Abstract
Purpose
Five strategies were recommended by the American Association of Clinical Endocrinologists/American College of Endocrinology (AACE/ACE) guidelines for the treatment of postmenopausal osteoporosis (PMO) patients with a very high fracture risk. We aimed to assess their cost-effectiveness in the United States (US).
Methods
A microsimulation Markov model was created to compare the cost-effectiveness of five treatment strategies, including zoledronate, denosumab, abaloparatide, teriparatide, and romosozumab in PMO patients with a recent fracture from the healthcare perspective of the US. The data used in the model were obtained from published studies or online resources. Base-case analysis, one-way deterministic sensitivity analysis (DSA) and probability sensitivity analysis (PSA) were conducted for 65-, 70-, 75-, and 80-year-old patients.
Results
In base case, at 65 years, zoledronate was the cheapest strategy. The incremental cost-effectiveness ratios (ICER, which represent incremental costs per QALY gained) of denosumab, teriparatide, abaloparatide, and romosozumab against zoledronate were $13,020/QALY (quality-adjusted years), $477,331 /QALY, $176,287/QALY, and $98,953/QALY, respectively. Under a willing-to-pay (WTP, which means the highest price a consumer will pay for one unit of a good of service) threshold of $150,000/QALY, denosumab and romosozumab were cost-effective against zoledronate. The PSA results showed that denosumab was the most cost-effective option with WTP thresholds of $50,000/QALY, $100,000/QALY and $150,000/QALY. The results were similar in other age groups. The DSA results indicated that the most common parameters that have important influence on the outcome were drug persistence, incidence of adverse events, the efficacy of drugs on hip fractures and the cost of the drug.
Conclusion and relevance
Among PMO patients with a very high fracture risk in the US, zoledronate is the cheapest strategy and denosumab is the most cost-effective choice among these five strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporosis is a disease characterized by low bone mass and microarchitectural deterioration of bone [1], which has become a significant public health issue. There are about 6–11% and 32–46% adults, age 50 years and older, in the US who have, respectively, osteoporosis or low bone mass [2], and it is estimated that more than 3 million osteoporotic fractures will occur in 2025, causing more than $25 billion in Medicare costs [3].
In 2020, the AACE/ACE recommended that the treatment of osteoporosis should be stratified by the patient’s fracture risk. For patients with a very high fracture risk (e.g., patients with a recent fracture, fractures while on approved osteoporosis therapy or multiple fractures), zoledronate, denosumab, abaloparatide, teriparatide, or romosozumab are recommended for initial treatment [4], as these drugs are more effective in increasing bone mineral density (BMD) and reducing the incidence of fractures [5, 6]. As the guidelines did not establish priorities for drug selection, and it is unrealistic to conduct a head-to-head trial that includes all five drugs to compare differences in their efficacy to reduce fracture risk, clinicians may be uncertain when deciding on a patient’s specific treatment regimen.
The objective of this study was to assess the cost-effectiveness of zoledronate, denosumab, teriparatide, abaloparatide, and romosozumab for treating postmenopausal women in the US with osteoporosis and a very high fracture risk. It aimed to provide a reference for selecting drug treatments for this population, and help promote the rational allocation of medical resources to reduce the national financial burden.
We chose patients with recent fractures as a representative group of those with a very high fracture risk, as it is widely recognized that fracture risk decreases gradually over time since the last fracture occurred [7, 8]. Moreover, a number of studies have confirmed the importance of a recent fracture for imminent fracture risk [9, 10]. It is of great individual value to treat patients with osteoporosis who have had a recent fracture and this will help in reducing osteoporotic fracture incidence.
Methods
As osteoporotic fractures are ongoing over time, a microsimulation Markov model was constructed to model the progression of it [11, 12]. This report followed the Consolidated Health Economic Evaluation Reporting Standards (CHEERS) statement [13] and the guidelines for the conduct of economic evaluations of osteoporosis [14]. We adopted a 3% discount rate for both costs and effectiveness, in line with US guidelines for economic evaluations [15].
Target population
The population was postmenopausal women with osteoporosis of various ages (65, 70, 75, 80 years old) who had a recent fracture (including hip, vertebral, wrist or other osteoporotic fracture).
Model structure
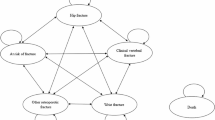
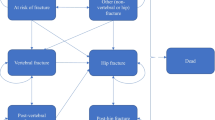
We constructed a microsimulation Markov model (Fig. 1) consisting of six health states: ‘At risk of fracture’, ‘Hip fracture’, ‘Clinical vertebral fracture’, ‘Wrist fracture’, ‘Other osteoporotic fracture’, and ‘Death’ from the US health care perspective. The horizon of the model was lifetime to capture the long-term effect of treatment. Patients were followed from the age they entered the model until 100 years old or death. The cycle length was 6 months [16, 17]. All the patients were supposed to start the cycle with a state based on their fracture site. Every 6 months, the patient would come to a state transition, based on the transition probabilities between states.
Fracture probabilities and mortality
Patients’ fracture risk was modeled based on three factors: the fracture risk of the general population, the relative risk of patients compared with the general population, and the risk reduced by treatment.
