Abstract
Background
The enhanced recovery after surgery (ERAS) is nowadays a widely accepted multimodal programme of care in colorectal surgery, but still there is some reluctance in its application to very elderly patients.
Aim
The aim of this study is to investigate short-term outcomes of laparoscopic resection for colorectal cancer in octogenarian patients within the ERAS programme.
Methods
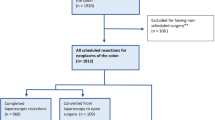
Data on 162 consecutive patients aged ≥ 80 years receiving elective minimally invasive colorectal resections within ERAS programme were collected in a multicentre, retrospective database in the period 2008–2017 in Italy. Univariate and multivariate analyses were performed to assess possible risk factors for poor clinical outcomes.
Results
The postoperative minor morbidity rate (Clavien–Dindo 1 and 2) was 25.9%. The incidence of postoperative major morbidity rate (severe medical and surgical complications defined as Clavien–Dindo 3 and 4) accounted 6.1% and only 1.8% had an anastomotic leakage. Reoperation rate was 5.5%, perioperative 30-day mortality was 1.8%, and 30-day readmission rate was 6.8%. On average, patients were released after 6 days. A univariate analysis showed that possible risk factors for severe medical complications were: low preoperative albumin level, high Charlson Age Comorbidity Index Score and number of days in the intensive care unit (ICU); risk factors for severe surgical complications were: low preoperative albumin level; risk factors for late hospital discharge were: multivisceral resections, number of days in ICU and body mass index (BMI) > 25 kg/m2. The multivariate analysis confirmed a low level of preoperative albumin and a longer ICU stay as independent risk factors for both postoperative severe surgical complications and late hospital discharge.
Discussion
The minimal invasive nature of the laparoscopic approach together with a multimodal analgesia therapy, the early resumption to oral diet and mobilisation could minimize the surgical stress and play an essential role in order to reduce medical morbidity in high-risk patients.
Conclusion
Colorectal surgery within ERAS programme in octogenarians is a safe and flexible treatment in high-volume centres.
Similar content being viewed by others
References
Kehlet H, Wilmore DW (2002) Multimodal strategies to improve surgical outcome. Am J Surg 183:630–641
Greco M, Capretti G, Beretta L et al (2014) Enhanced recovery program in colorectal surgery: a metaanalysis of randomized controlled trials. World J Surg 38:1531–1541
Varadhan KK, Neal KR, Dejong CH et al (2010) The enhanced recovery after surgery (ERAS) pathway for patients undergoing major elective open colorectal surgery: a meta-analysis of randomized controlled trials. Clin Nutr 29:434–440
Roulin D, Donadini A, Gander S et al (2013) Cost-effectiveness of the implementation of an enhanced recovery protocol for colorectal surgery. Br J Surg 100:1108–1114
Fearon KC, Ljungqvist O, Von Meyenfeldt M et al (2005) Enhanced recovery after surgery: a consensus review of clinical care for patients undergoing colonic resection. Clin Nutr 24:466–477
Eurostat (2018) Causes of death—standardised death rate by residence. http://ec.europa.eu/eurostat/statisticsexplained/index.php/Causes_of_death_statistics/it. Accessed 23 November 2018
Owens WD, Felts JA, Spitznagel EL Jr (1978) ASA physical status classifications: a study of consistency of ratings. Anesthesiology 49:239–243
Sobin LH, Compton CC (2010) TNM seventh edition: what’s new, what’s changed communication from the International Union Against Cancer and the American Joint Committee on Cancer. Cancer 116:5336–5339
Charlson M, Szatrowski TP, Peterson J et al (1994) Validation of a combined comorbidity index. J Clin Epidemiol 47:1245–1251
Clavien PA, de Oliveira ML, Vauthey JN et al (2009) Clavien–Dindo classification of surgical complications: five-year experience. Ann Surg 250:187–196
National Comprehensive Cancer Network. (2018) Clinical practice Guidelines in Oncology. https://www.nccn.org/professionals/physician_gls/default.aspx. Accessed 19 October, 2018
AIOM (2016) I numeri del cancro in Italia 2016. Il Pensiero Scientifico Editore, Roma, pp 28–29, ISBN 978-88-490-0568-4
Hermans E, van Schaik PM, Prins HA et al (2010) Outcome of colonic surgery in elderly patients with colon cancer. J Oncol. https://doi.org/10.1155/2010/865908
De Marco MF, Janssen-Heijnen ML, van der Heijden LH et al (2000) Comorbidity and colorectal cancer according to subsite and stage: a population-based study. Eur J Cancer 36:95–99
Zhu Q, Mao Z, Jin J et al (2010) The safety of CO2 pneumoperitoneum for elderly patients during laparoscopic colorectal surgery. Surg Laparosc Endosc Percutan Tech 20:54–57
Xie M, Qin H, Luo Q et al (2015) Laparoscopic colorectal resection in octogenarian patients is it safe? A Systematic review and meta-analysis. Medicine 94(42):e1765
