Abstract
Running economy (RE) has a strong relationship with running performance, and modifiable running biomechanics are a determining factor of RE. The purposes of this review were to (1) examine the intrinsic and extrinsic modifiable biomechanical factors affecting RE; (2) assess training-induced changes in RE and running biomechanics; (3) evaluate whether an economical running technique can be recommended and; (4) discuss potential areas for future research. Based on current evidence, the intrinsic factors that appeared beneficial for RE were using a preferred stride length range, which allows for stride length deviations up to 3 % shorter than preferred stride length; lower vertical oscillation; greater leg stiffness; low lower limb moment of inertia; less leg extension at toe-off; larger stride angles; alignment of the ground reaction force and leg axis during propulsion; maintaining arm swing; low thigh antagonist–agonist muscular coactivation; and low activation of lower limb muscles during propulsion. Extrinsic factors associated with a better RE were a firm, compliant shoe–surface interaction and being barefoot or wearing lightweight shoes. Several other modifiable biomechanical factors presented inconsistent relationships with RE. Running biomechanics during ground contact appeared to play an important role, specifically those during propulsion. Therefore, this phase has the strongest direct links with RE. Recurring methodological problems exist within the literature, such as cross-comparisons, assessing variables in isolation, and acute to short-term interventions. Therefore, recommending a general economical running technique should be approached with caution. Future work should focus on interdisciplinary longitudinal investigations combining RE, kinematics, kinetics, and neuromuscular and anatomical aspects, as well as applying a synergistic approach to understanding the role of kinetics.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Running biomechanics during ground contact, particularly those related to propulsion, such as less leg extension at toe-off, larger stride angles, alignment of the ground reaction force and leg axis, and low activation of the lower limb muscles, appear to have the strongest direct links with running economy. |
Inconsistent findings and limited understanding still exist for several spatiotemporal, kinematic, kinetic, and neuromuscular factors and how they relate to running economy. |
1 Introduction
For competitive runners, decreasing the time needed to complete a race distance is crucial. Consequently, there is a need to understand the determinants of running performance. Several physiological determinants have been identified, which include a high maximal oxygen uptake (\( \dot{V}{\text{O}}_{{ 2 {\text{max}}}} \)) [1, 2], lactate threshold [3, 4], and running economy (RE) [5, 6].
In a heterogeneous group of runners, \( \dot{V}{\text{O}}_{{ 2 {\text{max}}}} \) is strongly related to running performance [7]. However, in a group of runners with a similar \( \dot{V}{\text{O}}_{{ 2 {\text{max}}}} \), \( \dot{V}{\text{O}}_{{ 2 {\text{max}}}} \) cannot be used to discern between those who out-perform others [6]. A measure that can distinguish between good and poor running performers is the rate of oxygen consumed at a given submaximal running velocity, termed RE [5, 8, 9], with lower oxygen consumption (\( \dot{V}{\text{O}}_{2} \)) indicating better RE during steady-state running. For a group of runners with a similar \( \dot{V}{\text{O}}_{{ 2 {\text{max}}}} \), RE can differ by as much as 30 % and is a better predictor of running performance than \( \dot{V}{\text{O}}_{{ 2 {\text{max}}}} \) [6, 8, 10]. Several researchers have reported strong associations between RE and running performance [5, 7, 11, 12]. Additionally, RE differs substantially between elite, trained (recreational), and untrained runners and also between males and females [13–17]. Saunders et al. [18] proposed the following determinants of RE: training, environment, physiology, anthropometry, and running biomechanics.
Studies utilizing interventions show RE can be improved [19], meaning it is a ‘trainable’ parameter [20]. Improvements in RE have ranged from 2 to 8 % using various short-term training modes, such as plyometric [21–23], strength and resistance [24–27], whole-body vibration [28], interval [29–31], altitude [32, 33], and endurance running [34, 35]. In comparison, long-term physiological training can improve RE by 15 % [12]. Jones [12] reported that such an improvement over 9 years was probably a crucial factor in the elite marathon runner’s continued improvement in running performance. For intervention studies concerned with improving RE, the initial fitness level of participants is particularly important [18], with a high initial fitness level perhaps explaining why not all interventions have successfully improved RE [36–39]. Nevertheless, the trainability of RE suggests certain factors affecting RE can be modified. One such factor that can influence RE is an individual’s running biomechanics.
Understanding what constitutes an economical running technique has been the focus of much research. Specific factors include spatiotemporal factors [40, 41], lower limb kinematics [34, 42], kinetics [9, 43, 44], neuromuscular factors [45–48], the shoe–surface interaction [49–54], and trunk and upper limb biomechanics [55–57]. Synthesizing the literature within this field of research has received limited attention, with some still drawing upon descriptors provided up to 20 years ago [18, 58]. Much research has been conducted since, in an attempt to answer the question: is there an economical running technique? Therefore, the purposes of this review are to (1) examine the intrinsic and extrinsic modifiable biomechanical factors affecting RE; (2) assess training-induced changes in RE and running biomechanics; (3) evaluate whether an economical running technique can be recommended; and (4) discuss potential areas for future research directions.
2 Modifiable Biomechanical Factors Affecting Running Economy
Several modifiable biomechanical factors may affect RE. Each factor can be considered either intrinsic (internal) or extrinsic (external). Intrinsic factors refer to an individual’s running biomechanics. These factors can be further categorised as spatiotemporal (parameters relating to changes in and/or phases of the gait cycle, such as ground contact time and stride length); kinematics (the movement patterns, such as lower limb joint angles); kinetics (the forces that cause motion, such as ground reaction force [GRF]); and neuromuscular (the nerves and muscles, such as the activation and coactivation of muscles). The extrinsic factors covered in this review relate to the shoe–surface interaction and focus on footwear, orthotics, and running surface. Evidence for how each factor affects RE is reviewed and discussed.
3 Spatiotemporal Factors
Stride frequency and stride length are mutually dependent and define running speed. If running speed is kept constant, increasing either stride frequency or stride length will result in a decrease of the other. Runners appear to naturally choose a stride frequency or stride length that is economically optimal, or at least very near to being economically optimal. This innate, subconscious fine-tuning of running biomechanics is referred to as self-optimization [34, 42]. Studies supporting this self-optimizing theory generally use acute manipulations of stride frequency or stride length and mathematical curve-fitting procedures to derive the most economical stride frequency and length [40, 59–61].
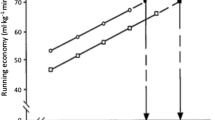
Interestingly, a trained runner’s mathematical optimal stride frequency or stride length is, on average, 3 % faster or 3 % shorter than their preferred frequency or length [40, 59, 61]. Acute and short-term manipulations whereby stride length has been shortened by 3 % show RE to be unaffected [50, 62], whereas stride length deviations greater than 6 % are detrimental to RE [59]. Collectively, these results suggest there is an optimal stride length ‘range’ that trained runners can acutely adopt without compromising their RE. This range appears to be the preferred stride length minus 3 % to the preferred stride length. Importantly, even in a fatigued state, trained runners reduce their stride frequency compared with a non-fatigued state and produce a preferred stride frequency that is similar to their optimal stride frequency achieved in a fatigued state [60]. These results imply that trained runners can dynamically self-optimize their running biomechanics in response to their physiological state. For novice runners, the difference between preferred and mathematically optimal stride frequencies is greater than for trained runners (8 vs. 3 %) [59] (Fig. 1). Therefore, generalizing the principle of an optimal stride length range to all runners should be done with caution, as self-optimization appears to be a physiological adaptation resulting from greater running experience.
Individual differences (selected-optimal) in stride frequency (a) and running cost (b) for novice (left) and trained runners (right) on day 1 (black bars) and day 2 (grey bars). 2 test days were used to assess the reliability of measures and were separated by at least 48 h. RCopt running cost of optimal stride frequency, RCsel running cost of self-selected stride frequency, SFopt optimal stride frequency based on minimal running cost, SFsel self-selected stride frequency. X denotes that optimal stride frequency and, consequently, optimal running cost could not be established in these five trials. Reproduced from de Ruiter et al. [59] by permission of Taylor & Francis Ltd, http://www.tandfonline.com
Similar to stride frequency and stride length, vertical oscillation can be altered. Acute interventions have shown that increasing vertical oscillation leads to increases in \( \dot{V}{\text{O}}_{2} \) [41, 63]. Additionally, vertical oscillation increases when running to exhaustion. However, vertical oscillation changes are minimal and increases in \( \dot{V}{\text{O}}_{2} \) are large [64, 65], meaning several other physiological and biomechanical factors contribute to increases in \( \dot{V}{\text{O}}_{2} \) during fatigue [66, 67]. Furthermore, decreases in vertical oscillation have been shown when individuals run barefoot and their RE improves [50], probably due to a smaller vertical displacement during stance [52]. Yet, it must be noted that shoe mass and other biomechanical changes associated with barefoot running also influence such RE improvements (see Sect. 3.4). Another study has shown that decreasing vertical oscillation can slightly improve RE, but only if the absolute height of the body’s center of mass (CoM) is not changed [68]. Collectively, these results imply that reducing the magnitude of vertical displacement should be encouraged. It is possible that reducing vertical displacement improves RE by reducing the metabolic cost associated with supporting body weight, as a smaller vertical impulse would be produced [69]. Additionally, it could make a runner more mechanically efficient, as a low displacement of the body’s CoM produces a low mechanical energy cost, since the body is performing less work against gravity [70].