The annual incidence rates of hip, vertebral and wrist fractures in the general population were based on data used to adjust the incidence of fractures of the US version of FRAX [18]; other osteoporotic fracture rates were calculated based on other published research [3]. Then, we calculated the incidence of fractures among postmenopausal women with osteoporosis without prior fractures, using the relative risk (RR) described by Mori et al. [19]. The fracture incidence of patients with a prior fracture was calculated according to a Swedish study, which considered the impact of the number of prior fractures and the time since the last fracture on the risk of subsequent fractures at the same time [20]. The RR for any fracture in that study was used for wrist and other osteoporotic fractures here.
Background mortality was obtained from the 2018 US life table [21]. The mortality rate of patients without hip fracture was equal to the background rate of mortality. When a hip fracture occurred, there would be excess mortality during the year of the fracture and subsequent years [22]. As comorbidities play a significant role in excess mortality after a hip fracture [23, 24], we assumed 25% of excess mortality was directly caused by fractures. Increased mortality following clinical vertebral fractures has been found to be very similar than those of a hip fracture [25, 26]. A scenario analysis that also considered excess mortality caused by clinical vertebral fracture was conducted.
Treatment and efficacy
Five strategies, which were recommended by the AACE/ACE guidelines [4], were compared in the model: (a) a 5 mg intravenous injection of zoledronate for 6 years (ZOLE); (b) a 60 mg subcutaneous injection of denosumab every 6 months for 10 years, followed by alendronate, po, weekly for 10 years (DENO/ALN); (c) an 80 μg subcutaneous injection of abaloparatide daily for 2 years, followed by 70 mg of alendronate, po, weekly, for 10 years (ABLA/ALN); (d) a subcutaneous injection of 20 μg of teriparatide daily for 2 years, followed by 70 mg of alendronate, po, weekly for 10 years (TPTD/ALN); and (e) a 210 mg subcutaneous injection of romosozumab monthly for 1 year, followed by 70 mg of alendronate, po, weekly for 10 years (ROMO/ALN).
Treatment efficacy was obtained from a meta-analysis and data used in prior models [26,27,28]. The efficacy of non-vertebral fractures was used for ‘Other osteoporotic fracture’ for all the drugs [14], as well as wrist fractures for zoledronate and romosozumab because other data were not available [16].
After treatment was completed, efficacy decreased linearly to zero over a period of time equal to the length of treatment [19, 29]. As BMD returned to baseline level within 1 years after discontinuation of denosumab [30], the offset time of denosumab was set to 1 years. For DENO/ALN, ABAL/ALN, TPTD/ALN, and ROMO/ALN, we assumed that efficacy of denosumab, teriparatide, abaloparatide, and romosozumab would be maintained during sequential therapy [26, 31, 32], and would start to decline after treatment was completed or the patient dropped out of treatment.
Persistence
Persistence refers to the duration of time from the initiation to the discontinuation of therapy [33]. Persistence data were obtained from published research [34, 35]. Because of a lack of long-term persistence data for zoledronate, denosumab, and alendronate, we assumed the persistence of patients who completed 2 or 5years of treatment would continue until the end of the prescribed treatment.
Adverse events
The adverse events that were considered in the model included gastrointestinal events for alendronate, acute-phase reaction for zoledronate, cellulitis for denosumab and hypercalcemia for both teriparatide and abaloparatide. Other adverse events, such as subtrochanteric femoral fracture, were excluded because of their very low incidence [36], and they were supposed to have little influence on the results. The incidence of adverse events was obtained from published studies [37,38,39].
Costs
Only direct costs were included in the model. The costs in the model were the costs of drugs, fractures and administration and management of adverse events. We obtained drug prices from average sale prices or red book [40, 41].The fracture costs were obtained from published Medicare claims data [42].The costs of managing adverse events were referred to previously published economic analysis models [32, 37, 43]. It was assumed that 12% of patients with a hip fracture required long-term care and the cost directly attributable to fractures was 25% [29]. Patients receiving treatment were assumed to make a physician visit and have a DXA test every year, as guidelines recommend [4]. For the costs of physician visits (Current Procedural Terminology [CPT] codes 99,213) and DXA scans (CPT codes 77,080), we used the allowable charges based on national payment amounts from the 2021 Medicare Physician Fee Schedule [44].A renal function test was required for patients treated with zoledronate ever year [43, 44]. For patients taking alendronate, 0·041 and 0·021 extra physician visits were required during the first 6 months and each subsequent cycle, and a proton pump inhibitor was prescribed at each visit [37].
All costs were reported in US dollars and converted to 2020 US dollars based on the Consumer Price Index (CPI).
Utility
Background utility was equal to that of the health of US women based on the EQ-5D [45]. There was a disutility in the first and subsequent years after hip and vertebral fractures [46], and when a gastrointestinal event [37], acute-phase reaction [47] or cellulitis [37] occurred. The disutility of hip and vertebra fracture were obtained from a systematic review [46]. The disutility of gastrointestinal event [37] and acute-phase reaction [47] were obtained from published economic analysis. Utility of cellulitis were measured using time trade-off methods [37, 48].
