition, including the use of beauty products and desire to wear sexy clothes (Batres et al., 2018; Durante et al., 2008; Saad & Stenstrom, 2012). Adding additional support for an association between hormones and competitiveness comes from research suggesting that women using HCs display a reduced level of competitive motivation compared to NC women (e.g., Bradshaw et al., 2020; Buser, 2012; Casto et al., 2020, 2021a), suggesting that exogenous hormones in HCs may suppress underlying (likely non-conscious) competitive motivations.
Critically, in recent years, several well-powered non-replications have cast doubt on the association between fluctuating fertility and psychological and behavioral outcomes (e.g., Arslan et al., 2018; Hahn et al., 2016; Jones et al., 2018a; Ranehill et al., 2018). The repeated non-replication of early research has sparked ongoing debate in the literature, including critiques of methodological designs, analyses and resulting theories (Jones et al., 2019; Stern et al., 2019; but see, Gangestad et al., 2019a, b). Further, a recent review by Arthur et al. (2022) concludes that the association between HCs and competitiveness is highly variable and that critical methodological limitations restrict researchers from drawing firm conclusions about HCs. Taken together, there is a need to conduct large-scale, methodologically robust studies examining menstrual cycle effects on competitiveness with both NC women and HC users.
The aims of this paper were twofold. First, we aimed to investigate the effect of fertility probability on four competitive orientations (self-developmental competition, hyper competitiveness, competition avoidance, and lack of interest toward competition) in NC women and HC users. Second, we sought to address limitations in previous research that would help provide a better understanding of mixed findings. The current study contains several key strengths. Using a longitudinal within-subjects design and continuous measure of fertility, we provide a clearer and more complete picture of self-report competitiveness across the cycle compared to sampling from only fertile and non-fertile phases (Gangestad et al., 2016; Roney, 2018). The decreased reliability of fertile-phase identification is also compensated for by the increase in sample size (Arslan et al., 2018). By using a nuanced measure of competition that includes four competitive orientations, we demonstrate that cycle phase is associated with some, but not all, competitive orientations. Finally, our sample contains over 3900 observations from 21 countries, providing this field with much-needed cross cultural data.
The Menstrual Cycle and Hormonal Contraceptives
The average menstrual cycle occurs over a 28-day period, with 21-to-35-day cycles considered within the normal range (Azari & Kaminski, 2019). Each ovulatory cycle is made up of three key phases – the follicular, ovulatory, and luteal phases – which are each characterized by fluctuations in estradiol and progesterone. Based on the average 28-day cycle, the first 14 days (i.e., the follicular phase) are marked by low progesterone and rising estradiol, which peaks during ovulation (roughly day 14). The final 14 days (i.e., the luteal phase) are characterized by progesterone dominance, which subsides again before the start of the next cycle. Different behaviors have been observed at different times in the cycle, leading researchers to believe that those behaviors may be associated with cycling levels of estradiol and progesterone.
For HC users, the natural menstrual cycle is disrupted through the provision of exogenous hormones. HCs contain progestin (a synthetic form of progesterone) or a combination of progestin and synthetic estradiol (Melo & Creinin, 2016). Several methods of administration are used, including an oral pill taken daily (widely referred to as “the pill”), devices inserted into the body (e.g., the intrauterine device [IUD], the implant/rod), contraceptive injection or the vagina ring. Regardless of the administration method, HCs usually prevent pregnancy by suppressing ovulation or creating an inhospitable environment for a fertilized embryo (Baird & Glasier, 1993). Given that HCs largely disrupt naturally occurring fluctuations in endogenous estradiol and progesterone, this suggests that if the menstrual cycle is associated with fluctuating competitiveness, that HCs may disrupt this pattern.
Hormones and Competition
Given the reproductive relevance of the menstrual cycle, research investigating the relationship between hormones and female behavior has often focused on shifts in mating relevant psychology and behavior. As briefly outlined above, the ovulatory competition hypothesis (Durante et al., 2014; Nikiforidis et al., 2017) posits that ovulation is associated with an increase in intrasexual competitiveness, with a focus on mate-seeking. Support for this theory comes from studies demonstrating that fertile (compared to non-fertile) women self-report increased intrasexual competitiveness, assertiveness, and use of mate attraction strategies, including heightened use of make-up, other grooming behaviours, and desire to wear sexy clothing (Batres et al., 2018; Blake et al., 2017; Durante et al., 2008; Piccoli et al., 2013; Saad & Stenstrom, 2012). However, several well-powered non-replications (e.g., Arslan et al., 2018; Hahn et al, 2016; Schleifenbaum et al., 2021) have since failed to find a robust association between fertility and increased intrasexual competition for mates, casting doubt on the reliability of this association.
Another way researchers have conceptualized competition across the menstrual cycle is by looking at fluctuations in mate preferences. While both sexes compete for access to mates, males see greater fitness gains by securing more mating opportunities, whereas females see greater gains by accessing higher quality mates (Clutton-Brock & Huchard, 2013; Rosvall, 2011). The ovulatory shift hypothesis (Gangestad & Thornhill, 1998, 2008) proposes that during phases of high fertility, women display heightened attraction to men with traits indicating high genetic quality (e.g., masculinity). A meta-analysis of women’s mating preferences indeed suggests that when selecting partners for a short-term relationship, fertility is associated with a small (Hedge’s g = 0.21) preference for masculine partners (Gildersleeve et al., 2014a). Meanwhile, a second meta-analysis (using similar papers) and two critical commentaries argue that the effect of fertility on masculinity preferences is subject to publication bias and “p-hacking” through the use of flexible fertility estimates (Harris et al., 2014; Wood & Carden, 2014; Wood et al., 2014; but see Gildersleeve et al., 2014b). In addition to the contradictory meta-analyses, more recent well-powered research reports no association between cycle phase and masculinity preferences (e.g., Jones et al., 2018a; Marcinkowska et al., 2019; Stern et al., 2021; van Stein et al., 2019), though rebuttal commentaries have been proposed (Gangestad et al., 2019a, b).
Given high variability within the mate seeking literature, other theories may better explain cycle shifts in competition for mates, such as the motivational shift hypothesis (Roney & Simmons, 2017). According to Roney and Simmons (2017), women experience cycle related shifts in motivational priorities between reproductive and other survival behaviors (e.g., feeding). The mid-cycle increase in estradiol is thought to be associated with a general increase in sexual motivation and intrasexual competition, while this same period is associated with a decrease in self-reported food intake. Several well-powered studies find support for this theory, arguing that fertility may be associated with a broad increase in sexual motivation, rather than a specific preference for mates with masculine features (Jünger et al., 2018a, b; Stern et al., 2020).
Given that HCs reduce hormonal variation across the ovulatory cycle, researchers have also investigated whether HCs are associated with systematic differences in intrasexual competition for mates, though the research is again mixed. Batres et al. (2018) found that HC users spend less time putting on make-up compared to NC women. Similarly, Schwarz and Hassebrauck (2008) found that although NC women increased the use of sexualized clothing during the middle of their cycle, the same pattern is not observed in HC users. Regarding mate selection, there is some evidence that HC users do not report a general preference for masculine men (Feinberg et al., 2008; Gori et al., 2014), suggesting that they are not motivated to compete for high quality genes for their offspring. However, mirroring the mate seeking literature for NC women, contradictory research can be found regarding the association between HCs and intrasexual competition (e.g., Jones et al., 2018a; Marcinkowska et al., 2019; for review, Arthur et al., 2022).
Competition for Status
In addition to competition for mates (which may increase status indirectly; for review, Buss & Schmitt, 2019), some research suggests that the menstrual cycle is associated with fluctuations in status-seeking through other pathways. Durante et al. (2014) demonstrated that as women approached the fertile phase of the cycle, they preferred higher relative status (i.e., more value relative to others), while Fisher (2004) and Piccoli et al. (2013) reported that derogation of other women increased with fertility risk in NC women. These three studies suggest that fertility is associated with a desire to maximize one’s social position relative to other women. When status is achieved through direct competition with others, HC users displayed poorer competitive persistence compared to NC women, as seen in both physically and psychologically difficult tasks (Bradshaw et al., 2020; Casto et al., 2020). This work collectively suggests that endogenous hormones may lead to fluctuations in status motivation and that HCs may blunt this effect.
The literature regarding competition for economic resources (which can also enhance status) is again mixed. In a novel study, Miller et al. (2007) recorded tip earnings for professional lap dancers, finding that HC users earned less than NC women in the fertile phase. Although the authors conclude that men are more attracted to fertile women, it is also possible that HCs suppress a mid-cycle increase in competitiveness experienced by NC women. Other researchers have used a combination of tasks from social psychology (e.g., social value orientation; Anderl et al., 2015) and behavioral economics, such as the dictator and ultimatum games (Eisenbruch & Roney, 2016; Lucas & Koff, 2013), auction games (Chen et al., 2013; Pearson & Schipper, 2013; Schipper, 2015), and the piece-rate or tournament entry scheme (Buser, 2012; Ranehill et al., 2018; Wozniak et al., 2014), many of which report contradictory or null associations between HCs and competitive outcomes. A detailed analysis of this literature is presented in Arthur et al. (2022) and Nikiforidis et al. (2017).
Despite the importance of accurately measuring female competitiveness, researchers in this field face social and methodological constraints. For example, experimental lab competitions are often male-biased and fail to account for gender differences in the socialization of competitive behavior (Casto & Prasad, 2017). Further, in the mate competition literature, researchers often make assumptions about what is motivating female behavior. For example, women’s beautification (e.g., provocative dress, use of make-up) has often been used as evidence of increased mating motivation. While mating likely motivates some appearance enhancement, women’s beautification also leads to benefits beyond mating, including improved social interactions and personal status acquisition (see Blake, 2022; Bradshaw & DelPriore, 2022). It is important to consider these limitations when reviewing the existing research and when designing future investigations. By measuring competitive inclinations directly, researchers are able to bypass these limitations.
