Abstract
Introduction
BI 695501 has shown similar efficacy, safety, and immunogenicity to the adalimumab reference product, Humira®. We present two phase 1 studies comparing the pharmacokinetics, safety, and immunogenicity of BI 695501 delivered via autoinjector (AI) vs. prefilled syringe (PFS).
Methods
Both trials were randomized, open-label, parallel-group studies undertaken in subjects aged ≥ 18–65 years. VOLTAIRE®-AI (NCT02606903) recruited healthy, Caucasian, male, non-athletic volunteers with BMI ≥ 18 to ≤ 30 kg/m2. VOLTAIRE®-TAI (NCT02899338) recruited healthy men and women with BMI > 17.5 to < 35 kg/m2. In both studies, a single dose of BI 695501 40 mg was administered via AI or PFS to the abdomen (VOLTAIRE®-AI) or thigh (VOLTAIRE®-TAI). The observation period was 43/57 days and the safety follow-up was 70 days. Co-primary endpoints were AUC0–1032 or AUC0–1368, Cmax, and AUC0–∞. Safety and immunogenicity were assessed.
Results
Subjects (VOLTAIRE®-AI: N = 71; VOLTAIRE®-TAI: N = 162) were randomized to AI (n = 35; n = 81) or PFS (n = 36; n = 81). Baseline characteristics were balanced between treatment groups in each study. Total exposure of BI 695501 was similar for both groups; adjusted geometric mean ratios for AUC0–∞, AUC0–1032, and Cmax were 106.17, 104.09, and 114.83%, respectively, for VOLTAIRE®-AI; 103.19, 101.71 (AUC0–1368), and 100.11% for VOLTAIRE®-TAI. In both studies, similar immunogenicity was observed between groups in terms of frequency of binding and neutralizing anti-drug antibody-positive subjects. Incidence of adverse events was similar for both groups.
Conclusions
Pharmacokinetics and immunogenicity of BI 695501 delivered via AI were similar to administration using a PFS, independent of injection site. No differences are expected between AI and PFS use in clinical practice.
Funding
Boehringer Ingelheim.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Adalimumab is a recombinant human monoclonal antibody that binds specifically to tumor necrosis factor (TNF)α and neutralizes its biological function by blocking its interaction with TNFα receptors 1 and 2 [1]. It is an efficacious treatment for several autoimmune and inflammatory disease conditions, including rheumatoid arthritis (RA), psoriasis, Crohn’s disease, ulcerative colitis, hidradenitis suppurativa, and non-infectious uveitis [1].
BI 695501 is a biosimilar to Humira® [2, 3]. BI 695501 was demonstrated to be structurally and functionally highly similar to Humira® (data on file). In a three-way comparative pharmacokinetic (PK) trial (VOLTAIRE®-PK) conducted in healthy subjects, bioequivalence (BE) was established between BI 695501, US-, and EU-approved Humira® [4]. Similarity of efficacy, safety, and immunogenicity between BI 695501 and Humira® was demonstrated in the VOLTAIRE®-RA study [5].
Adalimumab is administered via subcutaneous (SC) injection to either the abdomen or the thigh [1]. Drugs that require regular SC administration can be administered via prefilled syringe (PFS) or, via autoinjector (AI). Selection of the appropriate presentation should be based on individual patient (or carer) characteristics and preferences [6]. AIs have demonstrated improved comparative usability over PFSs in patients with severe RA, in terms of pain experienced, ease of use, and convenience, with similar tolerability [6]. These factors are of considerable importance, especially when manual dexterity is compromised, for example, when RA causes pain in the fingers or damages finger joints [7].
US FDA guidance [8] states that approval of an AI presentation requires a PK bridging study to demonstrate similar PK profiles across a range of body weights, alongside human factor studies and real-life patient handling experience. The aim of the VOLTAIRE®-AI and VOLTAIRE®-TAI studies was to assess the relative bioavailability of BI 695501 administered via AI vs. PFS across a broad range of body mass index (BMI) and body weights, with two different administration locations. Safety, tolerability, and immunogenicity were also assessed.