For wrist and other osteoporotic fractures, we only assumed a disutility during the first year of fracture [46]. We did not consider a disutility associated with hypercalcemia, as per a previous study [32].
Analysis
Consistent with the recommendations for conducting economic evaluations [14], we conducted base case, one-way deterministic sensitivity analysis and probabilistic sensitivity analysis for each age group, and quality-adjusted life years (QALY) were used as the outcome. Costs and QALY were calculated for each strategy. DSA was conducted on drug, fracture, cost of the administration and management of adverse events, background utility and disutility caused by fracture and adverse events, excess mortality after fractures, discount rate, treatment effects, and persistence. In the DSA, all the variables varied over a plausible range, which were obtained from 95% confidence intervals or a variance of 20% from base-case values. PSA was conducted using distributions to assess the influence of the joint uncertainty surrounding the model variables. Most of the parameters were contained in the PSA. The range and distribution of the variables are presented in Table 1.
The incremental cost-effectiveness ratio (ICER) was calculated from the difference in average costs divided by the difference in the average effectiveness of two strategies. And we used willing-to-pay thresholds of $50,000, $100,000 and $150,000 in PSA.
Analyses were conducted using TreeAge Pro 2021 software (TreeAge, Williamstown, Massachusetts) and R software (version 4·1·0, http://www.r-project.org).
Results
Model validation
The model predicted that 99·5%, 99·5%, 99·4% and 99·3% of people died before age 100 in a starting age of 65, 70, 75 and 80 years old, separately. It was close to the US life table [21]. Consistent with the incidence of new osteoporotic fractures among Medicare beneficiaries in the US [3, 4], for patients with a recent fracture, 17% were hip fractures, 23% were vertebral fractures, 13% were wrist fractures, and 47% were other osteoporotic fractures. According to previous researches, 2.4% and 10.2% of women ≥ 65 years had a subsequent hip fracture within 1 and 5 years, respectively, following their prior clinical fracture regardless the site of the prior fracture. [49] And the 25-year incidence rates of subsequent hip were 23.8% [50]. Our model predicted that without an intervention, the probabilities for 65, 70, 75 and 80 year-old patients to have a hip fracture within 1 year following prior fracture were 1.9%, 2.5%, 4.0% and 4.6%, respectively. And the probability to have a hip fracture within 5 years were 6.3%, 8.0%, 11.4% and 13.3%, respectively. Lifetime probability to have a hip fracture following prior fracture in 65, 70, 75 and 80 year-old patients with recent fracture was 17.80%, 19.80%, 22.40% and 23.50%. Overall, refracture rates predicted by our model were similar to epidemiological data.
Base case analysis
The results of the base-case analysis of 65-, 70-, 75- and 80-year-old patients are shown in Table 2. For the 65-year-old patients, zoledronate had the lowest cost and utility. Compared with zoledronate, the ICER of denosumab/alendronate, romosozumab/alendronate, abaloparatide/alendronate and teriparatide/alendronate were $13,020, $98,953, $176,287 and $477,331 per QALY gains, respectively. Teriparatide/alendronate was absolutely dominated by abaloparatide/alendronate, which means it had lower effectiveness, but higher costs. Extended domination means that the ICER for a given alternative strategy is higher than that of the next more effective alternative. Romosozumab/alendronate exhibited extended domination because, when compared with denosumab/alendronate, it had a higher ICER than abaloparatide/alendronate did. The results were similar in other age groups.
Sensitivity analysis
DSA was performed separately to compare undominated strategies against zoledronate. In 65-year-old patients, for the comparison of denosumab/alendronate against zoledronate (Fig. 2), the parameters that had a significant effect on the outcome were the price of zoledronate, the persistence of denosumab and zoledronate. For the comparison of abaloparatide/alendronate against zoledronate, the parameters that had a significant effect on the outcome were the price of abaloparatide, the efficacy of abaloparatide on hip fractures and persistence of zoledronate. The results for patients in the 70-, 75- and 80-year-old age groups are shown in Online Resource 1 (Figs. 4–6).
The scenario analysis that considered excess mortality caused by hip as well as clinical vertebral fracture showed very similar results to those of base case analysis. For 65-, 70-, 75- and 80-year-old age group, zoledronate had the lowest cost and utility. Teriparatide/alendronate was absolutely dominated by abaloparatide/alendronate. And romosozumab/alendronate exhibited extended domination. (Online Resource 1 Table 6). When dominated strategies were eliminated from the PSA, the results showed, with a 100% probability, that denosumab/alendronate was the most cost-effective strategy at three WTP thresholds ($50,000/QALY, $100,000/QALY and $150,000/QALY) (Fig. 3).
