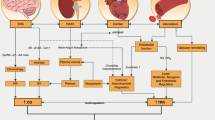
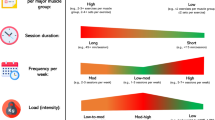
Abstract
Objectives
This umbrella review aimed to review the effects of resistance training on patients with end-stage renal disease and assess the methodological quality of the available literature.
Methods
An umbrella review and meta-meta-analysis was performed. A systematic search was conducted until May 2022. Article selection, quality assessment, and risk of bias assessment were performed by two independent reviewers. The meta-meta-analyses were performed with a random-effects model and the summary statistics were presented in the form of a forest plot with a weighted compilation of all standardized mean differences and corresponding 95% confidence interval. Twenty-four reviews were eventually included. The protocol was registered in the international registry PROSPERO (CRD42022321702).
Results
Resistance training showed positive effects on functional capacity (g = 0.614), aerobic capacity (g = 0.587), health-related quality of life (g = 0.429), and peak force (g = 0.621). Fifteen of the included studies (63%) presented low risk of bias, and the remaining studies (37%) showed unclear risk of bias.
Conclusion
Resistance training in patients undergoing hemodialysis is an intervention that shows positive results regarding physical and functional outcomes. The quality level of the literature is inconclusive, but the included studies present low risk of bias.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
End-Stage Renal Disease (ESRD) is an increasing worldwide concern that involves significant healthcare costs [1]. Global estimates reveal 8–16% prevalence rates that are progressively growing and will end up presenting a major challenge for healthcare systems [2].
Among the possible treatment options, most patients suffering from ESRD receive hemodialysis [3], a treatment that is often related to several impairments, such as decreased functional capacity, decreased levels of physical activity and increased mortality rates [4]. The most significant predictor of mortality rates in this population is skeletal muscle wasting [5, 6]. This is very common in patients with ESRD due to sedentary behavior [7], acidosis, comorbid illnesses, corticosteroid usage, aging, oxidative stress, insulin resistance, chronic inflammation and problems related to restrictions in dietary protein intake [8]. All these factors lead to loss of muscle strength and the development of functional impairments [7, 9].
To revert and prevent the development of skeletal muscle wasting, exercise has been extensively introduced as a complement to daily dialysis care routines. Exercise modalities include aerobic, resistance and combined programs, which are delivered during dialysis sessions or on non-dialysis days. Among them, resistance training has become a well-established, safe, and effective modality to overcome skeletal muscle wasting [10, 11], being the choice of preference of many authors when implementing exercise sessions during dialysis.
Several resistance training programs [10], such as the “Progressive Exercise for Anabolism in Kidney Disease (PEAK)” or clinical exercise guidelines such as the one developed by The Life Options Rehabilitation Advisory Council (LORAC), “Exercise. A guide for people on dialysis” have also been created to support these implementations [12]. To better understand the benefits and the effects of resistance training in patients with ESRD, several clinical trials, and further systematic reviews and meta-analyses, have been conducted. However, to the best of our knowledge, there is no review that involves the assessment of the overall effect and quality of these studies, which could help to establish guidelines and recommendations when implementing resistance training in these patients. Therefore, our research team conducted an umbrella review based on the evidence found involving resistance training in ESRD patients. This study aims to provide an overview of the benefits of resistance training in patients suffering from ESRD and an assessment of the quality of the published literature.
Methods
Design
A systematic review of reviews was conducted in accordance with the Preferred Reporting Items for Overviews of Systematic Reviews including harm checklist (PRIO-harms), which consists of 27 items and 56 sub-items, followed by a 5-stage process flow diagram [13]. The protocol was registered in the international registry PROSPERO (CRD42022321702).
Inclusion criteria
The criteria used in this search involved methodological and clinical outcome factors, including population, intervention, comparison, and study type (PICOS) criteria [14].
Participants (population) included in this search were adult (> 18 years old) patients that suffered from CKD who were undergoing maintenance hemodialysis. The trial (intervention) had to include either a resistance exercise training program or a combined exercise program that included resistance exercises. Comparator groups (control) received no-exercise, sham, or placebo exercise interventions. Assessments (outcomes) involved functional capacity assessed through the 6-Minute Walk Test (6MWT), aerobic capacity assessed through maximum oxygen intake (VO2max), peak force assessed through dynamometry, health-related quality of life (HRQOL) assessed through the Short-Form 36 (SF-36) questionnaire, depression symptoms assessed through the Beck Depression Inventory (BDI), and changes in systolic and diastolic blood pressures. Papers included systematic reviews with a meta-analysis of randomized controlled, randomized uncontrolled or controlled clinical trials.
