Abstract
Drug-induced movement disorders (DIMDs) are associated with use of dopamine receptor blocking agents (DRBAs), including antipsychotics. The most common forms are drug-induced parkinsonism (DIP), dystonia, akathisia, and tardive dyskinesia (TD). Although rare, neuroleptic malignant syndrome (NMS) is a potentially life-threatening consequence of DRBA exposure. Recommendations for anticholinergic use in patients with DIMDs were developed on the basis of a roundtable discussion with healthcare professionals with extensive expertise in DIMD management, along with a comprehensive literature review. The roundtable agreed that “extrapyramidal symptoms” is a non-specific term that encompasses a range of abnormal movements. As such, it contributes to a misconception that all DIMDs can be treated in the same way, potentially leading to the misuse and overprescribing of anticholinergics. DIMDs are neurobiologically and clinically distinct, with different treatment paradigms and varying levels of evidence for anticholinergic use. Whereas evidence indicates anticholinergics can be effective for DIP and dystonia, they are not recommended for TD, akathisia, or NMS; nor are they supported for preventing DIMDs except in individuals at high risk for acute dystonia. Anticholinergics may induce serious peripheral adverse effects (e.g., urinary retention) and central effects (e.g., impaired cognition), all of which can be highly concerning especially in older adults. Appropriate use of anticholinergics therefore requires careful consideration of the evidence for efficacy (e.g., supportive for DIP but not TD) and the risks for serious adverse events. If used, anticholinergic medications should be prescribed at the lowest effective dose and for limited periods of time. When discontinued, they should be tapered gradually.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Anticholinergic medications can be effective in treating drug-induced parkinsonism and dystonia, but they are not recommended for the treatment of tardive dyskinesia, akathisia, or neuroleptic malignant syndrome. |
Appropriate use of anticholinergics requires careful consideration of effectiveness and risk of side effects, especially in older adults and other vulnerable populations, and should be prescribed at the lowest effective dose. |
When discontinuing use of anticholinergics, they should be tapered gradually. |
1 Introduction
Antipsychotic medications have achieved widespread utilization on the basis of a robust body of evidence for the treatment of psychiatric conditions, and use of these medications is on the rise. In the USA, antipsychotic prescriptions in noninstitutionalized individuals increased from 2.2 million in 1997 to 6.1 million in 2018 [1, 2]. Moreover, in North America, antipsychotic polypharmacy for schizophrenia increased from the 1980s to the 2000s, reflecting an uptick in the combined use of second-generation antipsychotics (SGAs) with first-generation antipsychotics (FGAs) [3]. Contributing to the expanding use of antipsychotics are the on-label uses for approved indications such as bipolar disorder, major depressive disorder, and agitation associated with dementia due to Alzheimer’s disease, along with the off-label uses for unapproved indications such as dementia-related psychosis and personality disorders [4,5,6].
Although antipsychotics are highly effective for many conditions and have positive impacts on patients’ lives, they can have significant side effects. Moreover, greater antipsychotic exposure due to higher cumulative doses and polypharmacy can increase these risks [7]. Drug-induced movement disorders (DIMDs) are among the more debilitating adverse consequences associated with antipsychotics and other dopamine receptor blocking agents (DRBAs), including certain agents used for gastrointestinal disorders such as metoclopramide. The DIMDs described in this review are all associated with exposure to DRBAs, including currently available FGAs and SGAs. As new antipsychotic therapies emerge, including those that target muscarinic receptors, the potential efficacy and safety of emerging antipsychotic therapies will also need to be considered [8].
DIMDs are classically categorized by phenomenology, with different times to onset and varying responses to DRBA discontinuation and treatment. Accurate diagnosis is therefore imperative to achieve optimal clinical outcomes [9]. Anticholinergic medications have been shown to be effective for treating drug-induced parkinsonism (DIP) and acute dystonic reactions, but not for treating tardive dyskinesia (TD), akathisia, or neuroleptic malignant syndrome (NMS). Anticholinergics are also not generally recommended for the prophylaxis of DIMDs except in certain clinical situations (e.g., when combined with an intramuscular FGA in patients with a history of DIMDs). Misuse and over-prescription of anticholinergics, especially in older adults and patients taking multiple medications with anticholinergic properties (Fig. 1), contribute to high anticholinergic burden and even higher risks of cognitive dysfunction and peripheral adverse effects.
Appropriate use of anticholinergics in patients with DIMDs requires careful consideration of several important factors: the evidence for efficacy; the potential for various peripheral and central adverse events that may impact tolerability of each anticholinergic medication, especially with long-term use in at-risk populations; and the cumulative adverse effects (i.e., anticholinergic burden) that may occur in patients taking multiple medications with anticholinergic properties. The need for continued education on these topics was recently illustrated in a real-world study that found inappropriate and potentially harmful use on the basis of a survey of 315 healthcare professionals (HCPs) and a pharmacy claims analysis of 738,207 patients with benztropine prescriptions [10]. In the survey, 159 (50.5%) HCPs reported using benztropine to treat TD and 153 (48.6%) reported using this medication in adults ≥ 65 years of age. In the claims analysis, 55.7% of patients took benztropine for > 3 months and 59.1% were taking > 10 medications—many of which were associated with moderate or high anticholinergic burden, as determined using the ACB calculator (https://www.acbcalc.com) [10].
