Abstract
Purpose of Review
In the field of periodontology, numerous studies have been conducted to observe the effects of sex hormones on the periodontal tissues and the oral mucosa. The balance in the circulating hormonal levels in the female body ensures proper homeostasis of the periodontium. Therefore, this literature review focuses on the more recent concepts related to changes in the endogenous and exogenous female hormones on the periodontal health in the lifetime of a woman.
Recent Findings
Previous studies have reported severe gingival inflammation in women using oral contraceptives. But present day oral contraceptives have decreased dosages in contrast to the older formulations and therefore only mildly affect the periodontium with the majority reporting lack of gingival inflammation. Periodontal treatment strategies for each stage in a woman’s life is also described in this review, and research pertaining to periodontal therapy during pregnancy has shown conflicting results in terms of reducing the risk of adverse pregnancy outcomes.
Summary
The changes seen in the periodontium seen in each milestone of a woman’s life correlates to the fluctuating levels in the female sex hormones. Maintaining oral health during the peak of hormone release such as pregnancy helps alleviate the symptoms of periodontal disease as well as reduce the risk of adverse pregnancy outcomes, although studies report conflicting results.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In the past decade, the focus on women’s health has undergone drastic reformation. Though women and men share comparable health challenges, the metamorphoses are such that the health of women deserves attention. Among the biological systems, endocrines have influenced the overall health, especially with reference to hormones that play an integral molecular role in creating balance between the host and the external environment. Their influence not only reflects on the general body but also displays concerns in the oral cavity. This literature review is structured around the effect of sex hormone fluctuations and its impact on the periodontium, along with therapeutic information concerning different conditions seen during the lifetime of a woman.
Methods
In the current literature review, scientific articles were searched in Google Scholar, Scopus, and PubMed electronic databases until October 2020. The following search terms and their combinations were used: “female sex hormones”, “periodontal health”, “periodontal disease”, “menstrual cycle”, “pregnancy”, “oral contraceptives”, “adverse pregnancy outcomes”, “polycystic ovarian syndrome”, “menopause”, “hormone replacement therapy”, “periodontal management”.
Current State of Knowledge
Female Sex Hormones: Metabolism, Function, Receptors
The anterior pituitary gland, at the time of puberty, begins secreting gonadotropins: follicle-stimulating hormones and the luteinizing hormone, which act on the ovaries to begin cyclical production and secretion of the two main female sex hormones: estrogen and progesterone [1].
The main functions of estrogen are in the development and maintenance of the primary and secondary sex characteristics, uterine growth, and increased bone mineral density, etc. Among the naturally occurring estrogens, the most potent is Estradiol. Its actions are conveyed through certain receptors within the target cell, which have been identified as ERα and ERβ [2].
The other vital female hormone is progesterone. They belong to a group of steroid hormones called progestins. The natural progestins are derived from a 21-carbon saturated steroid hydrocarbon known as pregnane, and like estrogen, they mediate their actions through certain progesterone receptors. Two isoforms of these receptors have been identified in most rodents and humans: PR-A and PR-B [3].
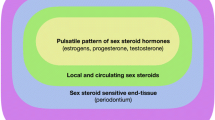
Sex steroid hormone receptors are not ubiquitous but are found at high concentrations in hormone-sensitive tissues, termed target tissues. Cytoplasmic and nuclear receptors that bind specific hormones lead to the preferential accumulation and their retention within these tissues. One of the target tissues is the periodontium with specific estrogen and progesterone receptors present in human gingiva [4, 5].
There is sufficient evidence showing female sex hormones and their metabolic products within the gingival tissues and their ability to produce or modify the gingival response to dental plaque [6, 7, 8•]. Certain enzymatic mechanisms involved in the metabolism of sex hormones have been identified within the gingiva, and the presence of an inflammatory component significantly affects its metabolism. The conversion of estrone to estradiol is three times higher in inflamed tissues, and the metabolism of progesterone is more efficient in a chronically inflamed gingiva. The duration of inflammation also determines the rate of metabolism. Chronic inflammation is shown to have a higher degree of conversion of estrone to estradiol when compared to that of an acutely inflamed tissue due to increase in the number of fibroblasts which are known to contribute greatly to the enzymatic conversion [8•].
Effects of Female Sex Hormones on the Periodontal Tissues
-
1.
Estrogen:
Estrogen is known to decrease keratinization, increase epithelial glycogen, and alter the polymerization of the ground substance which results in decrease of the effectiveness of the epithelial barrier. Similarly, fibroblast cell proliferation and protein production are also affected.
Lindhe and Brånemark studied the effect of estrogen on hamster models. The authors observed increased proliferation of blood vessels which provoked disturbances in the plasma-endothelial wall by inducing adhesion between granulocytes, platelets, and endothelium [9, 10].
Likewise, it influences the immune complex by reducing T-cell–mediated inflammation, inhibiting polymorphonuclear leukocyte chemotaxis, IL6 suppression and has a bimodal action on prostaglandin E2 synthesis.
-
2.
