Abstract
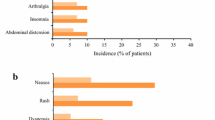
Pirfenidone (Esbriet®), a synthetic pyridine compound with antifibrotic, anti-inflammatory and antioxidant properties that inhibit the progression of tissue fibrosis, is indicated for the treatment of idiopathic pulmonary fibrosis (IPF). In clinical trials in patients with mild to moderate IPF, oral pirfenidone generally provided a reduced rate of decline in forced vital capacity, while prolonging progression-free survival and reducing the risk of IPF-related and all-cause mortality. Pirfenidone has a manageable tolerability profile; however, gastrointestinal and skin-related events are common. Data from longer-term and real-world studies of pirfenidone use in patients with IPF are consistent with those in clinical trials.

Similar content being viewed by others
References
Raghu G, Rochwerg B, Zhang Y, et al. An official ATS/ERS/JRS/ALAT clinical practice guideline: treatment of idiopathic pulmonary fibrosis. An update of the 2011 clinical practice guideline. Am J Respir Crit Care Med. 2015;192(2):e3–19.
Cottin V, Maher T. Long-term clinical and real-world experience with pirfenidone in the treatment of idiopathic pulmonary fibrosis. Eur Respir Rev. 2015;24(135):58–64.
Sandbo N. Mechanisms of fibrosis in IPF. In: Meyer KC, Nathan SD, editors. Idiopathic pulmonary fibrosis: a comprehensive clinical guide: Humana Press; 2014. p. 161–205.
Kim ES, Keating GM. Pirfenidone: a review of its use in idiopathic pulmonary fibrosis. Drugs. 2015;75(2):219–30.
Esbriet (pirfenidone) 267 mg hard capsules: summary of product characteristics. London: European Medicines Agency; 2016.
Oku H, Shimizu T, Kawabata T, et al. Antifibrotic action of pirfenidone and prednisolone: Different effects on pulmonary cytokines and growth factors in bleomycin-induced murine pulmonary fibrosis. Eur J Pharmacol. 2008;590(1–3):400–8.
Esbriet (pirfenidone) capsules: US prescribing information. South San Francisco: Genetech USA, Inc.; 2016.
King TE Jr, Bradford WZ, Castro-Bernardini S, et al. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med. 2014;370(22):2083–92.
Noble PW, Albera C, Bradford WZ, et al. Pirfenidone in patients with idiopathic pulmonary fibrosis (CAPACITY): two randomised trials. Lancet. 2011;377(9779):1760–9.
Azuma A, Nukiwa T, Tsuboi E, et al. Double-blind, placebo-controlled trial of pirfenidone in patients with idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 2005;171(9):1040–7.
Taniguchi H, Ebina M, Kondoh Y, et al. Pirfenidone in idiopathic pulmonary fibrosis. Eur Respir J. 2010;35(4):821–9.
Noble PW, Albera C, Bradford WZ, et al. Pirfenidone for idiopathic pulmonary fibrosis: analysis of pooled data from three multinational phase 3 trials. Eur Respir J. 2016;47(1):243–53.
Nathan SD, Albera C, Bradford WZ, et al. Effect of continued treatment with pirfenidone following clinically meaningful declines in forced vital capacity: analysis of data from three phase 3 trials in patients with idiopathic pulmonary fibrosis. Thorax. 2016;71(5):429–35.
Lancaster L, Albera C, Bradford WZ, et al. Safety of pirfenidone in patients with idiopathic pulmonary fibrosis: integrated analysis of cumulative data from 5 clinical trials. BMJ Open Respir Res. 2016;3(1):e000105.
Valeyre D, Albera C, Bradford WZ, et al. Comprehensive assessment of the long-term safety of pirfenidone in patients with idiopathic pulmonary fibrosis. Respirology. 2014;19(5):740–7.
Costabel U, Albera C, Bradford WZ, et al. Analysis of lung function and survival in RECAP: an open-label extension study of pirfenidone in patients with idiopathic pulmonary fibrosis. Sarcoidosis Vasc Diffuse Lung Dis. 2014;31(3):198–205.
Ravaglia C, Gurioli C, Romagnoli M, et al. Pirfenidone treatment in idiopathic pulmonary fibrosis: an Italian case series [abstract no. P3370]. Eur Respir J. 2013;42(Suppl. 57):688s.
Bonella F, Wessendorf TE, Costabel U. Clinical experience with pirfenidone for the treatment of idiopathic pulmonary fibrosis [article in German]. Dtsch Med Wochenschr. 2013;138(11):518–23.
Oltmanns U, Kahn N, Palmowski K, et al. Pirfenidone in idiopathic pulmonary fibrosis: real-life experience from a German tertiary referral center for interstitial lung diseases. Respiration. 2014;88:199–207.
Lützkendorf L, Vogler M, Hoffken G, et al. Real-life experience with pirfenidon in idiopathic pulmonary fibrosis: does early treatment start predict better therapy adherence? [abstract no. PA3043]. Eur Respir J. 2015;46(Suppl 59).
Wijsenbeek M, Van Beek FT, Geel AL, et al. Pirfenidone in daily clinical use in patients with idiopathic pulmonary fibrosis in the Netherlands [abstract no. A4340]. Am J Respir Crit Care Med. 2013;187(abstract issue).
Chaudhuri N, Duck A, Frank R, et al. Real world experiences: pirfenidone is well tolerated in patients with idiopathic pulmonary fibrosis. Respir Med. 2014;108(1):224–6.
Wijsenbeek M, Grutters J, Wuyts W. Early experience of pirfenidone in daily clinical practice in Belgium and the Netherlands: a retrospective cohort analysis. Adv Ther. 2015;32(7):691–704.
Loeh B, Drakopanagiotakis F, Bandelli GP, et al. Intraindividual response to treatment with pirfenidone in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 20151;191(1):110–3.
Harari S, Caminati A, Albera C, et al. Efficacy of pirfenidone for idiopathic pulmonary fibrosis: an Italian real life study. Respir Med. 2015;109(7):904–13.
Nieto A, Valenzuela C, Rivera I, et al. Pirfenidone in idiopathic pulmonary fibrosis (ipf): Spanish experience with the European named patient program (npp) and Spanish patient progam (spp) [abstract no. A1418]. Am J Respir Crit Care Med. 2014;189(abstract issue).
Koschel D, Cottin V, Sköld M, et al. Pirfenidone post-authorization safety registry (PASSPORT)—interim analysis of IPF treatment [abstract no. 850348]. Eur Respir J. 2014;44(Suppl 58).
Acknowledgments
The review was updated from Drugs 2015;75(2):219–30 [4], and was reviewed by: T.E. Wessendorf, Interstitial and Rare Lung Disease Unit, Ruhrlandklinik, University Hospital, University of Duisburg-Essen, Essen, Germany. During the peer review process, the manufacturer of pirfenidone was also offered an opportunity to review this article. Changes resulting from comments received were made on the basis of scientific and editorial merit.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The preparation of this review was not supported by any external funding.
Conflict of interest
S.L. Greig, K.A. Lyseng-Williamson, E.S. Kim and G.M. Keating are salaried employees of Adis/Springer, are responsible for the article content and declare no relevant conflicts of interest.
Rights and permissions
About this article
Cite this article
Greig, S.L., Lyseng-Williamson, K.A., Kim, E.S. et al. Pirfenidone in idiopathic pulmonary fibrosis: a guide to its use in the EU. Drugs Ther Perspect 32, 323–329 (2016). https://doi.org/10.1007/s40267-016-0321-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40267-016-0321-6