Li Y, Wang S, Gao S et al (2016) Laparoscopic colorectal resection versus open colorectal resection in octogenarians: a systematic review and meta-analysis of safety and efficacy. Tech Coloproctol 20:153–162
Devoto L, Celentano V, Cohen R et al (2017) Colorectal cancer surgery in the very elderly patient: a systematic review of laparoscopic versus open colorectal resection. Int J Colorectal Dis 32:1237–1242
Bagnall NM, Malietzis G, Kennedy RH et al (2014) A systematic review of enhanced recovery care after colorectal surgery in elderly patients. Colorectal Dis 16:947–956
Wang Q, Suo J, Jiang J et al (2012) Effectiveness of fast-track rehabilitation vs conventional care in laparoscopic colorectal resection for elderly patients: a randomized trial. Colorectal Dis 14:1009–1013
Jia Y, Jin G, Guo S et al (2013) Fast-track surgery decreases the incidence of postoperative delirium and other complications in elderly patients with colorectal carcinoma. Langenbecks Arch Surg 399:77–84
Jung WB, Shin JY, Suh B (2017) The short-term outcome and safety of laparoscopic colorectal cancer resection in very elderly patients. Korean J Gastroenterol 69:291–297
Forsmo HM, Erichsen C, Rasdal A et al (2017) Enhanced recovery after colorectal surgery (ERAS) in elderly patients is feasible and achieves similar results as in younger patients. Gerontol Geriatr Med 3:2333721417706299
Gonzalez Ayora S, Pastor C, Guadalajara H et al (2016) Enhanced recovery care after colorectal surgery in elderly patients: compliance and outcomes of a multicenter study from the Spanish working group on ERAS. Int J Colorectal Dis 31:1625–1631
Baek SJ, Kim SH, Kim SY et al (2013) The safety of a “fast-track” program after laparoscopic colorectal surgery is comparable in older patients as in younger patients. Surg Endosc 27:122–123
Stocchi L, Nelson H, Young-Fadok TM et al (2000) Safety and advantages of laparoscopic vs. open colectomy in the elderly: matched-control study. Dis Colon Rectum 43:326–332
Franklin ME Jr, Rosenthal D, Abrego-Medina D et al (1996) Prospective comparison of open vs. laparoscopic colon surgery for carcinoma. 5-year results. Dis Colon Rectum 39:S35–S46
Rumstadt B, Guenther N, Wendling P et al (2009) Multimodal perioperative rehabilitation for colonic surgery in the elderly. World J Surg 33:1757–1763
Pędziwiatr M, Pisarska M, Wierdak M et al (2015) The use of the enhanced recovery after surgery (ERAS) protocol in patients undergoing laparoscopic surgery for colorectal cancer: a comparative analysis of patients aged above 80 and below 55. Pol Przegl Chir. 87:565–572
Kehlet H (2018) ERAS Implementation-Time To Move Forward. Ann Surg 267:998–999
Fish DR, Mancuso CA, Garcia-Aquilar JE et al (2017) Readmission after ileostomy creation retrospective review of a common and significant event. Ann Surg 265:379–387
Messaris E, Sehgal R, Deiling S et al (2012) Dehydration is the most common indication for readmission after diverting ileostomy creation. Dis Colon Rectum 55:175–180
Acknowledgements
The research presented here was the product of a team-work. Doctor CD and doctor DN contributed to the design and supervised all the research’s process. Doctor GM was in charge of the statistical analysis and also provided a general support to the manuscript. Thanks go to doctor FM, AA and BV for their assistance with data collection. We gratefully acknowledge the support and professionalism of doctor BG and VM who performed all the surgical procedures.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The present paper is not based on any previous communication to a society or meeting. The authors declare they have no conflict of interest.
Ethical approval
This study was approved by the institutional Ethics Committee and conducted according to the declaration of Helsinki.
Informed consent
Informed consent was obtained from all patients. All authors gave their consentment for paper publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix 1: surgical technique
Right colectomy
Authors started with the separation of the omentum from the transverse colon, mesentery using a medial to lateral approach. The hepatic flexure was taken down with different energy devices. The right paracolic gutter was divided along the Toldt’s line with blunt dissection until the third portion of the duodenum was clearly visible. The ileocolic and right colic vessels were divided and sectioned at their origin between clips, performing a complete mesocolic excision10. An intracorporeal anastomosis was accomplished with 60 mm laparoscopic linear staple loads. No drain was usually placed in the abdomen.
Transverse resection
The intervention consisted in three types of procedures:
-
Segmentary resection of the splenic flexure, defined as a resection of the colon from the last third of the transverse colon to the first third of the descending colon. An intracorporeal mechanical latero-lateral colo-colic anastomosis was performed.