Notwithstanding these encouraging results, findings show that female runners have a lower vertical oscillation than their male counterparts, but findings are conflicting regarding whether females are more or less economical than males [13, 16, 71]. Eriksson et al. [72] demonstrated that vertical oscillation could be successfully lowered using visual and auditory feedback, and that runners found it more natural to change vertical oscillation than step frequency. However, to date, only one study has assessed the effect of specifically decreasing a runner’s vertical oscillation. This means research has not tried to manipulate vertical oscillation, in a similar manner to stride frequency and stride length, to determine whether runners have an optimal magnitude of vertical oscillation or whether runners would simply benefit from lowering their vertical oscillation to improve RE.
The time the foot spends in contact with the ground has equivocal results regarding its association with RE. Several studies have failed to find any relationship between ground contact time and RE [9, 42, 73, 74], whilst some have observed a better RE to be associated with longer contact times [75, 76] and others have found the opposite to be true [11, 77]. It is suggested that short ground-contact times incur a high metabolic cost because faster force production is required, meaning metabolically expensive fast twitch muscle fibers are recruited [78, 79]. Conversely, long ground-contact times may incur a high metabolic cost because force is produced slowly, meaning longer braking phases when runners undergo deceleration [77]. Whilst both arguments appear plausible, it has been argued that being able to reduce the amount of speed lost during ground contact is the most important aspect rather than the time in contact with it [77, 80–82]. Combining this with evidence that individuals can produce shorter ground-contact times, but similar deceleration times and RE when forefoot striking compared with rearfoot striking [83], suggests that the time spent decelerating may influence RE.
Another factor that may affect the body’s deceleration is how far ahead of the body the foot strikes the ground. Evidence from step rate manipulation investigations and global gait re-training studies instructing runners to adopt a Pose running method, suggest that significantly decreasing the horizontal distance between the body’s CoM and foot at initial ground contact reduces peak braking and propulsive forces [84, 85] and braking impulses (less speed lost) applied by the runner [86, 87]. Yet, both performance and RE were unaffected during the gait re-training [85], potentially because too many running biomechanics were modified at once. Others have suggested that a runner’s optimal stride frequency is a trade-off between the metabolic cost associated with braking impulses and those associated with swinging the leg [87]. Further work into this braking strategy is required to understand the implications for RE.
Increasing the absolute time spent in the swing phase has been associated with better RE by several researchers [11, 42, 43]. However, others have failed to find any relationship between the two [43, 71]. Findings from Barnes et al. [43] suggest that sex also affects this relationship; however, this has not been corroborated by others [11, 71]. It is conceivable that a longer absolute swing time means runners spend a smaller proportion of the gait cycle in contact with the ground, which is believed to be the metabolically expensive phase of the cycle. It is important to note that the swing and ground contact times will impact the stride frequency and stride length of a runner, and it is perhaps the relationship between all these aspects that should be considered.
3.1 Lower Limb Kinematic Factors
Various kinematic parameters have been identified as being associated with better RE in cross-comparison studies; greater plantarflexion velocity [75], greater horizontal heel velocity at initial contact [75], greater maximal thigh extension angle with the vertical [75], greater knee flexion during stance [42], reduced knee range of motion during stance [88], reduced peak hip flexion during braking [88], slower knee flexion velocity during swing [42, 71], greater dorsiflexion and faster dorsiflexion velocity during stance [71], slower dorsiflexion velocity during stance [88], and greater shank angle at initial contact [42]. Intra-individual comparisons have identified later occurrence of peak dorsiflexion, slower eversion velocity at initial contact, and less knee flexion at push-off as being associated with improved RE [34].
One of the few kinematic variables to have strong support from both cross- and intra-individual comparisons as being beneficial for RE is a less extended leg at toe-off [34, 42, 50, 71, 75, 89]. Evidence has shown that this can be achieved through less plantarflexion and/or less knee extension as the runner pushes off the ground (Fig. 2). Hip extension is also likely to contribute, but studies have typically focused on the knee and ankle angles. Less leg extension could produce greater propulsive force, as identified by Moore et al. [34], by potentially allowing the leg extensor muscles to operate at a more favorable position on the force–length curve and higher gear ratios (GRF moment arm to muscle–tendon moment arm) being obtained. Both strategies could maximize force production [90, 91]. Additionally, less leg extension would reduce the amount of flexion needed during swing by already being partially flexed and potentially reduce the leg’s moment of inertia, lowering the energy required to flex the leg during the swing phase. Previous research has shown that reduced leg moment of inertia lowers the leg’s mechanical demand during the swing phase, as well as the metabolic demand, of walking [92]. Therefore, it is conceivable that a similar relationship exists when running, but this needs investigating.
Differences in knee angle (top) and ankle angle (bottom) at toe-off between pre and post measurements. Pre refers to baseline running biomechanics and post refers to running biomechanics after 10 weeks of running whereby beginner runners improved their running economy and altered their running technique. Reproduced from Moore et al. [34], with permission
Another kinematic during the push-off phase that has been associated with better RE is stride angle, which is defined as the angle of the parable tangent of the CoM at toe-off [11, 93, 94]. Larger stride angles appear to be beneficial for lowering \( \dot{V}{\text{O}}_{2} \) and can be achieved by either increasing swing time or decreasing stride length. However, the system (Optojump Next) used by each study [11, 93, 94] only tracks the foot during ground contact and not the CoM. Therefore, only inferences can be made regarding the trajectory angle of the CoM and other possible kinematic changes. Future work focusing on the push-off phase should assess CoM trajectory in relation to kinematics and kinetics, as increasing swing time would also increase the vertical displacement of the CoM based on previous calculations [11, 93, 94] and observations [95]. Crucially, research suggests that increases to these spatiotemporal parameters appear to have contradictory relationships with RE [11, 41–43, 63].
Foot strike patterns have been implicated as a modifiable factor affecting RE [96], with some researchers arguing that the most economical strike pattern is forefoot striking, even when RE is not assessed [97–99]. However, empirical evidence refutes this claim. Findings shows no difference in RE between rearfoot and forefoot striking at slow (≤3 m·s−1) [51, 83, 100, 101], medium (3.1–3.9 m·s−1) [83, 100, 101], and fast speeds (≥4.0 m·s−1) [83, 100] or rearfoot and midfoot striking at medium speeds [76]. However, others have shown rearfoot striking to be more economical than midfoot striking at slow running speeds [102]. Interestingly, habitual forefoot strikers can change to a rearfoot strike without detrimental consequences to RE, while an imposed forefoot strike in habitual rearfoot strikers produces worse RE at slow and medium speeds [100]. Based on the current literature, foot strike appears to have a negligible effect upon RE, with only habitual rearfoot strikers likely to experience a worsening of RE by switching foot strike patterns.
3.2 Kinetic Factors
Early research reported that RE was proportional to the vertical component of GRF (e.g., force required to support body weight) and was termed the ‘cost of generating force’ hypothesis [79, 103, 104]. However, later investigations have used a task-by-task approach to partition RE into individual biomechanical tasks [105]. Such work has demonstrated that braking (decelerating the body) and propulsive (accelerating the body) forces also incur metabolic costs [105]. Typically, the three components of GRF (anterior-posterior, medial–lateral, and vertical) have been independently assessed, with evidence suggesting lower vertical impact force [42], lower peak medial–lateral force [42, 75], lower anterior–posterior braking force [73], and higher anterior–posterior propulsive force [34] are economical. However, numerous studies have also failed to identify similar associations between RE and individual GRF components [26, 73, 74].