Search strategy
A systematic search was carried out in the following scientific databases for related papers published by May 27, 2022: MEDLINE (Pubmed), The Cochrane Library (CENTRAL), EMBASE, CINAHL, PEDro and Google Scholar. The search strategy combined medical subject headings (MeSH) through Boolean operators “OR” and/or “AND”. The following headings were applied: “hemodialysis”, “renal dialysis”, “dialysis” “kidney failure, chronic”, “renal insufficiency, chronic”, “strength training” and “resistance training”.
Two independent researchers (BPD and LSM) carried out the search strategy following the same methods, and differences were resolved by consensus, mediated through a third researcher (JCG). Researchers manually searched journals that had published related papers as well as the reference lists of the included papers. Duplicates were hand-checked manually before being removed.
Selection criteria and data extraction
Two independent researchers conducted a data analysis to assess the relevance of the reviews according to the search question. Initially, this analysis was carried out by extracting information regarding the title, abstract and keywords of the systematic reviews. If there was no consensus between the researchers or if the title and abstract contained insufficient information, the full text was reviewed. After this first analysis, the researchers screened the reviews to assess whether they met the inclusion criteria. Differences between researchers were resolved through discussion and mediation by a third researcher.
The extraction protocol was based on the Cochrane Review Groups: (1) study characteristics, including author, title, and year of publication, (2) review information, including sample size, patients, and types of trials, (3) description of the intervention and control, including types of exercise, parameters of frequency, intensity and time, and study outcomes such as functional capacity, aerobic capacity, strength, health-related quality of life, depression symptoms, and blood markers.
Risk of bias analysis
Two independent researchers (BPD and LSM) assessed the quality of the included reviews by analyzing their risk of bias through the Risk of Bias in Systematic Reviews (ROBIS) tool [15]. This tool assesses quality across four domains: (1) study eligibility, (2) study identification and selection, (3) data collection and study appraisal and (4) synthesis of findings. ROBIS provides an overall assessment of high, low, or unclear risk of bias. Both researchers assessed the quality of the systematic reviews using the same method. Disagreements between the researchers were resolved by consensus mediated by a third researcher. Interrater reliability was determined using the kappa coefficient (κ), using κ > 0.7 to indicate a high level of agreement; κ = 0.5–0.7 to indicate a moderate level of agreement; and κ < 0.5 to indicate a low level of agreement [16].
Statistical analysis
Statistical analysis was performed using the “metaumbrella” app and its associated package in R, which is a tool that allows users to perform umbrella reviews with stratification of evidence [17]. Meta-analytical procedures were carried out following recommendations on how to conduct umbrella reviews [18]. We used the same inclusion criteria for the systematic review and meta-analysis but added two criteria: (1) The Results section contained detailed information on the comparative statistical data (mean, standard deviation, and/or 95% confidence interval [CI]) of the main variables, and (2) data for the analyzed variables were represented in at least three meta-analyses. Common effect sizes were estimated using standardized mean differences (SMDs), assessed through Hedge’s g to compare continuous variables, and their 95% confidence intervals using a random-effects model. The heterogeneity of each meta-analysis was estimated using the I-squared statistic (I2), establishing an I2 of 25% to represent a small degree of heterogeneity, 50% a moderate degree and 75% a large degree [19]. To obtain a pooled estimate of the effect in the meta-analysis of the heterogeneous studies, we performed a random-effects model, as described by DerSimonian and Laird [20]. The estimated SMDs were interpreted as described by Hopkins et al. an SMD of 4.0 was considered to represent an extremely large clinical effect; 2.0–4.0 a very large effect; 1.2–2.0 a large effect; 0.6–1.2 a moderate effect; 0.2–0.6 a small effect; and 0.0–0.2 a trivial effect [21].
Overall strength of the evidence
Strength of the evidence across the systematic reviews was assessed using the Physical Activity Guidelines Advisory Committee (PAGAC) [22]. For the PAGAC analysis, the findings were evaluated according to five criteria: (1) applicability of the study sample, exposures, and outcomes to the research question; (2) generalizability to the population of interest; (3) risk of bias or study limitations; (4) quantity and consistency of findings across studies; and (5) magnitude and precision of the effect. The strength of the evidence was classified according to the PAGAC as strong, moderate, limited or not assignable.