Theoretical Justification for Shifting Levels of Competitiveness
As with all evolved traits and behaviors, engaging in competition can be costly and often involves trade-offs with other adaptive behaviors. For women, an underlying tension regarding the costs of competition is that securing valuable resources requires some level of competition, but if a woman is outwardly perceived by others as a threat, then she may herself become the victim of competition. For example, despite the advantages conferred upon high-status individuals, blatant status-seeking in women is often met with backlash due to gender norm violations (Benenson, 2013; Campbell, 2013). Women and girls also express distress when other women outperform them in terms of appearance, popularity, employment and academic success (Benenson & Benarroch, 1998; Simmons, 2002; Vigil, 2007; for review, Reynolds, 2021). Maintaining a continuous level of high competition may therefore inhibit a woman’s ability to maintain positive relationships with others, especially other women.
Instead, we propose that women alternate (or, ‘trade-off’) between periods of high and low competitive motivation, a process which may be mediated by underlying hormonal mechanisms (Arthur et al., 2022). Specifically, it is possible that women limit competition to conditions in which it is most beneficial, such as periods of high fertility when potential reproductive gains are highest. Elsewhere the costs of competing may outweigh the benefits, resulting in an energetic shift away from competition and toward other motivational priorities (e.g., affiliation). This theoretical framework presumes that high competitiveness during the fertile window is a byproduct of increased mating competition during this period. It bears distinct conceptual cohesion with the motivational shift hypothesis (Roney & Simmons, 2017) which posits that when reproductive gains are highest (i.e., the fertile phase) women experience an increase in sexual motivation, while the non-reproductive phase is associated with increases in survival (i.e., feeding) behavior. Understanding how competition fluctuates across the menstrual cycle is the one step towards unpacking this theoretical model.
The Current Study
In the current study, we sought to resolve existing uncertainty regarding menstrual cycle shifts in competition for NC women and HC users. Using an online self-report diary study, we collected data regarding HC use, menstrual cycle phase and self-report competitiveness across four competitive orientations: self-developmental competitiveness, hypercompetitive, competitive avoidance, and lack of interest toward competition (Orosz et al., 2018). We used menstrual onset dates to form a continuous measure of fertility probability, providing a clearer understanding of how competitiveness changes across the full menstrual cycle. The method and analytic strategy reported here was pre-registered as part of a separate investigation from this project (https://osf.io/zw8qx). The predictions here were not preregistered, however the first author formulated the hypotheses without prior access to the data and before any analyses had been conducted.
To avoid making assumptions about what women are motivated to compete for, we used a general measure of competition that assessed competitive inclinations without specifying the competitive strategy or competitive outcome. Measuring competition through four sub-domains further broadened understanding whether some competitive orientations demonstrate a stronger association with certain cycle phases relative to others. Self-development (i.e., relating to achievement motivation and success) and hyper-competitiveness (i.e., a strong results-based motivation and a desire to dominate others) are both approach-based orientations, with high scores representing a desire to compete. In contrast, high scores in competitive avoidance (i.e., an avoidance of competitions) and lack of interest in competition (i.e., an overall disinterest in competition) represent a desire to avoid competitive situations. Based on existing research demonstrating a mid-cycle increase in competitiveness in naturally cycling women but not hormonal contraceptive users, we predicted:
-
Hypothesis 1: An ovulatory increase in self-reported a) self-development competitiveness and b) hyper competitiveness, in naturally cycling women but not hormonal contraceptive users.
-
Hypothesis 2: An ovulatory decrease in self-reported a) competition avoidance and b) lack of interest toward competition, in naturally cycling women but not hormonal contraceptive users.
Method
Participants
Three hundred and thirty-six cis-gender women (Mage = 26.51, SD = 6.25) were recruited from the University of Melbourne undergraduate participation pool and the general community for a study involving daily surveys on the menstrual cycle and psychological phenomena. Selection criteria for eligibility were fluency in English; regular menstrual cycles; self-reported confidence about menstrual cycle length exceeding the scale mid-point (i.e., ‘3 – somewhat confident’ and above); menstrual cycle length between 22–35 days (M = 28.13, SD = 2.94); aged between 18–45 years; pre-menopausal; no emergency contraception or breastfeeding or pregnancy use within the past three months; no polycystic ovarian syndrome or endometriosis; and no medically diagnosed fertility or endocrine issues, leaving n = 278 (Mage = 26.34, SD = 6.08).
Most participants were exclusively heterosexual (62.78%), 25.94% were occasionally or more than occasionally homosexual, 4.51% were bisexual, 5.64% were pansexual, 1.88% were exclusively homosexual and 1.13% were asexual. More than half (56.5%) of participants reported being in a relationship (n = 157) and the remaining 43.5% were single (n = 121). The largest proportion of participants of participants (18.96%) were from Australia/New Zealand, 18.59% reported mixed ethnicity, 15.24% were South-East Asian, 10.41% were North American, 7.43% were Southern European, 7.06% East Asian, 6.69% were Northern European and the remaining 15.61% each occupied less than 4% each and were collectively from Southern, Western or Central Asia, Latin America, Eastern or Western Europe, African or unspecified. Most participants reported average relative socio-economic status (57.25%), 4.09% reported low socio-economic status, and 38.66% reported high socio-economic status. For religion, 65.43% report no religious affiliation, 24.91% report being religious and 9.67% did not respond to this question.
Hormonal Contraceptive Use
The final sample included 69.63% NC women (n = 192) and 30.37% HC users (n = 86). Of the HC users, 74.39% used combined oral contraceptives, 4.88% used the vaginal ring, 3.66% used the contraceptive implant, 2.44% used the contraceptive patch and 14.63% (n = 12) did not provide sufficient information to determine contraceptive type. Most HC users (65.85%) used contraceptives containing androgenic progestins, 19.51% contained anti-androgenic progestins and 14.63% (n = 12) did not provide sufficient information to determine androgen type.
Procedure
Participants were invited to join the study in exchange for a personalized feedback report, including information about how their menstrual cycle relates to individual changes in a range of variables from the ongoing study (e.g., mood, big-5 personality, health, assertiveness, self-esteem and impulsiveness). The undergraduate participants were also provided with course credit. This study was conducted using formR (Arslan et al., 2020), an open-source survey framework that was specifically designed for longitudinal diary studies. A brief prescreening and baseline survey were used to collect demographic and menstrual cycle characteristics, followed by 28-days of daily surveys. During the daily survey stage, participants were automatically issued a daily email link at 5 pm local time. After 28 days, participants were offered the opportunity to complete the study or continue collecting data for an additional 28 days (median number of daily entries = 10, M = 14.34, SD = 12.87, range = 1–53). Following the final daily survey and a 10 day pause, participants were emailed a link to the follow-up survey. Feedback reports were generated and shared with participants shortly after the final survey. All surveys were accessed via participants personal devices (including laptops, tablets, and mobile phones). This study was approved by the University of Melbourne Human Research Ethics. All data were collected between September 2020 to June 2021.
Measures
Two sets of measures were used in this study: person-level variables (measured during the prescreening, baseline or follow up survey) and within-person variables (measured daily). Person-level variables included questions regarding HC use and the menstrual cycle. To identify group level differences between NC women and HC users, person-level data was also collected for depression, anxiety, stress, mate value and sociosexual orientation. As part of a larger study, within-person measures included questions regarding current menstrual cycle status, competitiveness, self-objectification, affect, Big-5 personality (i.e., agreeableness, conscientiousness, extraversion, neuroticism, openness), agency, impulsiveness, self-regulation, health, and sexual behavior. Measures related to competitiveness are reported in the current paper. Examples of each scale measure are provided in Table 1.
Competitiveness
Competitiveness was measured using the Multidimensional Competitive Orientation Inventory (MCOI; Orosz et al., 2018), a 12-item scale with four subscales: hypercompetitive orientation (α = 0.83), self-developmental competitive orientation (α = 0.83), competition avoidance (α = 0.83), and lack of interest toward competition (α = 0.77). All items were ordinal and measured on a 5-point scale ranging from strongly disagree, disagree, neither, agree, strongly agree. One item within each subscale was randomly presented each day.
Cronbach’s alpha after reverse coding where appropriate found low internal consistency across the four sub-scales (α = 0.495) and Pearson’s correlations between the two approach (R = 0.33) and two avoidant (R = 0.20) competitive orientations were small suggesting that retaining the four orientation types (as opposed to averaging values across orientations) was appropriate for these data.
Sociosexual Orientation (SOI-R)
Sociosexual orientation was measured using the Revised Sociosexual Orientation Scale (SOI-R; Penke & Asendorpf, 2008). In the baseline survey, participants were asked a total of nine questions about their sexual behavior, attitudes and desires.
Mate Value
Mate value was measured using the Mate Value Scale (Edlund & Sagarin, 2014). In the baseline survey, participants were asked a total of nine questions about their perceptions of themselves as a romantic partner.
Depression, Anxiety and Stress
Mood disorders were measured in the baseline survey using the short-form Depression, Anxiety and Stress Scale (DASS-21; Lovibond & Lovibond, 1995).