To understand the metabolic costs incurred during running Arellano and Kram [106] advocate using a synergistic approach, rather than the ‘cost of generating force’ hypothesis or task-by-task approach. Using this approach, the vertical force (supporting body weight) and forward propulsive force (accelerating the body) incur the greatest metabolic cost (Fig. 3). However, very few biomechanical studies have utilized such an approach. Storen et al. [74] demonstrated that it could be usefully applied as they found significant relationships between the summation of peak vertical and anterior–posterior forces and 3-km performance (r = −0.71) and RE (r = −0.66). Their findings show that lower forces were associated with a better running performance and RE. Additionally, Moore et al. [107] reported near perfect alignment of the angle of the resultant GRF vector (all three components) with the angle of the longitudinal leg axis vector during propulsion when novice runners improved their RE. This change in alignment was associated with a change in RE (r s = 0.88), suggesting that minimizing the muscular effort of generating force during propulsion is beneficial to RE [107].
The a cost of generating force, b individual task-by-task, and c synergistic task-by-task approach partition the net metabolic cost of human running into its biomechanical constituents. The cost of generating force approach and the individual task-by-task approach both illustrate that body weight support is the primary determinant of the net metabolic cost of human running. In the individual task-by-task approach, forward propulsion represents the second largest determinant. The individual task-by-task approach leads to an overestimation, while the synergistic task-by-task approach suggests that the synergistic tasks of body weight support and forward propulsion are the primary determinants of the net metabolic cost of human running. Note that leg swing and lateral balance exact a relatively small net metabolic cost. If we sum all the biomechanical tasks, the synergistic task-by-task approach accounts for 89 % of the net metabolic cost of human running, leaving 11 % of unexplained metabolic cost, and the cost of generating force accounts for 80 %, leaving 20 % of unexplained metabolic cost. Reproduced from Arellano and Kram [106], with permission from Oxford University Press
Associations have also been found between GRF impulses and RE, with lower braking [87], total, and net vertical impulses related to a better RE [9]. However, this finding is not consistent in the literature [77]. Through collectively considering the deceleration and acceleration (anterior–posterior) impulses, a runner’s change in momentum can be determined. One pilot study has utilized this technique, but reported similar changes in momentum pre and post a 10-week running program that improved RE [107]. The authors suggested that such a short-term training program might not have been long enough to induce modifications in momentum [107]. It is also conceivable that a synergistic approach should be applied to momentum and speed lost during braking.
The magnitude of the GRF during running has a linear relationship with the body’s vertical displacement [108], suggesting the leg acts like a spring during ground contact [44]. Therefore, use of the spring-mass model to describe the body’s bounce during the support phase of running has been widespread. The springs’ stiffness is the ratio of deformation (vertical displacement) to the force applied to it (vertical GRF) and therefore represents the stiffness of the whole body’s musculoskeletal system [109]. Leg stiffness represents the ratio of maximal vertical force to maximal vertical leg spring compression [110]. Greater leg stiffness has been associated with a better RE [44], whilst fatiguing runs to volitional exhaustion have led to reductions in leg stiffness [64, 65]. Furthermore, alterations to extrinsic factors, such as increasing surface compliance, can lead to decreases in leg stiffness, resulting in a worse RE [111]. Running in minimalist footwear can increase leg stiffness and improve RE compared with traditional and cushioned footwear [112, 113]. Interestingly, leg stiffness is predominately associated with ground-contact time rather than step frequency [114]. Thus, to try and increase leg stiffness, runners are advised to focus on shortening ground-contact time rather than increasing step frequency. Such an approach may be beneficial for RE improvements.
As leg stiffness represents the stiffness of the whole musculoskeletal system, several factors relating to stiffness are unmodifiable, such as muscle crossbridges and tendon stiffness. However, neuromuscular activation is a modifiable characteristic that can modulate stiffness.
3.3 Neuromuscular Factors
The preactivation of muscles prior to ground contact, termed muscle tuning, is believed to increase muscle–tendon stiffness [77], potentially enhance muscular force generation via the stretch–shortening cycle (SSC) [115], and affect leg geometry at initial ground contact [116–118]. Nigg et al. [119] studied the effect of shoe midsole characteristics on RE and preactivation, and, whilst no overall shoe-dependent changes were found in either variable, systematic individual changes in vastus medialis preactivation were evident. Runners who produced higher vastus medialis preactivation independent of shoe condition also had a higher \( \dot{V}{\text{O}}_{2} \) [119]. However, given the small changes in RE (<2 %) the differences may be due to test–retest measurement error and are unlikely to represent a meaningful change in RE [120].
Greater muscular activity of the lower limbs has been reported as a potential mechanism behind increasing \( \dot{V}{\text{O}}_{2} \) and thus is seen as detrimental to RE [73]. The intuitive link between muscle activity and RE stems from muscles needing to utilize oxygen to activate, and thereby, control movement patterns and stabilize joints. Therefore, greater muscle activation, as typically measured using surface electromyography (EMG), is thought to require a higher \( \dot{V}{\text{O}}_{2} \) and lead to a worsening of RE. In line with this, findings have shown a higher activation of the gastrocnemius during propulsion and of the biceps femoris during braking and propulsion to be associated with higher \( \dot{V}{\text{O}}_{2} \) [73]. Additionally, Abe et al. [45] found an increase in \( \dot{V}{\text{O}}_{2} \) during a prolonged run was associated with a decrease in the ratio of eccentric–concentric vastus lateralis activity. This change in eccentric–concentric ratio was due to an increase in activity during propulsion (concentric phase). Collectively, these findings suggest that needing to utilize greater muscle activation to propel the runner forwards, possibly due to a reduced efficiency of the SSC, is detrimental to RE.
Bourdin et al. [121] support this notion, as they found lower eccentric–concentric ratios of vastus lateralis activity were associated with a higher energetic cost of running. Importantly, however, this relationship was more prominent when inter-individual differences were being assessed and was weaker when intra-individual differences were considered. Sinclair et al. [88] also found a higher activity of the vastus medialis to be related to a worse RE when comparing different runners. Conversely, Pinnington and colleagues [122, 123] have suggested that intra-individual increases in \( \dot{V}{\text{O}}_{2} \) associated with running on sand compared with on a firm surface are partially due to increased activation of the quadriceps and hamstrings muscles involved in greater hip and knee range of motion. However, as \( \dot{V}{\text{O}}_{2} \) and EMG data were collected in separate studies, causal interpretations should be made with caution. Larger intra- and inter-individual variations in lower limb muscle activity duration and timing of peak activation have been reported in novice compared with experienced runners [124], suggesting that greater running exposure may alter neuromuscular control. However, longitudinal investigations are needed to confirm this.
Conflicting results have also been reported for the role of muscular coactivation in relation to RE [46–48], whereby muscular coactivation is defined as the simultaneous activation of two muscles. Heise et al. [47] found a negative relationship between RE and the coactivation of the rectus femoris and gastrocnemius, suggesting coactivation of biarticular muscles is economical, whereas Moore et al. [48] reported a positive relationship. Furthermore, muscular coactivation of the proximal agonist–antagonist leg muscles, rectus femoris and biceps femoris, has also been shown to have a positive association with RE, meaning such coactivation is detrimental to RE [46, 48]. Coactivation of the proximal thigh antagonist–agonist muscles occurs during the loading phase of stance as the knee flexes. Without such coactivation, it is likely that the leg would collapse [125], but essentially the muscles are performing opposing movements. Using two muscles to control such a movement would therefore incur a greater metabolic cost than using one muscle, potentially decreasing the efficiency of the SSC.
Investigations into the effect of orthotics on muscular activation during ground contact and RE have provided inconsistent findings. Kelly et al. [126] reported that alterations to muscular activity when wearing orthotics during a 1-h run were not accompanied by changes in RE. Contrastingly, Burke and Papuga [127] observed improvements in RE when runners ran in custom-made orthotics rather than shoe-fitted insoles, yet there were no changes in lower limb muscular activity. However, the mass of the different orthotics used by Burke and Papuga [127], and the potential effect the orthotics had on running biomechanics, were not assessed and may have influenced their findings.