Overlapping analysis
To assess the overlap of primary studies among systematic reviews the GROOVE (Graphical Representation of Overlap for OVErviews) methodological approach was employed. This tool is intended to be used mainly by authors of overviews of systematic reviews. The tool was made in Microsoft Excel, which we think could boost its wide usage [23].
Results
Figure 1 presents the PRISMA flow diagram, showing the different stages of the review process, including reasons for exclusion. One hundred fifteen initial studies were identified through databases and nine additional studies were identified through other sources. After removing duplicates and eliminating studies that the authors were unable to retrieve, 88 studies were screened for eligibility. Studies were excluded if they were not related to a population of patients suffering from ESRD that underwent treatment with dialysis or if the intervention did not include a form of resistance training. After excluding all the papers that did not meet the criteria, 24 studies were finally included in the analysis.
Characteristics of the included studies
Table 1 reports the characteristics of the studies included in the umbrella analysis (n = 24). Studies were published between 2010 and 2021 and included a total of 13,415 participants. Participants included in the studies were adults (> 18 years) suffering from ESRD under maintenance dialysis, who received an intervention of either a resistance training program or a combined training program that included resistance exercises. Specific details of the included interventions are summarized in Table 2. Exercise interventions lasted from 4 weeks to 1 year, exercise was mostly performed 2–3 times per week and lasted from 10 min to 2 h. Exercise intensity was mainly assessed with Borg’s Rating of Perceived Exertion scale. Control or comparison groups were defined as patient’s receiving regular dialysis treatment, considered to be usual care, or placebo or sham exercise, that consisted in stretching exercises or passive range of movement exercises.
Methodological quality analysis
To assess the level of risk of bias, two independent reviewers used the ROBIS tool, and discrepancies were resolved by discussion and consensus. Figure 2 shows a summary plot graph summarizing the results obtained for each of the included reviews. Overall results showed that most of the studies had low risk of bias (67.5%). The risk of bias in the remaining studies was unclear (25%) or high (7.5%). The level of agreement between the reviewers in the assessment was high (κ = 0.820).
Main findings
Functional capacity
Nine studies (38%) [25, 26, 29, 30, 32, 36, 37, 41, 45] including 2290 participants reported effects of resistance training on functional capacity. Functional capacity was assessed in every study through the 6MWT. Overall effect size of resistance training with respect to functional capacity showed statistically significant differences with a moderate clinical effect (SMD = 0.614, CI 95% 0.433–0.795), and heterogeneity among the studies was high (I2 = 74.43%) (Fig. 3). The certainty of evidence was moderate, showing that resistance training increases functional capacity in patients with ESRD (Table 3).
Aerobic capacity
Twelve studies (50%) [24, 26, 28, 30, 32, 33, 35, 37, 39, 40, 42, 43] including 4037 participants reported the effects of resistance training on aerobic capacity. Every included study assessed aerobic capacity through the VO2max. Overall effect size of resistance training with respect to aerobic capacity showed statistically significant results with small clinical effects (SMD = 0.587, CI 95% 0.328–0.845) and high heterogeneity among the studies (I2 = 89.82%) (Fig. 3). The certainty of evidence was moderate, showing that resistance training increases aerobic capacity in patients with ESRD (Table 3).
Peak force
Six studies (25%) [27, 32, 33, 36, 37, 41] including 2160 participants reported the effects of resistance training on peak force. Every included study assessed peak force through dynamometry. Overall effect size of resistance training with respect to muscle strength showed statistically significant differences with moderate clinical effects (SMD = 0.621, CI 95% 0.181–1.061) and heterogeneity among the studies was very high (I2 = 90.752%) (Fig. 3). The certainty of evidence was moderate, showing that resistance training increases muscle strength in patients with ESRD (Table 3).
Depression
Five studies (21%) [25, 28, 31, 32, 39] including 960 participants reported the effects of resistance training on depression symptoms. Depression was assessed through the Beck Depression Inventory. Overall effect size of resistance training with respect to depression did not show statistically significant differences (SMD = −0.759, CI 95% −1.545–0.027) and heterogeneity among the studies was high (I2 = 93.75%) (Fig. 3). The certainty of evidence was moderate, showing that resistance training does not decrease depression in patients with ESRD (Table 3).
