Abstract
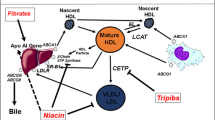
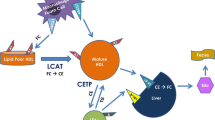
Earlier epidemiological studies have shown an inverse correlation between high-density lipoprotein cholesterol (HDLc) and coronary heart disease (CHD). This observation along with the finding that reverse cholesterol transport is mediated by HDL, supported the hypothesis that the HDL molecule has a cardioprotective role. More recently, epidemiological data suggest a U-shaped curve correlating HDLc and CHD. In addition, randomized clinical trials of drugs that significantly increase plasma HDLc levels, such as nicotinic acid and cholesterol ester transfer protein (CETP) inhibitors failed to show a reduction in major adverse cardiovascular events. These observations challenge the hypothesis that HDL has a cardioprotective role. It is possible that HDL quality and function is optimal only when de novo synthesis of apo A-I occurs. Inhibition of turnover of HDL with currently available agents yields HDL molecules that are ineffective in reverse cholesterol transport. To test this hypothesis, newer therapeutic drugs that increase de novo production of HDL and apo A-I should be tested in clinical trials.
Similar content being viewed by others
References
LaRosa JC, He J, Vupputuri S. Effect of statins on risk of coronary disease: a meta-analysis of randomized controlled trials. JAMA. 1999;282:2340–6.
Rosenson RS, Brewer HB, Davidson WS, et al. Cholesterol efflux and atheroprotection: advancing the concept of reverse cholesterol transport. Circulation. 2012;125:1905–19.
Bielicki JK, Oda MN. Apolipoprotein A-I(Milano) and apolipoprotein A-I(Paris) exhibit an antioxidant activity distinct from that of wild-type apolipoprotein A-I. Biochemistry. 2002;41:2089–96.
Cameron SJ, Morrell CN, Bao C, Swaim AF, Rodriguez A, Lowenstein CJ. A novel anti-inflammatory effect for high density lipoprotein. PLoS One. 2015;10(12):e0144372.
Ashby DT, Rye KA, Clay MA, Vadas MA, Gamble JR, Barter PJ. Factors influencing the ability of HDL to inhibit expression of vascular cell adhesion molecule-1 in endothelial cells. Arterioscler Thromb Vasc Biol. 1998;18:1450–5.
Yui Y, Aoyama T, Morishita H, Takahashi M, Takatsu Y, Kawai C. Serum prostacyclin stabilizing factor is identical to apolipoprotein A-I (Apo A-I). A novel function of Apo A-I. J Clin Investig. 1988;82:803–7.
Vaisar T, Couzens E, Hwang A, Russell M, Barlow CE, DeFina LF, Hoofnagle AN, Kim F. Type 2 diabetes is associated with loss of HDL endothelium protective functions. PLoS One. 2018;13(3):e0192616.
Chung DW, Chen J, Ling M, Fu X, Blevins T, Parsons S, Le J, Harris J, Martin TR, Konkle BA, Zheng Y, López JA. High-density lipoprotein modulates thrombosis by preventing von Willebrand factor self-association and subsequent platelet adhesion. Blood. 2016;127:637–45.
Gordon T, Castelli WP, Hjortland MC, Kannel WB, Dawber TR. High density lipoprotein as a protective factor against coronary heart disease: the Framingham study. Am J Med. 1977;62:707–14.
Miller M, Stone NJ, Ballantyne C, et al. Triglycerides and cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2011;123:2292–333.
Boekholdt SM, Arsenault BJ, Hovingh GK, et al. Levels and changes of HDL cholesterol and apolipoprotein A-I in relation to risk of cardiovascular events among statin-treated patients: a meta-analysis. Circulation. 2013;128:1504–12.
Vergès B, Adiels M, Boren J, Barrett PH, Watts GF, Chan D, et al. Interrelationships between the kinetics of VLDL subspecies and HDL catabolism in abdominal obesity: a multicenter tracer kinetic study. J Clin Endocrinol Metab. 2014;99:4281–90.
Haas MJ, Mazza AD, Wong NC, Mooradian AD. Inhibition of apolipoprotein A-I gene expression by obesity-associated endocannabinoids. Obesity (Silver Spring). 2012;20:721–9.
Mooradian AD, Haas MJ, Wehmeier KR, Wong NC. Obesity-related changes in high-density lipoprotein metabolism. Obesity (Silver Spring). 2008;16:1152–60.