To raise awareness about these issues and review the evidence of how and when anticholinergics should be used, a roundtable discussion with psychiatrists, neurologists, nurse practitioners, and physician assistants was conducted in June 2021. All roundtable participants had vast expertise in the management of DIMDs, and topics of discussion included the following: pharmacology of anticholinergic drugs and rationale for their use in DIMDs; adverse effects of anticholinergic medications; differentiation and appropriate treatment of DIMDs; prophylactic use of anticholinergics; discontinuation and tapering of anticholinergic medications; and treatment considerations for high-risk populations. This report summarizes key points from the roundtable discussion, supplemented by a literature review.
2 Pharmacology
2.1 Mechanism of Action of Anticholinergic Medications
Broadly speaking, anticholinergics block acetylcholine from binding to its receptors in the peripheral and central nervous systems. There are two major classes of acetylcholine receptors: G protein-coupled muscarinic receptors and ionotropic ligand-gated nicotinic receptors [11]. Muscarinic receptors comprise five subtypes (M1 through M5) that are associated with a wide range of functions, including cognition, smooth muscle contraction, sweating, and motor control, with M1 and M4 receptors implicated in disorders such as schizophrenia [12]. Nicotinic receptors are composed of various subunits (α1–α10, β1–β4, γ, δ, ε), with α7 and α4β2 receptors implicated in disorders such as schizophrenia, depression, Parkinson’s disease, and Alzheimer’s disease [13]. The centrally acting anticholinergic medications approved for all forms of parkinsonism (e.g., benztropine and trihexyphenidyl [11]) are nonselective muscarinic receptor antagonists, with the primary target being M1 receptors present on medium spiny neurons (MSNs) in the striatum and nearby cholinergic interneurons (CINs). Blocking acetylcholine M1 receptors restores the imbalance between dopamine and acetylcholine signaling on MSNs caused by insufficient dopaminergic signaling resulting from dopamine D2 receptor blockade by antipsychotics and other DRBAs [14].
2.2 Interactions Between Dopamine and Acetylcholine in Motor Circuitry
The nigrostriatal dopamine pathway, which projects from the substantia nigra to the dorsal striatum [11], is especially important for movement control. Decreased dopamine signaling is associated with hypokinetic movement disorders, while excessive dopamine signaling is associated with hyperkinetic disorders (Fig. 2) [15]. On the basis of early clinical experience with anticholinergic drugs in patients with Parkinson’s disease, it was postulated that dopamine and acetylcholine have opposing and balancing effects in the striatum; however, interactions between these neurotransmitter systems are more dynamic and complex than originally thought [16].
Hypokinetic movement disorders, such as drug-induced parkinsonism, are characterized by a decrease in dopamine signaling, which amplifies cholinergic activity. Anticholinergics can be effective in treating these disorders by restoring dopamine–acetylcholine balance. Hyperkinetic disorders, such as tardive dyskinesia, are characterized by an increase in dopamine signaling, which dampens cholinergic activity. Thus, anticholinergic medications are not effective in treating these disorders and may even worsen hyperkinetic symptoms
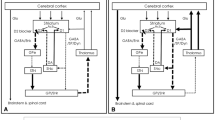
Dopaminergic activity is modulated by cholinergic neurons that project from the laterodorsal tegmentum and pedunculopontine tegmentum, two acetylcholine-rich nuclei located in the pons region of the brainstem, along with CINs located within the striatum [11, 15]. Cholinergic projections from the laterodorsal tegmentum are primarily involved in non-motor systems that control reward, motivation, cognition, and aversion via their interactions with dopaminergic neurons in the ventral tegmental area. Cholinergic projections from the pedunculopontine tegmentum directly and indirectly (via subthalamic nuclei) innervate dopamine-containing cells in the substantia nigra, thereby stimulating dopamine release into areas of the sensorimotor striatum that are primarily involved in motor control (Fig. 3).
Dopamine (DA) is released from the substantia nigra to the striatum. Within the striatum, antipsychotics prevent DA from binding to D2 receptors on indirect medium spiny neurons (MSNs) and cholinergic interneurons (CINs). Anticholinergics prevent the binding of acetylcholine (ACh) to muscarinic M1 and M4 receptors on direct MSNs and to M1 receptors on indirect MSNs. Adapted with permission from Lester 2010 [15] and Paz 2021 [17]
Dopamine input to MSNs within the striatum is also modulated by CINs, which account for 1–2% of striatal neurons. These CINs are the main source of striatal acetylcholine and have extensive modulatory effects on MSNs via their widespread projections [16, 17]. MSNs play key roles in the motor circuitry by releasing gamma-aminobutyric acid (GABA), which modulates the opposite motor effects of the direct and indirect pathways. The activation and inhibition of MSNs and CINs by dopamine from substantia nigra depend on dopamine receptor type and location (Fig. 3). Normal motor function requires that these dopamine-acetylcholine interactions remain in dynamic balance [16, 17].