Health-related quality of life
Ten studies (42%) [25, 27, 28, 32, 34, 37,38,39, 42, 43] including 2481 participants assessed the effects of resistance training on HRQOL, which was assessed using the Short-Form 36 questionnaire (SF-36), specifically with its Physical Component Scale. Overall effect size of resistance training with respect to HRQOL showed statistically significant differences with small clinical effects (SMD = 0.429, CI 95% 0.326–0.532) and small levels of heterogeneity among the studies (I2 = 36.09%) (Fig. 3). The certainty of evidence was moderate, showing that resistance training increases HRQOL in patients with ESRD (Table 3).
Systolic and diastolic blood pressures
Four studies (17%) [30, 33, 39, 40] including 1067 participants reported effects of resistance training on systolic blood pressure and diastolic blood pressure. Changes were assessed by analyzing the change in values in mmHg for both systolic and diastolic blood pressures. Overall effect size of resistance training with respect to systolic blood pressure did not show statistically significant differences (SMD = −0.173, CI 95% −0.512–0.167) and heterogeneity among the studies was high (I2 = 82.61%). Overall effect size of resistance training with respect to diastolic blood pressure did not show statistically significant differences (SMD = −0.377, CI 95% −0.902–0.147) and heterogeneity among the studies was high (I2 = 88.17%) (Fig. 3). The certainty of evidence was moderate, showing that resistance training does not influence systolic and diastolic blood pressures in patients with ESRD (Table 3).
Overlapping analysis
The overlap analysis performed using the GROOVE tool revealed a total corrected overlap area of 9.45%, showing a moderate level of overlap. The detail of the overlap is shown in Fig. 4.
In addition, an overlap analysis was performed for each variable. In the 6MWT, a total of nine reviews including 70 different primary studies were included, which represented a corrected covered area of 18.39% (very high overlap) (Fig. 5). Regarding VO2max, 12 reviews were included with 165 different primary studies, representing a corrected covered area of 19.21% (very high overlap) (Fig. 6). Similar results were found in peak force outcome, including a total of six reviews, 51 primary studies and a corrected covered area of 16.08% (very high overlap) (Fig. 7). In relation to HRQOL, a total of 10 reviews and 63 different primary studies were included, representing a corrected covered area of 15.34% (very high overlap) (Fig. 8). Regarding the depression variable, five reviews and 47 primary studies were included, representing a corrected covered area of 18.62% (very high overlap) (Fig. 9). Finally, in SBP and DBP, four reviews and 39 primary studies were included, representing a corrected covered area of 19.66% (very high overlap) (Fig. 10).
Discussion
This umbrella review analyzed the benefits and the effects of resistance training in patients with ESRD undergoing dialysis and assessed the quality of the published literature. Multiple outcomes that relate to patient’s overall well-being were analyzed, and positive effects were found in functional capacity, aerobic capacity, peak force and HRQOL. Twenty-four studies were eventually included in the review, presenting a substantial quantity of literature evidence that outlines the benefits of resistance training in patients undergoing dialysis.
Physical factors, such as functional capacity, aerobic capacity, and peak force, as well as psychosocial factors, such as HRQOL, showed positive results, reinforcing the idea that in this usually sedentary population, exercise interventions have an effect on several dimensions that intervene in the biopsychosocial well-being of these patients. Nonetheless, some of them showed small clinical effects, but still had a considerable margin of improvement, so developing adapted exercise interventions that are controlled and that follow a specific protocol for patients on dialysis could potentially result in even greater improvements.
Both functional and aerobic capacities, assessed through the 6MWT and VO2max, respectively, showed positive results. These assessments are widely used in the literature and are closely correlated [36], therefore it is logical to assume that an improvement in one will potentially generate improvement in the other. Because functional capacity showed bigger differences than aerobic capacity, the inclusion of aerobic exercise interventions would be relevant to more specifically improve this outcome [46].
HRQOL also showed positive effects, albeit with a small clinical effect. Functional impairment also contributes to impaired HRQOL [27], therefore, an improvement in functional outcomes could also imply an improvement in HRQOL, as shown in this review. Even though there is no standard definition of HRQOL, it is widely accepted that it includes physical, psychological and social domains of life [34], and therefore an improvement in this outcome could be more limited solely by performing exercise.
Significant differences were found regarding muscle peak force, with a moderate effect. The moderate effect might be due to insufficient stimulus in the training programs, that is unable to produce enough improvement. Results presenting smaller effects could require more complete programs that include sufficient exercise intensity in the interventions, together with adequate exercise volume (quantity of exercise measured in repetitions and sets), because of the overcautious approach when applying exercise in a frail population.