Contraceptive Use
Participants indicated their contraceptive use via the following multi-choice categories: hormonal contraceptives (e.g., the pill, hormonal implant/rod, depot injections, vaginal ring, hormone plasters), barrier method (e.g., condoms, diaphragm), period/fertility tracking app (e.g., Clue, Flo, Glow), fertility awareness method (e.g., diary, calendar, temperature), having no (or less) sexual intercourse when fertile, hormonal intrauterine device (e.g., Mirena), copper intrauterine device, morning-after pill, other contraceptive, or none. Participants were also offered an opportunity to write the name of their current HC. This information was used to determine details such as route of administration (e.g., pill, patch) and androgen content (see Participants section above). As noted in the Participants section, anyone indicating usage of the morning after pill within the past three months were excluded from the study.
Menstrual Cycle Characteristics
Fertility probability was estimated using the backward-counting method. This method counts backward from the reported or estimated onset of menses to the day on which the outcome variable is sampled. For example, a daily survey completed 5 days prior to a participant’s next menstrual onset would be assigned as backward-counted Day -5. Each backward-counted day corresponded to a specific fertility probability value based on the estimates generated by Stirnemann et al. (2013), a method advocated for by Gangestad et al. (2016). Fertility probability estimates were assigned to all participants (irrespective of HC and NC grouping), enabling us to compare patterns between HC users and NC women. Cycle-phase variation among HC participants would suggest variation unrelated to endogenous hormones.
To gather the necessary cycle data, the baseline survey asked participants to report their average cycle length and the start date of their current cycle. In each daily survey and at follow-up, participants were asked if and when their subsequent cycle had started. This survey question was accompanied by a calendar, as recommended by Welling and Burriss (2019) to assist recall accuracy. All responses were checked for consistency and where discrepancies emerged with multiple onsets reported within the same week, we used the median date. In a small number of cases where onsets were reported within the same fortnight (1.26%), we excluded these data from analysis. In cases where the next menstrual onset date was not reported (31.94% of cycles), we inferred it from the average cycle length reported by the participant at baseline (as recommended by Welling and Burriss 2019). Each day, participants also indicated whether they were currently menstruating. In some cases, participants indicated that their cycle had started but did not indicate that they were bleeding, thus we coerced the first day of each cycle to contain values indicative of menstruation.
Data Analysis
HCs and Menstrual Cycle Effects
Bayesian estimation for ordinal mixed models is required to test the effects of fertility and HCs on ordinal outcomes. We used default (uninformative) priors, which ensured that our parameter estimates were maximally influenced by the data and were asymptotically equivalent to those obtained under maximum likelihood estimation. Model convergence was determined by PSR values reaching < 1.05, after which the number of Bayesian iterations was doubled to ensure stable convergence was reached. We concluded there was evidence for an effect whenever the 95% credible intervals did not cross zero. Bayesian multilevel models correct for problems of multiple comparisons through the use of partial pooling and low group level variation (see Gelman et al., 2012). The inflation of type 1 errors is therefore unlikely. Due to the nature of Bayesian models and random number generation in r simulations (including brms), output can vary between replications of the same model. To create reproducible output we applied set.seed(123) to all primary models. By setting a uniform seed value simulations can be reproduced.
Our primary model controlled for menstruation and included a random slope for fertility probability and a random intercept for the individual (model 1). The main predictor of interest was the interaction between fertility probability and HC, with support for mid-cycle shifts in outcome variables indicated by an interaction where fertility probability affected the outcome variable in the NC group only.
In Wilkinson notation (Bates et al., 2014; Wilkinson & Rogers, 1973), model 1 equation can be formalised as:
Robustness Tests
In models including evidence of an interaction, we ran additional robustness tests. First, we ran analyses excluding participants who did not reported a next menstrual onset date, as fertility probability for these participants was inferred based on the reported cycle length (model 2). We then repeated models among participants who were most likely to ovulate (model 3) by excluding people who were aged over 40, had highly variable cycle lengths (varied by over 7 days), were heavy smokers (1 + pack per day), high self-reported high stress levels or had recently lost a large amount of weight (over 8 kgs), had a BMI below 18.5 or above 25, or were professional and semi-professional athletes. To account for possible progestogenic effects we also created a luteal phase variable by backwards counting four to ten days before the onset of the next menstrual cycle and included it as an additional control variable to the primary model (model 4). Finally, we ran additional models controlling for the potential confounders of age (model 5), relationship status (model 6) and sociosexual orientation (model 7). Previous research suggests competition may vary based on age (Mayr et al., 2012) and relationship status (Cobey et al., 2013), and HC users in our sample reported higher scores on the SOI than NC users (see Table 2 below). Datasets, model specification code and sample output are available in the Open Science Framework (https://osf.io/rs6bm).
Power and effect size estimates
To approximate a minimum detectable effect size, we simulated power for identical models to the Bayesian formula but modelled using the R package lme4. We used the simr package to estimate the power obtained to detect effects of particular sizes for the primary interaction of interest (HC × fertility probability). To do so, we imputed the average observed fixed effect for the other variables in the model (i.e., averaged across all outcome variables), then simulated power to detect an interaction at effect sizes ranging from 0.1–0.3 (at intervals of 0.05). One thousand simulations indicated that we had 80% power to detect a minimum effect of b = ~ 0.18. To estimate Marginal R2 and Conditional R2 values we calculated Nakagawa’s R2 (Lüdecke et al., 2021) for the GLMM models with identical formula specification to the Bayesian models. Marginal R2 provides variance explained only by fixed effects and conditional R2 provides the variance explained by both fixed and random effects (Nakagawa & Schielzeth, 2013).
Data Visualization
To continuously visualize outcome variation according to cycle phase without imposing discrete phases such as menstruation and the fertile window, we fit Bayesian mixed models with a Gaussian family and cyclic cubic splines over backward-counted cycle days by HC status. For slight regularization, we set half-normal priors with a SD of 1 on the random intercepts. We then took 100 random samples from the posterior and visualized the conditional means for the continuous splines by HC status, as well as the difference in splines between NC and HC groups. The resulting visualized variation reflects the average patterns in the data and includes the uncertainty resulting from the person-level clustering in the data.
Results
Descriptive Statistics
Compared to NC women, HC users had less restrictive sociosexual behavior and attitudes. No other differences emerged for person level variables (see Table 2).
Hypothesis Testing
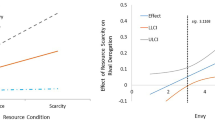
Table 3 contains the results of the primary mixed models (model 1) for competitive orientation. As hypothesized, there was evidence for an interaction between Fertility x HC use for self-development competitiveness (H1a; see Fig. 1). NC women (b = 0.73, SE = 0.28, CrI = 0.18, 1.29), but not HC users (b = -0.12, SE = 0.37, CrI = -0.85, 0.59), experienced a mid-cycle increase in self-report self-development competitiveness. Robustness tests indicated that this effect was 23% stronger when we restricted the sample to women who we didn’t infer ovulation without a next menstrual onset date (b = 0.90, SE = 0.32, CrI = 0.28, 1.52). However, restricting the sample to women most likely to ovulate did not provide evidence of an effect (b = 0.34, SE = 0.58, CrI = -0.81, 1.50), likely owing to the 59.6% reduction in sample size (n = 76). The interaction effect was not moderated by age (CrI = -0.04, 0.30), relationship status (CrI = -1.32, 3.00) or sociosexual orientation (CrI = -0.59, 1.40). For competitive avoidance (H2a), there was weak evidence for a Fertility x HC use interaction. Although this interaction suggests cycle shifts may vary between NC women and HC users, the large credible interval (which almost crosses zero) suggests a high degree of uncertainty and visual inspection of competitive avoidance (Fig. 2) produces no clear patterns for either group.
Levels of self-development competitiveness as a function of backward-counted cycle day. Note. This figure shows the conditional mean for self-development competitiveness (y-axis), plotted based on backward-counted cycle day (x-axis). Black = Hormonal contraceptive users; Red = Naturally cycling women; Blue = difference in splines between NC and HC groups; dashed line = estimated day of ovulation
Levels of hyper-competitiveness, lack of interest in competition and competitive avoidance as a function of backward-counted cycle day. Note. This figure shows the conditional mean for a) hyper-competitiveness; b) lack of interest in competition; c) competitive avoidance (y-axis), plotted based on backward-counted cycle day (x-axis). Black = Hormonal contraceptive users; Red = Naturally cycling women; Blue = difference in splines between NC and HC groups; dashed line = estimated day of ovulation
Counter to our hypotheses, hyper-competitiveness and lack of interest in competition did not provide evidence for a Fertility x HC interaction. Even so, a negative association between HCs and hyper-competitiveness was observed, indicating that HC users report less hyper-competitiveness compared to NC women. Conversely, a positive association between HCs and lack of interest in competition indicates that HC users may lack interest in competing compared to NC women. For both variables, large credible intervals (and credible intervals close to zero) suggest a high degree of uncertainty. Further, for hyper-competitiveness, competitive avoidance and lack of interest in competition, setting a different seed value (or resetting the model to randomly generate a seed value) produced some credible intervals that crossed zero.
Plotting the effects of backward counted cycle day on the remaining outcome variables, delineated by HC and NC subgroups, depicts an interesting, albeit exploratory, relationships between variables (see Fig. 2). Hyper-competitiveness visually peaked for NC women around the estimated day of ovulation, where such effects are not seen for HC users. In contrast, lack of interest in competition and competitive avoidance displayed no clear patterns for HC users of NC women.