2.3 Adverse Consequences of Dopamine Receptor Blockade on Motor Circuitry
In patients with DIP, blockade of D2 receptors by DRBAs leads to diminished dopamine signaling [18]. Findings from preclinical studies suggest that blocking D2 receptors may also have the following effects: (1) disinhibition of indirect MSNs, leading to increased release of inhibitory GABA from these MSNs; and (2) disinhibition of CINs, leading to greater binding of acetylcholine to excitatory M1 receptors on indirect and direct MSNs (and further augmenting GABA release from indirect MSNs) (Fig. 3) [16, 17, 19]. These cumulative effects of D2 receptor blockade on MSN and CIN activity may contribute to the abnormal movements associated with DIP and other acute, reversible movement disorders. For these conditions, anticholinergics are believed to restore the dynamic balance between dopamine and acetylcholine (Fig. 2) [16].
Furthermore, for the case of hyperkinetic movements such as TD, it has been hypothesized that chronic blockade of postsynaptic D2 receptors induces upregulation of D2 receptors on MSNs, along with conformational changes in the receptors themselves that render a state of hypersensitivity [18, 20]. The net result is enhanced dopamine signaling, leading to increased inhibition of indirect MSNs and CINs, along with decreased cholinergic and GABAergic signaling [21]. Clinically, these effects present as the potentially persistent and excessive abnormal involuntary movements associated with TD. Further inhibition of acetylcholine activity could exacerbate the imbalance between dopamine and acetylcholine (Fig. 2), which is consistent with the lack of evidence supporting the use of anticholinergics in TD [22, 23] and reports of TD worsening with anticholinergics and improving after anticholinergic discontinuation [24,25,26,27].
3 Adverse Effects of Anticholinergic Medications
Medications with anticholinergic properties are used to treat a wide range of peripheral somatic and central nervous system conditions, including parkinsonism, chronic obstructive pulmonary disease (COPD), overactive bladder, hyperhidrosis, poor sleep, flu/cold symptoms, and allergies [28]. Anticholinergics have long been used to treat Parkinson’s disease, even before levodopa was introduced; however, their cognitive side effects may be particularly concerning in this older patient population [29]. Evidence on the efficacy of anticholinergics in DMDs, as covered in greater detail below, has generally been low quality in previous reviews [18, 30, 31]. Ideally, an evidence-based approach should be taken when prescribing anticholinergic medication for any condition. Recent meta-analyses, such as those for COPD [32, 33] and overactive bladder [34, 35], are valuable resources for such information.
More than 600 prescription and over-the-counter medications with anticholinergic properties are currently available, including commonly used antipsychotics, antidepressants, antihistamines, antiemetics, antispasmodics, and nonprescription flu/cold remedies (Fig. 1). Anticholinergic burden is increased when these drugs are used concomitantly, and accidental or intentional overexposure to one or more of these medications can lead to anticholinergic toxicity [36]. For M1/M4 receptor agonists in development for schizophrenia [8, 37], as well as M4 receptor positive allosteric modulators [38], the potentially deleterious effects of centrally acting anticholinergics on antipsychotic efficacy remains to be determined.
The range of adverse effects with anticholinergics is extensive. Peripheral or systemic effects include dry mouth (associated with dental caries), anhidrosis (contributing to heatstroke), tachycardia, constipation or decreased bowel motility, vomiting, urinary retention, blurred vision, and narrow-angle glaucoma [28, 39]. Central effects include impaired cognition and memory loss [28]. Acute anticholinergic toxicity can present as a medical emergency presenting with arrhythmias, seizures, hyperthermia, agitation, and delirium [40]. Treatment of acute toxicity consists of supportive medical care including hemodynamic support, fluid resuscitation, treatment of dysrhythmias, cooling elevated temperatures, treatment of seizures, and consideration of benzodiazepines or physostigmine for agitation and delirium [41].
These central adverse effects of anticholinergics are particularly concerning in older individuals due to decreasing cholinergic reserves in the aging brain [42]. Studies have shown that centrally acting anticholinergics can exacerbate cognitive dysfunction related to schizophrenia, with negative repercussions for cognitive and psychosocial rehabilitation and activities of daily living [43, 44]. In one study, high serum anticholinergic activity was associated with decrements in verbal working memory, verbal learning, and response to cognitive training, independent of age, intelligence quotient, or disease severity [45]. In another study, use of highly anticholinergic medications was associated with poorer cognition, especially immediate memory recall and executive function, along with reduced cortical volume and temporal lobe atrophy [46]. These results may be linked to synaptic loss and neurodegeneration in the cortex and medial temporal lobe caused by decreased cholinergic innervation and activity in these areas due to cholinergic receptor blockade by anticholinergic medications.