Additionally, these results might be influenced by other factors involved in muscle performance, such as dietary restrictions and metabolic inflammation. Even though resistance training is considered a potent insulin-like growth factor inducer [36], restrictions in protein intake, along with enhanced proteinuria, limit the synthesis of proteins in patients with ESRD, especially in those with associated prevalent metabolic comorbidities, such as diabetes mellitus [47]. Also, the associated inflammatory environment could create abnormalities in muscle fibers due to cellular adaptations, including enzymatic and contractile protein changes, also limiting protein synthesis [41] and impairing muscle performance.
No statistically significant effects were found in depression outcomes. This might be due to the biopsychosocial nature of depression, and the necessity to deal with this problem from a more multidisciplinary approach, including psychotherapy, for instance. Also, results in this analysis show high variability, meaning that, even though depression has high prevalence rates in this population [31, 32], patients on dialysis might not suffer from it, hence not perceiving the benefits of resistance training in this outcome.
No statistically significant changes were found in either systolic or diastolic blood pressure levels. Our results are consistent with those found by Heiwe et al. [33]. Even though intradialytic exercise interventions have been proposed to improve blood pressure levels [30, 39], these results should be cautiously interpreted because these reviews do not isolate different types of exercise interventions, so their conclusions are not strictly based on resistance training interventions alone. Also, resistance training protocols that elicit the most favorable blood pressure level benefits remain elusive, and populations taking antihypertensive medications, such as patients with ESRD, show smaller reductions in blood pressure levels [48]. In patients on hemodialysis, hypertension is closely related to overload, caused by retention due both to the dialytic treatment and the disease’s pathophysiology (uremic state, anemia, electrolyte imbalance) [24]. The authors of some studies even suggest that fluid overload (> 5% above dialysis body weight) must be considered an exclusion criterion, as they deem it makes patients unable to exercise during dialysis sessions [25].
Risk of bias was assessed with the ROBIS tool, a domain-based resource supported by signaling questions. Results from this analysis showed that most studies (63%) included had low risk of bias levels. However, it is important to outline that the ROBIS tool is not a fully objective assessment resource, and some of the items that the tool includes depend on the examiner’s interpretation, so careful conclusions must be extracted from this analysis.
The overlap analysis performed using the GROOVE tool showed a total moderate level of overlap. However, when analyzing the overlap for each individual outcome, high overlapping levels were observed in every outcome (6MWT (18.39% of overlap), VO2max (19.21% of overlap), peak force (16.08% of overlap), HRQOL (15.34% of overlap), Depression (18.62% of overlap), and SBP and DBP (19.66% of overlap)). When conducting clinical trials implementing exercise interventions in participants with ESRD, throughout the literature it is common to assess the same outcomes through the same assessment methods, and this increases the chances that the same clinical trial will be included in several reviews. This could potentially create an overpowering of the results in a specific outcome, therefore creating a bias that might lead to false assumptions about the results. However, overlap can also be considered an advantage as it simplifies the meta-reviewer’s work and reinforces the conclusions that can be extracted [49]. The assessment of study overlapping is important, as systematic reviews must have well-defined search strategies and should be conducted only when necessary, avoiding repetitive reviews that might lead to unreliable and overpowered clinical results.
Our study shows several strengths. There is a considerable number of included studies, and these show appropriate methodological quality levels. Studies included in the review were consistent with the assessment method choice when analyzing every outcome of this review (functional capacity was assessed with the 6MWT, aerobic capacity with the VO2max and so on), facilitating further analysis. Finally, even though resistance training programs were inconsistent due to the lack of following a standardized developed protocol, the included exercise programs were able to show positive results, implying that even more positive results can be potentially achieved if proper strength training protocols are followed.
Finally, this review also presents limitations. Even though the research was conducted in several scientific databases and other resources were also consulted, not every existing scientific database was accessed, meaning that not every study assessing resistance training in patients undergoing dialysis might have been included. Additionally, clinical characteristics of the participants enrolled in the exercise programs, detailed descriptions of the exercise programs used, and adherence rates were not thoroughly reported or should be carefully interpreted, and this may have influenced the interpretation of the results. For instance, even though we believe that this does not have a significant effect on the results, one of the studies included in this review [27] considered a study in pre-dialysis patients.