Duvillard L, Pont F, Florentin E, Gambert P, Vergès B. Inefficiency of insulin therapy to correct apolipoprotein A-I metabolic abnormalities in non-insulin-dependent diabetes mellitus. Atherosclerosis. 2000;152:229–37.
Mooradian AD, Haas MJ, Wong NC. Transcriptional control of apolipoprotein A-I gene expression in diabetes. Diabetes. 2004;53:513–20.
Murao K, Wada Y, Nakamura T, Taylor AH, Mooradian AD, Wong NC. Effects of glucose and insulin on rat apolipoprotein A-I gene expression. J Biol Chem. 1998;273:18959–65.
Lam JK, Matsubara S, Mihara K, Zheng XL, Mooradian AD, Wong NC. Insulin induction of apolipoprotein AI, role of Sp1. Biochemistry. 2003;42:2680–90.
Mooradian AD, Albert SG, Haas MJ. Low serum high-density lipoprotein cholesterol in obese subjects with normal serum triglycerides: the role of insulin resistance and inflammatory cytokines. Diabetes Obes Metab. 2007;9:441–3.
Haas MJ, Mooradian AD. Inflammation, high-density lipoprotein and cardiovascular dysfunction. Curr Opin Infect Dis. 2011;24:265–72.
Haas MJ, Mooradian AD. Regulation of high-density lipoprotein by inflammatory cytokines: establishing links between immune dysfunction and cardiovascular disease. Diabetes Metab Res Rev. 2010;26:90–9.
Beers A, Haas MJ, Wong NC, Mooradian AD. Inhibition of apolipoprotein AI gene expression by tumor necrosis factor alpha: roles for MEK/ERK and JNK signaling. Biochemistry. 2006;45:2408–13.
Parseghian S, Onstead-Haas LM, Wong NC, Mooradian AD, Haas MJ. Inhibition of apolipoprotein A-I expression by TNF-alpha in HepG2 cells: requirement for c-jun. J Cell Biochem. 2014;115:253–60.
Haas MJ, Horani M, Mreyoud A, Plummer B, Wong NC, Mooradian AD. Suppression of apolipoprotein AI gene expression in HepG2 cells by TNF alpha and IL-1beta. Biochim Biophys Acta. 2003;1623(2–3):120–8.
Palacio C, Alexandraki I, Bertholf RL, Mooradian AD. Transient dyslipidemia mimicking the plasma lipid profile of Tangier disease in a diabetic patient with gram negative sepsis. Ann Clin Lab Sci. 2011;41:150–3.
Naem E, Alcalde R, Gladysz M, Mesliniene S, Jaimungal S, Sheikh-Ali M, Haas MJ, Wong NC, Mooradian AD. Inhibition of apolipoprotein A-I gene by the aryl hydrocarbon receptor: a potential mechanism for smoking-associated hypoalphalipoproteinemia. Life Sci. 2012;91:64–9.
Mooradian AD. Is high-density lipoprotein cardioprotective or simply a marker of cardiovascular disease? Am J Ther. 2014;21:438–9.
Newman AB, Glynn NW, Taylor CA, et al. Health and function of participants in the Long Life Family Study: a comparison with other cohorts. Aging (Albany, NY). 2011;3:63–76.
Rahilly-Tierney CR, Spiro A, Vokonas P, Gaziano JM. Relation between high-density lipoprotein cholesterol and survival to age 85 years in men (from the VA Normative Aging Study). Am J Cardiol. 2011;107:1173–7.
Odden MC, Shlipak MG, Whitson HE, et al. Risk factors for cardiovascular disease across the spectrum of older age: the Cardiovascular Health Study. Atherosclerosis. 2014;237:336–42.
Stampfer MJ, Sacks FM, Salvini S, Willett WC, Hennekens CH. A prospective study of cholesterol, apolipoproteins, and the risk of myocardial infarction. N Engl J Med. 1991;325:373–81.
Cohen JC, Wang Z, Grundy SM, Stoesz MR, Guerra R. Variation at the hepatic lipase and apolipoprotein AI/CIII/AIV loci is a major cause of genetically determined variation in plasma HDL cholesterol levels. J Clin Investig. 1994;94:2377–84.
Reguero JR, Cubero GI, Batalla A, et al. Apolipoprotein A1 gene polymorphisms and risk of early coronary disease. Cardiology. 1998;90:231–5.