Methods
Study Designs
VOLTAIRE®-AI (NCT02606903)
This randomized, single-dose, parallel-arm, open-label, phase 1 trial of BI 695501, delivered via AI or PFS in the abdomen, was conducted at a single site in Belgium between October 29, 2015 and October 4, 2016. An initial screening period of up to 28 days was followed by a single-dose administration of BI 695501 via AI or PFS. After drug administration, there was a 43-day observation period and a safety follow-up period of up to 70 days.
VOLTAIRE®-TAI (NCT02899338)
This randomized, single-dose, parallel-arm, open-label, phase 1 trial of BI 695501, delivered via AI or PFS in the thigh, was conducted at two sites: one in Belgium and one in the Netherlands. The date of first enrollment was September 22, 2016; the last subject completed on February 23, 2017. An initial screening period of up to 28 days was followed by a single-dose administration of BI 695501 via AI or PFS. After drug administration, there was a 57-day observation period and a safety follow-up period of up to 70 days.
Both studies were approved by independent ethics committees and the competent authorities, and conducted in accordance with the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use Good Clinical Practice (GCP) guidelines and the Declaration of Helsinki [9]. All subjects provided written, informed consent prior to participation.
Study Populations
VOLTAIRE®-AI
The study enrolled healthy, non-athletic, adult, Caucasian males aged 18–65 years with a BMI of 18–30 kg/m2. Exclusion criteria included prior exposure to a biologic, or exposure to drugs with a half-life (t1/2) > 24 h, within 30 days or < 5 half-lives prior to administration of trial medication.
VOLTAIRE®-TAI
The study enrolled healthy male and female volunteers aged 18–65 years with a BMI of 17.5–35 kg/m2. Subjects were excluded if they had any prior exposure to adalimumab, or drug use within 10 days prior to administration that might affect the results of the study.
The tissue into which a drug is injected can affect PK, and this in turn can be influenced by the amount of SC fat. As the quantity of SC fat tissue correlates with body weight and BMI [10], randomization in VOLTAIRE®-AI was stratified by BMI category (18.0–< 20.0; 20.0–< 25.0, 25.0–≤ 30.0 kg/m2) and in VOLTAIRE®-TAI by body weight categories (low: ≤ 60.0 kg, medium: > 60.0–< 90.0 kg, and high: ≥ 90.0 kg). The trial populations therefore covered a broad range of subjects with respect to BMI and body weight.
Detailed inclusion and exclusion criteria for both trials are provided in the supplemental digital content.
Interventions
VOLTAIRE®-AI
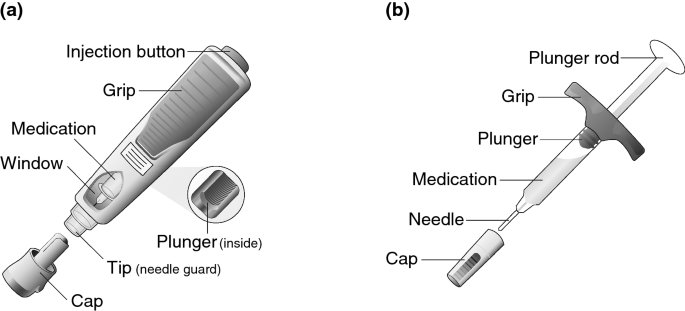
Subjects were randomized 1:1 to receive a single SC administration of BI 695501 40 mg/0.8 ml, via either AI or PFS (Fig. 1). Injections were to the lower abdomen with the subjects in a supine position. Subjects were placed under close, residential supervision for the first 24 h following drug administration.
VOLTAIRE®-TAI
Subjects were randomized 1:1 to receive a single SC administration of BI 695501 40 mg/0.8 ml, via either AI or PFS. Injections were to the front of the thigh.
For both studies, the spring-powered AI (Fig. 1a) and the standard PFS (Fig. 1b) included a 1-ml syringe. Details of the injection processes are presented in the supplemental digital content.
The BI 695501 40-mg/0.8-ml dose was deemed to have an acceptable risk/benefit ratio in healthy subjects and reflected the standard clinical dose.