4 Differentiation of Drug-Induced Movement Disorders and Their Response to Anticholinergic Treatment
With their wide-ranging phenomenology, differentiating one DIMD from another can be challenging, especially in patients who exhibit simultaneous abnormal movements [4, 9]. DIMDs related to antipsychotic exposure include DIP, akathisia, dystonia, and TD. Historically, these sequelae of D2 receptor antagonism have collectively been referred to as “extrapyramidal symptoms” or “EPS.” The panelists agreed, however, that this terminology is non-specific and encompasses a range of abnormal movements with distinct pathophysiology and clinical presentation, thereby requiring divergent treatment approaches. As such, “EPS” is not useful for conveying a precise clinical characterization, which is critical for effective treatment. However, “extrapyramidal disorder” remains in use in the Medical Dictionary for Regulatory Activities (MedDRA) terminology and is therefore included in product labeling.
The panelists agreed that standards for DIMD evaluation and treatment are covered in the 2020 American Psychiatric Association (APA) guidelines for the treatment of adults with schizophrenia (Table 1) [47]. Per these guidelines, all antipsychotic-treated individuals should be evaluated for DIMDs at every clinical encounter. More formal assessments with a structured instrument, such as the Abnormal Involuntary Movement Scale (AIMS) for TD, are recommended at least every 12 months or every 6 months in patients with high risk for TD (e.g., older age).
4.1 Drug-Induced Parkinsonism
DIP is a form of secondary parkinsonism that is predominantly associated with the use of D2 receptor-blocking antipsychotics. Older adults may be at particular risk, even if they have tolerated these types of medications in the past. Motor symptoms, which include bradykinesia, rigidity, and tremors, generally emerge within days to weeks of antipsychotic initiation or dosage increase [4, 9, 18]. These symptoms often subside when the medication is stopped, the dose is reduced, or the treatment is switched to a different antipsychotic with a lower propensity to cause DIP (e.g., from FGA to SGA) [18]. However, remission of symptoms can take weeks to months, especially in older patients, and symptoms may persist in up to 15% of patients possibly due to tardive parkinsonism or to unmasking of underlying Parkinson’s disease [30].
Anticholinergics that are FDA approved for all forms of parkinsonism include benztropine, trihexyphenidyl, and biperiden. These medications can be used for short-term management while adjustments are made to the patient’s antipsychotic regimen [47]. The panelists agreed that in such cases, anticholinergic treatment could be considered at the lowest effective dose. Although anticholinergic-like symptoms such as dry mouth and constipation have also been reported in previous trials with amantadine, it is a weak uncompetitive N-methyl-D-aspartate (NMDA) receptor with no measurable direct anticholinergic receptor activity. Amantadine has shown beneficial effects in secondary parkinsonism with relatively less peripheral or central adverse effects compared with anticholinergic medications [31]. An extended-release form of amantadine is specifically approved for the treatment of “drug-induced extrapyramidal reactions in adults” in addition to Parkinson’s disease [48]. Strong consideration should be given to preferential use of amantadine for managing DIP, especially in patients who cannot tolerate anticholinergic medications or are at risk for their central and peripheral adverse effects.
4.2 Dystonia
Acute dystonia can occur within hours of initiating antipsychotic treatment or incremental dose adjustment [30]. Risk factors for dystonia include parenteral administration, high doses of high-potency FGAs, younger age, male sex, or recent cocaine use [47]. Dystonia is manifested as the contraction of both agonist and antagonist muscles, resulting in abnormal postures, pain, and cramping sensations [4, 9, 47]. These contractions often occur in the head, neck, face, and back, although the trunk and extremities can also be affected. Characteristic symptoms include torticollis, retrocollis, trismus, grimacing, oculogyric crisis, blepharospasm, and opisthotonos. In rare instances, laryngeal dystonia could impact breathing and swallowing, requiring emergency treatment [4, 47].
Per APA recommendations, intramuscular administration of diphenhydramine or benztropine—both of which have anticholinergic properties—can provide rapid relief of acute dystonic symptoms [47, 49]. In emergency situations where an injectable antipsychotic is indicated (e.g., for a highly agitated patient), an anticholinergic medication is often administered in combination with an antipsychotic and benzodiazepine for rapid control [49]. Alternative approaches for the emergency treatment of agitation include an injectable SGA that has reduced risk of dystonia (e.g., olanzapine, ziprasidone) or sublingual dexmedetomidine [50, 51].
After dystonia is suppressed, oral anticholinergics can be continued for 1–2 days if the antipsychotic was discontinued and for several days if the antipsychotic was not discontinued [30]. The anticholinergic medications should be used at the minimal effective dose and tapered off as soon as changes in the antipsychotic regimen are made to lower the risk of dystonic reactions (e.g., switch to SGA). As discussed in section 5, anticholinergics can also be used prophylactically, although the potential benefits need to be weighed against the risks associated with anticholinergic burden.