Voight BF, Peloso GM, Orho-Melander M, et al. Plasma HDL cholesterol and risk of myocardial infarction: a mendelian randomisation study. Lancet. 2012;380(9841):572–80.
Mooradian AD, Haas MJ. Targeting high-density lipoproteins: increasing de novo production versus decreasing clearance. Drugs. 2015;75:713–22.
Pastore L, Belalcazar LM, Oka K, et al. Helper-dependent adenoviral vector-mediated long-term expression of human apolipoprotein A-I reduces atherosclerosis in apo E-deficient mice. Gene. 2004;327:153–60.
Patel S, Di Bartolo BA, Nakhla S, et al. Anti-inflammatory effects of apolipoprotein A-I in the rabbit. Atherosclerosis. 2010;212:392–7.
Nissen SE, Tsunoda T, Tuzcu EM, et al. Effect of recombinant ApoA-I Milano on coronary atherosclerosis in patients with acute coronary syndromes. JAMA. 2003;290:2292.
Nissen SE, Tuzcu EM, Schoenhagen P, et al. Effect of intensive compared with moderate lipid-lowering therapy on progression of coronary atherosclerosis: a randomized controlled trial. JAMA. 2004;291:1071–80.
Zanoni P, Khetarpal SA, Larach DB, et al. Rare variant in scavenger receptor BI raises HDL cholesterol and increases risk of coronary heart disease. Science. 2016;351:1166–71.
Schwartz CC, VandenBroek JM, Cooper PS. Lipoprotein cholesteryl ester production, transfer, and output in vivo in humans. J Lipid Res. 2004;45:1594–607.
Allard-Ratick M. Association between very high levels of HDL-c and increased risk of CV events. In: Atlanta: ESC; 2018. https://ipccs.org/2018/09/04/association-between-very-high-levels-of-hdl-c-and-increased-risk-of-cv-events/. Accessed Aug 6, 2019.
Haase CL, Tybjærg-Hansen A, Ali Qayyum A, Schou J, Nordestgaard BG, Frikke-Schmidt R. LCAT, HDL cholesterol and ischemic cardiovascular disease: a mendelian randomization study of HDL cholesterol in 54,500 individuals. J Clin Endocrinol Metab. 2012;97:E248–56.
Tan JTM, Prosser HCG, Dunn LL, et al. High-density lipoproteins rescue diabetes-impaired angiogenesis via scavenger receptor class B type I. Diabetes. 2016;65:3091–103.
Gille A, Easton R, D’Andrea D, Wright SD, Shear CL. CSL112 enhances biomarkers of reverse cholesterol transport after single and multiple infusions in healthy subjects. Arterioscler Thromb Vasc Biol. 2014;34:2106–14.
Tricoci P, D’Andrea DM, Gurbel PA, et al. Infusion of reconstituted high-density lipoprotein, CSL112, in patients with atherosclerosis: safety and pharmacokinetic results from a phase 2a randomized clinical trial. J Am Heart Assoc. 2015;4(8):e002171.
Tardif J-C, Grégoire J, L’Allier PL, et al. Effects of reconstituted high-density lipoprotein infusions on coronary atherosclerosis: a randomized controlled trial. JAMA. 2007;297(15):1675–82.
Kootte RS, Smits LP, van der Valk FM, et al. Effect of open-label infusion of an apoA-I-containing particle (CER-001) on RCT and artery wall thickness in patients with FHA. J Lipid Res. 2015;56:703–12.
Hovingh GK, Smits LP, Stefanutti C, et al. The effect of an apolipoprotein A-I–containing high-density lipoprotein–mimetic particle (CER-001) on carotid artery wall thickness in patients with homozygous familial hypercholesterolemia: The Modifying Orphan Disease Evaluation (MODE) study. Am Heart J. 2015;169:736–42.
Tardif J-C, Ballantyne CM, Barter P, et al. Effects of the high-density lipoprotein mimetic agent CER-001 on coronary atherosclerosis in patients with acute coronary syndromes: a randomized trial. Eur Heart J. 2014;35:3277–86.
Nicholls SJ, Puri R, Ballantyne CM, et al. Effect of infusion of high-density lipoprotein mimetic containing recombinant apolipoprotein A-I Milano on coronary disease in patients with an acute coronary syndrome in the MILANO-PILOT Trial: a randomized clinical trial. JAMA Cardiol. 2018;3:806–14.