Result of probability sensitivity analysis comparing zoledronate, denosumab/alendronate and abaloparatide/alendronate in 65 (a), 70 (b), 75 (c) and 80 (d) years old. Circle (blue) represents for zoledronate, square(red) represents for denosumab/alendronate and cross (yellow) represents for abaloparatide/alendronate
Discussion
In this study, we evaluated the cost-effectiveness of zoledronate, denosumab, abaloparatide, teriparatide, and romosozumab in the treatment of postmenopausal women with osteoporosis and a recent fracture. The base-case analysis results indicate that zoledronate has the lowest cost and utility in 65-, 70-, 75-, and 80-year-old patients. The cost-effectiveness of strategies when compared with zoledronate differed under different WTP thresholds. Under a WTP threshold of $150,000/QALY, denosumab and romosozumab were cost-effective compared with zoledronate. But when WTP thresholds was lowered to $100,000/QALY or $50,000/QALY, only denosumab was cost-effective compared with zoledronate. The DSA results indicate that drug persistence, incidence of acute-phase reactions, the efficacy of drugs on hip fractures and the cost of the drugs are the most common parameters that have important influences on outcomes. The PSA results indicate that the probability of denosumab being the most cost-effective strategy is 100% in all age groups under different WTP thresholds.
Some previous US studies that analyzed the cost-effectiveness of osteoporosis treatment included the same drugs we investigated in our study. Stuart et al. [51] evaluated the cost-effectiveness of denosumab versus zoledronate, teriparatide and other osteoporotic treatments in 78 year-old men who had a BMD T-score of − 2·12 and a vertebral prevalence of 23%. The results showed that denosumab was dominant over zoledronate and teriparatide. Parthan et al. [37], which evaluated the cost-effectiveness of denosumab versus oral bisphosphonates for postmenopausal osteoporosis, suggested that denosumab was cost-effective or dominant compared with other strategies. A study by Mori and colleagues compared two strategies, including alendronate alone and teriparatide followed by alendronate [29]. The results indicated that for women with osteoporosis who had a previous vertebral fracture, the ICERs of sequential teriparatide/alendronate over alendronate varied from $288,200/QALY to $441,700/QALY from ages 65 to 80, which all exceeded the WTP threshold of $150,000/QALY. Another study that compared the cost-effectiveness of abaloparatide and teriparatide in postmenopausal women with osteoporosis, which used discrete-event simulation [32], showed that abaloparatide was a dominant treatment strategy over teriparatide and yielded an ICER of $18,891/QALY against alendronate.
It should be noted that the results of the cost-effectiveness analysis are affected by the choice of target population and the strategies being compared, as well as the model used in the analyses. Hence, our findings cannot be directly compared with the findings of previous studies. However, to some extent, they are consistent with previous findings. Our results also agree that the cost-effectiveness of denosumab may be better compared with other strategies, and that teriparatide seems to be a strategy that is not cost-effective because of its high cost and being dominated by abaloparatide.
This is the first study to investigate the cost-effectiveness of romosozumab in the US, and it shows the US costs and QALYs of 1-year treatment of romosozumab followed by 10-year treatment of alendronate in 70 year-old women with osteoporosis with a recent fracture was $37,975 and 8·54 QALY. Research in Japan showed that in 78-year-old women with a T score ≤ − 2·5 and a previous fragility fracture, the costs and QALY for a 1 year of romosozumab followed by 4 years of alendronate were, respectively, $35,178 and 7·290 QALY [52]. In Sweden, the costs and QALY were $60,396 and 8·547 in 74-year-old women with a recent major osteoporotic fracture [35].
In line with the summary of a systematic review of cost-effectiveness analyses of osteoporosis [53], the DSA results of our study confirmed that drug effect, drug cost, and medication persistence were key drivers of cost effectiveness. Our results also indicate that the incidence of adverse events during treatment and its influence on utility play an important role in outcomes. In previous studies, one of the most frequently unreported recommendations from the guidelines for economic evaluations of osteoporosis is adverse events [53]. Our study further illustrated the importance of including adverse events in cost-effectiveness analyses.
There are a few limitations associated with our study. First, because epidemiological data and WTP thresholds vary across countries, our findings should be generalized to other countries, conservatively. Second, we used efficacy of non-vertebral fracture for other osteoporosis fracture and vertebral fracture for clinical vertebral fracture. But we included the efficacy in DSA and PSA, and the results showed that denosumab always was the most cost-effective strategy when parameters vary. Third, although most of the data in our model were extracted from US data sets, some parameters were obtained from other countries, such as the 5-year persistence of alendronate and the RR of fractures, which varies with the number of prior fractures and the stage after the last fracture. Besides, the RR of subsequent fracture after prior fracture did not adjust for BMD which might cause fracture risk being over-evaluated. Fourth, the long-term persistence of zoledronate, denosumab and alendronate was assumed in this study. This assumption was based on research indicating that dropout rates are highest shortly after the initiation of treatment and remain stable for more than 5 years after that [54, 55]. The results should be updated when better data are available. At last, we used alendronate as a sequential treatment because it is the most widely used drug for the treatment of osteoporosis and it is also the drug that has been most frequently investigated in studies of the cost-effectiveness of sequential therapy [26, 35, 52]. However, in clinical practice, patients might choose other drugs. Thus, cost-effectiveness analyses using drugs other than alendronate as sequential therapy should be conducted in the future. Finally, we chose patients with a recent fracture to represent patients with a very high fracture risk, so the results may differ for patients meeting other standards.