3.4 Shoe–Surface Interaction Factors
There is a general consensus that running in traditional running trainers is detrimental to RE compared with running barefoot or in lightweight, minimalist trainers, due to the added shoe mass [49–52, 128, 129]. A recent meta-analysis suggested that a shoe mass (per pair) of less than 440 g does not affect RE, but a shoe mass greater than 440 g negatively affects RE [129]. However, when shoe mass is taken into account, evidence regarding footwear effects on RE is equivocal due to different methodologies used. Mathematically correcting for different footwear mass when expressing \( \dot{V}{\text{O}}_{2} \) in relative terms supports the above statement that running in traditional trainers is detrimental to RE compared with barefoot or minimalist footwear running [50]. However, strapping weights equal to the mass of a shoe to participants’ feet results in either similar RE [52] or worse RE when barefoot compared with shod [49]. One reason for this discrepancy is that mathematically adjusting \( \dot{V}{\text{O}}_{2} \) technically adjusts the whole body’s mass rather than the foot’s mass and does not take into account the decrease in lower limb moment of inertia. When the foot’s CoM is altered (weights strapped to the top of foot) \( \dot{V}{\text{O}}_{2} \) is worse when barefoot [49], but when the foot’s CoM is unchanged (weights evenly distributed on the foot), \( \dot{V}{\text{O}}_{2} \) is similar between barefoot and shod conditions [52]. Therefore, changes to lower limb moment of inertia, and not just shoe mass, appear to affect RE. Findings from Scholz et al. [130] support this notion by showing greater lower limb moment of inertia was associated with higher \( \dot{V}{\text{O}}_{2} \). Other shoe characteristics, such as stiffness [131], comfort [132], and cushioning [133], are likely to effect RE and thus, may have also contributed to the equivocal findings regarding footwear effects on RE when shoe mass is taken into account. However, if shoe mass is not adjusted for, running barefoot or in lightweight, minimalist trainers improves RE compared with traditional running trainers (shoe mass >440 g).
Changing footwear can also change the level of cushioning underfoot. Frederick et al. [134] proposed the ‘cost of cushioning’ hypothesis, stating that actively cushioning the body whilst running may incur a metabolic cost. Therefore, shoes with limited cushioning or no cushioning (such as being barefoot) would result in an individual having to actively cushion the body using the lower limb muscles [117] and lead to an increase in \( \dot{V}{\text{O}}_{2} \). Some evidence to support this claim is provided by Franz et al. [49], who found that running in shoes with increasing mass had a lower metabolic power demand than running barefoot with increasing mass strapped to their feet. These results therefore show that running without cushioning has a higher metabolic demand than running with cushioning, even when added shoe mass is similar. However, results from Divert et al. [52] suggest it may be mechanical energy that is increased rather than \( \dot{V}{\text{O}}_{2} \) when barefoot. This means that barefoot running leads to mechanical efficiency improvements due to greater work being done for the same \( \dot{V}{\text{O}}_{2} \) compared with shod running.
Further, it appears there is an ‘optimal’ level of surface cushioning for good RE. When running barefoot on a treadmill, 10 mm of surface cushioning was more beneficial for RE than no surface cushioning and 20 mm of surface cushioning [53]. When considering natural running terrain, Pinnington and Dawson [122] found running on grass elicited a lower \( \dot{V}{\text{O}}_{2} \) than running on sand. This is likely due to the damping effects of sand, leading to an increase in mechanical work done during stance [135]. Therefore, a firmer surface that returns the energy it absorbs will benefit a runner’s RE. Moreover, a firm surface with reduced stiffness, and thus greater compliance, will return more energy due to the surface’s elastic rebound and improve RE [111].
This theory can also be applied to running shoes, as Worobets et al. [54] showed that a softer shoe, which was more compliant and lost less energy during impact than a control shoe, improved RE. Additionally, shoes with a high forefoot bending elasticity can increase propulsive force and reduce contact time and gastrocnemius muscle activation during slow (<3 m·s−1), but not medium (3.1–3.9 m·s−1), running speeds compared with a flexible forefoot region [136]. Such shoes may therefore improve RE due to enhancing propulsion; however, no \( \dot{V}{\text{O}}_{2} \) data were gathered during the study, so direct associations cannot be made. Consequently, it is likely that a medium level of cushioning, that returns energy, is beneficial for RE compared with the shoe–surface cushioning being too compliant or too hard.
Footwear (or lack of) can also affect running biomechanics. Several modifications to running biomechanics may potentially benefit RE, whilst others may not. For example, in comparison with shod running, barefoot running can shorten ground contact time and stride length [49–52, 128, 137–140], increase knee flexion at initial contact [139], increase leg stiffness [52, 139, 141, 142], decrease vertical oscillation [50, 138], increase propulsive force [143], and reduce plantarflexion at toe-off [50, 139]. The most commonly cited change when running barefoot is a more anterior foot strike pattern brought about by a flatter foot, such as switching from a rearfoot to a forefoot strike pattern [50, 98, 137, 139, 140, 142, 144]. However, evidence shows many confounding variables affect foot strike, including speed [97, 145], surface stiffness [146], stride length [50], and familiarization with barefoot running [147]. Therefore, footwear (or lack of) alone cannot explain changes in foot strike. Based on the several findings above, it can be suggested that acute exposure to running barefoot may be beneficial for RE, especially if performed on a surface with a medium level of cushioning. Aside from acute exposure, the effect of individual adaptations due to short- and long-term exposure to barefoot running on RE and running biomechanics is currently unknown.
3.5 Trunk and Upper Limb Biomechanical Factors
The relationship between RE and trunk and upper body biomechanics has received limited research attention compared with lower limb biomechanics. Swinging the arms during running plays an important role as it contributes to vertical oscillation [55, 56]; counters vertical angular momentum of the lower limbs [148]; and minimizes head, shoulder, and torso rotation [149, 150]. Eliminating arm swing by placing the hands on top of the head can be detrimental to RE [41, 149], whilst placing the hands behind the back or across the chest has provided inconsistent findings [41, 56, 63, 149, 150]. However, there is no evidence to suggest that individuals can alter arm kinematics to improve RE and thus, running performance. Therefore, based on current evidence, individuals are encouraged to maintain their natural arm swing whilst running.
Suppressing arm swing can alter several lower limb biomechanics and kinetics. For example, restraining the arms behind the back and across the chest decreases peak vertical force, increases peak hip and knee flexion angles during stance, and reduces knee adduction during stance [151]. These biomechanical changes appear to be due to the loss of arm motion rather than the body’s CoM moving position [151], suggesting that arm motion plays an integral role in an individual’s running technique. Further, the greater knee flexion and reduced peak vertical force observed when arm swing is suppressed suggests that leg stiffness decreases, which may explain the change in RE found in some studies [41, 56, 149]. However, currently, the relationship between leg stiffness and arm motion during running is unknown.
It has been suggested that a forward trunk lean during running improves RE [58], based on findings from Williams and Cavanagh [42]. Yet, a forward lean has also been implicated as detrimental to RE. Hausswirth et al. [57] compared the \( \dot{V}{\text{O}}_{2} \) during a marathon run (2 h, 15 min) with that during a 45-minute run and found the marathon run had a higher \( \dot{V}{\text{O}}_{2} \) and greater forward trunk lean. However, this finding should be interpreted in light of the other modifications to running biomechanics when comparing the marathon run with the 45-min run, such as the 13 % shorter stride lengths. It is possible that shortening the stride lengths by this amount incurred the highest \( \dot{V}{\text{O}}_{2} \) rather than the forward lean. Additionally, the biomechanical changes could be due to muscular fatigue resulting from the difference in running time between the two conditions (1 h, 30 min), meaning muscular fatigue could have led to increases in \( \dot{V}{\text{O}}_{2} \).
For women runners, breast kinematics also have the potential to affect RE and running biomechanics. Evidence shows that breast kinematics can affect running kinetics [152], trunk lean via changes in breast support [153], and lower limb biomechanics, in particular knee angle and step length [154]. These findings imply there may be alterations to RE, particularly if the changes in step length are greater than 3 % of the preferred step length. Further work that simultaneously assesses RE, breast kinematics, breast support, and lower limb biomechanics is warranted to assess whether there is a direct association between the measures.
4 Simultaneously Modifying Running Biomechanics and Running Economy Through Training
Short- and mid-term training interventions (3–12 weeks) have been conducted to assess relationships between running biomechanics and RE. But to date, no long-term training interventions have been performed. Early interventions primarily focused on spatiotemporal factors, with Morgan et al. [155] showing that trained runners with uneconomical stride lengths could be retrained using audio-feedback over 3 weeks to produce mathematically derived optimal stride lengths and improved RE. In contrast, Messier and Cirillo [95] failed to find improvements in RE when using verbal and visual feedback for 5 weeks to change specific running biomechanics, such as longer stride lengths, shorter ground-contact time, and reduced vertical oscillation. However, optimal stride length was not mathematically determined prior to the intervention, meaning suitable procedures were not used and several running biomechanics either were not modified or, in the case of vertical oscillation, actually increased after the intervention. Bailey and Messier [156] also found that if runners were able to freely choose their stride length over 7 weeks, there was no change in RE. Similarly, if runners were restricted to their initial freely chosen stride length over 7 weeks, RE was unaffected [156].