Tardive dystonia, sometimes described as belonging to a “tardive syndrome” along with tardive akathisia and TD, can be distinguished from acute dystonic reactions by the delayed time of onset and persistence despite antipsychotic discontinuation [9, 47]. Although controlled clinical trials are needed to determine the best first-line approach for tardive dystonia, early studies suggest that either a vesicular monoamine transporter 2 (VMAT2) inhibitor or relatively high anticholinergic dose may benefit some [52,53,54]. However, VMAT2 inhibitors may be preferred if clinicians want to avoid serious adverse effects of high-dose anticholinergics or potential worsening of concomitant stereotypic TD movements with these medications. Other options for tardive dystonia include antipsychotic modification (e.g., decreasing or increasing dosing, switching to clozapine), botulinum toxin, and neurosurgical interventions [9, 55].
4.3 Akathisia
Patients with akathisia usually experience overwhelming feelings of restlessness and the urge to move, resulting in pacing, walking in place, rocking back and forth, leg swinging, toe tapping, squirming, and fidgeting [4, 9, 30, 56]. Due to similarity in symptoms, akathisia may be misdiagnosed as anxiety, potentially resulting in exacerbation of symptoms if the antipsychotic dosage is increased [56]. Although related to DRBA exposure, akathisia responds poorly to anticholinergics. The lack of supportive evidence for anticholinergic medication in patients with akathisia is documented in a 2006 Cochrane review [57].
Acute akathisia can occur within hours to a few weeks after initiation of antipsychotic treatment or dose change [58]. For many patients, akathisia symptoms improve when the antipsychotic is stopped, the dose is reduced, or the medication is switched to another antipsychotic agent. However, the benefits of these strategies need to be weighed against the possibility of worsening psychiatric symptoms. As a tardive syndrome, tardive akathisia usually can be distinguished from the acute disorder by delayed onset and persistence after antipsychotic discontinuation.
If changing the antipsychotic regimen is not feasible or effective, adjunctive treatment with propranolol, low-dose mirtazapine (7.5–15 mg at bedtime), or a high-potency benzodiazepine (e.g., clonazepam) may be considered for acute akathisia [47, 56]. However, the warnings, precautions, adverse reactions, and drug interactions associated with these medications also need to be considered. Data on treatment of tardive akathisia are limited and may differ from the acute form in the response to DRBA dose modification or specific agents. An early clinical report suggested that reserpine (irreversible and nonselective VMAT2 inhibitor) and tetrabenazine (reversible VMAT2 inhibitor) may be beneficial in the tardive form [59], but they would be expected to worsen acute akathisia. Although a few early clinical reports suggested amantadine may be beneficial in acute persistent akathisia [60], and anticholinergics may reduce symptoms in some patients with acute akathisia especially if experiencing concurrent parkinsonism, the evidence is limited and benefits may be outweighed by risks of anticholinergic side effects [57].
4.4 Tardive Dyskinesia
TD usually emerges after three or more months of DRBA exposure, although some at-risk patients including older adults may develop symptoms more quickly [61]. TD is typically characterized by abnormal involuntary movements in the orofacial region, such as lip smacking, chewing, tongue protrusions, excessive blinking or squinting, and jaw clenching. Symptoms can also arise in other body regions, with writhing, twisting, rocking, and finger/toe tapping among the more common types of movements found in the trunk, limbs, and extremities [4, 9, 18, 62].
There is insufficient evidence for changing antipsychotic regimens or discontinuing antipsychotics to treat TD [63,64,65]. Maintenance of antipsychotic treatment is often necessary in patients with severe psychotic disorders who are at high risk for relapse, stable on their current antipsychotic therapy, and experiencing mild or localized TD symptoms with minimal self-reported impact. Dose reduction can temporarily unmask or worsen motor symptoms while increasing the risk of psychotic decompensation [9, 66]. Antipsychotics with lower D2 receptor affinity or occupancy still carry a risk for TD; however, switching to these medications can be considered in patients who have developed TD, particularly if there are overriding benefits (e.g., switching to clozapine in patients who have treatment-resistant schizophrenia or who are at risk for suicidal behavior) [47, 67]. Discontinuation of antipsychotics has been used in clinical practice, especially in younger patients who have early onset of mild TD, but with limited supportive evidence for remission. This type of approach should only be considered in patients who are likely to safely tolerate withdrawal and have conditions that might not require continuous antipsychotic exposure (e.g., adjunctive treatment for mood disorders).
As reported in a 2018 Cochrane review, there is an absence of compelling evidence to recommend the use of anticholinergics in patients with TD [22]. Moreover, the prescribing labels for both benztropine and trihexyphenidyl indicate that they should not be used for TD and may even aggravate TD (Fig. 2) [68,69,70]. Withdrawal of anticholinergics in patients with TD should be done with cautious tapering after considering the impact on concurrent conditions that may worsen upon anticholinergic withdrawal (e.g., DIP, tardive dystonia), [53].