Despite of these limitations, our study has notable strengths. This is the first study to investigate the cost-effectiveness of treatment strategies for US patients with osteoporosis who have a very high fracture risk. In addition, the strategies we compared were developed according to the recommendation of the AACE/ACE guidelines that zoledronate, denosumab, abaloparatide, teriparatide, and romosozumab can be considered as initial options for patients in a very high fracture risk, and that these patients should be treated with zoledronate for 6 years or receive a sequential treatment that consists of 10 years of bisphosphonate, p.o., following 10 years of denosumab, 2 years of abaloparatide or teriparatide or 1 year of romosozumab [4]. Also, we considered sequential treatment for denosumab for the first time, as recommended by the AACE/ACE guidelines. In addition, to the best of our knowledge, this is first study to assess the cost-effectiveness of romosozumab in the US. Finally, we used and referenced the guidelines for economic evaluations in osteoporosis, which helps to improve the transparency and quality of the study [14].
It should be noted that the pandemic of Coronavirus disease 19 (COVID-19) has brought some impact on the management of osteoporosis [56,57,58]. For example, the prevalence of non-adherence of denosumab was higher during the COVID-19 lockdown than before [56]. Since medicine persistence is one of the key drivers of cost effectiveness. The cost-effective of osteoporosis drug treatment might be overestimated during COVID-19 period. Therefore, results should be interpreted cautiously under this special situation.
In conclusion, this study provides economic results comparing zoledronate, denosumab, teriparatide, abaloparatide, and romosozumab in the US, which indicate zoledronate is the cheapest strategy. When compared with zoledronate, the cost-effectiveness of other strategies was dependent on the WTP threshold. Denosumab is the most cost-effective option when all the strategies are compared together under WTP thresholds of $50,000/QALY, $100,000/QALY and $150,000/QALY.
References
WHO Study Group (1994) Assessment of fracture risk and its application to screening for postmenopausal osteoporosis report of a WHO study group. World Health Organ Tech Rep Ser 843:1–129
Looker AC, Sarafrazi Isfahani N, Fan B, Shepherd JA (2017) Trends in osteoporosis and low bone mass in older US adults, 2005–2006 through 2013–2014. Osteoporos Int 28(6):1979–1988. https://doi.org/10.1007/s00198-017-3996-1
Burge R, Dawson-Hughes B, Solomon DH, Wong JB, King A, Tosteson A (2007) Incidence and economic burden of osteoporosis-related fractures in the United States, 2005–2025. J Bone Miner Res 22(3):465–475. https://doi.org/10.1359/jbmr.061113
Camacho PM, Petak SM, Binkley N, Diab DL, Eldeiry LS, Farooki A et al (2020) American association of clinical endocrinologists/American college of endocrinology clinical practice guidelines for the diagnosis and treatment of postmenopausal osteoporosis-2020 update. Endocr Pract 26(Suppl 1):1–46. https://doi.org/10.4158/gl-2020-0524suppl
Saag KG, Shane E, Boonen S, Marín F, Donley DW, Taylor KA et al (2007) Teriparatide or alendronate in glucocorticoid-induced osteoporosis. N Engl J Med 357(20):2028–2039. https://doi.org/10.1056/NEJMoa071408
Saag KG, Petersen J, Brandi ML, Karaplis AC, Lorentzon M, Thomas T et al (2017) Romosozumab or alendronate for fracture prevention in women with osteoporosis. N Engl J Med 377(15):1417–1427. https://doi.org/10.1056/NEJMoa1708322
van Geel TA, van Helden S, Geusens PP, Winkens B, Dinant GJ (2009) Clinical subsequent fractures cluster in time after first fractures. Ann Rheum Dis 68(1):99–102. https://doi.org/10.1136/ard.2008.092775
Johansson H, Siggeirsdóttir K, Harvey NC, Odén A, Gudnason V, McCloskey E et al (2017) Imminent risk of fracture after fracture. Osteoporos Int 28(3):775–780. https://doi.org/10.1007/s00198-016-3868-0
Adachi JD, Berger C, Barron R, Weycker D, Anastassiades TP, Davison KS et al (2019) Predictors of imminent non-vertebral fracture in elderly women with osteoporosis, low bone mass, or a history of fracture, based on data from the population-based Canadian multicentre osteoporosis study (CaMos). Arch Osteoporos 14(1):53. https://doi.org/10.1007/s11657-019-0598-x