Progesterone
Similar to estrogen, this hormone has the ability to influence the periodontium alone or in combination. They increase vascular permeability, lower the synthesis of glycosaminoglycans, alter the rate and pattern of collagen production and in high concentrations, and downregulate IL6 production. Along with estrogen, it has shown to increase the rate of folate metabolism in the oral mucosa thereby affecting tissue repair and also enhancing the production of prostaglandin E2 [11,12,13].
Hormonal Effects on the Periodontium in Different Age Groups
-
1.
Adolescence
Puberty
Puberty is the period of maturation into adulthood and is associated with changes in the physical appearance and behavior with a major increase in the secretions of the sex steroid hormones. In relation to the periodontium, several changes occur during puberty with an increased prevalence of gingivitis followed by remission. Puberty gingivitis is clinically characterized by exuberant inflammation and gingival overgrowth which may be associated with gingival bleeding. Interestingly, gingival inflammation may not be consistent with the plaque levels, rather may be closely related to the fluctuations in the hormonal levels [14, 15].
However, this concept was contradicted as per the study that showed the influence of oral hygiene on the gingival condition of pubertal children was more beneficial than the rising level of steroid hormones [16]. The microbial changes seen during this age are the result of the responses of the gingival tissue to the sex hormones that alter its microenvironment along with the ability of certain species of bacteria to capitalize on the higher concentration of the hormones present as described in Table 1 [17, 18•, 19].
The Menstrual Cycle
The menstrual or reproductive cycle is characterized by the onset of increased production and secretion of estrogen and progesterone in a cyclic pattern coincident with the onset of puberty. Significant gingival inflammatory changes have been documented and are aggravated by an imbalance in the sex hormones. Mühlemann presented a “gingivitis intermenstrualis” which was characterized by bright red hemorrhagic lesions of the interdental papilla developed prior to the menstrual period [20]. Gingival changes including variations in the gingival crevicular fluid (GCF) flow during menstruation are mostly attributed to those with pre-existing gingivitis compared to that of a healthy gingiva [21, 22]. Additionally, inflammatory cytokines like interleukin 1β (IL1β) levels were also reported to have significant changes, i.e., steady increase in the GCF from the day of menstruation to the peak progesterone day. This may be explained by the contribution of progesterone to stimulate the inflammatory cells to express more IL1β [23•]. Other inflammatory cytokines like IL6 and tumor necrosis factor alpha may also contribute to the inflammatory changes seen in the gingiva during the menstrual cycle.
-
2.
Womanhood
Pregnancy
Estrogen and progesterone are the predominant pregnancy hormones. During this fascinating phase, the periodontium may cater to pathological conditions like gingivitis, pregnancy granuloma, periodontitis, and dental caries. Pregnancy gingivitis is prevalent in approximately 35% to 100% woman which may be non-specific, vascularizing, and proliferative inflammation with increased cell infiltration that begins to appear in the 2nd month of pregnancy and continues till the 8th month with maximum effect seen between the 1st and 2nd trimester [24, 25]. Another common oral finding is tooth mobility including alterations in probing depth which seemed to increase as a result of edema or the hyperplastic changes of the gingiva [26]. Furthermore, microbial alterations have been noticed in both supra- and subgingival environment which has been described in Table 2 [27, 28•, 29]. Reports have also stated some important immune-modulators like T-lymphocytes are also affected during pregnancy [28•, 29, 30]. Accumulation of active hormone within the gingival tissue and its increased levels are immunosuppressive, preventing acute inflammatory reaction to plaque. Thus, a chronic type of inflammatory reaction occurs, resulting in an exaggerated appearance of gingival inflammation. The combined effects of the immunosuppressive actions of progesterone, its increased levels in the gingiva, and the microbial action contribute to the periodontal disease state during pregnancy. An inflammatory gingival lesion, “pyogenic granuloma” synonym “pregnancy tumor,” and/or “epulis gravidarum” may appear during the 1st or 2nd trimester. It is clinically and histologically similar to any pyogenic granuloma, but the initiation of this proliferative lesion is due to a combination of factors such as plaque, altered hormonal balance or trauma. The cause could be attributed to the general effects of progesterone and estrogen on the immune system, gingival vasculature, and ability of progesterone to inhibit collagenase. The lesion either regresses or develops into a residual fibrous mass postpartum. But some of them may be painful, continue to bleed or suppurate, and might interfere with mastication. The following case describes a gingival lesion developed during the 2nd trimester of pregnancy.
Case Report
A 31-year-old female patient, reported to the Department of Periodontology with a chief complaint of a swelling in the maxillary anterior region, which interfered with mastication, speech, and esthetics. The swelling gradually increased in size over the period of 4 months and was associated with bleeding on brushing. Intra-oral examination revealed an ovoid sessile gingival overgrowth measuring 11 × 8 mm, which extended onto the buccal gingiva in relation to left lateral incisor and canine. The surface appeared red, smooth with no ulcerations. Oral hygiene was graded as fair (Fig. 1).