Study Endpoints
VOLTAIRE®-AI
Three co-primary endpoints were investigated for BI 695501: area under the plasma concentration–time curve (AUC) from 0 to 1032 h post-dose (AUC0–1032); maximum plasma concentration (Cmax); and AUC from 0 extrapolated to infinity (AUC0–∞), based on observed concentrations at the last observation.
VOLTAIRE®-TAI
Three co-primary endpoints were investigated for BI 695501: AUC from 0 to 1368 h post-dose (AUC0–1368); Cmax; and AUC0–∞, based on observed concentrations at the last observation.
In both studies, the secondary endpoint was the number of subjects with drug-related treatment-emergent adverse events (AEs) occurring from day 1 to day 70. Additional PK, safety, and immunogenicity parameters were also assessed.
PK Methodology and Immunogenicity Assays
Blood samples for PK analyses were drawn daily on days 0–8, and then on days 10, 15, 22, 29, 36, and 43 (and at day 57 in VOLTAIRE®-TAI) after an overnight (at least 10-h) fast. BI 695501 plasma concentration was determined via a validated enzyme-linked immunosorbent assay, as described by Wynne et al. 2016. Blood samples for immunogenicity analyses were drawn at baseline (pre-dose) and throughout the studies [days 22 and 43 (VOLTAIRE®-AI); days 22 and 57 (VOLTAIRE®-TAI)]. Anti-drug antibodies (ADA) and neutralizing antibodies (nAb) measurements were performed with validated assays [a single bridging electrochemiluminescence assay on the MSD platform (Meso Scale Diagnostics LLC, Rockville, MA, USA) for ADA measurements, and a cell-based, antibody-dependent cell-mediated cytotoxicity method for nAb measurements], as described by Wynne et al. 2016 [4].
Statistical Analyses
In agreement with regulatory advice at the time of study design, the sample size for VOLTAIRE®-AI was set at 66 subjects to compare the bioavailability of BI 695501 administered by AI and PFS with sufficient precision. For VOLTAIRE®-TAI, it was planned to enter a total of 160 subjects for the primary bioavailability comparison. Updated regulatory requirements in comparison to VOLTAIRE®-AI required higher precision for estimation of effects, thus, the higher sample size was selected for VOLTAIRE®-TAI to assess the primary objective. The sample size calculations were performed with nQuery Version 2.0.1.0 (Statistical Solutions Ltd, Cork, Ireland).
All PK analyses were conducted using the PK analysis set, which included all subjects who received the single dose of study medication, had ≥ 1 evaluable primary PK endpoint, and had no protocol deviations considered relevant to affect PK assessments.
Safety and immunogenicity evaluations were conducted on the safety analysis set, consisting of all subjects who received the single dose of study medication.
Exploratory analyses were performed to estimate the relative bioavailability of BI 695501 administered via AI compared with PFS, using an analysis of variance (ANOVA) model of the logarithmically transformed primary PK parameters (Cmax, AUC0–∞, and AUC0–1032/AUC0–1368), with fixed effects for treatment and BMI group (VOLTAIRE®-AI) or body weight (VOLTAIRE®-TAI). Point estimates of relative bioavailability for PK parameters and their two-sided 90% confidence intervals (CIs) were calculated [ratio of geometric means (gMean); AI vs. PFS]. In VOLTAIRE®-AI, an additional sensitivity analysis was conducted to estimate the primary PK endpoints for BI 695501 using an analysis of covariance (ANCOVA) model on a logarithmic scale, with a fixed effect for treatment and baseline BMI as a continuous covariate.
CIs were compared using the standard BE acceptance range, even though no formal BE testing was required.
Scatterplots and boxplots were used to graphically evaluate Cmax, AUC0–∞, and AUC0–1032 (AUC0–1368 for VOLTAIRE®-TAI) for AI vs. PFS administration.
Descriptive statistics were provided for additional PK parameters, safety, tolerability, and immunogenicity.