Yusuf AA, Hu Y, Chandler D, Crittenden DB, Barron RL (2020) Predictors of imminent risk of fracture in medicare-enrolled men and women. Arch Osteoporos 15(1):120. https://doi.org/10.1007/s11657-020-00784-7
Sonnenberg FA, Beck JR (1993) Markov models in medical decision making: a practical guide. Med Decis Making 13(4):322–338. https://doi.org/10.1177/0272989x9301300409
Briggs A, Sculpher M (1998) An introduction to Markov modelling for economic evaluation. Pharmacoeconomics 13(4):397–409. https://doi.org/10.2165/00019053-199813040-00003
Husereau D, Drummond M, Petrou S, Carswell C, Moher D, Greenberg D et al (2013) Consolidated health economic evaluation reporting standards (CHEERS) statement. Value Health 16(2):e1-5. https://doi.org/10.1016/j.jval.2013.02.010
Hiligsmann M, Reginster JY, Tosteson ANA, Bukata SV, Saag KG, Gold DT et al (2019) Recommendations for the conduct of economic evaluations in osteoporosis: outcomes of an experts’ consensus meeting organized by the European society for clinical and economic aspects of osteoporosis, osteoarthritis and musculoskeletal diseases (ESCEO) and the US branch of the international osteoporosis foundation. Osteoporos Int 30(1):45–57. https://doi.org/10.1007/s00198-018-4744-x
Sanders GD, Neumann PJ, Basu A, Brock DW, Feeny D, Krahn M et al (2016) Recommendations for conduct, methodological practices, and reporting of cost-effectiveness analyses: second panel on cost-effectiveness in health and medicine. JAMA 316(10):1093–1103. https://doi.org/10.1001/jama.2016.12195
Jönsson B, Ström O, Eisman JA, Papaioannou A, Siris ES, Tosteson A et al (2011) Cost-effectiveness of denosumab for the treatment of postmenopausal osteoporosis. Osteoporos Int 22(3):967–982. https://doi.org/10.1007/s00198-010-1424-x
Söreskog E, Borgström F, Lindberg I, Ström O, Willems D, Libanati C et al (2021) A novel economic framework to assess the cost-effectiveness of bone-forming agents in the prevention of fractures in patients with osteoporosis. Osteoporos Int 32(7):1301–1311. https://doi.org/10.1007/s00198-020-05765-7
Ettinger B, Black DM, Dawson-Hughes B, Pressman AR, Melton LJ 3rd (2010) Updated fracture incidence rates for the US version of FRAX. Osteoporos Int 21(1):25–33. https://doi.org/10.1007/s00198-009-1032-9
Mori T, Crandall CJ, Ganz DA (2017) Cost-effectiveness of combined oral bisphosphonate therapy and falls prevention exercise for fracture prevention in the USA. Osteoporos Int 28(2):585–595. https://doi.org/10.1007/s00198-016-3772-7
Söreskog E, Ström O, Spångéus A, Åkesson KE, Borgström F, Banefelt J et al (2020) Risk of major osteoporotic fracture after first, second and third fracture in Swedish women aged 50 years and older. Bone 134:115286. https://doi.org/10.1016/j.bone.2020.115286
Arias E, Xu J (2020) United States life tables, 2018. Natl Vital Stat Rep 69(12):1–45
Haentjens P, Magaziner J, Colón-Emeric CS, Vanderschueren D, Milisen K, Velkeniers B et al (2010) Meta-analysis: excess mortality after hip fracture among older women and men. Ann Intern Med 152(6):380–390. https://doi.org/10.7326/0003-4819-152-6-201003160-00008
Kanis JA, Oden A, Johnell O, De Laet C, Jonsson B, Oglesby AK (2003) The components of excess mortality after hip fracture. Bone 32(5):468–473. https://doi.org/10.1016/s8756-3282(03)00061-9
Parker MJ, Anand JK (1991) What is the true mortality of hip fractures? Public Health 105(6):443–446
Johnell O, Kanis JA, Odén A, Sernbo I, Redlund-Johnell I, Petterson C et al (2004) Mortality after osteoporotic fractures. Osteoporos Int 15(1):38–42. https://doi.org/10.1007/s00198-003-1490-4
Hiligsmann M, Williams SA, Fitzpatrick LA, Silverman SS, Weiss R, Reginster JY (2019) Cost-effectiveness of sequential treatment with abaloparatide vs teriparatide for United States women at increased risk of fracture. Semin Arthritis Rheum 49(2):184–196. https://doi.org/10.1016/j.semarthrit.2019.01.006
Barrionuevo P, Kapoor E, Asi N, Alahdab F, Mohammed K, Benkhadra K et al (2019) Efficacy of pharmacological therapies for the prevention of fractures in postmenopausal women: a network meta-analysis. J Clin Endocrinol Metab 104(5):1623–1630. https://doi.org/10.1210/jc.2019-00192