We believe that the meta-analytic results in this study reinforce the idea of recommending resistance training exercise interventions as part of the daily management of patients undergoing hemodialysis, due to the effectiveness in improving biopsychosocial dimensions of this disease. Future studies should focus on thoroughly reporting exercise parameters to determine the optimal loading.
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
KDIGO 2012 (2013) Clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int Suppl 3(1):73–90
Kramer A, Pippias M, Noordzij M et al (2018) The European renal association—European dialysis and transplant association (ERA-EDTA) registry annual report 2015: a summary. Clin Kidney J 11(1):108–122
Jha V, Garcia-Garcia G, Iseki K et al (2013) Chronic kidney disease: global dimension and perspectives. Lancet 382(9888):260–272
Saran R, Robinson B, Abbott KC et al (2018) US renal data system 2017 annual data report: epidemiology of kidney disease in the United States. Am J Kidney Dis 71(3):A7
Bündchen DC, Sousa H, Afreixo V, Frontini R, Ribeiro O, Figueiredo D, Costa E (2021) Intradialytic exercise in end-stage renal disease: an umbrella review of systematic reviews and/or meta-analytical studies. Clin Rehabil 35(6):812–828. https://doi.org/10.1177/0269215520986784. (Epub 2021 Feb 3 PMID: 33530715)
Castaneda C (2002) Muscle wasting and protein metabolism. J Anim Sci 80(Suppl. 2):E98–E105
Desmeules S, Levesque R, Jaussent I, Leray-Moragues H, Chalabi L, Canaud B (2004) Creatine index and lean body are excellent predictors of long-term survival in haemodiafiltration patients. Nephrol Dial Transplant 19:1182–1189
Johansen KL, Chertow GM, Ng AV et al (2000) Physical activity levels in patients on hemodialysis and healthy sedentary controls. Kidney Int 57:2564–2570
Caso G, Garlick P (2005) Control of muscle protein kinetics by acidbase balance. Curr Opin Clin Nutr Metab Care 8(1):73–76
John SG, Sigrist MK, Taal MW et al (2013) Natural history of skeletal muscle mass changes in chronic kidney disease stage 4 and 5 patients: an observational study. PLoS ONE 8(5):e65372
Johansen KL, Shubert T, Doyle J et al (2003) Muscular atrophy in patients receiving hemodialysis: effects on muscle strength, muscle quality, and physical function. Kidney Int 63:291–297
Cheema BS, O’Sullivan AJ, Chan M, Patwardhan A, Kelly J, Gillin A, Fiatarone Singh MA (2006) Progressive resistance training during hemodialysis: rationale and method of a randomized-controlled trial. Hemodial Int 10(3):303–310. https://doi.org/10.1111/j.1542-4758.2006.00112.x. (PMID: 16805893)
Chan M, Cheema B, Fiatarone Singh M (2007) Progressive resistance training and nutrition in renal failure. J Ren Nutr 17:84–87
Cheema B, Abas H, Smith B, O’Sullivan A, Chan M, Patwardhan A, Kelly J, Gillin A, Pang G, Lloyd B, Singh MF (2007) Progressive exercise for anabolism in kidney disease (PEAK): a randomized, controlled trial of resistance training during hemodialysis. J Am Soc Nephrol 18(5):1594–1601. https://doi.org/10.1681/ASN.2006121329. (Epub 2007 Apr 4 PMID: 17409306)
Painter PL (2004) Exercise for dialysis patients. In: Hörl WH, Koch KM, Lindsay RM, Ronco C, Winchester JF (eds) Replacement of renal function by dialysis. Springer, Dordrecht
Liberati A, Altman DG, Tetzlaff J et al (2009) The PRISMA statement for reporting systematic reviews and metanalyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol 6(10):e1–e34
Stone PW (2002) Popping the (PICO) question in research and evidence-based practice. Appl Nurs Res 15(3):197–198
Whiting P, Savović J, Higgins JPT et al (2016) ROBIS: a new tool to assess risk of bias in systematic reviews was developed. J Clin Epidemiol 69:225–234. https://doi.org/10.1016/j.jclinepi.2015.06.005
McHugh ML (2012) Interrater reliability: the kappa statistic. Biochem Medica 22(3):276–282. https://doi.org/10.11613/bm.2012.031