Discussion
We used a longitudinal diary study to investigate the relationship between competitiveness, menstrual cycle shifts and HC use. We investigated fertility probability effects for four competitive orientations: self-developmental competition, hyper competitiveness, competition avoidance, and lack of interest toward competition. Each orientation captured a different aspect of competition and was domain general, providing nuanced insights regarding hormones and competition that avoided making inferences about the outcome of competition or the competitive strategy. Much like the menstrual cycle literature more broadly, results were mixed across the four competitive orientations. We found evidence of a mid-cycle increase in self-development competitiveness for NC women but not HC users. We also found preliminary evidence for a negative association between HCs and hyper-competitiveness and a positive association between HCs and lack of interest in competition, but note that large credible intervals (and credible intervals close to zero) suggested a high degree of uncertainty. Competitive avoidance was not reliably associated with fertility probability, HC use or their interaction, with credible intervals for the interaction likewise suggesting a high degree of uncertainty and visual inspection of competitive avoidance yielding no clear patterns. Below we review each orientation within the context of existing literature and in relation to the theoretical framework we presented in the introduction.
Self-development competition is an approach-based orientation, related to achievement motivation and success (Orosz et al., 2018). We found evidence of this outcome being associated with the interaction between HC use and fertility probability. Sub-group analysis revealed that when fertility probability was high compared to low, NC women but not HC users self-reported higher self-development competitiveness. To our knowledge, this is the first study to identify a mid-cycle increase in a domain-general motivation to compete and achieve in order to benefit the self, a pattern that is not observed in HC users. Compared to fertility x HC interactions observed in similar studies (e.g., Arslan et al., 2018; Schleifenbaum et al., 2021), the size of this fixed effect (marginal R2 = 0.003) was of a similar magnitude, while the size of the fixed and random effects (conditional R2 = 0.484) and the size of the unstandardizedFootnote 2 regression coefficient of the interaction was considerably larger. The conditional R2 reflects the between level variance (i.e., differences in intercepts among subjects), and variance around the effect of fertility probability for each individual (i.e., if the slope varies among different people). This suggests that we had considerably more heterogeneity at the between-person level, which may reflect cross-cultural differences in our diverse participant pool compared to similar studies (e.g., Arslan et al., 2018; Schleifenbaum et al., 2021). From a theoretical perspective, mid-cycle increases in self-development competition that is only observed in NC women provides support for both the ovulatory competition hypothesis (Durante et al., 2014; Nikiforidis et al., 2017) and the theory of HCs as endocrine mediated behavioral disruptors (Arthur et al., 2022).
Competitive avoidance is an avoidance-based orientation, with scores indicating a general anxiety towards competitive situations (Orosz et al., 2018). In our study, weak evidence for an interaction between fertility and HC use revealed that the effect of fertility on competitive avoidance differed for NC women and HC users. However, analyzing each group separately revealed no evidence for an association between fertility and competitive avoidance. In other words, while the slope of fertility differed between the groups, neither slope was significantly different from zero. This result could indicate that the current work was insufficiently powered to detect a small true effect, though sensitivity analysis suggested that our work was powered to detect a minimum effect size of b = ~ 0.18. It is also possible that there is no notable association between fertility or HCs and competitive avoidance, replicating past work (e.g., Hahn et al., 2016; Lucas & Koff, 2013; Ranehill et al., 2018). Given the large credible interval for this interaction, and that the interval approached zero (or included zero, depending on the seed value), our data suggests that competitive avoidance is probably not associated with fertility or HC use to any meaningful magnitude. We note that this does not rule out the possibility that competitive avoidance is associated with other endogenous hormones and expand upon this possibility in the ‘Future Directions’ below.
Results for hyper-competitiveness and lack of interest in competition provided weak evidence in favor of lower competitive motivation in HC users. Specifically, HC use was negatively associated with hyper-competitiveness and positively associated with a lack of interest in competition. A general reduction in competitiveness for HC users is in line with previous research demonstrating that HCs are associated with reduced competitive motivation. For instance, Casto et al., (2020, 2021a) reported that when competing in a physical task, NC women were willing to compete longer than HC users. Bradshaw et al. (2020) reported a similar association between reduced competitive persistence and HCs, whereby NC women persist for longer and thus outperform HC users on a series of tasks. For both of our findings, however, credible intervals were large and close to zero, suggesting a high degree of uncertainty. We recommend that these results be interpreted as preliminary and suggest that future research should aim to replicate these effects in a larger sample.
As highlighted by a reviewer, visual inspection of Fig. 1 (self-developmental) and Fig. 2 (specifically, hyper-competitive and competitive avoidance) indicates some variability around menstruation. Although our statistical analysis did not detect evidence for an association between menstruation and any competitive orientation, it is possible that there are other cycle related peaks (or troughs) in competitiveness that our data did not detect. Future investigations into patterns across the entire cycle, including statistical operationalization of other cycle events (i.e., luteal phase, pre-menstrual phase) may provide further insight into shifting motivational states.
Several null findings are also worth investigating. Counter to our hypotheses, hyper-competitiveness, competitive avoidance and lack of interest in competition were not associated with fertility probability in NC women, nor was there an interaction between fertility probability and HC for hyper-competitiveness or lack of interest in competition. Women are known to face societal pressure in competitive contexts (see Benenson, 2013; Campbell, 2013; Reynolds 2021) and self-report surveys may have been vulnerable to impression management when responding to questions about hyper-competitiveness. In support of this possibility, the intercept for hyper-competitiveness was lower than self-development competitiveness (see Table 3), indicating that women were less likely to rate themselves as hyper-competitive overall. Another possibility is that desire to win and to beat others is not associated with cycle phase or HC use. Critically, previous studies reporting that cycle phase and HCs are not associated with overt competitiveness (e.g., Hahn et al., 2016; Ranehill et al., 2018) have used measurement tools that are undermined by the socialization of competition in women, including self-report measures or male-biased experimental designs tasks (for review, Casto & Prasad, 2017). Research investigating behavioral avoidance of (or disinterest in) competitions (as opposed to competitive motivation) is lacking and limits our ability to theorize regarding hormonal correlates of competitive avoidance. Using behavioral measures to investigate associations between cycle phase and the avoidance of socially competitive situations may provide a novel approach to measuring competition in women.
Nuanced Competitive Strategies in Women: Insights for Status Seeking Among Users and Non-Users
The results reported here demonstrate that when studying competition in women, it is important to differentiate between different types of competition. Although self-developmental competitiveness and hyper-competitiveness are both approach-based orientations, each orientation was associated with different behavioral strategies and status outcomes. One theory of status-seeking in humans indicates that there are two main pathways to status attainment: the prestige route and the dominance route (Cheng et al., 2013, 2014; Henrich & Gil-White, 2001). Prestige-based strategies involve displays of skills and knowledge, while dominance-based strategies involve using power to coerce or influence others. Of the two approach-based competitive strategies studied here, self-development competition aligns with prestige while hyper-competitiveness aligns with dominance. For instance, self-development competition is centered on the improvement of personal skills and abilities, while hyper-competitiveness represents a desire to win at any cost (Orosz et al., 2018). Research also shows that both prestige and self-development competition are negatively correlated with aggression (Johnson et al., 2007; Ryckman et al., 1996).
Within the context of status research, a potential implication of our findings is that HC use may decrease the mid-cycle peak in prestige-seeking observed in NC women, and that HC use may decrease dominance-based status-seeking strategies overall. Although both dominance and prestige are effective in obtaining status (Cheng et al., 2013; McClanahan et al., 2021), women are more likely to use prestige (Hays, 2013). A mid-cycle suppression of prestige-motivated competitiveness may thus have implications on HC users’ ability to gain status. For instance, while fertile women may use prestige to signal that they are competent and skilled, an attenuation of this relationship suggests that HC use diminishes the desire to signal competence and instrumental value to others. Given that we did not measure dominance and prestige, we acknowledge that the link between our research and these constructs is highly speculative and requires direct investigation. Even so, given the importance of status in human society this is an important area for future research.
Proposed Hormonal Mechanisms Affecting Hormones and Competition
Although we did not measure hormone levels directly, the fertile phase is accompanied by rising estradiol until ovulation occurs (Baird & Fraser, 1974). A fertile phase increase in self-development competition may thus be driven by an increase in estradiol. Existing hormone research has illustrated several ties between estradiol and competitive behaviors in humans. In adolescent girls, estradiol is positively correlated with aggressive risk taking (Vermeersch et al., 2008), while women show positive correlations between estradiol and implicit power motivation (Stanton & Edelstein, 2009; Stanton & Schultheiss, 2007) and assertiveness (Blake et al., 2017). In men, estradiol has also been associated with psychological aggression (Eriksson et al., 2003), which includes behaviors women commonly use to compete with others (e.g., social isolation, humiliation). For these reasons, we propose further studies investigating the role of estradiol on self-developmental competition.
The mechanisms through which HCs are associated with competition is not yet well understood, though it is possible that HCs influence competition through multiple pathways (for review, Arthur et al., 2022). One possibility is that HCs suppress a mid-cycle increase in estradiol. This possibility is supported by previous research demonstrating both a positive correlation between endogenous estrogens and competition, and that HCs attenuate the relationship between estradiol and power that is experienced by NC women (Stanton & Edelstein, 2009; Stanton & Schultheiss, 2007). Here we find that HCs attenuated the mid-cycle increase in self-developmental competition, which may support the role of estradiol in achievement-based competitiveness. Direct tests of the relationship between estradiol and competition in HC users presents a fruitful opportunity for research.
