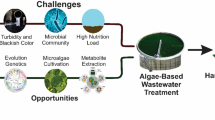
Abstract
Algae cultivation complements wastewater treatment (WWT) principles as the process uptakes nutrients while assimilates CO2 into biomass. Thus, the application of algae-based WWT is on the upward trajectory as more attention for recovery nutrients and CO2 capture while reducing its economic challenge in the circular economy concept. However, the complexity of wastewater and algal ecological characteristics induces techno-economic challenges for industry implementation. Algae-based WWT relies totally on the ability of algae to uptake and store nutrients in the biomass. Therefore, the removal efficiency is proportional to biomass productivity. This removal mechanism limits algae applications to low nutrient concentration wastewater. The hydraulic retention time (HRT) of algae-based WWT is significantly long (i.e. > 10 days), compared to a few hours in bacteria-based process. Phototrophic algae are the most used process in algae-based WWT studies as well as in pilot-scale trials. Application of phototrophic algae in wastewater faces challenges to supply CO2 and illumination. Collectively, significant landscape is required for illumination. Algae-based WWT has limited organic removals, which require pretreatment of wastewaters before flowing into the algal process. Algae-based WWT can be used in connection with the bacteria-based WWT to remove partial nutrients while capturing CO2. Future research should strive to achieve fast and high growth rate, strong environmental tolerance species, and simple downstream processing and high-value biomass. There is also a clear and urgent need for more systematic analysis of biomass for both carbon credit assessment and economic values to facilitate identification and prioritisation of barriers to lower the cost algae-based WWT.
Graphical abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Algae are a diverse group of photosynthetic aquatic organisms. They can be either unicellular (microalgae) or multicellular (macroalgae). Unlike higher plants, algae do not have roots, stems, and leaves [1]. Microalgae are microscopic eukaryotes ranging from a few micrometres to a few hundred of micrometres. They can grow extremely fast, reproduce every few hours under favourable autotrophic or mixotrophic conditions, and efficiently uptake nutrients into the body cells [2••, 3••, 4•].
Macroalgae are also fast growing in aquatic ecosystems, and can size up to tens of meters in lengths. They are composed of thallus (leaf-like body) that have enclosed gas-filled structures (float) to help in buoyancy and a specialised basal structure to provide attachment to a surface (holdfast) [5•]. Because of their wide range of physiological and biochemical characteristics, microalgae and macroalgae are naturally capable of accumulating nutrients (N and P) from different aquatic environments to produce many different bioactive compounds (e.g. pigments, carbohydrates, proteins, and lipids) for commercial applications [5•, 6, 7].
Algae-based wastewater treatment emerged from the concept to reduce the production cost in algae cultivation (i.e. alternative nutrient and water sources) while finding a sustainable way of wastewater treatment (i.e. alternative to conventional activated sludge). As photosynthetic organisms, algae generate oxygen and assimilate CO2, unlike bacteria-based WWT where significant amount of CO2 is emitted. The cultivation of algae in wastewater can simultaneously provide the wastewater treatment service and generate valuable biomass [8]. Direct utilisation of wastewater also provides an inexpensive and effective source of nutrients that also reduces freshwater use [9].
Proof-of-concept studies have proven the feasibility of algae-based WWT process to remove nutrients using different wastewater sources (e.g. municipal, agricultural, aquaculture, and industrial wastewaters) [7, 10••, 11•, 12••]. However, further work is required to ensure the suitability of wastewater for subsequent algae cultivation. For example, wastewater is susceptible to indigenous bacteria and virus contamination, which can inhibit algae growth. Culture crush increases operating cost and system downtime, which cannot be compromised for wastewater treatment service.
There are considerable technical challenges to the commercialisation of algae-based WWT, associated with cost competitiveness, scalability, and process efficiency. This review aims to provide a comprehensive insight into the algae-based WWT process. It describes the nutrient removal difference between algae and bacteria and the ecological characteristics of algae in each cultivation methods. The paper reviews lessons learned from pilot and large-scale trials and emphasises the challenges of the current algae-based WWT. It is expected that this paper will provide a broad view for researchers, engineers, and relevant industry with interest in algae process.
Algae
Algae Mechanisms to Uptake Nutrients
Algae remove nutrients from the culture environment by nitrogen assimilation and phosphorylation. Nitrogen assimilation is the process of converting inorganic nitrogen (e.g. nitrate, nitrite, ammonium, and ammonia) to its organic form, which is the building block of peptides, proteins, enzymes, chlorophylls, and energy transfer molecules such as adenosine diphosphate (ADP) and adenosine triphosphate (ATP and genetic materials such as RNA and DNA [13]. During assimilation, nitrate and nitrite are reduced ultimately to ammonium with the help of nitrate and nitrite reductase, respectively [14•]. The incorporation of ammonium into the intracellular amino acid glutamine is then facilitated by glutamate (Glu) and ATP [4•, 15••]. This nitrogen uptake strategy by algae is different to that adopted by bacteria (Fig. 1). Nitrogen removal by bacteria can be achieved in two sequential steps of nitrification and then denitrification or by anaerobic ammonium oxidation, which is often called ANAMMOX [16•, 17].
In addition to nitrogen, phosphorous also plays a key role in microalgae metabolism and growth. Inorganic phosphorous (H2PO4− and HPO42−) is incorporated into intracellular organic compounds (e.g. nucleic acids, lipids, and proteins) via phosphorylation [4•, 14•, 18•]. This process involves multiple phosphate transporters located at the plasma membrane of microalgae to uptake inorganic P for cellular phosphorous transformation. The transformation under light condition (i.e. photosynthesis) includes the generation of ATP from ADP and the synthesis of polyphosphate (e.g. acid-soluble and acid-insoluble polyphosphate) by polyphosphate kinase [4•].
Nutrient availability can affect the patterns of nutrient uptake for algae [4•]. For example, slow-growing macroalgae develop large nutrient stored pools by N and P accumulation during nutrient-rich periods for growth during nutrient-deplete time (i.e. luxury uptake) [19, 20••]. When the nutrients are limited, these slow-growing algae minimise their nutrient demand by reducing growth rates and optimise their carbon uptake capacity, profiting from sunlight abundance [21]. The ability to accumulate nutrients in excess of these species is useful for bioremediation. Freshwater Oedogonium was successfully cultured in situ for 12 months in secondary effluent at a municipal treatment plant and achieved biomass productivity of 9 to 15 g/m2/day, 36 and 65% for TN and TP removal, respectively [20••]. On the other hand, opportunistic macroalgae with high nutrient demands for fast growth incline to high-affinity uptake systems instead of storing nutrient pools [19, 22]. The uptake and assimilation of nutrients in these species occur simultaneously to sustain high growth and the synthesis of amino acids, proteins, and other nutrient-rich organic compounds [19]. This strategy makes opportunistic algae such as Cladophora glomerata, Enteromorpha ahlneriana, and Scytosiphon lomentaria well suited for cultivation in culture with high nutrient loading such as wastewater. Increased growth with protein-rich biomass was observed for green, brown, and red macroalgae cultivated in wastewaters [23•], especially for green macroalgae which are often opportunistic species with high tolerance to environmental fluctuations [24].