Interventions concerned with instructing runners to retrain their running biomechanics towards a specific global running technique, such as Pose, Chi and midstance to midstance running, has generally resulted in either no improvement in RE [62, 85] or a worsening of RE [157]. Whilst these techniques are often advocated as efficient forms of running [157, 158], and all the interventions led to modified running biomechanics, currently there appears to be no evidence to substantiate the claims that they benefit RE. It is conceivable that the failure of global running techniques to improve RE is because they are not targeting the right running biomechanics or because they are trying to change too many at the same time.
Running gait retraining has also focused on reducing injury risk [159–162], but only one study has assessed the effect of such retraining on RE as well [163]. Clansey et al. [163] provided trained runners with gait re-training using real-time visual feedback over 3 weeks to modify impact-loading variables associated with tibial stress fracture risk. Runners reduced peak tibial acceleration and loading rates without changing RE. Thus, gait re-training to reduce injury risk can be performed without necessarily affecting running performance. This is possibly because the gait alterations were predominantly during the impact phase and have minimal effect on RE, as individuals increased plantarflexion at initial contact and exhibited a more anterior foot strike.
Moore et al. [34] reported that novice runners could self-optimize their running gait over 10 weeks of running training, with 94 % of the variance of change in RE explained by less knee extension at toe-off, a later occurrence of peak dorsiflexion, and slower eversion velocity at initial contact. Furthermore, trained, habitually shod runners can improve their RE when running in minimalist footwear after a 4-week intervention exposing them to running in minimalist footwear [96]. Although very few running gait parameters were assessed by Warne and Warrington [96], runners did exhibit a more anterior foot strike when more economical. Whilst collectively these results support short-term biomechanical self-optimization to running training, a previous investigation failed to find RE improvements and biomechanical changes in trained runners after 6 weeks of running [36]. Consequently, novice runners may be more responsive to self-optimization in the short-term than trained runners; however providing trained runners with a novel stimulus, such as different footwear, can lead to short-term self-optimization. Thus, self-optimization is a physiological adaptation to running acquired through greater experience of the stimulus. For trained runners, the majority of this physiological adaptation may have already occurred. A summary of how training interventions have affected RE is presented in Fig. 4.
5 Is there an Economical Running Technique?
Based on the literature, several modifiable factors that can potentially improve RE have been identified, as well as factors that have conflicting or limited findings regarding their relationship with RE (Table 1). From this summary, it is clear that biomechanics during ground contact play an important role. Furthermore, evidence shows that many of the running biomechanics identified occur during propulsion, suggesting that this phase has the strongest direct links with RE. However, theoretical deceleration strategies, such as short braking times and minimizing the speed lost during braking, may translate to more economical strategies in the propulsive phase and mediate the relationship between propulsion and RE. Therefore, utilizing the principles of the SSC is encouraged.
Considering the empirical evidence, one economical running strategy could be aiming to shorten ground-contact times whilst maintaining stride frequency, which may facilitate greater leg stiffness, larger stride angles, and longer swing times. However, such a strategy may increase vertical oscillation and encourage greater muscular activity during propulsion. Another strategy could involve aligning the resultant GRF more closely with the leg axis during propulsion. This may help minimize muscular activity and agonist–antagonist coactivation and could be produced as a result of reducing leg extension at toe-off.
An experienced runner’s naturally chosen stride length is self-optimized to within 3 % of the mathematically derived optimal. Deviating between naturally chosen and mathematically optimal will only have a negligible effect on RE. However, novice runners have not acquired the running experience necessary to self-optimize as effectively. Therefore, a short-term running training program for novice runners can lead to running biomechanics being modified to benefit RE. However, long-term running training has seldom been investigated. Consequently, longitudinal investigations assessing the development of running biomechanics in both novice runners and experienced runners are required to better understand self-optimization for RE improvements.
Notwithstanding the identified modifiable factors affecting RE, prescribing an economical way of running has its limitations based on the current empirical evidence. The majority of studies have used cross-comparison methodologies or are restricted to one running population. Additionally, it is evident from the numerous studies analyzing intra-individual changes that group differences, which statistically hold more power, provide limited conclusions of modifications to running biomechanics [88, 119, 164]. Also, very few studies have assessed running biomechanics during the swing phase, even though current findings indicate the position of the CoM and leg during this phase may be crucial to conserving energy and reducing \( \dot{V}{\text{O}}_{2} \). Exploring running biomechanics during swing and the interaction with stance-phase biomechanics is recommended in future work. Furthermore, the role of unmodifiable factors and how they may interact with modifiable factors is an area requiring investigation. For example, Cavanagh and Williams [40] reported that individuals with long legs had a larger increase in \( \dot{V}{\text{O}}_{2} \) when shortening their strides compared with lengthening them. In contrast, individuals with shorter legs had a larger increase in \( \dot{V}{\text{O}}_{2} \) when lengthening their stride than when shortening it.
Biomechanical case studies of economical runners have not been published, but could provide interesting findings if an in-depth runner profile was provided. Such a profile would need to encompass factors such as running biomechanics, anatomical structures, functional capacity (e.g., flexibility, muscular strength, and stiffness), shoe degradation, injury history, and training protocols [165]. Whilst only the former have been discussed here, the interaction between an individual’s anatomical structures—such as foot morphology, leg length, and tendon stiffness—and their running biomechanics is likely to be influential upon RE. This is certainly a direction for future research to pursue, as it could identify novel relationships and interactions that inform larger, cohort studies.
6 Conclusion
One of the determining factors of running performance is RE. Modifiable running biomechanical factors that affect RE include spatiotemporal factors, lower limb kinematics, kinetics, neuromuscular factors, shoe–surface interactions, and trunk and upper limb biomechanics. Several intrinsic factors that appear to benefit RE are a self-selected stride length with a 3 % shorter stride length range, lower vertical oscillation, greater leg stiffness, low lower limb moment of inertia, alignment of the GRF and leg axis vectors, less leg extension at toe-off, larger stride angles, maintaining arm swing, low muscle activation during propulsion, and low antagonist–agonist thigh coactivation. In regards to extrinsic factors, better RE was found to be associated with a firm, compliant shoe-surface interaction and being barefoot or wearing lightweight shoes. Other modifiable biomechanical factors, such as ground contact time, impact force, anterior–posterior forces, trunk lean, lower limb biarticular muscle coactivation, and orthotics, presented inconsistent relationships with RE. Collectively, the evidence shows that many of the running biomechanics identified occur during propulsion, suggesting that this phase has the strongest direct links with RE. However, recurring methodological problems exist within the literature, such as cross-comparisons, assessing variables in isolation, and acute to short-term interventions. Further, intra-individual differences due to unmodifiable factors limit the findings of cross-comparisons, and future research should look to investigate longitudinal interventions and assess runners on an individual basis. Consequently, recommending an economical running technique should be approached with caution. Directions for further work within the field should focus on a synergistic approach to assessing kinetics as well as integrated approaches combining \( \dot{V}{\text{O}}_{2} \), kinematics, kinetics, and neuromuscular and anatomical aspects to increase our understanding of economical running technique.
References
Billat VL, Demarle A, Slawinski J, et al. Physical and training characteristics of top-class marathon runners. Med Sci Sports Exerc. 2001;33:2089–97.
Foster C. VO2 max and training indices as determinants of competitive running performance. J Sports Sci. 1983;1:13–22.
Farrell PA, Wilmore JH, Coyle EF, et al. Plasma lactate accumulation and distance running performance. Med Sci Sports. 1979;11:338–44.
Tanaka K, Matsuura Y. Marathon performance, anaerobic threshold, and onset of blood lactate accumulation. J Appl Physiol Respir Environ Exerc Physiol. 1984;57:640–3.
Conley DL, Krahenbuhl GS. Running economy and distance running performance of highly trained athletes. Med Sci Sports Exerc. 1980;12:357–60.
Morgan DW, Baldini FD, Martin PE, et al. Ten kilometer performance and predicted velocity at VO2max among well-trained male runners. Med Sci Sports Exerc. 1989;21:78–83.
Pollock ML. Submaximal and maximal working capacity of elite distance runners. Part I: cardiorespiratory aspects. Ann N Y Acad Sci. 1977;301:310–22.
Daniels JT. A physiologist’s view of running economy. Med Sci Sports Exerc. 1985;17:332–8.
Heise GD, Martin PE. Are variations in running economy in humans associated with ground reaction force characteristics? Eur J Appl Physiol. 2001;84:438–42.
Costill DL, Thomason H, Roberts E. Fractional utilization of the aerobic capacity during distance running. Med Sci Sports. 1973;5:248–52.
Santos-Concejero J, Tam N, Granados C, et al. Stride angle as a novel indicator of running economy in well-trained runners. J Strength Cond Res. 2014;28:1889–95.
Jones AM. The physiology of the world record holder for the women’s marathon. Int J Sports Sci Coach. 2006;1:101–16.