Another possible mechanism that HCs may disrupt competition is through the relationship between HCs and testosterone. Testosterone has been linked to competition in men (for review, Archer, 2006) and, to a lesser extent, women (Casto et al, 2019; Edwards & Casto, 2013). A well-documented effect of HCs is the lowering of serum total and free testosterone levels by an average of 31% and 61%, respectively (Zimmerman et al., 2014). Linking HCs and testosterone in a sample of NC women and a subset of HC users (oral contraceptive users only), Casto et al., (2021a, b) revealed that lower testosterone levels in HC users explained differences in competitiveness between HC users and NC women. In our study, the main effect of HC use on hyper-competitiveness may thus be explained by the depression of testosterone across the full cycle for HC users. Some women also experience a small-to-moderate mid-cycle increase in testosterone (Bui et al., 2013; Rothman et al., 2011) which may explain the mid-cycle increases in self-development competitiveness in NC women but not HC users. However, few studies have explicitly tested the effect of testosterone and competitiveness in fertile NC women, limiting our ability to draw further conclusions.
One final possibility is that HCs disrupt competition through the extended delivery of synthetic progestins. During reproduction, an important physiological role of progesterone is to maintain a healthy pregnancy. Some research suggests that the luteal phase is associated with increased health-related risks. For example, pathogen avoidance is elevated for women in the luteal phase compared to follicular phase (Miłkowska et al., 2021; but see, Jones et al., 2018b; Stern & Shiramizu, 2022), and for female mice following injection with progesterone (Bressan & Kramer, 2022). Progesterone is also associated with higher subjective anxiety in general (Reynolds et al., 2018; but see, Hahn et al., 2020). It may be that progesterone also inhibits risk taking that is specific to social settings, including competition (Casto et al., 2021b). For example, in the case of hyper-competitiveness, high levels of synthetic progesterone in HC may lead HC users to avoid situations that require them to compete with others. It is important to note that few studies have directly investigated the link between competition and progesterone, providing an opportunity for future research.
Though we have presented several possible mechanisms through which hormones may influence competition, it is likely that competition is mediated through a range of complex and specialized neuroendocrine pathways. For instance, mating relevant competition may be concentrated in the fertile phase, when estradiol is high, and conception is most likely. In contrast, testosterone levels may influence baseline levels of competitiveness and not competitive fluctuations across the cycle. And finally, progesterone may drive avoidance of competitions that are physically or psychologically risky to a woman (or her future offspring). An important takeaway point is that these questions warrant further investigation and that investing time and money in understanding female physiology is important because it affects a significant proportion of the world’s population.
Limitations and Future Directions
The research presented here has several limitations. Our design relied on self-report measures of competition, raising concerns about social desirability bias and self-enhancement (Paulhus & Vazire, 2007). Future research testing the link between the self-report measures used in this study (i.e., MCOI; Orosz et al., 2018) and actual behavior would enhance the strength and validity of our conclusions. Our design also uses a general measure of competition (which could mean different things to different people) and assumes that participants are aware of changes to their own competitive motivations, despite the fact that hormones may mediate behavior through non-conscious mechanisms. Each day, our survey structure presented participants with one of three possible questions for each of the four competitive orientations. Although we did not use a single item measure (which are less reliable than scales with multiple items; Diamantopoulos et al., 2012), the presentation of questions did vary across participants and days, which may have led to an increase in measurement error. A replication of this study using a larger number of survey items or demonstrating changes to competitive behavior (in place of competitive motivation) would strengthen the impact and validity of these findings.
Our design also had limitations in terms of fertility estimates and sample size. In addition to estimating fertility using daily diary entries, employing non-counting methods (e.g., luteinizing hormone tests) would have further improved the validity of our estimates. Even so, research has shown that studies with large samples and repeated measures have many strengths, including accounting for reduced accuracy in identifying the fertile phase (Arslan et al., 2022). Although our analyses included almost 3,900 observations, sensitivity power analyses indicated that we were only sufficiently powered to detect effect sizes of b = ~ 0.18. Robustness tests which restricted our analyses to women who reported a next menstrual onset date (i.e., excluding participants that we did not need to infer a menstrual onset date; model 2) and women most likely to ovulate (model 3), increased the validity of our cycle phase estimates but further reduced the overall samples size. A larger sample of HC users would have allowed us to investigate within-group differences among HC users, including comparisons of administration type, chemical formula and duration of HC use. These limitations could be addressed in future research by introducing direct hormone measurement (e.g., salivary assays, luteinizing hormone tests) and increasing the total sample.
A final limitation of this study is that causal effects of HCs cannot be concluded based on the current quasi-experimental design. It is possible that HC users differ from non-users in a meaningful way, leading to unobserved selection effects that may influence competitive motivation. Botzet et al. (2021) attempted to disentangle causal effects from selection effects in a study investigating the relationship between HCs and measures of relationship quality and sexual function. In their analysis, they found that age and relationship duration were significant predictors of contraceptive choice (e.g., hormonal vs non-hormonal), suggesting that HC users and non-users vary in meaningful ways. In any case, to truly determine causation, a randomized control trial or longitudinal design where participants are tested both on and off HCs would be needed.
Theoretical and Practical Implications
The theory presented in our introduction proposes that women alternate (or, ‘trade-off’) between periods of high and low competitive motivation. Our data partially supports this theory while also highlighting limitations in the way competition is researched among female populations. To better understand the relationship between the menstrual cycle and competitiveness, researchers should consider using (or developing) measures which bypass (or are sensitive to) the socialization of female competitiveness. Further, although this theoretical framework presumes that high competitiveness during the fertile window is a byproduct of increased mating competition during this period, the data collected in the current study cannot test this hypothesis. To investigate what drives increased competitiveness during the fertile phase, researchers may seek to correlate mating related competition with other forms of competition.
One final caveat is that our study looked at possible high and low phases of competition, though a more developed theoretical framework requires a trade with other biologically relevant behaviours. As outlined above, the motivational shift hypothesis (Roney & Simmons, 2017) proposes that the fertile phase is associated with an increase in sexual motivation and decrease in feeding behavior. Investigating a trade-off between competition and affiliation, or competition and cooperation may better inform our understanding of the functional benefits of cycling competitive motivation. Future research should continue to investigate the possibility of an endocrine-mediated trade-off between competition and other key behaviours.
Understanding how the menstrual cycle and HCs influence competition is important because it contributes to our holistic understanding of female competition. Engaging in competitive situations provides women with a host of benefits that are only available to those willing to compete against others. For example, achieving gender equality requires that men and women compete for access to the same resources. Likewise, the finite number of mates in an environment means that only women who compete effectively will gain access to high-quality mates and the resources that they hold. Failing to investigate the way behavior changes across the menstrual cycle limits our understanding of an important biological correlate of status behaviors, and suppressing it risks the perpetuation of masculinity as the dominant biological framework for humans. Identifying potential barriers to competition – be it social, political or biological – is of value to women around the world.
Conclusion
Our research suggests that NC women experience fluctuations in achievement-oriented competitiveness across the cycle. HC use eliminated the mid-cycle peak in self-developmental competitiveness and may be associated with reduced competitive motivation overall. By understanding the effect of HCs and fertility on competitiveness, this information can be used to inform women of the possible costs and benefits of HC use. This study improves our understanding of hormones and competition while providing new information regarding the potential psychological consequences of HCs.
Data and Code Availability
The data generated during the current study are available in the Open Science Framework repository, https://osf.io/rs6bm
Notes
We use women/woman throughout this manuscript to indicate human persons with a menstrual cycle, however we acknowledge that not all people who experience a menstrual cycle identify as women.
References
Adkins-Regan, E. (2005). Hormones and animal social behavior. Princeton University Press.
Anderl, C., Hahn, T., Notebaert, K., Klotz, C., Rutter, B., & Windmann, S. (2015). Cooperative preferences fluctuate across the menstrual cycle. Judgment and Decision Making, 10(5), 400.
Anderson, C., Kraus, M. W., Galinsky, A. D., & Keltner, D. (2012). The Local-Ladder Effect: Social Status and Subjective Well-Being. Psychological Science, 23(7), 764–771. http://www.jstor.org/stable/23262493
Archer, J. (2006). Testosterone and human aggression: An evaluation of the challenge hypothesis. Neuroscience & Biobehavioral Reviews, 30(3), 319–345. https://doi.org/10.1016/j.neubiorev.2004.12.007
Arslan, R. C., Schilling, K. M., Gerlach, T. M., & Penke, L. (2018). “Using 26,000 diary entries to show ovulatory changes in sexual desire and behavior”: Correction to Arslan et al. (2018). Journal of Personality and Social Psychology. Advance online publication. https://doi.org/10.1037/pspp0000251
Arslan, R. C., Walther, M. P., & Tata, C. S. (2020). formr: A study framework allowing for automated feedback generation and complex longitudinal experience-sampling studies using R. Behavior Research Methods, 52, 376–387. https://doi.org/10.3758/s13428-019-01236-y
Arslan, R. C., Blake, K., Botzet, L., Bürkner, P., DeBruine, L. M., Fiers, T., … Stern, J. (2022). Not within spitting distance: salivary immunoassays of estradiol have subpar validity for cycle phase. https://doi.org/10.31234/osf.io/5r8mg
Arthur, L. C., Casto, K. V., & Blake, K. R. (2022). Hormonal contraceptives as disruptors of competitive behavior: Theoretical framing and review. Frontiers in Neuroendocrinology, 66, 101015. https://doi.org/10.1016/j.yfrne.2022.101015
Azari, Z., & Kaminski, V. (2019). Textbook of Female Reproductive Endocrinology. Scholars’ Press.