Nitrogen has been identified as the limiting factor to the growth rate of both microalgae and macroalgae. It has also been observed that ammonium is often the preferred N source and more readily uptake by algae due to the lower energy requirement for cells to assimilate ammonium [15••]. High N concentrations increase the internal N content in the algal biomass, leading to improved assimilation of inorganic N into amino acids and proteins. Wastewater rich in ammonium is then a promising source for algal cultivation. Algae that grow in N-rich wastewater had 4 times higher crude protein content when compared to seawater controls [25]. N-replete culture can lead to algae accumulating N-containing photosynthetic pigments such as chlorophyll, thus obtaining a darker and more vibrant green colour at the end of growth. However, too high ammonia/ammonium concentration may be inhibitory to algal growth [28] by triggering intracellular oxidative stress and disturbing cell metabolisms [29]. Ammonium tolerance levels are reportedly at < 100 mg NH4+-N/L for microalgae and < 250 mg NH4+-N/L for macroalgae [30, 31•], but some species might possess superior ammonium tolerance than others [28].
Nutrient Uptakes Rate by Algae
Due to their diverse physiology and morphologies, algae species have shown varied effectiveness and efficiency in removing nitrogen and phosphorous from different wastewater streams (Table 1). This is partly due to the nitrogen to phosphorous (N/P) ratio in wastewaters, which plays a critical role in ensuring an effective and simultaneous uptake of nutrients and biomass growth in algae species [15••]. The suitable ratio of N and P for macroalgae growth is between 10 N:1P and 80 N:1P [25] with optimal ratio of 30 N:1P, while it is 5–30 N:1P for microalgae [26, 27]. The optimal N/P ratio is strain-dependent; for example, it is reportedly 7 and 30 for Chlorella sp. and Scenedesmus sp., respectively [15••, 32••]. Wastewaters with too low N/P ratio (e.g. secondary effluent) or too high N/P ratio (e.g. centrate) might hinder algal growth; thus, strain selection is an important step when cultivating algae in wastewater. Because of this N/P ratio, faster uptake of nitrogen over phosphorous has been observed consistently for the growth of algae in wastewater (Table 1). This aligns with the empirical formula for microalgae (C106H263O110N16P), which indicates more nitrogen is needed for cell synthesis [33]. Strains within Chlorella sp. and Scenedesmus sp. have been widely used and found to be suitable for cultivation in wastewater due to their fast growth rate and high adaptability to different wastewater streams [8].
The nutrient uptake rate of macroalgae is further determined by the morphological structure together with transfer processes across the thalli rather than phylogenetic affinity. Opportunistic, filamentous, delicately branched, or monostromatic macroalgae (e.g. Cladophora glomerata, Enteromorpha ahlneriana, and Scytosiphon lomentaria) possess better nutrient uptake rates due to a higher thallus surface area to volume ratio and larger numbers of hairs protruding from the thallus [34]. An increase in water flow in the culture system also increases the nutrient flux to the surface of the thalli, thus increasing the nutrient uptake rate [35]. However, excessive water flow rate can be counterproductive due to lower water and nutrient retention time near the thalli. Nitrate and phosphate removals of Spirogyra sp. cultivated in photobioreactors were fourfold and twofold higher under low air mixing (18 ± 2 cm3/min) compared to high mixing (206 ± 14 cm3/min) rate [36•].
Cultivation Methods for Algae-Based WWT
The application of algae-based WWT relies on the method of cultivation, which influences biomass growth, nutrient removal, and the downstream process. Cultivation methods also impact on the overall capital and operation costs. Algae including micro- and macroalgae are highly diverse groups of organisms that can be found in many habitats. Their ecological diversity includes phototrophic, mixotrophic, and even heterotrophic metabolisms. This section will focus on the three metabolisms and their effects on the cultivation of algae for nutrient removal from wastewater.
Phototrophic Cultivation of Algae
Phototrophic algae utilise visible light as a primary energy source, dissolved carbon dioxide (i.e. mainly bicarbonate), and nutrients for metabolism, a process known as photosynthesis. The application of phototrophic algae for wastewater treatment therefore relies on the ability to supply effectively dissolved carbon dioxide and light energy (photon) to algae cells to boost their metabolisms [2••, 4•].
Previous studies on phototrophic algae for nutrient removal from wastewater have adapted most of the techniques in algae culture. These include open ponds (e.g. raceways, high rate algal ponds) and closed photobioreactors (i.e. horizontal tube, vertical tube, flat plate, floating film bag, helical type). However, pilot and full-scale trials mainly used the open ponds or high rate algal ponds (the “Pilot Trials of Algae-Based WWT” section). The main benefits of open ponds over the closed photobioreactors are simple cultivation systems, low capital and operating costs, and low energy requirement. However, the open ponds require large land surface and often have low biomass productivity [2••, 43]. They are also sensitive to bacterial contaminations, high water evaporation rate, and challenging to maintain stable culture conditions.
One technical challenge for phototrophic algae cultivation is to supply light to algae cells. Light penetration is often poor, and the penetration further falls with increasing cell density and depth of culture due to self-shading effects. Nguyen et al. [2••] summarised three innovative methods to enhance light exposure to algae. These include a thin-layer cascade design, submerged illumination, and airlift-lop reactor. In the thin-layer cascade, the ratio of exposed surface over total culture volume could be above 100 [44, 45••]. In the thin-layer cascade system, microalgae culture is distributed evenly at less than 5 cm at a flow rate of 0.4 to 0.5 m/s. This configuration harnesses the benefits of open systems (i.e. direct light irradiance, easy heat diffusion, rapid light/dark cycle, simple cleaning, and efficient degassing) as well as those of the closed photobioreactors (i.e. high biomass densities and high volumetric productivity) [44]. Morillas-España et al. [46•] appeared to be the first group conducting a pilot scale thin-layer cascade for nutrient removals from primary effluent. The group achieved an average of 24.8 g/m2/day of Scenedesmus sp. biomass (82 tone/ha per year) which is significantly higher than other culture setup. Thin-layer cascade also allowed a 1.4 times higher ammonium removal rate than that of the raceway pond [47]. However, it is noted that the treated volume per m2 space by thin-layer cascade is lower than the raceway pond [47]. Submerged illumination is typically designed for indoor algae cultivation. The first concept was developed by Ogbonna et al. [48•]. Until recently, its application has achieved more attention over sideway or external illumination. The notable feature of the submerged illumination system is the lighting source that is placed vertically into the culture condition. Industrial Phycology (https://i-phyc.com/the-iphyc-solution/) currently adapted this cultivation for bioremediation of secondary effluent.
Another technical challenge of the phototrophic cultivation is the CO2 availability. Dissolved CO2 level has been considered the major limiting factor in mass cultivation of algae. Generally, higher CO2 concentration to about 10 to 20 vol% is better than ambient CO2 for algae growth. Utilisation of flue gas (10% CO2) in Scenedesmus sp. cultivation with secondary municipal wastewater showed higher nutrient removal [49]. Therefore, an efficient method to supply CO2 for fast growing biomass in phototrophic cultivation would increase the nutrient removal efficiency. Previously, sparging with air or flue gas, bicarbonate solution, or carbonation with membranes has been tried in algae cultivation. However, sparging is the most common method in algae-based WWT due to its simplicity [2••].