Results
Subject Demographics and Baseline Characteristics
VOLTAIRE®-AI
A total of 122 subjects were screened and initially 66 were randomized (original population data set; Fig. 2a). Due to lower-than-required recruitment in the lowest BMI group (< 20 kg/m2), an additional five subjects were enrolled (full population data set). All data presented here are from this full population data set (N = 71; 35 to AI and 36 to PFS), other than for the primary endpoint analysis, for which the original analysis plan (N = 66) was retained per protocol. All subjects completed the trial (Fig. 2a).
VOLTAIRE®-TAI
A total of 362 subjects were screened, and 162 were randomized to AI (n = 81) or PFS (n = 81) (Fig. 2b). One hundred and fifty-seven subjects completed the trial per protocol; two out of five who were prematurely discontinued were replaced.
In both studies, demographics and baseline characteristics were balanced between treatment groups (Table 1).
Pharmacokinetics—Co-primary Endpoints
VOLTAIRE®-AI
Assessment of relative bioavailability of BI 695501 administered via either AI or PFS showed that the total exposure of BI 695501 for the two treatment groups was similar (Table 2a). Adjusted gMean ratios for AUC0–∞ and AUC0–1032 fell within the BE acceptance range 80–125%. The upper 90% CI limit for AUC0–∞ was 130.56%, slightly above the upper BE acceptance limit of 125%, while the 90% CI for AUC0–1032 (123.39%) was contained within the standard acceptance range of BE. The adjusted gMean ratio point estimate for Cmax of BI 695501 administered via AI compared with PFS (114.83%) was within the standard BE acceptance range, while the upper 90% CI limit (130.75%) was again slightly above the upper BE acceptance limit. The primary analysis for the full population data set (N = 71) is shown in Table 2b. The 90% CI limits for AUC0–∞, AUC0–1032, and Cmax were all within the BE acceptance range of 80–125%, except for the upper 90% CI limit for Cmax (125.44%).
Primary PK parameters were also estimated treating baseline BMI as a continuous covariate (rather than as a categorical variable). In this analysis, gMean ratio point estimates of the three primary PK parameters were lower compared with the primary analysis and 90% CIs were all entirely within the 80–125% standard BE acceptance range (Table 3).
Pharmacokinetics—Further Assessments
For the overall population, mean plasma concentration–time profiles for BI 695501 administered via AI and via PFS were similar over the entire observation period (Fig. 3a). On average, concentrations of BI 695501 rose relatively rapidly over the first 48–60 h and continued to rise gradually until a median tmax of approximately 4.5–5.5 days (108–132 h) (Fig. 3a; Table 4a). Afterwards, concentrations declined slowly and were still measurable for most subjects at the final sampling time point.
Variability in primary PK parameters was moderate to high [geometric coefficient of variation (gCV): 27.4–58.2%; Table 4a]. Two subjects had unexpected PK profiles: one person in the PFS group had particularly low exposure to BI 695501, not reflective of levels generally seen in this study; a second subject, also in the PFS group, had a profile indicative of accidental intravenous rather than SC injection. In a post hoc sensitivity analysis (Table 5) that excluded these two subjects, the adjusted gMean ratio for Cmax decreased from 110.19% to 108.47% and the 90% CI (97.86–120.22%) fell within the standard 80–125% range. For the first subject, AUC calculations could not be performed due to the lack of a real terminal phase; exclusion of the second subject caused a slight increase in the adjusted gMean ratio for AUC0–∞ (100.22–102.76%) and AUC0–1032 (100.14–102.01%).
Pharmacokinetics in BMI Groups
As a secondary analysis of the primary PK endpoints, relative bioavailability was evaluated within each BMI group, to assess the influence of body fat content on exposure. As expected, the 90% CIs widened, primarily due to a smaller number of subjects per group. In the low BMI group, gMeans were higher with PFS presentation than with AI presentation, whereas in the high BMI group, gMeans were higher with AI vs. PFS presentation (see supplemental material).
Scatterplots of AUC0–∞ and Cmax by BMI category (Fig. 4a, b) show a slight inverse correlation between BMI and exposure, which was similar regardless of presentation.