Non-surgical periodontal therapy was initiated to eliminate the inflammatory component. Following parturition, the lesion persisted; therefore, complete excision was carried out under local anesthesia. The histopathologic examination revealed, “Pyogenic granuloma”. Hematoxylin and eosin–stained section showed para-keratinized stratified squamous epithelium with endothelial lined vascular spaces seen within the epithelium. Connective tissue showed bundles of collagen fibers, numerous endothelial lined vascular spaces, proliferating endothelial cells, infiltrate of chronic inflammatory cells, and few areas of dystrophic calcification (Fig. 2). Post-operative visit 1 month after the excision showed good wound healing and improvement in function and esthetics (Fig. 3).
Adverse Pregnancy Outcomes
Adverse pregnancy outcomes are those complications that occur rather than normal live birth, namely preterm birth, stillbirth, low birth weight, and preeclampsia. In pregnancy, immune response plays a vital role in maintaining a healthy equilibrium between mother and fetus. There is a shift in the specific immune response towards a Th2 type as well as activation of the inflammatory cascade during normal pregnancy. An exacerbation of this inflammatory response may lead to adverse outcomes. A correlation between periodontitis and the birth of preterm low-birth-weight (PLBW) infants has been identified where patients with clinical evidence of periodontitis were 7.5 times more likely to have PLBW infants than the control [31]. The proposed theory stated that periodontal infections serve as reservoirs for gram-negative anaerobic organisms and inflammatory mediators which might pose a threat to the fetal-placental unit through hematogenous transmission. Women with PLBW demonstrate worse periodontal parameters and significantly higher GCF levels of IL6, PGE2 and tumor necrosis factor alpha compared to women with full-term birth infants [32]. Periodontitis may also contribute to overall increase of contractions, in turn resulting in pre-term labor onset. However, a recent systematic review and meta-analysis failed to confirm if periodontal disease was an independent risk factor for these adverse pregnancy outcomes [33•]. The evidence that does state the correlation between periodontal disease and adverse pregnancy outcomes justifies the need for non-surgical periodontal therapy in pregnant women to prevent such complications. Contradictorily, a study reported that even though nonsurgical periodontal therapy during pregnancy improved periodontal clinical parameters and decreased periodontal inflammatory biomarker levels, it did not influence inflammatory biomarker level from cord blood nor consistently reduced preterm birth or low birth weight occurrence [34•]. Another common adverse pregnancy outcome that has been associated with periodontal disease is preeclampsia which clinically manifests as hypertension and proteinuria and is usually seen during the 2nd trimester. Since periodontal disease is a multi-factorial condition with low-grade chronic inflammation, it can be hypothesized that patients with periodontal disease have an increased risk of developing preeclampsia. Severe preeclamptic women were 3.78 times more likely to present periodontal diseases, and the elevated levels of pro-inflammatory cytokines like PGE2, tumor necrosis factor alpha, and IL1β in the GCF and serum were consistent with the worsening preeclamptic state [35•].
Acute phase proteins like C-reactive proteins which are seen to increase in chronic periodontitis have shown to manifest preeclamptic complications in pregnant women. Even though periodontal therapy during pregnancy generally demonstrates a positive impact on periodontal disease, its impact on various pregnancy outcomes is conflicting. Thus, there is a need to evaluate novel strategies of preventive and therapeutic approaches at early stages of pregnancy to prevent adverse outcomes.
Oral Contraceptives
Oral contraceptives are among the most widely used classes of drugs in the world today. In the early sixties, the concentrations of estrogen in the oral contraceptives were high resulting in multiple systemic side effects like cardiovascular disorders and malignancy. Therefore, currently, the concentrations of the hormone have been greatly reduced and consist of low doses of estrogens (0.05 mg/day) and/or progestins (1.5 mg/day); {1 mg norethisterone acetate + 0.05 mg ethinylestradiol/mestranol}. The effects of estrogen and progesterone on the periodontium are enhanced during oral contraceptive therapy. Early studies on the effect of oral contraceptives on the periodontium have shown increased gingival exudate, hyperplastic gingivitis, and marked gingival inflammation [36, 37]. The changes on the gingival tissues not only were dependent on the dosage but also the duration of use and are cumulative with time [38]. Oral contraceptives also tend to influence the subgingival microflora and certain Candida species which were able to survive the conditions created by the sex hormones even after 3 years of consumption of the drugs [39]. The gingiva was not the only structure to be affected by these drugs but also the underlying alveolar bone. Along with the gingiva being affected, a stimulatory effect on bone metabolism was identified with decreasing serum levels of bone biomarkers like alkaline phosphatase and higher mean bone mineral density especially with long-term use of oral contraceptives [40, 41]. Thus, suggesting women on long-term oral contraceptives tend to have a lower risk of osteoporosis.
The presence of inflammatory changes in gingival tissues due to oral contraceptives noted in early studies is in contrast to current findings of a lack of gingival inflammation in women using present-day oral contraceptive formulations [42•, 43]. However, gingival tissues may have an exaggerated response to local irritants.