Heterotrophic Cultivation of Algae
Heterotrophic algae can grow in the dark using organic compounds for carbon and energy sources. Previously, heterotrophic algae are cultured in fermenter with periodical or continuous supply of carbon sources (e.g. glucose, volatile fatty acids) to generate high-lipid content biomass in for subsequent biofuel production. The heterotrophic cultivation of algae enables high-density biomass, consistent productivity, and quantities. Kim et al. [50] reported a biomass productivity rate of 5.4 g/L/day with a maximum concentration of 43 g/L in a culture with supplement of 72 and 8 g/L of glucose and NaNO3, respectively. Heterotrophic production of algae has already been commercialised technology given the high lipid content of heterotrophically grown algae and the ability to manipulate the cell’s biochemistry. However, heterotrophic cultivation requires inputs that are more expensive and the supply of oxygen in aerobic submerged cultivation.
Heterotrophic cultivation of algae for nutrient removal leverages on the conditions that wastewater presents many types of organic compounds such as glucose, volatile fatty acids, glycerol, and ethanol. These organic carbon sources are typical for algae uptake [51]. However, there only limited number of algae species (e.g. Chlorella vulgaris, Haematococcus pluvialis, Chlorella sorokiniana, and Botryococcus braunii) with ability to use organic carbon as source of energy and carbon due to the lack of uptake mechanisms or transport pathway [4•, 52]. For example, glucose is metabolised via the pentose phosphate pathway and the Embden-Meyerhof Pathway without and with light conditions, respectively [51]. It is also noted that high nitrogen wastewater in the form of ammonium promotes the pentose phosphate pathway. Thus, heterotrophic cultivation may be selected for high nitrogen wastewater source. To date, there are only a few studies on heterotrophic cultivation of algae in wastewater. Guldhe et al. [53] utilised Chlorella sorokiniana culture for treatment of aquaculture wastewater. This heterotrophic culture achieved above 80% removal of ammonium, nitrates, phosphates, and chemical oxygen demand. Pretreatments of aquaculture wastewater by filtration and autoclave were required prior to microalgae culture. This study was only limited to 1 L culture volume, although the biomass production was 3 times of that phototrophic cultivation [53]. Likewise, Kim et al. [54•] observed a better removal of nutrient and biomass productivity with Chlorella sorokiniana under heterotrophic conditions.
Heterotrophic cultivation overcomes light and dissolved CO2 limitations while achieves better organic carbon removal from wastewater compared to phototrophic algae. Therefore, wastewater compositions should be considered when selecting carbon metabolisms in algae culture. Another important notification is that heterotrophic algae culture often requires sterile conditions to avoid bacteria contamination. This limits its application in wastewater treatment process. Currently, there has been no pilot or large-scale trial of heterotrophic cultivation of algae for nutrient removal in wastewater (the “Pilot Trials of Algae-Based WWT” section).
Mixotrophic Cultivation of Algae
Mixotrophic cultivation of algae is a combination of phototrophic and heterotrophic metabolisms where energy and C source are light and organic and inorganic carbon [55]. In mixotrophic cultivation, both light limitation and inhibition are less effective, while dissolved CO2 and organic carbon can be used for metabolisms. Thus, mixotrophic cultivation provides flexibility and higher efficiency in nutrient removal and biomass productivity.
Organic carbon sources have a great influence on nutrient removal in mixotrophic cultivation. Peng et al. [56] observed that nitrogen and phosphorus removal was better when supplemented with the mixotrophic culture of Chlorella vulgaris with glucose rather than protein or sodium acetate. Addition of glucose also enhanced the fixation of nitrogen and phosphorus in an artificial wastewater [57]. Higher glucose dose also generated lipid-rich biomass for biofuel production [57]. Mixotrophic cultivation often applies for high organic content wastewater. For example, Wang et al. [58] used green algae Chlorella pyrenoidosa for treatment of piggery wastewater with carbon content from 0.25 to 1 g/L and obtained an efficient ammonium removal of 90%. At 1 g/L carbon, the culture produced a maximum lipid of 6.3 mg/dL. The ability to use organic carbon in wastewater to produce high lipid content biomass is one advantage of mixotrophic cultivation. Nevertheless, pretreatment (e.g. dilution) to reduce the organic content, suspended solids, and nutrients to suitable level for algae growth is required [58]. Thus, it is envisioned that algae-based WWT would need to be strategically integrated into existing wastewater infrastructure.
Pilot Trials of Algae-Based WWT
Pilot and full-scale reports of algae-based WWT using microalgae and macroalgae are summarised in Table 2. These systems showed that nutrient removal efficiency varied significantly depending on species, initial nutrient concentrations, reactor sizes, and culture systems. It is also noticed that most of pilot and large-scale trials utilised phototrophic cultivation method.
A few lessons have been discussed from the pilot and large-scale trials. First, Sutherland et al. [59••] suggested that the size of high rate algal pond has significant impact on biomass growth and operational stability. In comparison of three reactor sizes (5 m2, 330 m2, and 1 ha), the 5 m2 pond provided better nutrient removal and biomass productivity than that of 330 m2 and 1 ha. The authors suggested that this observation might have implications for commercial scale development with respect to capital and operational cost.
Another lesson is the realisation of long hydraulic retention time (HRT) in algae-based WWT. This is directly related to the initial nutrient concentration and the nutrient uptake rate of selected algae. The HRT is also directly proportional to solar radiation. Thus, a dilution factor to shorten the HRT in summer and increase the HRT in wintertime can be applied when there are light limiting conditions and low temperatures. This strategy is important in regions with significant seasonal variations as Europe, California, and New Zealand.
The reported biomass productivity also varies significantly amongst studies (Table 2). Although a direct comparison cannot be made, some general observations can be highlighted. For example, a thin-layer cascade offered high biomass productivity [46•]. However, thin-layer cascade may be limited to a low treated volume. The obtained results from these experiments may not translate well to larger scale. It is also noticed that techno-economic analysis has been neglected in these studies. Thus, the validation of large-scale feasibility for algae-based WWT remains unclear.
Pilot and full-scale trials of macroalgae have recently received more attention because of its advantages over microalgae process. Macroalgae provides an easy harvesting process and greater resistance to bacteria and predation. The facilitation of biomass harvesting reduces the cost and complexity during the implementation into wastewater treatment. Macroalgae can also be retained easily in the reactor allows for a strong culture enrichment [60]. Future studies could focus on high-quality biomass and explore the chemical extraction from macroalgae derived from wastewater. This would potentially unlock the economic barriers in algae-based WWT.
Benefits of Algae-Based WWT
Carbon Credit Assessment
The National Greenhouse and Energy Reporting Scheme estimated an emission factor of 4.9 tonnes CO2-e per tonne of nitrogen removed [70]. Direct emissions at WWTPs are from biological carbon, nitrogen, and phosphate removal, sludge management, and off-gas from wastewater collection systems. In the conventional bacteria-based WWT process, Bao et al. [71] reported 0.97 kg of the direct emission of CO2 for treatment of 1 kg chemical oxygen demand in wastewater. This was equivalent to 0.34 g CO2 per m3 of treated wastewater. There are also indirect emissions due to the large energy consumptions, chemical usages, and waste disposal [72].
Algae-based WWT utilises the ability of algae to lock CO2 into their biomass, which contributes to the CO2 emission reduction in two ways. First, it is about 1.83 kg of CO2 for every 1 kg of algae biomass. Algae biomass production could consume 1.83, 0.05, and 0.01 ton of CO2, nitrogen, and phosphorus, respectively [73••]. Second, the algae biomass can be used as renewable source for energy, biochemical, or long-term products (i.e. bioplastics, brick, and biochar) to lock away CO2 [2••] (the “Biomass Applications” section). Preliminary estimates demonstrate that algae-based WWT may entirely offset the industry’s greenhouse gas footprint and make it a globally significant contributor of negative carbon emissions. However, there has no system to report the carbon emissions for algae-based WWT. Future studies on life cycle assessment and carbon balance analysis are required to support the claim of algae-based WWT for carbon capture (Fig. 2).