Once the patient’s antipsychotic and anticholinergic regimens are reassessed and stabilized, specific antidyskinetic strategies can be employed. In the USA, two medications are currently approved for TD in adults: valbenazine and deutetrabenazine. The APA recommends that these medications be considered for adults with moderate-to-severe TD and in adults with milder symptoms on the basis of factors such as patient preference, associated impairment, and impact on psychosocial functioning [47].
Valbenazine, the first medication approved in the USA for TD, is the valine ester of [+]-α-dihydrotetrabenazine, a highly potent and selective VMAT2 inhibitor [71]. Valbenazine is available in three dosing options (40, 60, and 80 mg), all of which are taken as a single capsule once daily [72]. Although patients have been observed to improve with a low dose of 40 mg/day [73, 74], escalation to the target dose of 80 mg/day can be achieved after 1 week of initial treatment with 40 mg [72]. The second VMAT2 inhibitor to be approved in the USA for TD is deutetrabenazine, a deuterated formulation of tetrabenazine. Deutetrabenazine is available as twice-daily tablets (6, 9, and 12 mg) or once-daily extended release tablets (6, 12, and 24 mg) [75]. Treatment for TD requires titration in 6-mg increments weekly after starting treatment at 12 mg/day. As evident from the clinical trials, the effective dose range for TD is 24–48 mg/day, with an observed mean dose of approximately 39 mg/day in a long-term, open-label extension study [76,77,78]. Both valbenazine and deutetrabenazine carry warnings about secondary parkinsonism as a potential adverse effect.
Treatment can be challenging when TD and DIP are both present. In these cases, medication selection should be guided by the primary and most bothersome phenomenology [18]. When dyskinetic movements are the predominant symptom, a VMAT2 inhibitor can be prescribed while monitoring DIP severity. When parkinsonian symptoms are most bothersome, an antiparkinsonian medication should be considered. VMAT2 inhibitors and anticholinergics can be prescribed concurrently, although valbenazine was the only VMAT2 inhibitor that allowed for concomitant anticholinergic use in TD studies. The combined risk of side effects from polypharmacy of VMAT2 inhibitors and anticholinergics should be carefully considered. Amantadine has been suggested as a possible alternative in patients who have both TD and DIP [18, 63]. If modification of the antipsychotic regimen is feasible, switching agents to reduce or eliminate DIP may be considered [18].
4.5 Neuroleptic Malignant Syndrome
NMS is a relatively rare and potentially life-threatening reaction to DRBAs, with high-potency FGAs posing the greatest risk [79]. The clinical manifestations include severe muscle rigidity and tremor along with catatonic symptoms (stupor, mutism) and hypermetabolic systemic signs (hyperthermia, autonomic instability) [30]. The primary recommended approach is DRBA cessation and supportive care [47, 80]. Treatments that have been tried empirically in observational studies, with variable success, include benzodiazepines, dopamine agonists, dantrolene, and electroconvulsive therapy [80].
Anticholinergics are contraindicated in NMS and other cases of hyperthermia or heat-related illness because they block sympathetic innervation of sweat glands, resulting in anhidrosis and potentially complete loss of ability to dissipate a heat load. Consequently, these medications are a major contributing factor to potentially fatal heatstroke in the elderly and in mentally ill patients during increasingly common summer heat waves [81, 82].
5 Prophylactic Use of Anticholinergic Medications
Historically, many clinicians were trained to prescribe anticholinergics concomitantly with antipsychotics—especially FGAs—to prevent the emergence of DIMDs. However, prophylaxis with anticholinergics is not generally recommended, particularly with SGAs that have a lower propensity for DIMDs [83, 84]. Early studies indicated effective prophylaxis with anticholinergics for acute dystonia [85,86,87], but findings have not been consistent and there is no evidence that these medications have an effect in preventing DIP, akathisia, TD, or NMS [83]. Moreover, patients at high risk for DIP are usually older and susceptible to cognitive impairment. Given the potentially deleterious effects of anticholinergics in patients with these characteristics, prophylactic use to prevent DIP should be avoided or monitored carefully [18, 30, 47].
The strongest evidence for prophylactic anticholinergic use is for the prevention of acute dystonia in at-risk individuals (e.g., young, male) [30, 47]. Although prophylactic anticholinergics can be problematic in older adults owing to adverse anticholinergic effects, the benefits of preventing dystonia outweigh the potential risks in younger patients who are receiving parenteral high-potency FGAs and in patients who are paranoid or ambivalent about treatment [9, 30, 47]. The risk of acute dystonia can be largely mitigated by using SGAs [30].
6 Discontinuation and Dose Reduction
The panelists noted that clinicians may not appreciate the need to discontinue anticholinergics if the patient’s clinical status appears unchanged, but they emphasized the need for routine evaluation of anticholinergic need and pharmacological burden. Evaluation can be performed using instruments such as the Anticholinergic Drug Scale (ADS) or Anticholinergic Risk Scale (ARS) [88, 89]. Both scales include a list of medications that are assigned different levels of anticholinergic burden, ranging from 0 to 3 (highest burden). Regular evaluation of each patient’s total medication regimen is needed because many medications have anticholinergic properties (e.g., antipsychotics, antidepressants) that can contribute to anticholinergic burden, even if they are not typically classified as an anticholinergic agent. High anticholinergic burden has been associated with serious adverse outcomes, including increased risks for mortality, fractures, falls, delirium, and dementia or other cognitive impairments [68, 90,91,92,93,94,95,96,97]. Studies have shown that anticholinergic medications can be safely discontinued in those with severe mental illnesses such as schizophrenia, with beneficial outcomes in terms of cognition, mood, and quality of life, along with reduced fall risk in older patients [98,99,100].