Polycystic Ovary Syndrome
Polycystic ovary syndrome (PCOS) is the most common specific metabolic syndrome affecting 6.5–8% of women of reproductive age according to the National Institutes of Health criteria; women with PCOS have increased prevalence of insulin resistance, central obesity, dyslipidemia, and cardiovascular risk factors. Periodontal disease has also been associated with these systemic conditions. High levels of oxidative stress and inflammatory cytokines like IL6 and C-reactive protein have been identified in both PCOS and periodontal disease, and this might be suggestive of a common pathophysiological mechanism for both these conditions. PCOS and gingival inflammation appear to act in a synergistic manner by increasing the expression of proinflammatory cytokines IL6, IL17, and tumor necrosis factor alpha, and PCOS may have an impact on gingival inflammation and vice versa [44]. But periodontal breakdown and inflammation may be dependent on systemic inflammation [45]. PCOS may affect the composition of the oral microflora and modify the systemic antibody responses and thus potentiating the interaction between the oral microorganisms and gingival inflammation [46]. Considering oxidative stress conditions, both chronic periodontitis and PCOS result in an increase in serum levels of oxidative products and reduced antioxidant status suggesting a link between the two [47•]. A positive correlation between PCOS and periodontal disease has been established and that females with PCOS are at a higher risk of developing periodontitis.
-
3.
Geriatrics
Menopause and its Effects
Menopause is the natural cessation of a woman’s menstrual cycle usually seen between the ages of 45–55 years of age and marks the end of fertility. Due to the declining ovarian function, the changes seen in the oral cavity during menopause is in contrast to the one seen during pregnancy. Oral disturbances are not a common feature of menopause, but few women do suffer from certain conditions like menopausal gingivostomatitis. It is characterized by dry and shiny oral mucosa and gingiva which may appear abnormally pale or bright red with a tendency to bleed easily [48]. More commonly reported gingival changes seen in women of this age are desquamative in nature and are characterized by a smooth and polished gingival surface, mottled appearance. But it is difficult to link sex hormones to its pathogenesis because of its multifactorial etiologies.
Osteoporosis and Menopause
The most common finding seen during the menopausal phase is the increased tendency towards the development of osteoporosis. Reduced levels of estrogen affects the overall collagen metabolism and bone maintenance. Thirty-three percent of the women over the age of 60 years have postmenopausal osteoporosis which includes reduced bone mass, density, and strength and are at a high risk of fractures. Cross-sectional studies have demonstrated decreased bone mass, density, and mineral content in post-menopausal women with the mandible being more affected than maxilla and show proportionality with age [49, 50]. Post-menopausal women are at a higher risk of developing osteoporosis and therefore may also be susceptible to periodontal disease, but the correlation between the two is controversial. Evidence have shown a positive correlation between osteoporosis and periodontal disease in the form of clinical attachment loss, alveolar crest height loss, and tooth loss [51, 52]. Studies linking osteoporosis and periodontal disease are described in Table 3 [53,54,55, 56•, 57]. Despite many studies that display positive correlation between the two conditions, there are a few which states otherwise, especially in terms of bone mineral density and clinical attachment levels [56•, 58]. The possible mechanisms linking osteoporosis and periodontal disease have been discussed. The reduced systemic bone mineral density due to decreased estrogen levels may be associated with the low bone mineral density in the alveolar bones leading to accelerated bone loss following an insult by the periodontopathogenic microorganisms. The second hypothesis is the systemic release of pro-inflammatory cytokines like IL1 and IL6 in response to the skeletal bone loss which may modify the local tissue response to periodontal pathogen [59•]. Although there are a number of studies associating postmenopausal osteoporosis with periodontal disease, the exact cause-effect relationship is yet to be determined [53,54,55, 56•, 57, 58, 59•, 60].
Periodontal Management of the Female Patient
During Puberty
As mentioned previously, sex hormones tend to alter the periodontium during puberty with an exaggerated inflammatory response seen at sites with increased plaque or calculus deposits. Patient or parent education on the periodontal changes during puberty is an important aspect in its management. Preventive care, motivation, and rigorous oral hygiene maintenance form the basis of management of oral changes. Professional periodontal debridement along with frequent oral hygiene reinforcement could be done for managing mild cases of puberty gingivitis. Severe cases are dealt with antimicrobial mouthrinse or local delivery of antiseptic based on the microbial culture analysis [61•]. Depending on the risk of periodontal disease progression, recalls for maintenance should be more frequent. In subjects with hyperplastic gingiva, the inflammatory component can be removed by scaling and root planing, but the fibrotic tissue requires a surgical procedure, i.e. gingivectomy, which can be achieved using scalpel or lasers.
During Menstrual Cycle
The fluctuating levels of sex hormones during the menstrual cycle induce some changes in the periodontal tissues especially those with pre-existing gingivitis. Conventional non-surgical periodontal therapy is usually the mode of treatment for menstrual gingivitis along with reinforced training of oral hygiene measures. For patients with severe periodontal inflammation and tenderness, the frequency of periodontal recall should be 3–4 months [61•]. Some women also experience premenstrual syndrome which consists of various physical and emotional symptoms that may contribute to an exaggerated response to pain. Hence, the clinician may schedule the appointments for periodontal procedures, post menstrual cycle. Other conditions seen during this phase are recurrent aphthous ulcers, increased gag reflex, and other gastrointestinal symptoms. Most of the symptoms are self-limiting and resolves once the cycle begins.