Co-locations of wastewater treatment and algae cultivation for carbon capture and utilisation will bring opportunities for major CO2-emitting industries. In this concept, algae cultivation will supplement the main wastewater treatment to capture and recycle the nutrients and CO2 gas from anaerobic digester (Fig. 2). The harvested algae biomass can be recycled back into the digester for bioenergy production, thus closing the CO2 loop at WWTP.
Biomass Applications
There are many products that can be made with the whole algal biomass without further processing (i.e. after harvesting and drying stages). These include food (e.g. Spirulina supplements) [74], energy (e.g. syngas or bio-oil), soil additives (e.g. biostimulants), or feedstock for anaerobic digestion [75]. In the algae-based wastewater treatment, the generated biomass can be used for production of biocrude, biofuel [76•, 77], bioplastics [78••], and animal feed [2••, 79]. These applications could offset or reduce carbon emissions at wastewater treatment plant by replacing items currently derived from carbon-intensive practices using fossil fuel. For example, Naaz et al. [80•] demonstrated the ability of two algae species Chlorella pyrenoidosa and Phormidium cultivation in municipal wastewater at 100 L outdoor attached biofilm system. While the nutrient removal was 53 to 87% after 6 days HRT, the generated biomass was stable at 3.5 g/m2day with high lipid content (35% of total solid). Biomethane (i.e. anaerobic digestion of the generated biomass) and biocrude (hydrothermal liquefaction of the generated biomass) analysis showed the better net energy ratio of biocrude production [80•]. Although biocrude production has high-energy ratio, biomethane production from algae-based WWT could have multi-fold benefits. For example, the biogas generated from the anaerobic digester can be used to provide CO2 for algae cultivation. The digestate can be used to supply nutrients. This configuration was first introduced by Converti et al. [81], who combined a mixed sludge anaerobic digester with a photobioreactor leading to the production of a biogas with a CH4 content above 70%. This approach also allows direct utilisation of algae biomass.
Algae biomass from wastewater treatment process can potentially be used in several long-term products for carbon storage such as in cement and bioplastic production [78••, 82]. Unlike plant-based biomass (i.e. lignocellulosic biomass), algae biomass has low percentage of lignin and rich in long-chain hydrocarbons (i.e. high purity of cellulose) which can be extracted to make bioplastics [78••, 83]. Another potential usage of algae is the use of algal biochar, which can be added to the soil for more long-term carbon storage [84] and moving towards sustainable agriculture [74]. Algae could also be used as biostimulants to improve crop production, thereby reducing the need for fossil-based fertilisers [85].
The benefits of algae biomass have been well discussed in the literature review [2••, 72]. In general, the application of algae biomass product is technically feasible; their economic feasibility is still under discussion [73••, 86••]. The production cost includes cultivation, harvesting, pretreatment, and chemical extraction at scale that is still significant. Unfortunately, commercial techno-economic assessment models are not available. Currently, commercial algae biomass is mainly used to produce high-value ingredients for the cosmetic and nutraceutical industries.
Challenges for Adaptation of Algae-Based WWT
Scalability and Efficiency to Meet the Wastewater Treatment Process
Although there have been a number of pilot and full-scale trials around the world, the adaptation of algae-based WWT by industry is still very low. The main reason is that algae-based WWT currently faces significant challenges to scale up and maintain its treatment efficiency. The intrinsic physical limitation is that the light transmissivity is around 5 cm. Therefore, the ratio of effluent-treated water over surface area needed is significantly larger than that of other treatment methods. Nguyen et al. [87] provided an initial calculation of the required surface and volume of an algae membrane reactor to treat membrane bioreactor effluent at a hydraulic retention time of 1 days. With the assumption that nutrients are removed at 100% efficiency, the volume of an algae membrane reactor would be 37 times that of the membrane bioreactor. Likewise, Neveux et al. [66] used a nitrogen removal rate of 1 g/m2/day and estimated that a 3.5 ha of macroalgae cultivation area is required to remove 90% of nitrogen at 40 mg/L concentration from 1 ML/d treatment capacity. In the field study, Neveux et al. [66] reported that a 94-ha land surface is needed to treat 29 ML/d using Oedogonium culture. The large footprint will hinder the adaptation of algae-based WWT into the existing plants situated in urban and other space-deficient locations.
Nutrient removal by algae is mainly by cell assimilation. Thus, the removal rate is proportional to the biomass production rate (the “Cultivation Methods for Algae-Based WWT” section and the “Pilot Trials of Algae-Based WWT” section). This limitation of nutrient uptake (i.e. slow and low level) by algae requires a long HRT. The HRT of algae-based WWT is often above 10 days, compared to a few hours in bacteria-based WWT process. The long HRT future intensifies space requirements for algae-based WWT. Therefore, algae-based WWT has to be strategically integrated with existing wastewater infrastructure. One approach is to integrate algae-based WWT for tertiary wastewater treatment for polishing step (i.e. to remove the residual nutrients). Cole et al. [20••] utilised freshwater macroalgae as an in-line tertiary treatment of treated effluent from a municipal wastewater treatment in Australia. They used Oedogonium in open ponds to reduce 36 and 65% total nitrogen and total phosphorous in treated effluent. Another approach is to utilise submerged LED, which can reduce land requirement [2••]. The algal biological reactor developed by Industrial Phycology from the UK adapted to use internal illumination and treatment of tertiary effluent. In this process, the algae solution is mixed in a series of tanks. LED lamps are installed vertically into the tanks. This concept significantly improves the treated volume and reduces space requirement. Future research to improve the areal productivity and nutrient uptake rates are required to reduce the land requirements.
Resilience and Consistency in the Performance
The traditional objective of wastewater treatment is to remove carbon, nitrogen, phosphorus, and other contaminants so the effluent meets environmental quality regulation. In this context, algae-based WWT must be realised without compromising treatment efficacy given that protection of the local aquatic environment and public health will remain paramount for the wastewater industry. However, algae-based WWT still have uncertain performance reliability [7, 72].
Algae-based WWT faces seasonal performance due to mainly sunlight availability and temperature variation. In the pilot scale study, Cole et al. [20••] observed a maximum variation of 180% in biomass productivity (8.9 vs 15.8 g/m2/day) between winter and summer time during the cultivation of freshwater macroalgae Oedogonium in secondary treated water. Xu et al. [88] also observed that nutrient removal and biomass yield varied with seasonal conditions. In summer and autumn conditions, the highest removal of total nitrogen, total suspended solids, and specific growth rate were achieved. Morillas-España et al. [46•] reported a significant variation of nitrogen (695.4–2383.4) and phosphorus removal rates (70.4–111.8 mg/m2/day), respectively, from winter to summer. One approach to alleviate the seasonal variation is to rotate between high- and low-temperature tolerant strains. Culture mixing and reactor design (i.e. depth selection) could also improve performance when there is less sunlight availability. However, this approach needs to consider other broad factors such as space requirement and treatment capacity.