After anticholinergic burden is assessed and all potential beneficial and detrimental outcomes are considered, decisions can be made about which medications could be stopped and/or whether doses could be decreased. Abrupt discontinuation can lead to cholinergic rebound, which is characterized by symptoms such as nausea, vomiting, headache, sweating, muscle spasms, abdominal cramping, and urinary urgency [101]. Therefore, for elective discontinuation in stable outpatients without serious anticholinergic toxicity, anticholinergics should be tapered slowly when stopping treatment or reducing the dose.
Given the potential complexity of treating patients with DIMDs, especially those who have two or more distinct DIMDs, the panelists recommended taking an individualized approach when stopping or decreasing anticholinergics in this population. Factors to consider include age, sex, anticholinergic treatment duration, concomitant medications, and current medical/psychiatric conditions including acuity of anticholinergic toxicity. Moreover, patients should be monitored during the tapering process. Slow tapers may be considered in stable outpatients who chose to discontinue anticholinergics and are not currently in distress or experiencing adverse events (i.e., “elective” tapering). In studies that included outpatients with serious mental illness, tapering schedules ranging from approximately 1 to 4 months were associated with reduced anticholinergic burden and significant improvements in cognition and quality of life [99, 102,103,104]. However, a more rapid tapering schedule may be required in patients who are exhibiting signs of anticholinergic toxicity, including confusion, acute glaucoma, bladder obstruction, and heatstroke.
In patients who are experiencing cholinergic rebound or reemergence of DIMDs, tapering may require smaller dose-tapering reductions, longer intervals between dose reductions, and/or stopping anticholinergic withdrawal at the minimally effective dose needed to manage the DIMD. If parkinsonism reemerges, amantadine can be considered to help manage the movements while gradual tapering of the remaining anticholinergic medication continues. In the event that antipsychotics are also discontinued, anticholinergics should be continued for an additional 48–72 hours to prevent emergence or recurrence of acute dystonia. Due to the extended half-lives of most DRBAs, break-through dystonic reactions can occur if both drugs are discontinued simultaneously [105, 106].
7 High-Risk Populations
Older adults, women, and individuals with poor cytochrome P450 metabolism (due to genetic polymorphisms or aging) have an increased risk of incurring a high anticholinergic load [107]. Moreover, older patients often have additional risk factors (e.g., polypharmacy, cognitive decline, medical comorbidities) that contraindicate the use of anticholinergics in this population.
Although multiple medications may sometimes be indicated in severely ill patients, the widespread prevalence of polypharmacy in older adult populations has been well documented, especially in long-term care facilities [108]. However, polypharmacy among older adults in the general population is also common, as demonstrated in a recent study that included 2.12 billion outpatient visits in adults aged ≥ 65 years [109]. This study showed that 1.377 billion visits (65.1%) had older patients who were taking ≥ 2 medications of concern, as classified using American Geriatric Society Beers Criteria® for potentially inappropriate medication [110], and 779 million visits (36.9%) had older adults taking > 5 medications of concern. In a study that included 3.238 community-dwelling adults aged > 65 years, 1814 (56.0%) were taking > 3 medications [111]. Longitudinal analyses in this study showed that polypharmacy was associated with accelerated decline and the eventual need for assisted care. Other populations with high polypharmacy and increased exposure to anticholinergic medications include patients with intellectual disabilities, neurocognitive disorders, or serious mental illness [112,113,114].
As discussed by the expert panel, centrally acting anticholinergics should be used cautiously in patients with cognitive impairment, whether due to advancing age, neurodegenerative disorder, or serious mental illness such as schizophrenia. In a systematic review of patients with preexisting dementia, anticholinergic use was associated with poor clinical outcomes, including mortality, stroke, delirium, poor physical performance, reduced health-related quality of life, and longer hospital stays [115]. Another systematic review, based on studies of patients with schizophrenia, found anticholinergics to be associated with reduced cognitive ability, including impairment in learning, memory, processing speed, attention, and executive function [43].
The potential for anticholinergic abuse and misuse, particularly in forensic settings, is another important consideration. Abuse, defined as taking an anticholinergic for nonmedical or recreational purposes, is attributable to the euphoric, stimulant, and hallucinogenic properties of these drugs [116]. Routes of administration for anticholinergic abuse include ingestion, injection, and smoking (e.g., mixed with tobacco). Misuse is defined as taking an anticholinergic for medical reasons in nonprescribed ways, including for non-intended symptoms and at non-recommended doses. Anticholinergic misuse can occur for various reasons, but the perceived hedonic effects of these drugs likely contribute to why patients take them in nonprescribed ways. Given the potential for abuse or misuse, anticholinergics should be avoided or used with caution in patients with current or prior substance abuse or dependence. In forensic settings, cautionary measures should be taken to prevent diversion for recreational or other nonprescribed uses [117].