Baird, D. T., & Fraser, I. S. (1974). Blood production and ovarian secretion rates of estradiol-17 beta and estrone in women throughout the menstrual cycle. Journal of Clinical Endocrinology and Metabolism, 38(6). https://doi.org/10.1210/jcem-38-6-1009
Baird, D. T., & Glasier, A. F. (1993). Hormonal contraception. The New England Journal of Medicine, 328(21), 1543–1549. https://doi.org/10.1056/NEJM199305273282108
Baker, N. E. (2011). Cell competition. Current Biology, 21(1), R11–R15. https://doi.org/10.1016/j.cub.2010.11.030
Bates, D., Mächler, M., Bolker, B., & Walker, S. (2014). Fitting linear mixed-effects models using lme4. https://doi.org/10.48550/arXiv.1406.5823
Batres, C., Porcheron, A., Kaminski, G., Courrèges, S., Morizot, F., & Russell, R. (2018). Evidence that the hormonal contraceptive pill is associated with cosmetic habits. Frontiers in Psychology, 9, 1459. https://doi.org/10.3389/fpsyg.2018.01459
Benenson, J. F., & Benarroch, D. (1998). Gender differences in responses to friends’ hypothetical greater success. The Journal of Early Adolescence, 18(2), 192–208. https://doi.org/10.1177/0272431698018002004
Benenson, J. F., (2013). The development of human female competition: Allies and adversaries. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 368, 20130079. https://doi.org/10.1098/rstb.2013.0079
Bereshchenko, O., Bruscoli, S., & Riccardi, C. (2018). Glucocorticoids, sex hormones, and immunity. Frontiers in Immunology, 9, 1332. https://doi.org/10.3389/fimmu.2018.01332
Blake, K. R. (2022). Attractiveness helps women secure mates, but also status and reproductively relevant resources. Archives of Sexual Behavior, 51, 39–41. https://doi.org/10.1007/s10508-021-01949-2
Blake, K. R., Bastian, B., O’Dean, S. M., & Denson, T. F. (2017). High estradiol and low progesterone are associated with high assertiveness in women. Psychoneuroendocrinology, 75, 91–99. https://doi.org/10.1016/j.psyneuen.2016.10.008
Botzet, L. J., Gerlach, T. M., Driebe, J. C., Penke, L., & Arslan, R. C. (2021). Hormonal contraception and sexuality: Causal effects, unobserved selection, or reverse causality?. Collabra: Psychology, 7(1), 29039. https://doi.org/10.1525/collabra.29039
Bouman, A., Heineman, M. J., & Faas, M. M. (2005). Sex hormones and the immune response in humans. Human Reproduction Update, 11(4), 411–423. https://doi.org/10.1093/humupd/dmi008
Bradshaw, H. K., & DelPriore, D. J. (2022). Beautification is more than mere mate attraction: Extending evolutionary perspectives on female appearance enhancement. Archives of Sexual Behavior, 51, 43–47. https://doi.org/10.1007/s10508-021-01952-7
Bradshaw, H. K., Mengelkoch, S., & Hill, S. E. (2020). Hormonal contraceptive use predicts decreased perseverance and therefore performance on some simple and challenging cognitive tasks. Hormones and Behavior, 119, 104652. https://doi.org/10.1016/j.yhbeh.2019.104652
Bressan, P., & Kramer, P. (2022). Progesterone does raise disgust. Hormones and Behavior, 137, 104937. https://doi.org/10.1016/j.yhbeh.2021.104937
Bui, H. N., Sluss, P. M., Blincko, S., Knol, D. L., Blankenstein, M. A., & Heijboer, A. C. (2013). Dynamics of serum testosterone during the menstrual cycle evaluated by daily measurements with an ID-LC-MS/MS method and a 2nd generation automated immunoassay. Steroids, 78(1), 96–101. https://doi.org/10.1016/j.steroids.2012.10.010
Burrows, H. (2013). Biological actions of sex hormones. Cambridge University Press. https://doi.org/10.1017/CBO9781316530146
Buser, T. (2012). The impact of the menstrual cycle and hormonal contraceptives on competitiveness. Journal of Economic Behavior & Organization, 1–10. https://doi.org/10.1016/j.jebo.2011.06.006
Buss, D. M., & Schmitt, D. P. (2019). Mate preferences and their behavioral manifestations. Annual Review of Psychology, 70, 77–110. https://doi.org/10.1146/annurev-psych-010418-103408
Campbell, A. (2013). A mind of her own: The evolutionary psychology of women. OUP Oxford.
Casto, K. V., & Prasad, S. (2017). Recommendations for the study of women in hormones and competition research. Hormones and Behavior, 92, 190–194. https://doi.org/10.1016/j.yhbeh.2017.05.009
Casto, K. V., Hamilton, D. K., & Edwards, D. A. (2019). Testosterone and cortisol interact to predict within-team social status hierarchy among Olympic-level women athletes. Adaptive Human Behavior and Physiology, 5(3), 237–250. https://doi.org/10.1007/s40750-019-00115-2
Casto, K. V., Edwards, D. A., Akinola, M., Davis, C., & Mehta, P. H. (2020). Testosterone reactivity to competition and competitive endurance in men and women. Hormones and Behavior, 123, 104665. https://doi.org/10.1016/j.yhbeh.2019.104665
Casto, K. V., Arthur, L. C., Hamilton, D. K., & Edwards, D. A. (2021a). Testosterone, athletic context, oral contraceptive use, and competitive persistence in women. Adaptive Human Behavior and Physiology, 1–27. https://doi.org/10.1007/s40750-021-00180-6
Casto, K. V., Blake, K., & Arthur, L. C. (2021b). Hormonal contraceptive use and cycle phase effects on competitive persistence: Progesterone as a mediating mechanism (Stage 1 Registered Report). https://doi.org/10.31234/osf.io/vn8zu
Chen, Y., Katuscak, P., Ozdenoren, E., (2013). Why can’t a woman bid more like a man? Games and Economic Behavior, 77(1), 181–213. https://doi.org/10.1016/j.geb.2012.10.002
Cheng, J. T., Tracy, J. L., & Anderson, C. (2014). The psychology of social status. Springer.
Cheng, J. T., Tracy, J. L., Foulsham, T., Kingstone, A., & Henrich, J. (2013). Two ways to the top: Evidence that dominance and prestige are distinct yet viable avenues to social rank and influence, 104(1), 103–125https://doi.org/10.1037/a0030398
Clutton-Brock, T. H., & Huchard, E. (2013). Social competition and selection in males and females. Philosophical Transactions of the Royal Society b: Biological Sciences, 368(1631), 20130074. https://doi.org/10.1098/rstb.2013.0074
Cobey, K. D., Klipping, C., & Buunk, A. P. (2013). Hormonal contraceptive use lowers female intrasexual competition in pair-bonded women. Evolution and Human Behavior, 294–298.
Diamantopoulos, A., Sarstedt, M., Fuchs, C., Wilczynski, P., & Kaiser, S. (2012). Guidelines for choosing between multi-item and single-item scales for construct measurement: A predictive validity perspective. Journal of the Academy of Marketing Science, 40(3), 434–449. https://doi.org/10.1007/s11747-011-0300-3
Durante, K. M., Li, N. P., & Haselton, M. G. (2008). Changes in women’s choice of dress across the ovulatory cycle: Naturalistic and laboratory task-based evidence. Personality & Social Psychology Bulletin, 34(11), 1451–1460. https://doi.org/10.1177/0146167208323103
Durante, K. M., Griskevicius, V., Cantú, S. M., & Simpson, J. A. (2014). Money, status, and the ovulatory cycle. Journal of Marketing Research, 51(1), 27–39. https://doi.org/10.1509/jmr.11.0327
Edlund, J. E., & Sagarin, B. J. (2014). The mate value scale. Personality and Individual Differences, 64, 72–77. https://doi.org/10.1016/j.paid.2014.02.005
Edwards, D. A., & Casto, K. V. (2013). Women’s intercollegiate athletic competition: Cortisol, testosterone, and the dual-hormone hypothesis as it relates to status among teammates. Hormones and Behavior, 64(1), 153–160. https://doi.org/10.1016/j.yhbeh.2013.03.003
Eisenbruch, A. B., & Roney, J. R. (2016). Conception risk and the ultimatum game: When fertility is high, women demand more. Personality and Individual Differences, 98, 272–274. https://doi.org/10.1016/j.paid.2016.04.047
Eriksson, C. J. P., von der Pahlen, B., Sarkola, T., & Seppä, K. (2003). Oestradiol and human male alcohol-related aggression. Alcohol and Alcoholism, 38(6), 589–596. https://doi.org/10.1093/alcalc/agg117
Feinberg, D. R., DeBruine, L. M., Jones, B. C., & Little, A. C. (2008). Correlated preferences for men’s facial and vocal masculinity. Evolution and Human Behavior, 29(4), 233–241. https://doi.org/10.1016/j.evolhumbehav.2007.12.008
Fisher, M. L. (2004). Female intrasexual competition decreases female facial attractiveness. Proceedings of the Royal Society B, 271, S283–S285. https://doi.org/10.1098/rsbl.2004.0160
Gangestad, S. W., & Thornhill, R. (1998). Menstrual cycle variation in women’s preferences for the scent of symmetrical men. Proceedings of the Royal Society b: Biological Sciences, 265(1399), 927–933. https://doi.org/10.1098/rspb.1998.0380
Gangestad, S. W., & Thornhill, R. (2008). Human oestrus. Proceedings of the Royal Society B: Biological Sciences, 275(1638), 991–1000. https://doi.org/10.1098/rspb.2007.1425
Gangestad, S. W., Haselton, M. G., Welling, L. L., Gildersleeve, K., Pillsworth, E. G., Burriss, R. P., Larson, C. M., & Puts, D. A. (2016). How valid are assessments of conception probability in ovulatory cycle research? Evaluations, recommendations, and theoretical implications. Evolution and Human Behavior, 37(2), 85–96. https://doi.org/10.1016/j.evolhumbehav.2015.09.001
Gangestad, S. W., Dinh, T., Grebe, N. M., Del Giudice, M., & Thompson, M. E. (2019a). Psychological cycle shifts redux: Revisiting a preregistered study examining preferences for muscularity. Evolution and Human Behavior, 40(6), 501–516.