Bransford DR, Howley ET. Oxygen cost of running in trained and untrained men and women. Med Sci Sports Exerc. 1977;9:41–4.
Morgan DW, Bransford DR, Costill DL, et al. Variation in the aerobic demand of running among trained and untrained subjects. Med Sci Sports Exerc. 1995;27:404–9.
Daniels J, Daniels N. Running economy of elite male and elite female runners. Med Sci Sports Exerc. 1992;24:483–9.
Helgerud J, Storen O, Hoff J. Are there differences in running economy at different velocities for well-trained distance runners? Eur J Appl Physiol. 2010;108:1099–105.
Barnes KR, Kilding AE. Running economy: measurement, norms, and determining factors. Sports Med Open. 2015;1:8.
Saunders PU, Pyne DB, Telford RD, et al. Factors affecting running economy in trained distance runners. Sports Med. 2004;34:465–85.
Barnes KR, Kilding AE. Strategies to improve running economy. Sports Med. 2015;45:37–56.
Jones AM, Carter H. The effect of endurance training on parameters of aerobic fitness. Sports Med. 2000;29:373–86.
Saunders PU, Telford RD, Pyne DB, et al. Short-term plyometric training improves running economy in highly trained middle and long distance runners. J Strength Cond Res. 2006;20:947–54.
Spurrs RW, Murphy AJ, Watsford ML. The effect of plyometric training on distance running performance. Eur J Appl Physiol. 2003;89:1–7.
Turner AM, Owings M, Schwane JA. Improvement in running economy after 6 weeks of plyometric training. J Strength Cond Res. 2003;17:60–7.
Barnes KR, Hopkins WG, McGuigan MR, et al. Effects of resistance training on running economy and cross-country performance. Med Sci Sports Exerc. 2013;45:2322–31.
Guglielmo LGA, Greco CC, Denadai BS. Effects of strength training on running economy. Int J Sports Med. 2009;30:27–32.
Paavolainen L, Hakkinen K, Hamalainen I, et al. Explosive-strength training improves 5-km running time by improving running economy and muscle power. J Appl Physiol. 1999;86:1527–33.
Støren Ø, Helgerud J, Støa EM, et al. Maximal strength training improves running economy in distance runners. Med Sci Sports Exerc. 2008;40:1087–92.
Cheng CF, Cheng KH, Lee YM, et al. Improvement in running economy after 8 weeks of whole-body vibration training. J Strength Cond Res. 2012;26:3349–57.
Barnes KR, Hopkins WG, McGuigan MR, et al. Effects of different uphill interval-training programs on running economy and performance. Int J Sports Physiol Perf. 2013;8:639–47.
Denadai BS, Ortiz MJ, Greco CC, et al. Interval training at 95% and 100% of the velocity at VO2 max: effects on aerobic physiological indexes and running performance. Appl Physiol Nutr Metab. 2006;31:737–43.
Franch J, Madsen K, Djurhuus MS, et al. Improved running economy following intensified training correlates with reduced ventilatory demands. Med Sci Sports Exerc. 1998;30:1250–6.
Saunders PU, Pyne DB, Gore CJ. Endurance training at altitude. High Alt Med Biol. 2009;10:135–48.
Saunders PU, Telford RD, Pyne DB, et al. Improved running economy in elite runners after 20 days of simulated moderate-altitude exposure. J Appl Physiol. 2004;96:931–7.
Moore IS, Jones AM, Dixon SJ. Mechanisms for improved running economy in beginner runners. Med Sci Sports Exerc. 2012;44:1756–63.
Beneke R, Hutler M. The effect of training on running economy and performance in recreational athletes. Med Sci Sports Exerc. 2005;37:1794–9.
Lake MJ, Cavanagh PR. Six weeks of training does not change running mechanics or improve running economy. Med Sci Sports Exerc. 1996;28:860–9.
Ramsbottom R, Williams C, Fleming N, et al. Training induced physiological and metabolic changes associated with improvements in running performance. Br J Sports Med. 1989;23:171–6.
Ferrauti A, Bergermann M, Fernandez-Fernandez J. Effects of a concurrent strength and endurance training on running performance and running economy in recreational marathon runners. J Strength Cond Res. 2010;24:2770–8.
Roschel H, Barroso R, Tricoli V, et al. Effects of strength training associated with whole body vibration training on running economy and vertical stiffness. J Strength Cond Res. 2015;29:2215–20.
Cavanagh PR, Williams KR. The effect of stride length variation on oxygen uptake during distance running. Med Sci Sports Exerc. 1982;14:30–5.
Tseh W, Caputo JL, Morgan DW. Influence of gait manipulation on running economy in female distance runners. J Sports Sci Med. 2008;7:91–5.
Williams KR, Cavanagh PR. Relationship between distance running mechanics, running economy, and performance. J Appl Physiol. 1987;63:1236–45.
Barnes KR, McGuigan MR, Kilding AE. Lower-body determinants of running economy in male and female distance runners. J Strength Cond Res. 2014;28:1289–97.
Dalleau G, Belli A, Bourdin M, et al. The spring-mass model and the energy cost of treadmill running. Eur J Appl Physiol. 1998;77:257–63.
Abe D, Muraki S, Yanagawa K, et al. Changes in EMG characteristics and metabolic energy cost during 90-min prolonged running. Gait Posture. 2007;26:607–10.
Frost G, Dowling J, Dyson K, et al. Cocontraction in three age groups of children during treadmill locomotion. J Electromyogr Kinesiol. 1997;7:179–86.
Heise G, Shinohara M, Binks L. Biarticular leg muscles and links to running economy. Int J Sports Med. 2008;29:688–91.
Moore IS, Jones AM, Dixon SJ. Relationship between metabolic cost and muscular coactivation across running speeds. J Sci Med Sport. 2013;17:671–6.
Franz JR, Wierzbinski CM, Kram R. Metabolic cost of running barefoot versus shod: is lighter better? Med Sci Sports Exerc. 2012;44:1519–25.
Moore IS, Jones AM, Dixon SJ. The pursuit of improved running performance: can changes in cushioning and somatosensory feedback influence running economy and injury risk? Footwear Sci. 2014;6:1–11.
Perl DP, Daoud AI, Lieberman DE. Effects of footwear and strike type on running economy. Med Sci Sports Exerc. 2012;44:1335–43.
Divert C, Mornieux G, Freychat P, et al. Barefoot-shod running differences: shoe or mass effect? Int J Sports Med. 2008;29:512–8.
Tung KD, Franz JR, Kram R. A test of the metabolic cost of cushioning hypothesis during unshod and shod running. Med Sci Sports Exerc. 2014;46:324–9.
Worobets J, Wannop JW, Tomaras E, et al. Softer and more resilient running shoe cushioning properties enhance running economy. Footwear Sci. 2014;6:147–53.
Arellano CJ, Kram R. The energetic cost of maintaining lateral balance during human running. J Appl Physiol. 2012;112:427–34.
Arellano CJ, Kram R. The effects of step width and arm swing on energetic cost and lateral balance during running. J Biomech. 2011;44:1291–5.
Hausswirth C, Bigard AX, Guezennec CY. Relationships between running mechanics and energy cost of running at the end of a triathlon and a marathon. Int J Sports Med. 1997;18:330–9.
Anderson T. Biomechanics and running economy. Sports Med. 1996;22:76–89.
de Ruiter CJ, Verdijk PW, Werker W, et al. Stride frequency in relation to oxygen consumption in experienced and novice runners. Eur J Sport Sci. 2013;14:251–8.
Hunter I, Smith GA. Preferred and optimal stride frequency, stiffness and economy: changes with fatigue during a 1-h high-intensity run. Eur J Appl Physiol. 2007;100:653–61.
Connick MJ, Li FX. Changes in timing of muscle contractions and running economy with altered stride pattern during running. Gait Posture. 2014;39:634–7.
Craighead DH, Lehecka N, King DL. A novel running mechanic’s class changes kinematics but not running economy. J Strength Cond Res. 2014;28:3137–45.
Egbuonu ME, Cavanagh PR, Miller TA. Degradation of running economy through changes in running mechanics. Med Sci Sports Exerc. 1990;22:S17.
Fourchet F, Girard O, Kelly L, et al. Changes in leg spring behaviour, plantar loading and foot mobility magnitude induced by an exhaustive treadmill run in adolescent middle-distance runners. J Sci Med Sport. 2014;18:199–203.
Hayes PR, Caplan N. Leg stiffness decreases during a run to exhaustion at the speed at VO2max. Eur J Sport Sci. 2014;14:556–62.
McKenna MJ, Hargreaves M. Resolving fatigue mechanisms determining exercise performance: integrative physiology at its finest! J Appl Physiol. 2008;104:286–7.