-
Extended left colectomy, defined as a resection of the colon from the last third of the transverse colon to the colorectal junction. An intracorporeal mechanical end-to-end colorectal anastomosis was performed.
-
Extended right colectomy, defined as a resection of the colon from the last ileal limp to the first third of the descending colon. An intracorporeal mechanical latero-lateral ileo-colic anastomosis was performed.
Left colectomy and rectal resection
Left colon mobilization was started by a medial to lateral interruption of the gastrocolic and splenocolic ligaments followed by separation of the plane between Gerota’s and Toldt’s fascias. Left paracolic gutter was dissected along the Toldt’s line with blunt dissection up to middle rectum with Waldeyer nerve-sparing fascia dissection. Section of the inferior mesenteric vessels was always performed between metallic clips, 2 cm from the aortic plane in order to preserve hypogastric nerves. After circumferential dissection, the middle rectum was divided using a 60mm laparoscopic linear stapler. An intracorporeal circular end-to-end anastomosis was achieved inserting the stapler transanally. A hydropneumatic test was performed to check the integrity of the anastomosis and any leaks identified were sutured by interrupted stitches. A perianastomotic drain tube was placed in the pelvis.
Appendix 2
Fast-track protocol
Item | Study protocol |
|---|---|
Dedicated preoperative counselling | Anaesthesiologic, cardiologic and surgical counselling according to ESA guidelines |
Preoperative optimisation; smoking/alcohol cessation; physical exercise | Improving daily aerobic activity |
Nutritional supplementation | |
Avoidance of preoperative bowel preparation | Only for rectal resection: two rectal enemas the evening before surgery |
Preoperative fasting and carbohydrate treatment | Solids allowed up to 6 h prior surgery |
Clear fluids up to 2 h prior surgery | |
Carbohydrates oral loading | |
Avoidance of long- or short-acting premedication anaesthetics | Premedication with Midazolam 20 mcg/kg |
Prophylaxis against thromboembolism | Compression stockings |
Allen stirrups | |
LMWH according to Caprini score | |
Antimicrobial prophylaxis and skin preparation | Cefazoline 2 g and Metronidazole 500 mg, 60 min before surgery |
Two other infusions of metronidazole 500 mg are administered in the first 24 h | |
Multi-modal anaesthetic protocol | Laryngeal mask airway device or orotracheal intubation |
US-guided quadratus lomborum block or TAP block | |
Injection of local anaesthetics on the region of surgical wounds | |
PONV prophylaxis | Premedication with desametasone 8 mg + metoclopramide 10 mg + ondansetron 4 mg + ranitidine 50 mg |
Laparoscopy and modifications of surgical access | Laparoscopic approach always initially performed |
Avoidance of nasogastric intubation | Orogastric tube placed at the beginning of surgery, removed at the end of the procedure |
Prevention of intraoperative hypothermia | Warming device (Bair Hugger) |
Warmed intravenous fluids | |
Perioperative fluid management | Colloids avoided, if possible, in order to prevent coagulopathy |
0.5–1 ml/kg/h infusion, starting 2 h after surgery, stopped after 24 h | |
Avoidance of abdominal drainage | Abdominal drain/s often placed |
Urinary drainage for 1–2 days | Bladder catheter left in place for 24 h if operative time is > 120 min |
Prevention of postoperative ileus | If TAP block/quadratus lomborum block applied, if thoracic epidural analgesia notapplied |
Encouraging moderate coffee assumption | |
Multi-modal postoperative analgesia | Multimodal analgesia with TAP block |
Intravenous infusion of drugs other than opioids | |
Injection of local anaesthetics on the region of surgical wounds | |
Perioperative nutritional care | Nutritional screening highly recommended considering BMI and albumin level |
Preoperative oral nutrition support intake frequently necessary | |
Start oral feeding 1 day after surgery with semi-liquid diet (2 days after surgery in case of ICU stay) | |
Postoperative glucose control | A judiciously glycemic control is applied |
Personalized food intake, according to comorbidities | |
Early mobilisation | Mobilization assistance and physiokinesitherapy |
Familial support in early mobilisation | |
Prevention of cognitive impairment (not included in ERAS items) | Avoid any communicative barrier (i.e. scientifical words) |
Avoid postoperative sedative drugs | |
A caregiver should be encouraged to participate in the pre-/postoperative p |
Rights and permissions
About this article
Cite this article
Depalma, N., Cassini, D., Grieco, M. et al. Feasibility of a tailored ERAS programme in octogenarian patients undergoing minimally invasive surgery for colorectal cancer. Aging Clin Exp Res 32, 265–273 (2020). https://doi.org/10.1007/s40520-019-01195-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40520-019-01195-6