During Pregnancy
The periodontal management for pregnant women should be multidisciplinary. Thorough medical history which includes previous pregnancy complications should be taken and proper diagnosis and treatment planning should be discussed with the patient’s obstetrician. The main goals of treatment in a pregnant patient are to establish and maintain an optimal oral environment. Patient education about the increased tendency of gingival inflammation should be explained. Appropriate oral hygiene instructions should be reinforced, maintained, and monitored throughout pregnancy. In the 1st trimester, the focus is on full clinical periodontal examination and to understand the susceptibility to periodontal disease. Only emergency dental procedures are recommended during this phase, and all elective procedures are avoided as far as possible. Therapy in the 2nd trimester is aimed at controlling active disease and eliminating potential problems. Periodontal debridement should be performed at this stage including severe cases of pregnancy granuloma which is treated by surgical excision extending down to the periosteum to prevent recurrence. This has been described in the above case report. Other treatment modalities like use of diode laser, flash lamp pulsed dye laser, cryosurgery, intralesional injection of ethanol or corticosteroids, and sodium tetradecyl sulfate sclerotherapy have also been proposed [62•, 63]. Routine dental procedures can be done during the early phase of the 3rd trimester, but the most challenging and extensive surgical procedures should be done after delivery. A periodontal maintenance visit should be performed during the early- to mid-3rd trimester due to increased gingival inflammation at that time. The clinician should be aware of the risk category for each drug before prescribing it to a pregnant patient. Thorough knowledge of the medical history, the phase of pregnancy, and the pharmacological agents aids in formulating a treatment plan for managing periodontal diseases during pregnancy.
During Menopause
In contrast to pregnancy, the oral changes seen in menopause are relatively low. But one of the more common diseases seen at this age is osteoporosis. It may be a contributing factor in aggravating pre-existing periodontitis. Periodic review and update of medical history and oral status are essential for such patients. The gold standard for the prevention of severe osteoporosis in osteopenic postmenopausal women is hormone replacement therapy (HRT). The hormone replacements available now contain micronized 17β-estradiol including the standard conjugated equine estrogens and synthetic ethinylestrogens. The progestins that may be used in combination include the older nor-testosterone derivatives like nor-ethisterone acetate, progesterone derivatives like medroxyprogesterone acetate or the newer ones like levonorgestrel and micronized progesterone [64]. The use of HRT has been studied widely and is not only known to reduce the risk of osteoporosis and myocardial infarction and even delay the onset of Alzheimer’s disease, but it also affects the periodontium. Postmenopausal women not taking HRT are 2.1 times more likely to have periodontitis than premenopausal women [65]. Women with early menopause are more likely to develop osteoporosis due to a longer period of estrogen deficiency, and HRT could be more beneficial to such women. Adverse effects have been reported with HRT use such as thromboembolism, coronary artery disease, and breast and endometrial cancer and are highly related to the drugs, dosage, route of administration, and the duration and frequency of use. Therefore, HRT is customized to each individual after analysing the risks and benefits. A newer alternative to HRT called phytoestrogens, a group of plant-based aromatic sterol, has been introduced. They are classified into four main classes: isoflavones, lignans, coumestans, and stilbenes out of which the isoflavones and lignans are the most common. Phytoestrogens have shown lower incidences of menopausal symptoms and osteoporosis in women. They have also shown to affect the periodontal tissues through their antioxidant and estrogenic actions especially on the alveolar bone [66, 67]. Although phytoestrogens may affect the periodontal tissues, more long-term studies are required to analyze its benefits. Other drugs used to treat post-menopausal osteoporosis are bisphosphonates. They inhibit bone resorption by inducing osteoclast apoptosis and cell death and inhibit calcification and hydroxyapatite breakdown. Their ability to inhibit proinflammatory cytokines and modify bone turnover has made them a successful adjunct to mechanical debridement in the management of periodontal disease [68]. In postmenopausal women, bisphosphonates have shown positive results in the improvement of soft and hard tissue periodontal parameters independent of oral hygiene. They can also be considered prior to placing dental implants because they positively affect the quality and quantity of the cortical bone [69]. One of the major side effects involved in the long-term use of bisphosphonates is the risk of developing osteoradionecrosis of the jaw. But long-term clinical trials are necessary to assess its possible occurrence from adjunctive bisphosphonate therapy in periodontal disease. Osteoporosis in post-menopausal women is also associated with decreased calcium absorption and increased excretion and thereby increases the daily calcium requirement. To reduce the progression as well as the risk of future osteoporosis, vitamin D and calcium intake is an important factor. The recommendations for calcium are 1000 mg for premenopausal women and 1500 mg for post-menopausal women. For vitamin D, the doses recommended are 400 IU/day for premenopausal women and 800 IU/day for postmenopausal women. When vitamin D and calcium supplements were added as an adjunct to non-surgical periodontal therapy, women had 44% lesser prevalence of severe periodontitis [70•]. Therefore, calcium and Vitamin D supplements can be considered in managing periodontal disease in post-menopausal women especially in those with a risk of osteoporosis.