Gangestad, S. W., Dinh, T., Grebe, N. M., Del Giudice, M., & Emery Thompson, M. (2019b). Psychological cycle shifts redux, once again: Response to Stern et al., Roney, Jones et al., and Higham. Evolution and Human Behavior, 40(6), 537–542.
Gelman, A., Hill, J., & Yajima, M. (2012). Why we (usually) don’t have to worry about multiple comparisons. Journal of Research on Educational Effectiveness, 5(2), 189–211. https://doi.org/10.1080/19345747.2011.618213
Gildersleeve, K., Haselton, M. G., & Fales, M. R. (2014a). Do women’s mate preferences change across the ovulatory cycle? A Meta-Analytic Review. Psychological Bulletin, 140(5), 1205–1259. https://doi.org/10.1037/a0035438
Gildersleeve, K., Haselton, M. G., & Fales, M. R. (2014b). Meta-analyses and p-curves support robust cycle shifts in women’s mate preferences: Reply to Wood and Carden (2014b) and Harris, Pashler, and Mickes (2014b). Psychological Bulletin, 140(5), 1272–1280. https://psycnet.apa.org/doi/https://doi.org/10.1037/a0037714
Gori, A., Giannini, M., Craparo, G., Caretti, V., Nannini, I., Madathil, R., & Schuldberg, D. (2014). Assessment of the relationship between the use of birth control pill and the characteristics of mate selection. The Journal of Sexual Medicine, 11(9), 2181–2187. https://doi.org/10.1111/jsm.12566
Hahn, A. C., Fisher, C. I., Cobey, K. D., DeBruine, L. M., & Jones, B. C. (2016). A longitudinal analysis of women’s salivary testosterone and intrasexual competitiveness. Psychoneuroendocrinology, 64, 117–122. https://doi.org/10.1016/j.psyneuen.2015.11.014
Hahn, A. C., DeBruine, L. M., Pesce, L. A., Diaz, A., Aberson, C. L., & Jones, B. C. (2020). Does women’s anxious jealousy track changes in steroid hormone levels? Psychoneuroendocrinology, 113, 104553. https://doi.org/10.1016/j.psyneuen.2019.104553
Harris, C. R., Pashler, H., & Mickes, L. (2014). Elastic analysis procedures: an incurable (but preventable) problem in the fertility effect literature. Comment on Gildersleeve, Haselton, and Fales (2014). Psychological Bulletin, 140(5), 1260–1264. https://doi.org/10.1037/a0036478
Hays, N. A. (2013). Fear and loving in social hierarchy: Sex differences in preferences for power versus status. Journal of Experimental Social Psychology, 49(6), 1130–1136. https://doi.org/10.1016/j.jesp.2013.08.007
Henrich, J., & Gil-White, F. J. (2001). The evolution of prestige: Freely conferred deference as a mechanism for enhancing the benefits of cultural transmission. Evolution and Human Behavior, 22(3), 165–196. https://doi.org/10.1016/S1090-5138(00)00071-4
Johnson, R. T., Burk, J. A., & Kirkpatrick, L. A. (2007). Dominance and prestige as differential predictors of aggression and testosterone levels in men. Evolution and Human Behavior, 28(5), 345–351. https://doi.org/10.1016/j.evolhumbehav.2007.04.003
Jones, B., Hahn, A., Fisher, C., Wang, H., Kandrik, M., Han, C., Fasolt, V., Morrison, D., Lee, A., Holzleitner, I., O’Shea, K., Roberts, S., Little, A., & DeBruine, L. (2018a). No compelling evidence that preferences for facial masculinity track changes in women’s hormonal status. Psychological Science, 29(6), 996–1005. https://doi.org/10.1177/0956797618760197
Jones, B. C., Hahn, A. C., Fisher, C. I., Wang, H., Kandrik, M., Lee, A. J., Tybur, J. M., & DeBruine, L. M. (2018b). Hormonal correlates of pathogen disgust: Testing the Compensatory Prophylaxis Hypothesis. Evolution and Human Behavior, 39, 166–169. https://doi.org/10.1016/j.evolhumbehav.2017.12.004
Jones, B. C., Hahn, A. C., & DeBruine, L. M. (2019). Ovulation, sex hormones and women’s mating psychology. Trends in Cognitive Sciences, 23, 51–62. https://doi.org/10.1016/j.tics.2018.10.008
Jünger, J., Kordsmeyer, T. L., Gerlach, T. M., & Penke, L. (2018a). Fertile women evaluate male bodies as more attractive, regardless of masculinity. Evolution and Human Behavior, 39(4), 412–423. https://doi.org/10.1016/j.evolhumbehav.2018.03.007
Jünger, J., Motta-Mena, N. V., Cardenas, R., Bailey, D., Rosenfield, K. A., Schild, C., Penke, L., & Puts, D. A. (2018b). Do women’s preferences for masculine voices shift across the ovulatory cycle? Hormones and Behavior, 106, 122–134. https://doi.org/10.1016/j.yhbeh.2018.10.008
Lovibond, S.H. & Lovibond, P.F. (1995). Manual for the Depression Anxiety Stress Scales (2nd ed.) Sydney: Psychology Foundation
Lucas, M., & Koff, E. (2013). How conception risk affects competition and cooperation with attractive women and men. Evolution and Human Behavior, 34(1), 16–22. https://doi.org/10.1016/j.evolhumbehav.2012.08.001
Lüdecke, D., Ben-Shacker, M.S., Patil, I., Waggoner, P. & Makowski, D. (2021). performance: An R package for assessment, comparison and testing of statistical models. Journal of Open-Source Software, 6(60), 3139. https://doi.org/10.21105/joss.03139
Marcinkowska, U. M., Hahn, A. C., Little, A. C., DeBruine, L. M., & Jones, B. C. (2019). No evidence that women using oral contraceptives have weaker preferences for masculine characteristics in men’s faces. PLoS ONE, 14(1), e0210162.
Mayr, U., Wozniak, D., Davidson, C., Kuhns, D., & Harbaugh, W. T. (2012). Competitiveness across the life span: The feisty fifties. Psychology and Aging, 27(2), 278–285. https://doi.org/10.1037/a0025655
McClanahan, K. J., Maner, J. K., & Cheng, J. T. (2021). Two ways to stay at the top: Prestige and dominance are both viable strategies for gaining and maintaining social rank over time. Personality and Social Psychology Bulletin, 01461672211042319. 10.1177%2F01461672211042319
Melo, J., & Creinin, M. D. (2016). Combination oral contraceptive pills. In D. Shoupe & Mishell, Jr., Daniel R. (Eds.), The Handbook of Contraception: A Guide for Practical Management (pp. 61–77). Springer International Publishing. https://doi.org/10.1007/978-3-319-20185-6_4
Miłkowska, K., Galbarczyk, A., Klimek, M., Zabłocka-Słowińska, K., & Jasienska, G. (2021). Pathogen disgust, but not moral disgust, changes across the menstrual cycle. Evolution and Human Behavior, 42(5), 402–408. https://doi.org/10.1016/j.evolhumbehav.2021.03.002
Miller, G., Tybur, J. M., & Jordan, B. D. (2007). Ovulatory cycle effects on tip earnings by lap dancers: Economic evidence for human estrus? Evolution and Human Behavior, 28(6), 375–381. https://doi.org/10.1016/j.evolhumbehav.2007.06.002
Nakagawa, S., & Schielzeth, H. (2013). A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods in Ecology and Evolution, 4(2), 133–142. https://doi.org/10.1111/j.2041-210x.2012.00261.x
Nikiforidis, L., Arsena, A. R., & Durante, K. M. (2017). The effect of fertility on women’s intrasexual competition. The Oxford Handbook of Women and Competition.
Orosz, G., Tóth-Király, I., Büki, N., Ivaskevics, K., Bőthe, B., & Fülöp, M. (2018). The four faces of competition: The development of the Multidimensional Competitive Orientation Inventory. Frontiers in Psychology, 9, 779. https://doi.org/10.3389/fpsyg.2018.00779
Paulhus, D. L., & Vazire, S. (2007). The self-report method. Handbook of Research Methods in Personality Psychology, 1(2007), 224–239.
Pearson, M., & Schipper, B. C. (2013). Menstrual cycle and competitive bidding. Games and Economic Behavior, 78, 1–20. https://doi.org/10.1016/j.geb.2012.10.008
Penke, L., & Asendorpf, J. B. (2008). Beyond global sociosexual orientations: A more differentiated look at sociosexuality and its effects on courtship and romantic relationships. Journal of Personality and Social Psychology, 95, 1113–1135.