Fusar-Poli P, Radua J (2018) Ten simple rules for conducting umbrella reviews. Evid Based Ment Health 21(3):95–100. https://doi.org/10.1136/ebmental-2018-300014. (Epub 2018 Jul 13 PMID: 30006442)
Cohen J (1988) Statistical power analysis for the behavioral sciences, 2nd edn. Routledge, Milton Park
Higgins JP, Green S (2011) Cochrane handbook for systematic reviews of interventions. Version 5.1.0. The Cochrane Collaboration, London
Pérez-Bracchiglione J, Meza N, Bangdiwala SI et al (2022) Graphical Representation of overlap for overviews: GROOVE tool. Res Syn Methods 13:381–388. https://doi.org/10.1002/jrsm.1557
Andrade FP, de Rezende P, de Ferreira T, Borba GC, Müller AM, Rovedder PM (2019) Effects of intradialytic exercise on cardiopulmonary capacity in chronic kidney disease: systematic review and meta-analysis of randomized clinical trials. Sci Rep. https://doi.org/10.1038/s41598-019-54953-x
Bernier-Jean A, Beruni NA, Bondonno NP, Williams G, Teixeira-Pinto A, Craig JC, Wong G (2022) Exercise training for adults undergoing maintenance dialysis. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.cd014653
Bogataj P, Pajek M, Pajek J, ButurovićPonikvar J, Paravlic AH (2019) Exercise-based interventions in hemodialysis patients: a systematic review with a meta-analysis of randomized controlled trials. J Clin Med 9(1):43. https://doi.org/10.3390/jcm9010043
Cheema BS, Chan D, Fahey P, Atlantis E (2014) Effect of progressive resistance training on measures of skeletal muscle hypertrophy, muscular strength, and health-related quality of life in patients with chronic kidney disease: a systematic review and meta-analysis. Sports Med 44(8):1125–1138. https://doi.org/10.1007/s40279-014-0176-8
Chung YC, Yeh ML, Liu YM (2017) Effects of intradialytic exercise on the physical function, depression, and quality of life for haemodialysis patients: a systematic review and meta-analysis of randomised controlled trials. J Clin Nurs 26(13–14):1801–1813. https://doi.org/10.1111/jocn.13514
Clarkson MJ, Bennett PN, Fraser SF, Warmington SA (2019) Exercise interventions for improving objective physical function in patients with end-stage kidney disease on dialysis: a systematic review and meta-analysis. Am J Physiol Renal Physiol 316(5):F856–F872. https://doi.org/10.1152/ajprenal.00317.2018
Ferrari F, Helal L, Dipp T, Soares D, Soldatelli N, Mills AL, Paz C, Tenório MCC, Motta MT, Barcellos FC, Stein R (2019) Intradialytic training in patients with end-stage renal disease: a systematic review and meta-analysis of randomized clinical trials assessing the effects of five different training interventions. J Nephrol 33(2):251–266. https://doi.org/10.1007/s40620-019-00687-y
Ferreira TL, Ribeiro HS, Ribeiro ALA, Bonini-Rocha AC, Lucena JMS, de Oliveira PA, Amorim FRS, Ferreira AP, Magno LAV, Martins WR (2020) Exercise interventions improve depression and anxiety in chronic kidney disease patients: a systematic review and meta-analysis. Int Urol Nephrol 53(5):925–933. https://doi.org/10.1007/s11255-020-02612-w
Gomes Neto M, de Lacerda FFR, Lopes AA, Martinez BP, Saquetto MB (2018) Intradialytic exercise training modalities on physical functioning and health-related quality of life in patients undergoing maintenance hemodialysis: systematic review and meta-analysis. Clin Rehabil 32(9):1189–1202. https://doi.org/10.1177/0269215518760380
Heiwe S, Jacobson SH (2014) Exercise training in adults with CKD: a systematic review and meta-analysis. Am J Kidney Dis 64(3):383–393. https://doi.org/10.1053/j.ajkd.2014.03.020
Hu H, Liu X, Chau PH, Choi EPH (2021) Effects of intradialytic exercise on health-related quality of life in patients undergoing maintenance haemodialysis: a systematic review and meta-analysis. Qual Life Res 31(7):1915–1932. https://doi.org/10.1007/s11136-021-03025-7
Huang M, Lv A, Wang J, Xu N, Ma G, Zhai Z, Zhang B, Gao J, Ni C (2019) Exercise training and outcomes in hemodialysis patients: systematic review and meta-analysis. Am J Nephrol 50(4):240–254. https://doi.org/10.1159/000502447