Nicholls SJ, Andrews J, Kastelein JJP, et al. Effect of serial infusions of CER-001, a pre-β high-density lipoprotein mimetic, on coronary atherosclerosis in patients following acute coronary syndromes in the CER-001 Atherosclerosis Regression Acute Coronary Syndrome Trial: a randomized clinical trial. JAMA Cardiol. 2018;3:815–22.
Duffy D. Study to investigate CSL112 in subjects with acute coronary syndrome (AEGIS-II)—NCT03473223. https://clinicaltrials.gov/ct2/show/NCT03473223. Accessed Aug 8, 2019.
Chehade JM, Gladysz M, Mooradian AD. Dyslipidemia in type 2 diabetes: prevalence, pathophysiology and management. Drugs. 2013;73:327–39.
Mooradian AD. Diabetes and atherogenic dyslipidemia. In: Rodriguez-Saldana SJ, editor. The diabetes textbook. Clinical principles, patient management and public health issues. Basel: Springer Nature; 2019. p. 587–96.
Rubins HB, Robins SJ, Collins D, et al. Gemfibrozil for the secondary prevention of coronary heart disease in men with low levels of high-density lipoprotein cholesterol. N Engl J Med. 1999;341:410–8.
Frick MH, Elo O, Haapa K, et al. Helsinki Heart Study: Primary-Prevention Trial with gemfibrozil in middle-aged men with dyslipidemia. N Engl J Med. 1987;317:1237–45.
The BIP Study Group. Secondary prevention by raising HDL cholesterol and reducing triglycerides in patients with coronary artery disease. Circulation. 2000;102:21–7.
FIELD Study Investigators. Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (the FIELD study): randomised controlled trial. Lancet. 2005;366:1849–61.
The ACCORD Study Group. Effects of combination lipid therapy in type 2 diabetes mellitus. N Engl J Med. 2010;362:1563–74.
Elam MB, Ginsberg HN, Lovato LC, Corson M, Largay J, Leiter LA, et al. Association of fenofibrate therapy with long-term cardiovascular risk in statin-treated patients with type 2 diabetes. JAMA Cardiol. 2017;2:370–80.
The AIM-HIGH Investigators. Niacin in patients with low HDL cholesterol levels receiving intensive statin therapy. N Engl J Med. 2011;365:2255–67.
The HPS2-THRIVE Collaborative Group. Effects of extended-release niacin with laropiprant in high-risk patients. N Engl J Med. 2014;371:203–12.
Mooradian AD, Haas MJ, Wong NCW. The effect of select nutrients on serum high density lipoprotein cholesterol and apolipoprotein A-I levels. Endocr Rev. 2006;27:2–16.
Mooradian AD. The effect of nutrients on apolipoprotein AI gene expression. In: Fielding CJ, editor. High density lipoproteins. From basic biology to clinical aspects. Weinheim: Wiley-VCH; 2007. p. 399–423.
Haas MJ, Horani MH, Wong NCW, Mooradian AD. Induction of apolipoprotein AI promoter by Sp1 is repressed by saturated fatty acids. Metabolism. 2004;53:1342–8.
Mooradian AD. Evidence based cardiovascular risk management in diabetes. Am J Cardiovasc Drugs. 2019;19:439–48.
Mooradian AD. Dyslipidemia of type 2 diabetes mellitus. Nat Clin Pract Endocrinol Metab. 2009;5:150–9.
Bays HE, Maki KC, McKenney J, et al. Long-term up to 24-month efficacy and safety of concomitant prescription omega-3-acid ethyl esters and simvastatin in hyper-triglyceridemic patients. Curr Med Res Opin. 2010;26:907–15.
Bhatt D, Steg PG, Michael Miller M, et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med. 2019;380:11–22.
ORIGIN Trial Investigators, Bosch J, Gerstein HC, et al. n-3 fatty acids and cardiovascular outcomes in patients with dysglycemia. N Engl J Med. 2012;367:309–18.
ASCEND Study Collaborative Group, Bowman L, Mafham M. Effects of n-3 fatty acid supplements in diabetes mellitus. N Engl J Med. 2018;379:1540–50.
Manson JE, Cook NR, Lee IM, et al. Marine n-3 fatty acids and prevention of cardiovascular disease and cancer. N Engl J Med. 2019;380:23–32.