Freemantle N, Cooper C, Diez-Perez A, Gitlin M, Radcliffe H, Shepherd S et al (2013) Results of indirect and mixed treatment comparison of fracture efficacy for osteoporosis treatments: a meta-analysis. Osteoporos Int 24(1):209–217. https://doi.org/10.1007/s00198-012-2068-9
Mori T, Crandall CJ, Ganz DA (2019) Cost-effectiveness of sequential teriparatide/alendronate versus alendronate-alone strategies in high-risk osteoporotic women in the US: analyzing the impact of generic/biosimilar teriparatide. JBMR Plus 3(11):e10233. https://doi.org/10.1002/jbm4.10233
Bone HG, Bolognese MA, Yuen CK, Kendler DL, Miller PD, Yang YC et al (2011) Effects of denosumab treatment and discontinuation on bone mineral density and bone turnover markers in postmenopausal women with low bone mass. J Clin Endocrinol Metab 96(4):972–980. https://doi.org/10.1210/jc.2010-1502
Bone HG, Cosman F, Miller PD, Williams GC, Hattersley G, Hu MY et al (2018) ACTIVExtend: 24 months of alendronate after 18 months of abaloparatide or placebo for postmenopausal osteoporosis. J Clin Endocrinol Metab 103(8):2949–2957. https://doi.org/10.1210/jc.2018-00163
Le QA, Hay JW, Becker R, Wang Y (2019) Cost-effectiveness analysis of sequential treatment of abaloparatide followed by alendronate versus teriparatide followed by alendronate in postmenopausal women with osteoporosis in the United States. Ann Pharmacother 53(2):134–143. https://doi.org/10.1177/1060028018798034
Cramer JA, Roy A, Burrell A, Fairchild CJ, Fuldeore MJ, Ollendorf DA et al (2008) Medication compliance and persistence: terminology and definitions. Value Health. 11(1):44–47. https://doi.org/10.1111/j.1524-4733.2007.00213.x
Durden E, Pinto L, Lopez-Gonzalez L, Juneau P, Barron R (2017) Two-year persistence and compliance with osteoporosis therapies among postmenopausal women in a commercially insured population in the United States. Arch Osteoporos 12(1):22. https://doi.org/10.1007/s11657-017-0316-5
Söreskog E, Lindberg I, Kanis JA, Åkesson KE, Willems D, Lorentzon M et al (2021) Cost-effectiveness of romosozumab for the treatment of postmenopausal women with severe osteoporosis at high risk of fracture in Sweden. Osteoporos Int 32(3):585–594. https://doi.org/10.1007/s00198-020-05780-8
Brown JP, Morin S, Leslie W, Papaioannou A, Cheung AM, Davison KS et al (2014) Bisphosphonates for treatment of osteoporosis: expected benefits, potential harms, and drug holidays. Can Fam Physician 60(4):324–333
Parthan A, Kruse M, Yurgin N, Huang J, Viswanathan HN, Taylor D (2013) Cost effectiveness of denosumab versus oral bisphosphonates for postmenopausal osteoporosis in the US. Appl Health Econ Health Policy 11(5):485–497. https://doi.org/10.1007/s40258-013-0047-8
Reid IR, Gamble GD, Mesenbrink P, Lakatos P, Black DM (2010) Characterization of and risk factors for the acute-phase response after zoledronic acid. J Clin Endocrinol Metab 95(9):4380–4387. https://doi.org/10.1210/jc.2010-0597
Miller PD, Hattersley G, Riis BJ, Williams GC, Lau E, Russo LA et al (2016) Effect of abaloparatide vs placebo on new vertebral fractures in postmenopausal women with osteoporosis: a randomized clinical trial. JAMA 316(7):722–733. https://doi.org/10.1001/jama.2016.11136
2021 ASP Drug Pricing Files. https://www.cms.gov/medicare/medicare-part-b-drug-average-sales-price/2021-asp-drug-pricing-files2021 [cited 2021 Dec 1].
Thompson Healthcare M. Drugtopics red book 2018 (online version). http://truvenhealth.com/Products/ Micromedex/Product-Suites/Clinical-Knowledge/REDBOOK.
Kilgore ML, Morrisey MA, Becker DJ, Gary LC, Curtis JR, Saag KG et al (2009) Health care expenditures associated with skeletal fractures among medicare beneficiaries, 1999–2005. J Bone Miner Res 24(12):2050–2055. https://doi.org/10.1359/jbmr.090523
Stopeck A, Brufsky A, Kennedy L, Bhatta S, Bhowmik D, Buchanan J et al (2020) Cost-effectiveness of denosumab for the prevention of skeletal-related events in patients with solid tumors and bone metastases in the United States. J Med Econ 23(1):37–47. https://doi.org/10.1080/13696998.2019.1651122
Physician Fee Schedule. https://www.cms.gov/medicare/physician-fee-schedule/search?Y=0&T=4&HT=0&CT=3&H1=96413&M=5.