Conclusion
The complexities of the female patient are unique to each phase of her life. Female sex hormones play a crucial role, and their cyclic nature is often reflected on the periodontal structures. Substantial evidence collected in this narrative review reflects on the actions of female sex hormones on the periodontium. Although every patient differs, the dental professional should have sound knowledge and understanding of the mechanisms involved that link periodontal disease and female hormone fluctuations. These alterations in the levels of the hormones usually aggravate a pre-existing gingival or periodontal condition. Thus, it becomes important to educate patients about these changes and reinforce proper oral hygiene and maintenance. The management of female patients during various stages of life varies from individual to individual. Patient education, prevention, and oral hygiene care and maintenance form the basis of treating female patients.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Amar S, Chung KM. Influence of hormonal variation on the periodontium in women. Periodontol. 2000;1994(6):79–87.
Warner M, Nilsson S, Gustafsson JÅ. The estrogen receptor family. Curr Opin Obstet Gynecol. 1999;11:249–54.
Richer JK, et al. Differential gene regulation by the two progesterone receptor isoforms in human breast cancer cells. J Biol Chem. 2002;15(277):5209–18.
Vittek J, Hernandez MR, Wenk EJ, Rappaport SC, Southren AL. Specific estrogen receptors in human gingiva. J Clin Endocrinol Metab. 1982;1(54):608–12.
Vittek J, Gordon GG, Rappaport SC, Munnangi PR, Southren AL. Specific progesterone receptors in rabbit gingiva. J Periodontal Res. 1982;17:657–61.
El Attar TM, Roth GD, Hugoson A. Comparative metabolism of 4–14C-progesterone in normal and chronically inflamed human gingival tissue. J Periodontal Res. 1973;8:79–85.
ElAttar TM, Hugoson A. Comparative metabolism of female sex steroids in normal and chronically inflamed gingiva of the dog. J Periodontal Res. 1974;9:284–9.
• Sooriyamoorthy M, Gower DB. Hormonal influences on gingival tissue: relationship to periodontal disease. J Clin Periodontol. 1989;16:201–8. The manuscriptdescribes the mechanism of endogenous sex hormones on healthy and diseased periodontium.
Lindhe J, Brånemark PI. Changes in microcirculation after local application of sex hormones. J Periodontal Res. 1967;2:185–93.
Lindhe J, Brånemark PI. Changes in vascular permeability after local application of sex hormones. J Periodontal Res. 1967;2:259–65.
ElAttar TM. Prostaglandin E2 in human gingiva in health and disease and its stimulation by female sex steroids. Prostaglandins. 1976;11:331–41.
Thomson ME, Pack AR. Effects of extended systemic and topical folate supplementation on gingivitis of pregnancy. J Clin Periodontol. 1982;9:275–80.
Lundgren D, Magnusson B, Lindhe J. Connective tissue alterations in gingivae of rats treated with estrogen and progesterone. A histologic and autoradiographic study. Odontol Revy. 1973;24:49.
Bhat M. Periodontal health of 14–17-year-old US schoolchildren. J Public Health Dent. 1991;51:5–11.
Nakagawa S, Fujii H, Machida Y, Okuda K. A longitudinal study from prepuberty to puberty of gingivitis: correlation between the occurrence of Prevotella intermedia and sex hormones. J Clin Periodontol. 1994;21:658–65.
Tiainen L, Asikainen S, Saxén L. Puberty-associated gingivitis. Community Dent Oral Epidemiol. 1992;20:87–9.
Wojcicki CJ, Harper DS, Robinson PJ. Differences in periodontal disease-associated microorganisms of subgingival plaque in prepubertal, pubertal and postpubertal children. J Periodontol. 1987;58:219–23.
• Gusberti FA, Mombelli A, Lang NP, Minder CE. Changes in subgingival microbiota during puberty: a 4-year longitudinal study. J Clin Periodontol. 1990;17:685–92. The manuscript explains fluctuating levels of sex hormones during puberty that alter the subgingival environment.
Mombelli A, Lang NP, Bürgin WB, Gusberti FA. Microbial changes associated with the development of puberty gingivitis. J Periodontal Res. 1990;25:331–8.
Mühlemann HR. Intermenstrual gingivitis. SSO Schweiz Monatsschr Zahnheilkd. 1948;58:865–85.
Holm-Peedersen P, Löe H. Flow of gingival exudate as related to menstruation and pregnancy. J Periodontal Res. 1967;2:13–20.
Lindhe J, Attström R. Gingival exudation during the menstrual cycle. J Periodontal Res. 1967;2:194–8.
• Baser U, et al. Gingival inflammation and interleukin-1 β and tumor necrosis factoR-alpha levels in gingival crevicular fluid during the menstrual cycle. J Periodontol. 2009;80:1983–90. This manuscript depicts changes in the inflammatory markers in the GCF and gingival inflammation during the menstrual cycle.
Löe H, Silness J. Periodontal disease in pregnancy I. Prevalence and severity Acta Odontol Scand. 1963;21:533–51.