Algae-based WWT has limited organic removal. In fact, the presence of organic matter in wastewater can cause significant problem such as bacteria contaminations and light penetration limitation due to high turbidity. Therefore, most of algae-based WWT studies mainly utilised tertiary effluent, which has low organic carbon. Heterotrophic and mixotrophic cultivation can utilise the organic carbon present in wastewater. However, algae will be in competition with bacteria for this carbon. In this competition, bacteria often outcompete the algae populations [88]. Bacterial contamination and zooplankton grazing also reduce biomass productivity and reliability of nutrient removal efficiency and increase downtime [89]. The algae-bacteria consortium system is an alternative method for preventing contamination [90]. However, this system is difficult to implement in practice, and it takes at least 9 days for both microorganisms to adapt [91•]. Selection of algae species and operating conditions that support simultaneously the proliferation of algae and bacteria are also needed in this concept [3••].
The resilience of algae-based WWT process remains questionable. Algae-based process does not fit all types of wastewaters [8]. This is unlike bacteria-based process in which there are multiple of bacteria species to perform organic carbon and nutrients removal. Algae-based process largely relies on single species. Besides, wastewaters have inconsistent component event at daily timescale (i.e. various flow, peak hour, wet and dry weather). The component variation will impact algae performance significantly [4•, 8, 55].
Efficient Downstream Processes
The application of algae-based WWT also includes separation of algal biomass from treated water and downstream processing (i.e. chemical extraction or direct application). Currently, harvesting and chemical extraction are still expensive. Harvesting and extraction account for over 60% of the total operating cost. Due to the high cost of downstream processing, the full-scale algae-based WWT still have limited application.
The algae-based WWTs utilised microalgae have significant operation cost compared to that of macroalgae. Microalgae have small cell size (2 to 10 um), negative surface charge, and neutral buoyancy. To date, common harvesting methods include centrifugation, membrane filtration, flocculation, and flotation which are used for microalgae [92•]. Amongst these methods, flocculation and flotation appears to be more suitable due to their low operating cost and readily infrastructure at the wastewater treatment. Nguyen et al. [93•] demonstrated an efficient flocculation of freshwater Chlorella vulgaris using a cationic polyacrylamide flocculant which is commonly used for sludge dewatering at full-scale wastewater treatment plant (Fig. 3). Over 98% of algae biomass was recovered.
The utilisation of macroalgae species could overcome the harvesting challenge with microalgae due to its sizes [7]. Macroalgae can be easily separated from the treated water by filter screen. The pore size of the filter screen can be selected and changed based on the size of each macroalgae species [7, 20••]. Cole et al. [20••] developed a passive harvesting method for Oedogonium culture (Fig. 2). The algae biomass was filtered into a 750-µm filter screen twice a week for 18 h per time. The flow rate of harvesting was equivalent to the rate of the incoming water. They estimated that 50 to 75% of produced biomass was harvested. The remaining (0.18 to 0.37 g/L dry weight) continues the treatment until next harvesting. This passive harvesting required no energy input. The harvested biomass is dried or used freshly on a dairy farm.
Perspectives and Conclusions
Algae-based technology has been proposed as a sustainable solution for removal and recycle of nutrients in wastewater. Naturally, algae (both microalgae and macroalgae) can uptake nutrients from water for growth. Thus, laboratory studies have successfully demonstrated nutrient removal via algae cultivation. There have been also significant number of studies devoted to algae species selection, designing cultivation systems, assessing different wastewater sources, and evaluating the performance at pilot and large-scale systems. However, the pathway to implement algae-based WWT process still has several barriers.
There is limited full-scale adaptation of algae-based WWT both due to its technical and economic challenges. The technical challenge includes maintenance of viable algae culture for stable and long-time performance. Techniques to intensify algae cultivation for large-scale wastewater treatment are needed. Currently, the most suitable way is to co-locate algae cultivation with exiting wastewater treatment plant, where algae cultivation is mainly used to supplement part of nutrient removal. This approach could ensure minimal disturbance to the existing process. Co-location of algae cultivation also benefits to use CO2 from digester biomass as a way to intensify algae growth.
Life cycle analysis, carbon balance, and economic analysis have been neglected in the literature, particularly in pilot and large-scale trials. It is suggested that these analyses should be conducted parallel or early stage of the projects. The economic aspect would improve at the species selection process. For example, the selections should focus on the fast growth rate, high photosynthetic rate, and strong environmental tolerance species with simple downstream processing (e.g. harvesting) and high-value biomass. Algae are a highly diverse group of microorganisms (0.2–1 million recognised species), providing a significant biobank for selection. In addition, the development of algae biotechnology (e.g. genetic modifying organisms) can be applied to enhance its capacity.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Bajpai P. Characteristics of algae. In: Bajpai P, editor. Third Generation Biofuels. Singapore. Singapore: Springer; 2019. p. 11–5.
•• Nguyen LN, Vu MT, Vu HP, Johir MA, Labeeuw L, Ralph PJ, Mahlia TM, Pandey A, Sirohi R, Nghiem LD. Microalgae-based carbon capture and utilization: a critical review on current system developments and biomass utilization. Critical Rev Environ Sci Technol. 2022;1–23. This study provides a deep analysis on the current microalgal cultivation methods and biomass utilisation for carbon capture and utilisation.
•• Aditya L, Mahlia TMI, Nguyen LN, Vu HP, Nghiem LD. Microalgae-bacteria consortium for wastewater treatment and biomass production. Sci Total Environ. 2022;838: 155871. This critical review paper provides the application of mixed microalgae and bacteria for wastewater treatment. It indicates the complementary between algae and bacteria.
• Su Y. Revisiting carbon, nitrogen, and phosphorus metabolisms in microalgae for wastewater treatment. Sci Total Environ. 2021;762: 144590. This paper provides detailed explaination on different metabolisms in microalgae.
• Suganya T, Varman M, Masjuki HH, Renganathan S. Macroalgae and microalgae as a potential source for commercial applications along with biofuels production: a biorefinery approach. Renew Sustain Energy Rev. 2016;55:909–41. This review paper highlights the role of macroaglae and microaglae biomass for biorefinery.
Khoo CG, Dasan YK, Lam MK, Lee KT. Algae biorefinery: review on a broad spectrum of downstream processes and products. Biores Technol. 2019;292: 121964.
Liu J, Pemberton B, Lewis J, Scales PJ, Martin GJO. Wastewater treatment using filamentous algae - a review. Bioresour Technol. 2020;298: 122556.
Li K, Liu Q, Fang F, Luo R, Lu Q, Zhou W, Huo S, Cheng P, Liu J, Addy M, Chen P, Chen D, Ruan R. Microalgae-based wastewater treatment for nutrients recovery: a review. Bioresour Technol. 2019;291: 121934.
Yang J, Xu M, Zhang X, Hu Q, Sommerfeld M, Chen Y. Life-cycle analysis on biodiesel production from microalgae: water footprint and nutrients balance. Bioresour Technol. 2011;102(1):159–65.
•• Whitton R, Ometto F, Pidou M, Jarvis P, Villa R, Jefferson B. Microalgae for municipal wastewater nutrient remediation: mechanisms, reactors and outlook for tertiary treatment. Environment Technol Rev. 2015;4(1):133–48. This paper provides discussion on the nutrients removal efficiency in algae-based WWT.