Hanmer J, Lawrence WF, Anderson JP, Kaplan RM, Fryback DG (2006) Report of nationally representative values for the noninstitutionalized US adult population for 7 health-related quality-of-life scores. Med Decis Making 26(4):391–400. https://doi.org/10.1177/0272989x06290497
Hiligsmann M, Ethgen O, Richy F, Reginster JY (2008) Utility values associated with osteoporotic fracture: a systematic review of the literature. Calcif Tissue Int 82(4):288–292. https://doi.org/10.1007/s00223-008-9117-6
Davis S, Martyn-St James M, Sanderson J, Stevens J, Goka E, Rawdin A et al (2016) A systematic review and economic evaluation of bisphosphonates for the prevention of fragility fractures. Health Technol Assess 20(78):1–406. https://doi.org/10.3310/hta20780
Redekop WK, Stolk EA, Kok E, Lovas K, Kalo Z, Busschbach JJ (2004) Diabetic foot ulcers and amputations: estimates of health utility for use in cost-effectiveness analyses of new treatments. Diabetes Metab 30(6):549–556. https://doi.org/10.1016/s1262-3636(07)70154-4
Balasubramanian A, Zhang J, Chen L, Wenkert D, Daigle SG, Grauer A et al (2019) Risk of subsequent fracture after prior fracture among older women. Osteoporos Int 30(1):79–92. https://doi.org/10.1007/s00198-018-4732-1
Black DM, Cauley JA, Wagman R, Ensrud K, Fink HA, Hillier TA et al (2018) The ability of a single BMD and fracture history assessment to predict fracture over 25 years in postmenopausal women: the study of osteoporotic fractures. J Bone Miner Res 33(3):389–395. https://doi.org/10.1002/jbmr.3194
Silverman S, Agodoa I, Kruse M, Parthan A, Orwoll E (2015) Denosumab for elderly men with osteoporosis: a cost-effectiveness analysis from the US payer perspective. J Osteoporos 2015:627631. https://doi.org/10.1155/2015/627631
Hagino H, Tanaka K, Silverman S, McClung M, Gandra SR, Charokopou M et al (2021) Cost effectiveness of romosozumab versus teriparatide for severe postmenopausal osteoporosis in Japan. Osteoporos Int 32(10):2011–2021. https://doi.org/10.1007/s00198-021-05927-1
Li N, Cornelissen D, Silverman S, Pinto D, Si L, Kremer I et al (2021) An updated systematic review of cost-effectiveness analyses of drugs for osteoporosis. Pharmacoeconomics 39(2):181–209. https://doi.org/10.1007/s40273-020-00965-9
Huybrechts KF, Ishak KJ, Caro JJ (2006) Assessment of compliance with osteoporosis treatment and its consequences in a managed care population. Bone 38(6):922–928. https://doi.org/10.1016/j.bone.2005.10.022
Solomon DH, Avorn J, Katz JN, Finkelstein JS, Arnold M, Polinski JM et al (2005) Compliance with osteoporosis medications. Arch Intern Med 165(20):2414–2419. https://doi.org/10.1001/archinte.165.20.2414
De Vincentis S, Domenici D, Ansaloni A, Boselli G, D’Angelo G, Russo A et al (2022) COVID-19 lockdown negatively impacted on adherence to denosumab therapy: incidence of non-traumatic fractures and role of telemedicine. J Endocrinol Invest 19:1–11. https://doi.org/10.1007/s40618-022-01820-8
Falchetti A, Mohseni M, Tramontana F, Napoli N (2021) Secondary prevention of fragility fractures: where do we stand during the COVID-19 pandemic? J Endocrinol Invest 44(11):2521–2524. https://doi.org/10.1007/s40618-021-01552-1
Narla RR, Adler RA (2021) Osteoporosis care amidst the prolonged pandemic. J Endocrinol Invest 44(7):1353–1361. https://doi.org/10.1007/s40618-021-01542-3
Nayak S, Greenspan SL (2021) Cost-effectiveness of 3 versus 6 years of zoledronic acid treatment before bisphosphonate holiday for women with osteoporosis. Osteoporos Int. https://doi.org/10.1007/s00198-021-06010-5
Bureau of Labor Statistics Consumer Price Index. http://www.bls.gov/cpi/.
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China [grant numbers 81870622], the Changsha Municipal Natural Science Foundation [grant number kq2014251], Hunan Provincial Innovation Foundation For Postgraduate[grant number CX20210372], the Fundamental Research Funds for the Central Universities of Central South University[grant number 512191022], Degree& Postgraduate Education Reform Project of Central South University[grant number 512190112] and Scientific Research Project of Hunan Provincial Health Commission[grant number 202112070631].
Funding
The funding sources of the study had no role in the study design, data collection, analysis, interpretation, report writing or the decision to submit the paper for publication. National Natural Science Foundation of China, 81870622, Zhifeng Sheng, Changsha Municipal Natural Science Foundation,kq2014251, Zhifeng Sheng, Fundamental Research Funds for Central Universities of the Central South University,512191022, Zhifeng Sheng, Degree& Postgraduate Education Reform Project of Central South University, 512190112, Zhifeng Sheng, Scientific Research Project of Hunan Provincial Health Commission,202112070631, Zhifeng Sheng, Hunan Provincial Innovation Foundation for Postgraduate, CX20210372, Zhifeng Sheng.
Author information
Authors and Affiliations
Contributions
CL was involved in designing study, collecting data, constructing model, statistical analyzing, interpreting results and writing manuscript. S-XQ was involved in designing study, collecting data, constructing model and statistical analyzing. Q-LW, Y-FL, X-LQ, CY and LH were involved in designing study, collecting data and interpreting results. Z-FS and X-MW were involved in designing study, constructing model, interpreting results and supervising the whole study.
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare no conflicts of interest.
Ethical approval
Since this economic analysis was based on literature review and modeling techniques, there was no ethical issure involved.
Research involving human participants and/or animals
This research was conducted through literature review and modeling techniques, and does not contain any studies with human participants or animals.
Informed consent
For this study, informed consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Luo, C., Qin, SX., Wang, QY. et al. Cost-effectiveness analysis of five drugs for treating postmenopausal women in the United States with osteoporosis and a very high fracture risk. J Endocrinol Invest 46, 367–379 (2023). https://doi.org/10.1007/s40618-022-01910-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-022-01910-7