BMI vs. total exposure by method of administration (AI or PFS) (shown as log-transformed AUC0–∞, observed and Cmax) in a, b VOLTAIRE®-AI and c, d VOLTAIRE®-TAI. AI autoinjector, AUC0–∞, observed area under the plasma concentration–time curve from 0 extrapolated to infinity, based on observed concentrations at the last observation, BMI body mass index, Cmax maximum plasma concentration, Ln natural logarithm, PFS prefilled syringe
VOLTAIRE®-TAI
Assessment of relative bioavailability of BI 695501 administered via either AI or PFS showed that the total exposure of BI 695501 for the two administration methods was similar (Table 2c). Adjusted gMean ratios were 103.19% for AUC0–∞, 101.71% for AUC0–1368, and 100.11% for Cmax. All 90% CIs for the primary endpoints were within the standard BE acceptance range of 80–125%.
Pharmacokinetics—Further Assessments
Mean plasma concentration–time profiles for BI 695501 administered via AI and via PFS were similar over the entire observation period (Fig. 3b). Concentrations of BI 695501 rose rapidly over the first 48–60 h and continued to rise gradually until a median tmax of approximately 6–7 days (144–168 h) (Fig. 3b; Table 4b). Thereafter, concentrations declined slowly and were still measurable for approximately 60% of subjects at the final sampling time point.
gMean PK parameters from the overall trial population for BI 695501 were similar for AI and PFS (Table 4b). A similarly moderate inter-individual variability was observed for both presentations (gCV: 23.6–50.4%).
Pharmacokinetics by Body Weight
In scatterplots (Fig. 4c, d), the individual exposure values (AUC0–∞, AUC0–1368, and Cmax) overlapped for the AI and the PFS groups. The relationship between exposure and baseline body weight, particularly for Cmax, appeared to be similar for both treatment groups.
Safety
VOLTAIRE®-AI
In the AI and PFS groups, 29 (82.9%) and 29 (80.6%) of subjects reported ≥ 1 AE, respectively (Table 6). AEs reported in > 5% of subjects are shown in Table 7. No serious AEs were reported. One AE of special interest (rash) was reported by a subject in the PFS group. There were no AEs leading to discontinuation, or deaths reported during the trial.
Injection-site reactions (ISRs) were observed in a numerically greater proportion of subjects in the Al group [20 subjects (57.1%)] compared with the PFS group [14 subjects (38.9%)]. All were mild in intensity, and resolved within hours in the majority of subjects, without the need for corrective treatment.
VOLTAIRE®-TAI
In the AI and PFS groups, 61 (75.3%) and 85 (71.6%) of subjects reported ≥ 1 AE, respectively (Table 6). One or more drug-related AEs were reported in 31 (38.3%) and 30 (37.0%) of patients in the AI and PFS groups, respectively. AEs reported in > 5% of subjects are shown in Table 7. Three subjects had a serious AE: two subjects in the AI group (seizure and ligament rupture) and one subject in the PFS group (wrist fracture). None of these were related to trial drug and all led to discontinuation of the subjects. There were no deaths in the trial.
ISRs were observed in a numerically greater proportion of subjects in the Al group [26 subjects (32.1%)] compared with the PFS group [20 subjects (24.7%)]. All were non-serious; all except one were mild in intensity. The majority of events resolved within 1 or 2 days, without the need for treatment.
Immunogenicity
In both studies, similar frequencies of ADA-positive subjects, ADA titers, and frequencies of nAb-positive subjects were observed across AI and PFS groups (Fig. 5). In VOLTAIRE®-AI, by day 43, the median ADA titer in the AI and PFS groups were 8 and 4, respectively. In VOLTAIRE®-TAI, by day 57, the median ADA titer was 8 in both AI and PFS groups. Antibody titers in ADA-positive subjects evolved in a similar way across the two groups (Fig. 6).