Cohen DW, Friedman L, Shapiro J, Kyle GC. A longitudinal investigation of the periodontal changes during pregnancy. J Periodontol. 1969;40:563–70.
Tilakaratne A, et al. Periodontal disease status during pregnancy and 3 months post-partum, in a rural population of Sri-Lankan women. J Clin Periodontol. 2000;27:787–92.
Jensen J, Liljemark W, Bloomquist C. The effect of female sex hormones on subgingival plaque. J Periodontol. 1981;52:599–602.
• Raber-Durlacher JE, Van Steenbergen TJ, Van der Velden U, De Graaff J, Abraham-Inpijn L. Experimental gingivitis during pregnancy and post-partum: clinical, endocrinological, and microbiological aspects. J Clin Periodontol. 1994;21:549–58. This manuscript describes alteration in the subgingival microflora and inflammatory markers during pregnancy and post-partum with clinical correlation.
Muramatsu Y, Takaesu Y. Oral health status related to subgingival bacterial flora and sex hormones in saliva during pregnancy. Bull Tokyo Dent Coll. 1994;35:139–51.
O’Neil TC. Maternal T-lymphocyte response and gingivitis in pregnancy. J Periodontol. 1979;50:178–84.
Offenbacher S, et al. Periodontal infection as a possible risk factor for preterm low birth weight. J Periodontol. 1996;67:1103–13.
Perunovic ND, et al. The association between periodontal inflammation and labor triggers (elevated cytokine levels) in preterm birth: a cross-sectional study. J Periodontol. 2016;87:248–56.
• Corbella S, et al. Adverse pregnancy outcomes and periodontitis: a systematic review and meta-analysis exploring potential association. Quintessence Int. 2016;47:193–204. This manuscript illustrates that periodontal disease is a weak independent risk factor for adverse pregnancy outcomes.
• da Silva HE, et al. Effect of intra-pregnancy nonsurgical periodontal therapy on inflammatory biomarkers and adverse pregnancy outcomes: a systematic review with meta-analysis. Syst Rev. 2017;6:197. This manuscript describes that non-surgical periodontal therapy does not reduce the inflammatory markers associated with established disease during pregnancy.
• Canakci V, et al. Periodontal disease increases the risk of severe pre-eclampsia among pregnant women. J Clin Periodontol. 2007;34:639–45. This manuscript describes strong association between periodontal disease and preeclampsia.
Kaufman AY. An oral contraceptive as an etiologic factor in producing hyperplastic gingivitis and a neoplasm of the pregnancy tumor type. Oral Surg Oral Med Oral Pathol Oral Radiol. 1969;28:666–70.
Knight GM, Bryan WA. The effects of hormonal contraceptives on the human periodontium. J Periodontal Res. 1974;9:18–22.
Pankhurst CL, Waite IM, Hicks KA, Allen Y, Harkness RD. The influence of oral contraceptive therapy on the periodontium—duration of drug therapy. J Periodontol. 1981;52:617–20.
Brusca MI, et al. The impact of oral contraceptives on women's periodontal health and the subgingival occurrence of aggressive periodontopathogens and Candida species. J Periodontol. 2010;81:1010–8.
Sultana S, Choudhury S, Choudhury SA. Serum alkaline phosphatase and bone mineral density: to assess bone loss in oral contraceptive pill user. Mymensingh Med J. 2002;11:107–9.
Vescovi JD, VanHeest JL, De Souza MJ. Short-term response of bone turnover to low-dose oral contraceptives in exercising women with hypothalamic amenorrhea. Contraception. 2008;77:97–104.
• Mullally BH, Coulter WA, Hutchinson JD, Clarke HA. Current oral contraceptive status and periodontitis in young adults. J Periodontol. 2007;78:1031–6. This manuscript described current day oral contraceptives do not independently alter the periodontium.
Taichman LS, Eklund SA. Oral contraceptives and periodontal diseases: rethinking the association based upon analysis of National Health and Nutrition Examination Survey data. J Periodontol. 2005;76:1374–85.
Ozcaka O, et al. Is there an interaction between polycystic ovary syndrome and gingival inflammatioN? J Clin Periodontol. 2012;39:113.
Porwal S, Tewari S, Sharma RK, Singhal SR, Narula SC. Periodontal status and high-sensitivity C-reactive protein levels in polycystic ovary syndrome with and without medical treatment. J Periodontol. 2014;85:1380–9.
Akcalı A, et al. Association between polycystic ovary syndrome, oral microbiota and systemic antibody responses. PLoS ONE. 2014;9:e108074.
• Saglam E, et al. Evaluation of oxidative status in patients with chronic periodontitis and polycystic ovary syndrome: a cross-sectional study. J Periodontol. 2018;89:76–84. This manuscript describes a common mechanism linking periodontal disease with PCOS.
Friedlander AH. The physiology, medical management and oral implications of menopause. J Am Dent Assoc. 2002;133:73–81.
Kritz-Silverstein D, Barrett-Connor E. Early menopause, number of reproductive years, and bone mineral density in postmenopausal women. Am J Public Health. 1993;83:983–8.
von Wowern N. Bone mineral content of mandibles: normal reference values—rate of age-related bone loss. Calcif Tissue Int. 1988;43:193–8.