Levine BD. \( \dot{V}\)(O(2), max): what do we know, and what do we still need to know? J Physiol. 2008;586:25–34.
Halvorsen K, Eriksson M, Gullstrand L. Acute effects of reducing vertical displacement and step frequency on running economy. J Strength Cond Res. 2012;26:2065–70.
Teunissen LP, Grabowski A, Kram R. Effects of independently altering body weight and body mass on the metabolic cost of running. J Exp Biol. 2007;210:4418–27.
Slawinski JS, Billat VL. Difference in mechanical and energy cost between highly, well, and nontrained runners. Med Sci Sports Exerc. 2004;36:1440–6.
Williams KR, Cavanagh PR, Ziff JL. Biomechanical studies of elite female distance runners. Int J Sports Med. 1987;8(Suppl 2):107–18.
Eriksson M, Halvorsen KA, Gullstrand L. Immediate effect of visual and auditory feedback to control the running mechanics of well-trained athletes. J Sports Sci. 2011;29:253–62.
Kyrolainen H, Belli A, Komi PV. Biomechanical factors affecting running economy. Med Sci Sports Exerc. 2001;33:1330–7.
Storen O, Helgerud J, Hoff J. Running stride peak forces inversely determine running economy in elite runners. J Strength Cond Res. 2011;25:117–23.
Williams KR, Cavanagh PR. Biomechanical correlates with running economy in elite distance runners. Proceedings of the North American Congress on Biomechanics. Montreal; 1986. p. 287–8.
Di Michele R, Merni F. The concurrent effects of strike pattern and ground-contact time on running economy. J Sci Med Sport. 2013;17:414–8.
Nummela AT, Keranen T, Mikkelsson LO. Factors related to top running speed and economy. Int J Sports Med. 2007;28:655–61.
Roberts TJ, Kram R, Weyand PG, et al. Energetics of bipedal running. I. Metabolic cost of generating force. J Exp Biol. 1998;201:2745–51.
Kram R, Taylor CR. Energetics of running: a new perspective. Nature. 1990;346:265–7.
Kaneko M, Ito A, Fuchimoto T, et al. Influence of running speed on the mechanical efficiency of sprinters and distance runners. In: Winter DA, Norman RW, Wells RP, Heyes KC, Patla AE, editors. Biomechanics IX-B. Champaign: Human Kinetics; 1985. p. 307–12.
Nummela AT, Paavolainen L, Sharwood KA, et al. Neuromuscular factors determining 5 km running performance and running economy in well-trained athletes. Eur J Appl Physiol. 2006;97:1–8.
Kong PW, De Heer H. Anthropometric, gait and strength characteristics of Kenyan distance runners. J Sports Sci Med. 2008;7:499–504.
Ardigo LP, Lafortuna C, Minetti AE, et al. Metabolic and mechanical aspects of foot landing type, forefoot and rearfoot strike, in human running. Acta Physiol Scand. 1995;155:17–22.
Arendse RE, Noakes TD, Azevedo LB, et al. Reduced eccentric loading of the knee with the pose running method. Med Sci Sports Exerc. 2004;36:272–7.
Fletcher G, Bartlett R, Romanov N, et al. Pose® method technique improves running performance without economy changes. Int J Sports Sci Coach. 2008;3:365–80.
Heiderscheit BC, Chumanov ES, Michalski MP, et al. Effects of step rate manipulation on joint mechanics during running. Med Sci Sports Exerc. 2011;43:296–302.
Lieberman DE, Warrener AG, Wang J, et al. Effects of stride frequency and foot position at landing on braking force, hip torque, impact peak force and the metabolic cost of running in humans. J Exp Biol. 2015;218:3406–14.
Sinclair J, Taylor PJ, Edmundson CJ, et al. The influence of footwear kinetic, kinematic and electromyographical parameters on the energy requirements of steady state running. Mov Sport Sci. 2013;80:39–49.
Cavanagh PR, Pollock ML, Landa J. A biomechanical comparison of elite and good distance runners. Ann N Y Acad Sci. 1977;301:328–45.
Rassier DE, MacIntosh BR, Herzog W. Length dependence of active force production in skeletal muscle. J Appl Physiol. 1999;86:1445–57.
Carrier D, Heglund N, Earls K. Variable gearing during locomotion in the human musculoskeletal system. Science. 1994;265:651–3.
Royer TD, Martin PE. Manipulations of leg mass and moment of inertia: effects on energy cost of walking. Med Sci Sports Exerc. 2005;37:649–56.
Santos-Concejero J, Tam N, Granados C, et al. Interaction effects of stride angle and strike pattern on running economy. Int J Sports Med. 2014;35:1118–23.
Santos-Concejero J, Granados C, Irazusta J, et al. Differences in ground contact time explain the less efficient running economy in North African runners. Biol Sport. 2013;30:181–7.
Messier SP, Cirillo KJ. Effects of a verbal and visual feedback system on running technique, perceived exertion and running economy in female novice runners. J Sports Sci. 1989;7:113–26.
Warne JP, Warrington GD. Four-week habituation to simulated barefoot running improves running economy when compared with shod running. Scand J Med Sci Sports. 2014;24:563–8.
Hasegawa H, Yamauchi T, Kraemer WJ. Foot strike patterns of runners at the 15-km point during an elite-level half marathon. J Strength Cond Res. 2007;21:888–93.
Lieberman DE, Venkadesan M, Werbel WA, et al. Foot strike patterns and collision forces in habitually barefoot versus shod runners. Nature. 2010;463:531–5.
Jenkins DW, Cauthon DJ. Barefoot running claims and controversies: a review of the literature. J Am Podiatr Med Assoc. 2011;101:231–46.
Gruber AH, Umberger BR, Braun B, et al. Economy and rate of carbohydrate oxidation during running with rearfoot and forefoot strike patterns. J Appl Physiol. 2013;115:194–201.
Cunningham CB, Schilling N, Anders C, et al. The influence of foot posture on the cost of transport in humans. J Exp Biol. 2010;213:790–7.
Ogueta-Alday A, Rodriguez-Marroyo JA, Garcia-Lopez J. Rearfoot striking runners are more economical than midfoot strikers. Med Sci Sports Exerc. 2014;46:580–5.
Farley CT, McMahon TA. Energetics of walking and running: insights from simulated reduced-gravity experiments. J Appl Physiol. 1992;73:2709–12.
Taylor CR, Heglund NC, McMahon TA, et al. Energetic cost of generating muscular force during running: a comparison of large and small animals. J Exp Biol. 1980;86:9–18.
Chang YH, Kram R. Metabolic cost of generating horizontal forces during human running. J Appl Physiol. 1999;86:1657–62.
Arellano CJ, Kram R. Partitioning the metabolic cost of human running: a task-by-task approach. Integr Comp Biol. 2014;54:1084–98.
Moore IS, Jones AM, Dixon SJ. Reduced oxygen cost of running is related to alignment of the resultant GRF and leg axis vector: a pilot study. Scand J Med Sci Sports. 2015. doi:10.1111/sms.12514.
Cavagna GA, Franzetti P, Heglund NC, et al. The determinants of the step frequency in running, trotting and hopping in man and other vertebrates. J Physiol. 1988;399:81–92.
Butler RJ, Crowell HP 3rd, Davis IM. Lower extremity stiffness: implications for performance and injury. Clin Biomech. 2003;18:511–7.
Divert C, Baur H, Mornieux G, et al. Stiffness adaptations in shod running. J Appl Biomech. 2005;21:311–21.
Kerdok AE, Biewener AA, McMahon TA, et al. Energetics and mechanics of human running on surfaces of different stiffnesses. J Appl Physiol. 2002;92:469–78.
Lussiana T, Fabre N, Hebert-Losier K, et al. Effect of slope and footwear on running economy and kinematics. Scand J Med Sci Sports. 2013;23:246–53.
Lussiana T, Hébert-Losier K, Mourot L. Effect of minimal shoes and slope on vertical and leg stiffness during running. J Sport Health Sci. 2015;4:195–202.
Morin JB, Samozino P, Zameziati K, et al. Effects of altered stride frequency and contact time on leg-spring behavior in human running. J Biomech. 2007;40:3341–8.
Ruan M, Li L. Approach run increases preactivation and eccentric phases muscle activity during drop jumps from different drop heights. J Electromyogr Kinesiol. 2010;20:932–8.
Muller R, Grimmer S, Blickhan R. Running on uneven ground: leg adjustments by muscle pre-activation control. Hum Mov Sci. 2010;29:299–310.