• Vu MT, Nguyen LN, Mofijur M, Johir MAH, Ngo HH, Mahlia TMI, Nghiem LD. Simultaneous nutrient recovery and algal biomass production from anaerobically digested sludge centrate using a membrane photobioreactor. Bioresour Technol. 2022;343: 126069. This research paper highlighted the benefit of membrane photobioreactor for treatment of digested sludge centrate.
Vo T-D-H, Bui X-T, Dang B-T, Nguyen T-T, Nguyen V-T, Tran DPH, Nguyen P-T, Boller M, Lin K-YA, Varjani S, Show PL. Influence of organic loading rates on treatment performance of membrane bioreactor treating tannery wastewater. Environ Technol Innovation. 2021;24: 101810.
Barsanti L, Gualtieri P. Algae: anatomy, biochemistry, and biotechnology. CRC Press; 2005.
• Gonçalves AL, Pires JCM, Simões M. A review on the use of microalgal consortia for wastewater treatment. Algal Res. 2017;24:403–15. This review paper provided a summary of studies with microalgal consortia for WWT.
•• Cai T, Park SY, Li Y. Nutrient recovery from wastewater streams by microalgae: status and prospects. Renew Sustain Energy Rev. 2013;19:360–9. This paper provides a summary of research before 2013 on nutrient recovery by microalgae.
• Mishra S, Singh V, Cheng L, Hussain A, Ormeci B. Nitrogen removal from wastewater: a comprehensive review of biological nitrogen removal processes, critical operation parameters and bioreactor design. J Environ Chem Eng. 2022;10(3): 107387. This review paper provides some up to date literature on the nitrogen removal by different biological processes.
Ren Y, Hao Ngo H, Guo W, Wang D, Peng L, Ni B-J, Wei W, Liu Y. New perspectives on microbial communities and biological nitrogen removal processes in wastewater treatment systems. Biores Technol. 2020;297: 122491.
• Mohsenpour SF, Hennige S, Willoughby N, Adeloye A, Gutierrez T. Integrating micro-algae into wastewater treatment: a review. Sci Total Environ. 2021;752: 142168. This review paper provides some discussion on the methods to integrate algae into wastewater treatment.
Martínez B, Pato LS, Rico JM. Nutrient uptake and growth responses of three intertidal macroalgae with perennial, opportunistic and summer-annual strategies. Aquat Bot. 2012;96(1):14–22.
•• Cole AJ, Neveux N, Whelan A, Morton J, Vis M, de Nys R, Paul NA. Adding value to the treatment of municipal wastewater through the intensive production of freshwater macroalgae. Algal Res. 2016;20:100–9. This paper higlights some potential benefits of using wastwater for freshwater macroaglae cultivation.
Beuckels A, Smolders E, Muylaert K. Nitrogen availability influences phosphorus removal in microalgae-based wastewater treatment. Water Res. 2015;77:98–106.
Roleda MY, Hurd CL. Seaweed nutrient physiology: application of concepts to aquaculture and bioremediation. Phycologia. 2019;58(5):552–62.
• Stedt K, Pavia H, Toth GB. Cultivation in wastewater increases growth and nitrogen content of seaweeds: a meta-analysis. Algal Res. 2022;61: 102573. This paper provides some benefit of using wastewater to culture seaweeds.
Ye N-H, Zhang X-W, Mao Y-Z, Liang C-W, Xu D, Zou J, Zhuang Z-M, Wang Q-Y. ‘Green tides’ are overwhelming the coastline of our blue planet: taking the world’s largest example. Ecol Res. 2011;26(3):477.
Stedt K, Trigo JP, Steinhagen S, Nylund GM, Forghani B, Pavia H, Undeland I. Cultivation of seaweeds in food production process waters: evaluation of growth and crude protein content. Algal Res. 2022;63: 102647.
Choi HJ, Lee SM. Effect of the N/P ratio on biomass productivity and nutrient removal from municipal wastewater. Bioprocess Biosyst Eng. 2015;38(4):761–6.
Figler A, Márton K, B-Béres V, Bácsi I. Effects of nutrient content and nitrogen to phosphorous ratio on the growth, nutrient removal and desalination properties of the green alga Coelastrum morus on a laboratory scale. Energies. 2021;14(8).
Collos Y, Harrison PJ. Acclimation and toxicity of high ammonium concentrations to unicellular algae. Mar Pollut Bull. 2014;80(1):8–23.
Lu Q, Chen P, Addy M, Zhang R, Deng X, Ma Y, Cheng Y, Hussain F, Chen C, Liu Y, Ruan R. Carbon-dependent alleviation of ammonia toxicity for algae cultivation and associated mechanisms exploration. Biores Technol. 2018;249:99–107.
Nwoba EG, Moheimani NR, Ubi BE, Ogbonna JC, Vadiveloo A, Pluske JR, Huisman JM. Macroalgae culture to treat anaerobic digestion piggery effluent (ADPE). Biores Technol. 2017;227:15–23.
• Praveen P, Guo Y, Kang H, Lefebvre C, Loh K-C. Enhancing microalgae cultivation in anaerobic digestate through nitrification. Chem Eng J. 2018;354:905–12. This research article provides an assessment on the ability of microalgae culture in anaerobic digestate.
•• Shi J, Podola B, Melkonian M. Removal of nitrogen and phosphorus from wastewater using microalgae immobilized on twin layers: an experimental study. J Appl Phycol. 2007;19(5):417–23. This paper higlights a new method of algae immobilisation for nitrogen and phosphorus removal from wastewater.
Randrianarison G, Ashraf MA. Microalgae: a potential plant for energy production. Geology, Ecology, and Landscapes. 2017;1(2):104–20.
Wallentinus I. Comparisons of nutrient uptake rates for Baltic macroalgae with different thallus morphologies. Mar Biol. 1984;80(2):215–25.
Mawi S, Krishnan S, Din MFMD, Arumugam N, Chelliapan S. Bioremediation potential of macroalgae Gracilaria edulis and Gracilaria changii co-cultured with shrimp wastewater in an outdoor water recirculation system. Environ Technol Innov. 2020;17: 100571.
• Ge S, Madill M, Champagne P. Use of freshwater macroalgae Spirogyra sp. for the treatment of municipal wastewaters and biomass production for biofuel applications. Biomass Bioenerg. 2018;111:213–23. This is one of a few research paper on the utilisation of macroalgae for municipal wastewater
Liu Y, Lv J, Feng J, Liu Q, Nan F, Xie S. Treatment of real aquaculture wastewater from a fishery utilizing phytoremediation with microalgae. J Chem Technol Biotechnol. 2019;94(3):900–10.
Marinho-Soriano E, Nunes SO, Carneiro MAA, Pereira DC. Nutrients’ removal from aquaculture wastewater using the macroalgae Gracilaria birdiae. Biomass Bioenerg. 2009;33(2):327–31.
• Ruiz-Martinez A, Martin Garcia N, Romero I, Seco A, Ferrer J. Microalgae cultivation in wastewater: nutrient removal from anaerobic membrane bioreactor effluent. Bioresour Technol. 2012;126:247–53. This paper provides some good results on the application of algae for nutrient removal for anaerobic membrane bioreactor.
• Ge S, Champagne P. Cultivation of the marine macroalgae Chaetomorpha linum in municipal wastewater for nutrient recovery and biomass production. Environ Sci Technol. 2017;51(6):3558–66. Another good paper on the use of macroalgae for wastewater treatment.