ADA titers for ADA-positive subjects over time in a VOLTAIRE®-AI and b VOLTAIRE®-TAI. Box and whisker plots indicating the median (line) within the 25% and 75% percentile box, arithmetic mean (diamond) and outliers (circles), and 10% and 90% percentile for whiskers. ADA anti-drug antibody, AI autoinjector, nAb neutralizing antibody, PFS prefilled syringe
Discussion
The primary objective of both VOLTAIRE®-AI and VOLTAIRE®-TAI was to compare the PK of BI 695501 40 mg administered as a single SC injection using an AI vs. a PFS, in a broad range of BMI and body weights, and in two different administration locations. Total and peak exposure of BI 695501 for the two administration methods was similar regardless of injection site; the AI-to-PFS adjusted gMean ratio point estimates for the primary endpoints AUC0–1032, AUC0–∞, and Cmax were all within the 80–125% BE acceptance range (both studies).
CIs were compared using the standard BE acceptance range, even though no formal BE testing was required. All upper and lower CI limits in VOLTAIRE®-TAI were within the standard acceptance range. In VOLTAIRE®-AI, only the upper 90% CI limit for Cmax (125.44%) was slightly above the standard upper limit of 125%. This is unlikely to be clinically relevant; Pouw et al. [11] have shown that clinical efficacy improves with increasing adalimumab concentration then reaches a plateau at levels between 5 and 8 mg/ml. In a post hoc sensitivity analysis of VOLTAIRE®-AI with baseline BMI as a continuous covariate, the resulting upper 90% CI limit came within the standard 80–125% range (123.05%). These data suggest the PK of BI 695501 can be considered comparable whether administered via AI or PFS.
Humira® is approved for SC administration in the abdomen and the thigh, and patients are advised to rotate their injection sites [1]. The location of injection is thought to play a role in determining the therapeutic outcomes of SC injected biopharmaceuticals, based on differences in the physical, chemical, and physiological properties of the SC tissue [12]. As such, it was important to evaluate BI 695501 in both abdomen and thigh injection sites, necessitating the two replicate studies presented here.
Randomization was stratified by BMI category in VOLTAIRE®-AI, and by body weight category in VOLTAIRE®-TAI. Increasing thickness of abdominal subcutis reduces the chance of injections into the muscle, particularly if the free needle is > 4–5 mm [13]. As such, the tissue into which a drug is injected, and therefore PK, can be influenced by the amount of SC fat. There may also be a gender effect, since the distribution of fat and muscle tissue differs between men and women.
Overall, the relationship between BMI/body weight and the PK of BI 695501 was similar regardless of presentation. However, differences in the gMeans were observed when broken down by BMI group; in the low BMI group, gMeans were higher with PFS presentation than with AI presentation. This finding was reversed in the high BMI group. One reason for this observation may be that the PFS group tended to have more subjects at the lower end of the low BMI category, while the AI group had more subjects at the higher end of the low BMI group. The low sample size likely also contributes to this finding.
There is a clear relationship between body weight and systemic exposure to adalimumab, which is expected to be due to a difference in volume of distribution based on body weight rather than BMI, since Immunoglobulin G (IgG) is hydrophilic, and distributed equally in plasma and extracellular fluid in peripheral tissue (with relatively permeable endothelium).
In common with previous findings for the reference product [1], BI 695501 exposure was inversely related to BMI (VOLTAIRE®-AI) and body weight (VOLTAIRE®-TAI). This expected correlation has also been observed with other biologics [14]. Importantly, this relationship was independent of method of administration (i.e., AI or PFS), as seen by the considerable overlap in individual exposure values over the full BMI/body weight range following AI and PFS administration.
AEs observed during the study were primarily ISRs. Reactions of this type are common for anti-TNFα drugs [15] and are usually mild and resolve rapidly. The numerically greater proportion of ISRs in the AI group compared with the PFS group in the VOLTAIRE®-AI study may be explained by a relatively higher pressure applied with the AI against the skin; users may have pressed the AI firmly against the skin during injection, versus a more cautious administration with a PFS. Additionally, the AI has a standardized 3-s injection time whereas there is more variability possible with the PFS; subjects could slow down the injection speed in PFS if they found it painful, potentially leading to fewer ISRs. Patients have reported less pain with AI over PFS in other studies [6, 16], which appears to be at odds with the increased ISRs seen in the VOLTAIRE®-AI study.
With regard to immunogenicity, in both studies, ADAs were evident in approximately half to three-quarters of subjects in each group. A direct comparison of BI 695501 administered via AI or PFS did not show any relevant difference in terms of ADA frequency, titers, and the frequency of nAbs, thereby demonstrating no effect of the presentation on immunogenicity.