Mohammad AR, Brunsvold M, Bauer R. The strength of association between systemic postmenopausal osteoporosis and periodontal disease. Int J Prosthodont. 1996;9:479–83.
Hirai T, Ishijima T, Hashikawa Y, Yajima T. Osteoporosis and reduction of residual ridge in edentulous patients. J Prosthet Dent. 1993;69:49–56.
LaMonte MJ, et al. Five-year changes in periodontal disease measures among postmenopausal females: the Buffalo OsteoPerio study. J Periodontol. 2013;84:572–84.
Penoni DC, et al. Bone density and clinical periodontal attachment in postmenopausal women: a systematic review and meta-analysis. J Dent Res. 2017;96:261–9.
Penoni DC, et al. Association of osteoporosis and bone medication with the periodontal condition in elderly women. Osteoporos Int. 2016;27:1887–96.
• Niramitchainon C, Mongkornkarn S, Sritara C, Lertpimonchai A, Udomsak A. Trabecular bone score, a new bone quality index, is associated with severe periodontitis. J Periodontol. 2020;91:1264–73. This manuscript describes a new systemic bone quality index and its association with periodontal disease.
Savić Pavičin I, Dumančić J, Jukić T, Badel T. The relationship between periodontal disease, tooth loss and decreased skeletal bone mineral density in ageing women. Gerodontology. 2017;34:441–5.
Moeintaghavi A, Pourjavad M, Dadgar S, Tabbakh NS. Evaluation of the association between periodontal parameters, osteoporosis and osteopenia in post-menopausal women. J Dent (Tehran). 2013;10:443.
• Marjanovic EJ, et al. Do patients with osteoporosis have an increased prevalence of periodontal disease? A cross-sectional study Osteoporos Int. 2013;24:1973–9. This manuscript describes possible mechanisms linking periodontal disease and osteoporosis.
Wactawski-Wende J. Periodontal diseases and osteoporosis: association and mechanisms. Ann Periodontol. 2001;6:197–208.
• Otomo-Corgel J. Dental management of the female patient. Periodontol. 2000;2013(61):219–31. This manuscript describes dental treatment strategies in a healthy female patient with periodontal manifestations.
• Zhu YQ, Wang YQ, Tang YC, Li CZ. Initial periodontal therapy for the treatment of gingival pregnancy tumor. Genet Mol Res. 2016;28(15):10–4238. This manuscript describes management of pregnancy-associated pyogenic granuloma.
Zeng H, Yang R, Ding Y. Use of a water-cooled Nd: YAG pulsed laser in the treatment of giant gingival pyogenic granulomas during pregnancy. J Stomatol Oral Maxillofac. 2020;121:305–7.
Norderyd OM, et al. Periodontal status of women taking postmenopausal estrogen supplementation. J Periodontol. 1993;64:957–62.
Haas AN, Rösing CK, Oppermann RV, Albandar JM, Susin C. Association among menopause, hormone replacement therapy, and periodontal attachment loss in southern Brazilian women. J Periodontol. 2009;80:1380–7.
Tanaka K, Freshmen in Dietetic Courses Study II Group, et al. Relationship between soy and isoflavone intake and periodontal disease: the Freshmen in Dietetic Courses Study II. BMC Public Health. 2008;2008(8):39.
• de Cassia BC, et al. Assessing the action of phytoestrogens on alveolar bone tissue of female of ovariectomized rats with induced periodontal disease. Am Int J Contemp Res. 2016;6:5 <b>This manuscript describes the use of phytoestrogens and its beneficial role in reducing periodontal disease</b>undefined.
Akram Z, et al. Efficacy of bisphosphonate as an adjunct to nonsurgical periodontal therapy in the management of periodontal disease: a systematic review. Br J Clin Pharmacol. 2017;83:444–54.
Yajima N, Munakata M, Fuchigami K, Sanda M, Kasugai S. Influence of bisphosphonates on implant failure rates and characteristics of postmenopausal woman mandibular jawbone. J Oral Implantol. 2017;43:345–9.
• Passos-Soares JD, et al. Association between osteoporosis treatment and severe periodontitis in postmenopausal women. Menopause. 2017;24:789–95. This manuscript describes treatment strategies for periodontal disease in post-menopausal women at risk and with osteoporosis.
Funding
Open access funding provided by Manipal Academy of Higher Education, Manipal
Author information
Authors and Affiliations
Contributions
Study conception and design— J.V, A.K.S. Literature search: A.K.S. Writing—original draft: A.K.S. Writing—review and editing: J.V, A.K.S. Case report (surgical procedure and follow up): A.J.F.
Corresponding author
Ethics declarations
Institutional Review Board Statement
Not applicable.
Human and Animal Rights and Informed Consent
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Systemic Diseases
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sathish, A.K., Varghese, J. & Fernandes, A.J. The Impact of Sex Hormones on the Periodontium During a Woman’s Lifetime: a Concise-Review Update. Curr Oral Health Rep 9, 146–156 (2022). https://doi.org/10.1007/s40496-022-00321-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40496-022-00321-0