Álvarez-Díaz PD, Ruiz J, Arbib Z, Barragán J, Garrido-Pérez MC, Perales JA. Freshwater microalgae selection for simultaneous wastewater nutrient removal and lipid production. Algal Res. 2017;24:477–85.
•• Wu LF, Chen PC, Huang AP, Lee CM. The feasibility of biodiesel production by microalgae using industrial wastewater. Biores Technol. 2012;113:14–8. This study mentiones the cost and feasibility accessment on the biodiesel production by algae cultivation in wastewater.
Pugazhendhi A, Nagappan S, Bhosale RR, Tsai P-C, Natarajan S, Devendran S, Al-Haj L, Ponnusamy VK, Kumar G. Various potential techniques to reduce the water footprint of microalgal biomass production for biofuel—a review. Sci Total Environ. 2020;749: 142218.
Grivalský T, Ranglová K, da Câmara Manoel JA, Lakatos GE, Lhotský R, Masojídek J. Development of thin-layer cascades for microalgae cultivation: milestones (review). Folia Microbiol. 2019;64(5):603–14.
•• Celis-Plá PSM, Rearte TA, Neori A, Masojídek J, Bonomi-Barufi J, Álvarez-Gómez F, Ranglová K, Carmo da Silva J, Abdala R, Gómez C, Caporgno M, Torzillo G, Silva Benavides AM, Ralph PJ, Fávero Massocato T, Atzmüller R, Vega J, Chávez P, Figueroa FL. A new approach for cultivating the cyanobacterium Nostoc calcicola (MACC-612) to produce biomass and bioactive compounds using a thin-layer raceway pond. Algal Res. 2021;59: 102421. This study highlights the new cultivation system "a thin-layer raceway pond" for cyanobacterium cultivation.
• Morillas-España A, Lafarga T, Acién-Fernández FG, Gómez-Serrano C, González-López CV. Annual production of microalgae in wastewater using pilot-scale thin-layer cascade photobioreactors. J Appl Phycol. 2021;33(6):3861–71. This study provides a year of data on the thin-layer cascade reactor.
Raeisossadati M, Vadiveloo A, Bahri PA, Parlevliet D, Moheimani NR. Treating anaerobically digested piggery effluent (ADPE) using microalgae in thin layer reactor and raceway pond. J Appl Phycol. 2019;31(4):2311–9.
• Ogbonna JC, Yada H, Masui H, Tanaka H. A novel internally illuminated stirred tank photobioreactor for large-scale cultivation of photosynthetic cells. J Ferment Bioeng. 1996;82(1):61–7. This study provides a new approach to provide light to algae internally.
Posadas E, Morales MDM, Gomez C, Acién FG, Muñoz R. Influence of pH and CO2 source on the performance of microalgae-based secondary domestic wastewater treatment in outdoors pilot raceways. Chem Eng J. 2015;265:239-248.
Kim HS, Park WK, Lee B, Seon G, Suh WI, Moon M, Chang YK. Optimization of heterotrophic cultivation of Chlorella sp. HS2 using screening, statistical assessment, and validation. Sci Rep. 2019;9(1):19383.
Perez-Garcia O, Escalante FME, de-Bashan LE, Bashan Y. Heterotrophic cultures of microalgae: metabolism and potential products. Water Res. 2011;45(1):11-36.
Mitra D, van Leeuwen J, Lamsal B. Heterotrophic/mixotrophic cultivation of oleaginous Chlorella vulgaris on industrial co-products. Algal Res. 2012;1(1):40–8.
Guldhe A, Ansari FA, Singh P, Bux F. Heterotrophic cultivation of microalgae using aquaculture wastewater: a biorefinery concept for biomass production and nutrient remediation. Ecol Eng. 2017;99:47–53.
• Kim S, Park J-E, Cho Y-B, Hwang S-J. Growth rate, organic carbon and nutrient removal rates of Chlorella sorokiniana in autotrophic, heterotrophic and mixotrophic conditions. Bioresour Technol. 2013;144:8–13. This study provides data on the algae performance under three different cultivation conditions.
Yeh K-L, Chang J-S. Effects of cultivation conditions and media composition on cell growth and lipid productivity of indigenous microalga Chlorella vulgaris ESP-31. Bioresour Technol. 2012;105:120–7.
Peng Y-Y, Gao F, Hang W-JW, Yang H-L, Jin W-H, Li C. Effects of organic matters in domestic wastewater on lipid/carbohydrate production and nutrient removal of Chlorella vulgaris cultivated under mixotrophic growth conditions. J Chem Technol Biotech. 2019;94(11):3578–84.
Chandra R, Rohit MV, Swamy YV, Venkata Mohan S. Regulatory function of organic carbon supplementation on biodiesel production during growth and nutrient stress phases of mixotrophic microalgae cultivation. Bioresour Technol. 2014;165:279–87.
Wang H, Xiong H, Hui Z, Zeng X. Mixotrophic cultivation of Chlorella pyrenoidosa with diluted primary piggery wastewater to produce lipids. Bioresour Technol. 2012;104:215–20.
•• Sutherland DL, Park J, Heubeck S, Ralph PJ, Craggs RJ. Size matters – microalgae production and nutrient removal in wastewater treatment high rate algal ponds of three different sizes. Algal Res. 2020;45: 101734. This study highlights the challenge with up-scaling algae based WWT.
Liu J, Danneels B, Vanormelingen P, Vyverman W. Nutrient removal from horticultural wastewater by benthic filamentous algae Klebsormidium sp., Stigeoclonium spp. and their communities: from laboratory flask to outdoor Algal Turf Scrubber (ATS). Water Res. 2016;92:61–8.
Rossi S, Pizzera A, Bellucci M, Marazzi F, Mezzanotte V, Parati K, Ficara E. Piggery wastewater treatment with algae-bacteria consortia: pilot-scale validation and techno-economic evaluation at farm level. Biores Technol. 2022;351: 127051.
Zieliński M, Dębowski M, Kazimierowicz J. Outflow from a biogas plant as a medium for microalgae biomass cultivation—pilot scale study and technical concept of a large-scale installation. Energies. 2022;15(8):2912.
• Xie D, Ji X, Zhou Y, Dai J, He Y, Sun H, Guo Z, Yang Y, Zheng X, Chen B. Chlorella vulgaris cultivation in pilot-scale to treat real swine wastewater and mitigate carbon dioxide for sustainable biodiesel production by direct enzymatic transesterification. Biores Technol. 2022;349: 126886. This study provides some good findings on the ability of algae in swine wastewater.
• Díez-Montero R, Belohlav V, Ortiz A, Uggetti E, García-Galán MJ, García J. Evaluation of daily and seasonal variations in a semi-closed photobioreactor for microalgae-based bioremediation of agricultural runoff at full-scale. Algal Res. 2020;47: 101859. This is a good research paper. It reports the performance of algae based bioremediation for one year in environmentally relevant conditions.
Mhatre-Naik A, Pillai G, Savvashe P, Navale M, Palkar J, Lali AM, Pandit R. Developing efficient nutrient removal and resource recovery strategy towards synergistic MLW treatment using macroalgae in a flat panel photobioreactor. Sustainable Energy Technol Assess. 2021;47: 101475.
Neveux N, Magnusson M, Mata L, Whelan A, De Nys R, Paul NA. The treatment of municipal wastewater by the macroalga Oedogonium sp. and its potential for the production of biocrude. Algal Res. 2016;13:284–92.