Taken together, the data derived from these studies demonstrate that the PK of BI 695501 is independent of presentation, and therefore the therapeutic efficacy would remain the same. This is in line with recent data on the anti-TNFα agent, golimumab, for which no differences were found for PK parameters between AI and standard needle and syringe administration [17].
AIs are a popular means for self-administering SC agents. Their use may be particularly helpful for patients with limited manual dexterity, for example, with RA, psoriatic arthritis, or severe palmar psoriasis [18]. The availability of BI 695501 as both PFS and AI is expected to give options to patients with different preferences or abilities.
Limitations
These healthy subjects did not have limited function of the hand; therefore, the relative handling characteristics of the two presentations in patients with impaired dexterity remains uncertain. However, data from the VOLTAIRE®-RL study of AI use in patients with active RA has shown that patients are able to handle the AI without difficulty [19]. The open-label nature of the study has the potential to introduce bias into the reporting of the safety data.
Conclusions
Administration of BI 695501 with either an AI or PFS demonstrated similar PK and comparable immunogenicity. No specific safety concerns were raised with either presentation. Comparability of PK between presentations was maintained across the broad range of BMIs and body weights, indicating that AI and PFS can be used independently of those factors and administration location.
References
AbbVie Inc. Humira® US prescribing information October 2017. https://www.rxabbvie.com/pdf/humira.pdf. Accessed Jan 2018.
Boehringer Ingelheim International Gmbh. Cyltezo® US prescribing information August 2017 [cited January 2018].
Boehringer Ingelheim International Gmbh. Cyltezo® Summary of product characteristics November 2017 [cited January 2018].
Wynne C, Altendorfer M, Sonderegger I, Gheyle L, Ellis-Pegler R, Buschke S, et al. Bioequivalence, safety and immunogenicity of BI 695501, an adalimumab biosimilar candidate, compared with the reference biologic in a randomized, double-blind, active comparator phase I clinical study (VOLTAIRE®-PK) in healthy subjects. Expert Opin Investig Drugs. 2016;25:1361–70.
Cohen SB, Alonso-Ruiz A, Klimiuk PA, Lee EC, Peter N, Sonderegger I, et al. Similar efficacy, safety and immunogenicity of adalimumab biosimilar BI 695501 and Humira reference product in patients with moderately to severely active rheumatoid arthritis: results from the phase III randomised VOLTAIRE-RA equivalence study. Ann Rheum Dis. 2018;77(6):914–21.
Kivitz A, Cohen S, Dowd JE, Edwards W, Thakker S, Wellborne FR, et al. Clinical assessment of pain, tolerability, and preference of an autoinjection pen versus a prefilled syringe for patient self-administration of the fully human, monoclonal antibody adalimumab: the TOUCH trial. Clin Ther. 2006;28:1619–29.
Demary W, Schwenke H, Rockwitz K, Kastner P, Liebhaber A, Schoo U, et al. Subcutaneously administered methotrexate for rheumatoid arthritis, by prefilled syringes versus prefilled pens: patient preference and comparison of the self-injection experience. Patient Prefer Adherence. 2014;8:1061–71.
Food and Drug Administration. Guidance for industry. Rheumatoid arthritis: developing drug products for treatment. Revision 1 May 2013. http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM354468.pdf. Accessed Jan 2018.
World Medical Association. WMA Declaration of Helsinki—ethical principles for medical research involving human subjects 2013. http://www.wma.net/en/30publications/10policies/b3/index.html. Accessed Jan 2018.
Enzi G, Gasparo M, Biondetti PR, Fiore D, Semisa M, Zurlo F. Subcutaneous and visceral fat distribution according to sex, age, and overweight, evaluated by computed tomography. Am J Clin Nutr. 1986;44:739–46.
Pouw MF, Krieckaert CL, Nurmohamed MT, Van Der Kleij D, Aarden L, Rispens T, et al. Key findings towards optimising adalimumab treatment: the concentration–effect curve. Ann Rheum Dis. 2015;74:513–8.