Piccoli, V., Foroni, F., & Carnaghi, A. (2013). Comparing group dehumanization and intrasexual competition among normally ovulating women and hormonal contraceptive users. Personality & Social Psychology Bulletin, 39(12), 1600–1609. https://doi.org/10.1177/0146167213499025
Ranehill, E., Zethraeus, N., Blomberg, L., von Schoultz, B., Hirschberg, A. L., Johannesson, M., & Dreber, A. (2018). Hormonal contraceptives do not impact economic preferences: Evidence from a randomized trial. Management Science, 64(10), 4515–4532. https://doi.org/10.1287/mnsc.2017.2844
Reynolds, T. A. (2021). Our grandmothers’ legacy: Challenges faced by female ancestors leave traces in modern women’s same-sex relationships. Archives of Sexual Behavior, 1–32. https://doi.org/10.1007/s10508-020-01768-x
Reynolds, T. A., Makhanova, A., Marcinkowska, U. M., Jasienska, G., McNulty, J. K., Eckel, L. A., … & Maner, J. K. (2018). Progesterone and women’s anxiety across the menstrual cycle. Hormones and Behavior, 102, 34–40. https://doi.org/10.1016/j.yhbeh.2018.04.008
Roney, J. R. (2016). Theoretical frameworks for human behavioral endocrinology. Hormones and Behavior, 84, 97–110. https://doi.org/10.1016/j.yhbeh.2016.06.004
Roney, J. R. (2018). Hormonal mechanisms and the optimal use of luteinizing hormone tests in human menstrual cycle research. Hormones and Behavior, 106, A7–A9. https://doi.org/10.1016/j.yhbeh.2018.05.021
Roney, J. R., & Simmons, Z. L. (2017). Ovarian hormone fluctuations predict within-cycle shifts in women’s food intake. Hormones and Behavior, 90, 8–14. https://doi.org/10.1016/j.yhbeh.2017.01.009
Rosvall, K. A. (2011). Intrasexual competition in females: Evidence for sexual selection? Behavioral Ecology, 22(6), 1131–1140. https://doi.org/10.1093/beheco/arr106
Rothman, M. S., Carlson, N. E., Xu, M., Wang, C., Swerdloff, R., Lee, P., Goh, V. H. H., Ridgway, E. C., & Wierman, M. E. (2011). Reexamination of testosterone, dihydrotestosterone, estradiol and estrone levels across the menstrual cycle and in postmenopausal women measured by liquid chromatography-tandem mass spectrometry. Steroids, 76(1–2), 177–182. https://doi.org/10.1016/j.steroids.2010.10.010
Ryckman, R. M., Hammer, M., Kaczor, L. M., & Gold, J. A. (1996). Construction of a personal development competitive attitude scale. Journal of Personality Assessment, 66(2), 374–385. https://doi.org/10.1207/s15327752jpa6602_15
Saad, G., & Stenstrom, E. (2012). Calories, beauty, and ovulation: The effects of the menstrual cycle on food and appearance-related consumption. Journal of Consumer Psychology, 22(1), 102–113. https://doi.org/10.1016/j.jcps.2011.10.001
Schipper, B. C. (2015). Sex hormones and competitive bidding. Management Science, 61(2), 249–266. https://doi.org/10.1287/mnsc.2014.1959
Schleifenbaum, L., Driebe, J. C., Gerlach, T. M., Penke, L., & Arslan, R. C. (2021). Women feel more attractive before ovulation: Evidence from a large-scale online diary study. Evolutionary Human Sciences, 3. https://doi.org/10.1017/ehs.2021.44
Schwarz, S., & Hassebrauck, M. (2008). Self-perceived and observed variations in women’s attractiveness throughout the menstrual cycle—a diary study. Evolution and Human Behavior, 29(4), 282–288. https://doi.org/10.1016/j.evolhumbehav.2008.02.003
Scott, K. M., Al-Hamzawi, A. O., Andrade, L. H., Borges, G., Caldas-de-Almeida, J. M., Fiestas, F., Gureje, O., Hu, C., Karam, E. G., Kawakami, N., Lee, S., Levinson, D., Lim, C. C. W., Navarro-Mateu, F., Okoliyski, M., Posada-Villa, J., Torres, Y., Williams, D. R., Zakhozha, V., & Kessler, R. C. (2014). Associations between subjective social status and DSM-IV mental disorders: Results from the World Mental Health surveys. JAMA Psychiatry, 71(12), 1400–1408. https://doi.org/10.1001/jamapsychiatry.2014.1337
Sherwin, B. B. (2003). Estrogen and cognitive functioning in women. Endocrine Reviews, 24(2), 133–151. https://doi.org/10.1210/er.2001-0016
Sherwin, B. B. (2012). Estrogen and cognitive functioning in women: Lessons we have learned. Behavioral Neuroscience, 126(1), 123. https://psycnet.apa.org/doi/https://doi.org/10.1037/a0025539
Simmons, R. (2002). Odd girl out: The hidden culture of aggression in girls. Houghton Mifflin Harcourt.
Smolla, M., Gilman, R. T., Galla, T., & Shultz, S. (2015). Competition for resources can explain patterns of social and individual learning in nature. Proceedings of the Royal Society b: Biological Sciences, 282(1815), 20151405. https://doi.org/10.1098/rspb.2015.1405
Stanton, S.J., & Edelstein, R.S., (2009). The physiology of women’s power motive: Implicit power motivation is positively associated with estradiol levels in women. Journal of Research in Personality, 43(6), 1109–1113. https://psycnet.apa.org/doi/https://doi.org/10.1016/j.jrp.2009.08.002
Stanton, S. J., & Schultheiss, O. C. (2007). Basal and dynamic relationships between implicit power motivation and estradiol in women. Hormones & Behavior, 52(5), 571–580. https://doi.org/10.1016/j.jrp.2009.08.002
Stern, J., & Shiramizu, V. (2022). Hormones, ovulatory cycle phase and pathogen disgust: A longitudinal investigation of the Compensatory Prophylaxis Hypothesis. Hormones and Behavior, 138, 105103. https://doi.org/10.1016/j.yhbeh.2021.105103
Stern, J., Gerlach, T. M., & Penke, L. (2020). Probing ovulatory cycle shifts in women’s mate preferences for men’s behaviors. Psychological Science, 31, 424–436. https://doi.org/10.1177/0956797619882022
Stern, J., Kordsmeyer, T. L., & Penke, L. (2021). A longitudinal evaluation of ovulatory cycle shifts in women’s mate attraction and preferences. Hormones and Behavior, 128, 104916. https://doi.org/10.1016/j.yhbeh.2020.104916
Stern, J., Arslan, R.C., Gerlach, T.M., Penke, L., (2019). No robust evidence for cycle shifts in preferences for men's bodies in a multiverse analysis: A response to Gangestad et al. (2019). https://doi.org/10.1016/j.evolhumbehav.2019.08.005
Stirnemann, J. J., Samson, A., & Bernard, J.- P. & Thalabard, J.- C. (2013). Day-specific probabilities of conception in fertile cycles resulting in spontaneous pregnancies. Human Reproduction, 28(4), 1110–1116. https://doi.org/10.1093/humrep/des449
Toffoletto, S., Lanzenberger, R., Gingnell, M., Sundström-Poromaa, I., & Comasco, E. (2014). Emotional and cognitive functional imaging of estrogen and progesterone effects in the female human brain: A systematic review. Psychoneuroendocrinology, 50, 28–52. https://doi.org/10.1016/j.psyneuen.2014.07.025
van Stein, K. R., Strauß, B., & Brenk-Franz, K. (2019). Ovulatory shifts in sexual desire but not mate preferences: An LH-test-confirmed, longitudinal study. Evolutionary Psychology, 17, 1–10. https://doi.org/10.1177/1474704919848116
Vermeersch, H., T'Sjoen, G., Kaufman, J. M., & Vincke, J. (2008). The role of testosterone in aggressive and non-aggressive risk-taking in adolescent boys. Hormones and Behavior, 53(3), 463–471. https://doi.org/10.1016/j.yhbeh.2007.11.021
Vigil, J. M. (2007). Asymmetries in the friendship preferences and social styles of men and women. Human Nature, 18, 143–161. https://doi.org/10.1007/s12110-007-9003-3
Welling, L. L., & Burriss, R. P. (2019). Investigating the ovulatory cycle: An overview of research and methods. The Oxford handbook of evolutionary psychology and behavioral endocrinology. Oxford Publishing.
West-Eberhard, M. J. (1979). Sexual selection, social competition, and evolution. Proceedings of the American Philosophical Society, 123(4), 222–234. https://www.jstor.org/stable/986582
Wilkinson, G. N., & Rogers, C. E. (1973). Symbolic description of factorial models for analysis of variance. Journal of the Royal Statistical Society: Series C (applied Statistics), 22(3), 392–399.
Wood, W., & Carden, L. (2014). Elusiveness of menstrual cycle effects on mate preferences: Comment on Gildersleeve, Haselton, and Fales (2014). Psychological Bulletin, 140(5), 1265–1271. https://doi.org/10.1037/a0036722
Wood, W., Kressel, L., Joshi, P. D., & Louie, B. (2014). Meta-analysis of menstrual cycle effects on women’s mate preferences. Emotion Review, 6(3), 229–249. https://doi.org/10.1177/1754073914523073
Wozniak, D., Harbaugh, W. T., & Mayr, U. (2014). The menstrual cycle and performance feedback alter gender differences in competitive choices. Journal of Labor Economics, 32(1), 161–198. https://doi.org/10.1086/673324
Zimmerman, Y., Eijkemans, M. J. C., Coelingh Bennink, H. J. T., Blankenstein, M. A., & Fauser, B. C. J. M. (2014). The effect of combined oral contraception on testosterone levels in healthy women: A systematic review and meta-analysis. Human Reproduction Update, 20(1), 76–105. https://doi.org/10.1093/humupd/dmt038
Acknowledgements
We thank and acknowledge Meg McCartney for support with data collection and Ruben Arslan for support with formr, data visualization and statistical advice.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. No funding was received for conducting this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Competing Interests
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Open Science Framework—Data Repository Link (https://osf.io/rs6bm).
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Arthur, L.C., Blake, K.R. Fertility predicts self-development-oriented competitiveness in naturally cycling women but not hormonal contraceptive users. Adaptive Human Behavior and Physiology 8, 489–519 (2022). https://doi.org/10.1007/s40750-022-00198-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40750-022-00198-4