Kim T, Ren X, Chae K-J. High-rate algal pond coupled with a matrix of Spirogyra sp. for treatment of rural streams with nutrient pollution. J Environ Manage. 2018;213:297–308.
• Lawton RJ, Glasson CR, Novis PM, Sutherland JE, Magnusson ME. Productivity and municipal wastewater nutrient bioremediation performance of new filamentous green macroalgal cultivars. J Appl Phycol. 2021;33(6):4137–48. This is one of a few studies on the filamentous green macroaglae for nutrient removal.
Cole AJ, Paul NA, De Nys R, Roberts DA. Good for sewage treatment and good for agriculture: algal based compost and biochar. J Environ Manage. 2017;200:105–13. This study provides the benefits of algae biomass biochar and biocomposed.
NGER. National Greenhouse and Energy Reporting Scheme Measurement - Technical Guidelines for the estimation of emissions by facilities in Australia (August 2016). , (Ed.) A.G. Dept. of the Environment and Energy, Canberra, Australia. 2016.
Bao Z, Sun S, Sun D. Characteristics of direct CO2 emissions in four full-scale wastewater treatment plants. Desalin Water Treat. 2015;54(4–5):1070–9.
Lu L, Guest JS, Peters CA, Zhu X, Rau GH, Ren ZJ. Wastewater treatment for carbon capture and utilization. Nature Sustainability. 2018;1(12):750–8.
•• Dasan YK, Lam MK, Yusup S, Lim JW, Lee KT. Life cycle evaluation of microalgae biofuels production: effect of cultivation system on energy, carbon emission and cost balance analysis. Sci Total Environ. 2019;688:112–28. This study highlights the life cycle assessment of algae biofuel production.
Sutherland DL, McCauley J, Labeeuw L, Ray P, Kuzhiumparambil U, Hall C, Doblin M, Nguyen LN, Ralph PJ. How microalgal biotechnology can assist with the UN Sustainable Development Goals for natural resource management. Curr Opin Environ Sustain. 2021;3: 100050.
Choi YY, Patel AK, Hong ME, Chang WS, Sim SJ. Microalgae Bioenergy with Carbon Capture and Storage (BECCS): an emerging sustainable bioprocess for reduced CO2 emission and biofuel production. Bioresource Technology Reports. 2019;7: 100270.
• Mofijur M, Ashrafur Rahman SM, Nguyen LN, Mahlia TMI, Nghiem LD. Selection of microalgae strains for sustainable production of aviation biofuel. Bioresour Technol. 2022;345: 126408. This study shows methods to select algae for biofuel production.
Rana MS, Prajapati SK. Stimulating effects of glycerol on the growth, phycoremediation and biofuel potential of Chlorella pyrenoidosa cultivated in wastewater. Environ Technol Innovation. 2021;24: 102082.
•• Dang B-T, Bui X-T, Tran DPH, Hao H, Ngo LD, Nghiem T-K-D, Hoang P-T, Nguyen HH, Nguyen T-K-Q, Vo C, Lin YA, Lin K, Varjani S. Current application of algae derivatives for bioplastic production: A review Bioresour Technol. 2022;(347)126698. This review paper provides role of algae in bioplastic production.
Goswami RK, Mehariya S, Karthikeyan OP, Gupta VK, Verma P. Multifaceted application of microalgal biomass integrated with carbon dioxide reduction and wastewater remediation: A flexible concept for sustainable environment. J Cleaner Product. 2022;130654.
• Naaz F, Bhattacharya A, Pant KK, Malik A. Investigations on energy efficiency of biomethane/biocrude production from pilot scale wastewater grown algal biomass. Appl Energy. 2019;254:113656. This study compares the energy production between biomethane and biocrude production pathways at WWT via algae.
Converti A, Oliveira RPS, Torres BR, Lodi A, Zilli M. Biogas production and valorization by means of a two-step biological process. Bioresour Technol. 2009;100(23):5771–6.
Li P, Pan S-Y, Pei S, Lin YJ, Chiang P-C. Challenges and perspectives on carbon fixation and utilization technologies: an overview. Aerosol and Air Quality Research. 2016;16(6):1327–44.
Zanchetta E, Damergi E, Patel B, Borgmeyer T, Pick H, Pulgarin A, Ludwig C. Algal cellulose, production and potential use in plastics: challenges and opportunities. Algal Res. 2021;56: 102288.
Sayre R. Microalgae: the potential for carbon capture. Bioscience. 2010;60(9):722–7.
Mona S, Malyan SK, Saini N, Deepak B, Pugazhendhi A, Kumar SS. Towards sustainable agriculture with carbon sequestration, and greenhouse gas mitigation using algal biochar. Chemosphere. 2021;275: 129856.
•• Ali M, Masood A, Saleem M. Microalgae cultivation in wastewater for simultaneous nutrients removal and biomass production. Int J Energy Environ Eng. 2021;12(3):475–85. This study provides the nutrient removal rate by algae.
Nguyen LN, Truong MV, Nguyen AQ, Johir MAH, Commault AS, Ralph PJ, Semblante GU, Nghiem LD. A sequential membrane bioreactor followed by a membrane microalgal reactor for nutrient removal and algal biomass production. Environ Sci Water Res Technol. 2020;6(1):189–96.
Xu K, Zou X, Xue Y, Qu Y, Li Y. The impact of seasonal variations about temperature and photoperiod on the treatment of municipal wastewater by algae-bacteria system in lab-scale. Algal Res. 2021;54: 102175.
Park JBK, Craggs RJ, Shilton AN. Enhancing biomass energy yield from pilot-scale high rate algal ponds with recycling. Water Res. 2013;47(13):4422–32.
Parra-Saldivar R, Bilal M, Iqbal HM. Life cycle assessment in wastewater treatment technology. Current Opinion in Environmental Science & Health. 2020;13:80–4.
• Okurowska K, Karunakaran E, Al-Farttoosy A, Couto N, Pandhal J. Adapting the algal microbiome for growth on domestic landfill leachate. Biores Technol. 2021;319: 124246. This study shows the potential of algae for landfil leachate treatment.
• Vu HP, Nguyen LN, Vu MT, Labeeuw L, Emmerton B, Commault AS, Ralph PJ, Mahlia TMI, Nghiem LD. Harvesting Porphyridium purpureum using polyacrylamide polymers and alkaline bases and their impact on biomass quality. Sci Total Environ. 2021;755:142412. This research paper shows the difference on harvesting efficiency by polymer and pH addition. It focus on the biomass quality and potential application for both freash and marine algae cultivation.
• Nguyen LN, Labeeuw L, Commault AS, Emmerton B, Ralph PJ, Johir MAH, Guo W, Ngo HH, Nghiem LD. Validation of a cationic polyacrylamide flocculant for the harvesting fresh and seawater microalgal biomass. Environ Technol Innovation. 2019;16:100466. This research paper validates the performance of cationic polymer on algae flocculation efficiency.
Acknowledgements
Luong N. Nguyen acknowledges the University of Technology Sydney for the Vice Chancellor’s Postdoctoral Research Fellowship.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Water Pollution.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nguyen, L.N., Aditya, L., Vu, H.P. et al. Nutrient Removal by Algae-Based Wastewater Treatment. Curr Pollution Rep 8, 369–383 (2022). https://doi.org/10.1007/s40726-022-00230-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40726-022-00230-x