Kinnunen HM, Mrsny RJ. Improving the outcomes of biopharmaceutical delivery via the subcutaneous route by understanding the chemical, physical and physiological properties of the subcutaneous injection site. J Control Release. 2014;182:22–32.
Hirsch L, Byron K, Gibney M. Intramuscular risk at insulin injection sites—measurement of the distance from skin to muscle and rationale for shorter-length needles for subcutaneous insulin therapy. Diabetes Technol Ther. 2014;16:867–73.
Keizer RJ, Huitema AD, Schellens JH, Beijnen JH. Clinical pharmacokinetics of therapeutic monoclonal antibodies. Clin Pharmacokinet. 2010;49:493–507.
Murdaca G, Spano F, Puppo F. Selective TNF-alpha inhibitor-induced injection site reactions. Expert Opin Drug Saf. 2013;12:187–93.
Borras-Blasco J, Gracia-Perez A, Rosique-Robles JD, Castera MD, Abad FJ. Acceptability of switching adalimumab from a prefilled syringe to an autoinjection pen. Expert Opin Biol Ther. 2010;10:301–7.
Xu Z, Marciniak SJ Jr, Frederick B, Kim L, Zhuang Y, Davis HM, et al. Pharmacokinetic bridging approach for developing biologics-delivery devices: a case study with a golimumab autoinjector. Clin Ther. 2015;37:427–38.
Cojocaru M, Cojocaru IM, Silosi I, Vrabie CD, Tanasescu R. Extra-articular manifestations in rheumatoid arthritis. Maedica (Buchar). 2010;5:286–91.
Cohen S, Klimiuk PA, Kissel K, Jayadeva G, editors. The biosimilar candidate to Humira®, BI 695501, autoinjector (AI) detects successful self-administration in patients with rheumatoid arthritis (RA): Results from a Phase II AI patient-handling clinical study (VOLTAIRE®-RL). Stuttgart: DGRH; 2017.
Acknowledgements
Funding
These studies and this publication were supported by Boehringer Ingelheim International GmbH, Ingelheim am Rhein, Germany. All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis.
Medical Writing, Editorial, and Other Assistance
The authors wish to thank Nuala Peter (Boehringer Ingelheim, Biberach, Germany) for her critical review of the manuscript. Medical writing assistance, supported financially by Boehringer Ingelheim, was provided by Liam Sebag Montefiore of Watermeadow, and Christina Jennings of GeoMed, Ashfield companies, part of UDG Healthcare plc. The authors wish to thank the participants of the study.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
Steven Ramael is an employee of SGS and has received consultancy fees from SGS. Benjamin Van Hoorick is an employee of SGS. Renger Tiessen is an employee of PRA. Thijs van Iersel is an employee of PRA. Viktoria Moschetti is an employee of Boehringer Ingelheim. Benjamin Lang is an employee of Boehringer Ingelheim. Sabrina Wiebe is an employee of Boehringer Ingelheim. Bernd Liedert is an employee of Boehringer Ingelheim. Girish Jayadeva is an employee of Boehringer Ingelheim. Ivo Sonderegger was an employee of Boehringer Ingelheim at the time of the study and is now an employee of Takeda Pharmaceuticals International AG, Zurich.
Compliance with Ethics Guidelines
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Data Availability
All data generated or analyzed during this study are summarized in this published article/as supplementary information files.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Additional information
Steven Ramael and Benjamin Van Hoorick: Co-primary authors.
Enhanced digital features
To view enhanced digital features for this article go to https://doi.org/10.6084/m9.figshare.6456035.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Ramael, S., Van Hoorick, B., Tiessen, R. et al. Similar Pharmacokinetics of the Adalimumab (Humira®) Biosimilar BI 695501 Whether Administered via Subcutaneous Autoinjector or Prefilled Syringe (VOLTAIRE®-AI and VOLTAIRE®-TAI): Phase 1, Randomized, Open-Label, Parallel-Group Trials. Rheumatol Ther 5, 403–421 (2018). https://doi.org/10.1007/s40744-018-0119-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40744-018-0119-1