Boyer KA, Nigg BM. Muscle activity in the leg is tuned in response to impact force characteristics. J Biomech. 2004;37:1583–8.
Boyer KA, Nigg BM. Changes in muscle activity in response to different impact forces affect soft tissue compartment mechanical properties. J Biomech Eng. 2007;129:594–602.
Nigg BM, Stefanyshyn DJ, Cole G, et al. The effect of material characteristics of shoe soles on muscle activiation and energy aspects during running. J Biomech. 2003;36:569–75.
Saunders PU, Pyne DB, Telford RD, et al. Reliability and variability of running economy in elite distance runners. Med Sci Sports Exerc. 2004;36:1972–6.
Bourdin M, Belli A, Arsac LM, et al. Effect of vertical loading on energy cost and kinematics of running in trained male subjects. J Appl Physiol. 1995;79:2078–85.
Pinnington HC, Dawson B. The energy cost of running on grass compared to soft dry beach sand. J Sci Med Sport. 2001;4:416–30.
Pinnington HC, Lloyd DG, Besier TF, et al. Kinematic and electromyography analysis of submaximal differences running on a firm surface compared with soft, dry sand. Eur J Appl Physiol. 2005;94:242–53.
Chapman AR, Vicenzino B, Blanch P, et al. Is running less skilled in triathletes than runners matched for running training history? Med Sci Sports Exerc. 2008;40:557–65.
Montgomery WH 3rd, Pink M, Perry J. Electromyographic analysis of hip and knee musculature during running. Am J Sports Med. 1994;22:272–8.
Kelly LA, Girard O, Racinais S. Effect of orthoses on changes in neuromuscular control and eerobic cost of a 1-h run. Med Sci Sports Exerc. 2011;43:2335–43.
Burke JR, Papuga MO. Effects of foot orthotics on running economy: methodological considerations. J Manip Physiol Ther. 2012;35:327–36.
Burkett LN, Kohrt WM, Buchbinder R. Effects of shoes and foot orthotics on VO2 and selected frontal plane knee kinematics. Med Sci Sports Exerc. 1985;17:158–63.
Fuller JT, Bellenger CR, Thewlis D, et al. The effect of footwear on running performance and running economy in distance runners. Sports Med. 2014;45:411–22.
Scholz MN, Bobbert MF, Van Soest AJ, et al. Running biomechanics: shorter heels, better economy. J Exp Biol. 2008;211:3266–71.
Roy J-PR, Stefanyshyn DJ. Shoe midsole longitudinal bending stiffness and running economy, joint energy, and EMG. Med Sci Sports Exerc. 2006;38:562–9.
Luo G, Stergiou P, Worobets J, et al. Improved footwear comfort reduces oxygen consumption during running. Footwear Sci. 2009;1:25–9.
Frederick EC, Howley ET, Powers S. Lower oxygen demands of running in soft-soled shoes. Res Q Exerc Sport. 1986;57:174–7.
Frederick EC, Clarke TE, Larsen JL, et al. The effect of shoe cushioning on the oxygen demands on running. In: Nigg BM, Kerr BA, editors. Biomechanical aspects of sports shoes and playing surfaces. Calgary: University of Calgary; 1983. p. 107–14.
Lejeune TM, Willems PA, Heglund NC. Mechanics and energetics of human locomotion on sand. J Exp Biol. 1998;201:2071–80.
Chen C-H, Tu K-H, Liu C, et al. Effects of forefoot bending elasticity of running shoes on gait and running performance. Hum Mov Sci. 2014;38:163–72.
McCallion C, Donne B, Fleming N, et al. Acute differences in foot strike and spatiotemporal variables for shod, barefoot or minimalist male runners. J Sports Sci Med. 2014;13:280–6.
Vincent HK, Montero C, Conrad BP, et al. Metabolic responses of running shod and barefoot in mid-forefoot runners. J Sports Med Phys Fit. 2014;54:447–55.
De Wit B, De Clercq D, Aerts P. Biomechanical analysis of the stance phase during barefoot and shod running. J Biomech. 2000;33:269–78.
Moore IS, Pitt W, Nunns M, et al. Effects of a seven-week minimalist footwear transition programme on footstrike modality, pressure variables and loading rates. Footwear Sci. 2014;7:17–29.
Chambon N, Delattre N, Gueguen N, et al. Is midsole thickness a key parameter for the running pattern? Gait Posture. 2014;40:58–63.
Squadrone R, Gallozzi C. Biomechanical and physiological comparison of barefoot and two shod conditions in experienced barefoot runners. J Sports Med Phys Fitness. 2009;49:6–13.
Paquette MR, Zhang S, Baumgartner LD. Acute effects of barefoot, minimal shoes and running shoes on lower limb mechanics in rear and forefoot strike runners. Footwear Sci. 2013;5:9–18.
Hamill J, Russell E, Gruber A, et al. Impact characteristics in shod and barefoot running. Footwear Sci. 2011;3:33–40.
Breine B, Malcolm P, Frederick EC, et al. Relationship between running apeed and initial foot contact patterns. Med Sci Sports Exerc. 2014;46:1595–603. doi:10.249/MSS.0000000000000267.
Allison HG, JuliaFreedman S, Peter B, et al. Footfall patterns during barefoot running on harder and softer surfaces. Footwear Sci. 2013;5:39–44.
Moore IS, Dixon SJ. Changes in sagittal plane kinematics with treadmill familiarization to barefoot running. J Appl Biomech. 2014;30:626–31.
Hinrichs RN. Upper extremity function in running II: angular momentum considerations. J Appl Biomech. 1987;3:242–63.
Arellano CJ, Kram R. The metabolic cost of human running: is swinging the arms worth it? J Exp Biol. 2014;217:2456–61.
Pontzer H, Holloway JH 4th, Raichlen DA, et al. Control and function of arm swing in human walking and running. J Exp Biol. 2009;212:523–34.
Miller RH, Caldwell GE, Van Emmerik RE, et al. Ground reaction forces and lower extremity kinematics when running with suppressed arm swing. J Biomech Eng. 2009;131:124502.
White JL, Scurr JC, Smith NA. The effect of breast support on kinetics during overground running performance. Ergonomics. 2009;52:492–8.
Milligan A, Mills C, Corbett J, et al. The influence of breast support on torso, pelvis and arm kinematics during a five kilometer treadmill run. Hum Mov Sci. 2015;42:246–60.
Milligan A. The effect of breast support on running biomechanics. PhD thesis. University of Portsmouth; 2013. http://eprints.port.ac.uk/14846/. Accessed 10 Dec 2015.
Morgan DW, Martin P, Craib M, et al. Effect of step length optimization on the aerobic demand of running. J Appl Physiol. 1994;77:245–51.
Bailey SP, Messier SP. Variations in stride length and running economy in male novice runners subsequent to a seven-week training program. Int J Sports Med. 1991;12:299–304.
Dallam GM, Wilber RL, Jadelis K, et al. Effect of a global alteration of running technique on kinematics and economy. J Sports Sci. 2005;23:757–64.
Romanov N, Fletcher G. Runners do not push off the ground but fall forwards via a gravitational torque. Sports Biomech. 2007;6:434–52.
Crowell HP, Davis IS. Gait retraining to reduce lower extremity loading in runners. Clin Biomech. 2011;26:78–83.
Davis IS, Crowell HP, Fellin RE, et al. Reduced impact loading following gait retraining over a 6-month period. Gait Posture. 2009;30:S4–5.
Diebal AR, Gregory R, Alitz C, et al. Forefoot running improves pain and disability associated with chronic exertional compartment syndrome. Am J Physiol. 2012;40:1060–7.
Willy RW, Scholz JP, Davis IS. Mirror gait retraining for the treatment of patellofemoral pain in female runners. Clin Biomech. 2012;27:1045–51.
Clansey AC, Hanlon M, Wallace ES, et al. Influence of tibial shock feedback training on impact loading and running economy. Med Sci Sports Exerc. 2014;46:973–81.
Willwacher S, König M, Braunstein B, et al. The gearing function of running shoe longitudinal bending stiffness. Gait Posture. 2014;40:386–90.
Williams KR. Biomechanical factors contributing to marathon race success. Sports Med. 2007;37:420–3.
Acknowledgments
The author would like to thank Professor Andrew Jones and Dr. Victoria Stiles for their critical comments on earlier versions of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No sources of funding were used to assist in the preparation of this article.
Conflicts of interest
Isabel Moore declares she has no conflicts of interest relevant to the content of this review.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Moore, I.S. Is There an Economical Running Technique? A Review of Modifiable Biomechanical Factors Affecting Running Economy. Sports Med 46, 793–807 (2016). https://doi.org/10.1007/s40279-016-0474-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40279-016-0474-4