Lu Y, Wang Y, Lu Q (2019) Effects of exercise on muscle fitness in dialysis patients: a systematic review and meta-analysis. Am J Nephrol 50(4):291–302. https://doi.org/10.1159/000502635
Matsuzawa R, Hoshi K, Yoneki K, Harada M, Watanabe T, Shimoda T, Yamamoto S, Matsunaga A (2017) Exercise training in elderly people undergoing hemodialysis: a systematic review and meta-analysis. Kidney Int Rep 2(6):1096–1110. https://doi.org/10.1016/j.ekir.2017.06.008
Molsted S, Bjørkman ASD, Lundstrøm LH (2019) Effects of strength training to patients undergoing dialysis: a systematic review. Dan Med J 66(1):A5526 (PMID: 30573007)
Pu J, Jiang Z, Wu W, Li L, Zhang L, Li Y, Liu Q, Ou S (2019) Efficacy and safety of intradialytic exercise in haemodialysis patients: a systematic review and meta-analysis. BMJ Open 9(1):e020633. https://doi.org/10.1136/bmjopen-2017-020633
Scapini KB, Bohlke M, Moraes OA, Rodrigues CG, Inácio JF, Sbruzzi G, Leguisamo CP, Sanches IC, Tourinho Filho H, Irigoyen MC (2019) Combined training is the most effective training modality to improve aerobic capacity and blood pressure control in people requiring haemodialysis for end-stage renal disease: systematic review and network meta-analysis. J Physiother 65(1):4–15. https://doi.org/10.1016/j.jphys.2018.11.008
Schardong J, Stein C, dellaMéaPlentz R (2020) Neuromuscular electrical stimulation in chronic kidney failure: a systematic review and meta-analysis. Arch Phys Med Rehabil 101(4):700–711. https://doi.org/10.1016/j.apmr.2019.11.008
Segura-Ortí E (2010) Ejercicio en pacientes en hemodiálisis: revisión sistemática de la literatura. Nefrología (Madrid) 30(2):236–246
Sheng K, Zhang P, Chen L, Cheng J, Wu C, Chen J (2014) Intradialytic exercise in hemodialysis patients: a systematic review and meta-analysis. Am J Nephrol 40(5):478–490. https://doi.org/10.1159/000368722
Wu X, Yang L, Wang Y, Wang C, Hu R, Wu Y (2020) Effects of combined aerobic and resistance exercise on renal function in adult patients with chronic kidney disease: a systematic review and meta-analysis. Clin Rehabil 34(7):851–865. https://doi.org/10.1177/0269215520924459
Zhang F, Zhou W, Sun Q, Zhai Y, Zhang Y, Su H, Wang Z (2019) Effects of intradialytic resistance exercises on physical performance, nutrient intake, and quality of life among haemodialysis people: a systematic review and meta-analysis. Nurs Open 8(2):529–538. https://doi.org/10.1002/nop2.274
Sawant A, House AA, Overend TJ (2014) Anabolic effect of exercise training in people with end-stage renal disease on hemodialysis: a systematic review with meta-analysis. Physiother Can 66(1):44–53. https://doi.org/10.3138/ptc.2012-59
van Huffel L, Tomson CRV, Ruige J, Nistor I, van Biesen W, Bolignano D (2014) Dietary restriction and exercise for diabetic patients with chronic kidney disease: a systematic review. PLoS ONE 9(11):e113667. https://doi.org/10.1371/journal.pone.0113667
Macdonald HV, Johnson BT, Huedo-Medina TB et al (2016) Dynamic resistance training as stand-alone antihypertensive lifestyle therapy: a meta-analysis. JAHA. https://doi.org/10.1161/jaha.116.003231
Hennessy EA, Johnson BT (2019) Examining overlap of included studies in meta-reviews: guidance for using the corrected covered area index. Res Synth Methods 11(1):134–145. https://doi.org/10.1002/jrsm.1390
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
Ethical approval was not required as this umbrella review is a synthesis and analysis of existing studies.
Human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
For this type of study formal consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Perez-Dominguez, B., Suso-Marti, L., Dominguez-Navarro, F. et al. Effects of resistance training on patients with End-Stage Renal Disease: an umbrella review with meta-analysis of the pooled findings. J Nephrol 36, 1805–1839 (2023). https://doi.org/10.1007/s40620-023-01635-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-023-01635-7