Nicholls SJ, Lincoff AM, Bash D, Ballantyne CM, Barter PJ, Davidson MH, et al. Assessment of omega-3 carboxylic acids in statin-treated patients with high levels of triglycerides and low levels of high-density lipoprotein cholesterol: rationale and design of the STRENGTH trial. Clin Cardiol. 2018;41:1281–8.
Anon. Update on Phase III STRENGTH trial for Epanova in mixed dyslipidaemia (AstraZeneca press release dated January 13, 2020). https://www.astrazeneca.com/media-centre/press-releases/2020/update-on-phase-iii-strength-trial-for-epanova-in-mixed-dyslipidaemia-13012020.html. Accessed 22 Jan 2020.
Barter PJ, Caulfield M, Eriksson M, et al. Effects of torcetrapib in patients at high risk for coronary events. N Engl J Med. 2007;357:2109–22.
Schwartz GG, Olsson AG, Abt M, et al. Effects of dalcetrapib in patients with a recent acute coronary syndrome. N Engl J Med. 2012;367:2089–99.
Lincoff AM, Nicholls SJ, Riesmeyer JS, et al. Evacetrapib and cardiovascular outcomes in high-risk vascular disease. N Engl J Med. 2017;376:1933–42.
The HPS3/TIMI55–REVEAL Collaborative Group. Effects of anacetrapib in patients with atherosclerotic vascular disease. N Engl J Med. 2017;377:1217–27.
A phase III, double-blind, randomized placebo-controlled study to evaluate the effects of dalcetrapib on cardiovascular (CV) risk in a genetically defined population with a recent acute coronary syndrome (ACS): the Dal-GenE Trial. https://clinicaltrials.gov/ct2/show/NCT02525939. Accessed Jan, 2020.
Haas MJ, Mooradian AD. Therapeutic interventions to enhance apolipoprotein A-I-mediated cardioprotection. Drugs. 2010;70:805–21.
Haas MJ, Onstead-Haas L, Kurban W, et al. High-throughput analysis identifying drugs that regulate apolipoprotein A-I synthesis. Assay Drug Dev Technol. 2017;15:362–71.
Haas MJ, Plazarte M, Chamseddin A, et al. Inhibition of hepatic apolipoprotein A-I gene expression by histamine. Eur J Pharmacol. 2018;823:49–57.
Haas MJ, Jurado-Flores M, Hammoud R, et al. Regulation of apolipoprotein A-I gene expression by the histamine H1 receptor: Requirement for NF-κB. Life Sci. 2018;208:102–10.
Bailey D, Jahagirdar R, Gordon A, et al. RVX-208. J Am Coll Cardiol. 2010;55:2580–9.
Nicholls SJ, Gordon A, Johannson J, et al. ApoA-I induction as a potential cardioprotective strategy: rationale for the SUSTAIN and ASSURE studies. Cardiovasc Drugs Ther. 2012;26:181–7.
Nicholls SJ, Puri R, Wolski K, et al. Effect of the BET protein inhibitor, RVX-208, on progression of coronary atherosclerosis: results of the phase 2b, randomized, double-blind, multicenter, ASSURE Trial. Am J Cardiovasc Drugs. 2016;16:55–65.
Shishikura D, Kataoka Y, Honda S, et al. The effect of bromodomain and extra-terminal inhibitor apabetalone on attenuated coronary atherosclerotic plaque: Insights from the ASSURE Trial. Am J Cardiovasc Drugs. 2019;19(1):49–57. https://doi.org/10.1007/s40256-018-0298-8.
Nicholls SJ, Ray KK, Johansson JO, et al. Selective BET protein inhibition with apabetalone and cardiovascular events: a pooled analysis of trials in patients with coronary artery disease. Am J Cardiovasc Drugs. 2018;18(2):109–15. https://doi.org/10.1007/s40256-017-0250-3.
Effect of BET protein inhibition with apabetalone on cardiovascular outcomes in patients with acute coronary syndrome and diabetes—BETonMACE. https://www.acc.org/latest-in-cardiology/clinical-trials/2019/11/15/17/25/betonmace. Accessed Jan 2020.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No external funding was used in the preparation of this manuscript.
Conflict of interest
Julien J. Feghaly and Arshag D. Mooradian declare that they have no potential conflicts of interest that might be relevant to the contents of this article.
Rights and permissions
About this article
Cite this article
Feghaly, J.J., Mooradian, A.D. The Rise and Fall “ing” of the HDL Hypothesis. Drugs 80, 353–362 (2020). https://doi.org/10.1007/s40265-020-01265-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-020-01265-4