8 Conclusions
Although DIMDs share a common cause (i.e., exposure to antipsychotics, antiemetics, or other dopamine D2 receptor antagonists), they are neurobiologically and clinically distinct. As such, DIMDs require accurate diagnoses and appropriate treatment approaches. One common misconception is that anticholinergics can be used to treat, prevent, or reduce any DIMDs, despite the well-documented hazards of high anticholinergic burden and the evidence indicating that these medications are effective for DIP and acute dystonia but not for akathisia, TD, or neuroleptic malignant syndrome. As a result, anticholinergics continue to be overprescribed in conjunction with antipsychotics.
Part of the problem of anticholinergic overuse is the tendency to conflate all DIMDs as “EPS,” which is a non-specific term that encompasses a wide variety of clinical manifestations and distinct treatment strategies. It is challenging to clinically differentiate DIMDs, and the panelists agreed that continued training on the identification and appropriate treatment of these disorders is warranted. In addition, more education is needed on anticholinergic tapering strategies and the serious adverse effects associated with anticholinergic medications, especially in high-risk populations such as the elderly.
References
Stagnitti MN. Trends in antipsychotics purchases and expenses for the U.S. civilian noninstitutionalized population, 1997 and 2007. Statistical Brief #275. Agency for Healthcare Research and Quality, Rockville, MD. 2010.
Ahrnsbra R, Stagnitti MN. Comparision of antidepressant and antipsychotic utilization and expenditures in the U.S. civilian noninstitutionalized population, 2013 and 2018. Statistical Brief #534. Agency for Healthcare Research and Quality, Rockville, MD. 2021.
Gallego JA, Bonetti J, Zhang J, Kane JM, Correll CU. Prevalence and correlates of antipsychotic polypharmacy: a systematic review and meta-regression of global and regional trends from the 1970s to 2009. Schizophr Res. 2012;138:18–28.
Dilks S, Xavier RM, Kelly C, Johnson J. Implications of antipsychotic use: antipsychotic-induced movement disorders, with a focus on tardive dyskinesia. Nurs Clin N Am. 2019;54:595–608.
Muhlbauer V, Mohler R, Dichter MN, Zuidema SU, Kopke S, Luijendijk HJ. Antipsychotics for agitation and psychosis in people with Alzheimer’s disease and vascular dementia. Cochrane Database Syst Rev. 2021;12:CD013304.
Devanand DP. Management of neuropsychiatric symptoms in dementia. Curr Opin Neurol. 2023;36:498–503.
Iversen TSJ, Steen NE, Dieset I, Hope S, Mørch R, Gardsjord ES, et al. Side effect burden of antipsychotic drugs in real life - Impact of gender and polypharmacy. Prog Neuropsychopharmacol Biol Psychiatry. 2018;82:263–71.
Meyer JM, Correll CU. Increased metabolic potential, efficacy, and safety of emerging treatments in schizophrenia. CNS Drugs. 2023;37:545–70.
Hauser RA, Meyer JM, Factor SA, Comella CL, Tanner CM, Xavier RM, et al. Differentiating tardive dyskinesia: a video-based review of antipsychotic-induced movement disorders in clinical practice. CNS Spectr. 2022;27:208–17.
Chepke C, Benning B, Cicero S, Hull M, Giraldo E, Yeaw J, et al. Investigating real-world benztropine usage patterns in movement disorders: claims analysis and health care provider survey results. Prim Care Companion CNS Disord. 2023;25:22m03472.
Schatzberg AF, Nemeroff CB, American Psychiatric Association. The American Psychiatric Association Publishing textbook of psychopharmacology. 5th ed. Arlington: American Psychiatric Association Publishing; 2017.
Yohn SE, Weiden PJ, Felder CC, Stahl SM. Muscarinic acetylcholine receptors for psychotic disorders: bench-side to clinic. Trends Pharmacol Sci. 2022;43:1098–112.
Ho TNT, Abraham N, Lewis RJ. Structure-function of neuronal nicotinic acetylcholine receptor inhibitors derived from natural toxins. Front Neurosci. 2020;14: 609005.
Brocks DR. Anticholinergic drugs used in Parkinson’s disease: an overlooked class of drugs from a pharmacokinetic perspective. J Pharm Pharm Sci. 1999;2:39–46.
Lester DB, Rogers TD, Blaha CD. Acetylcholine-dopamine interactions in the pathophysiology and treatment of CNS disorders. CNS Neurosci Ther. 2010;16:137–62.
Ztaou S, Amalric M. Contribution of cholinergic interneurons to striatal pathophysiology in Parkinson’s disease. Neurochem Int. 2019;126:1–10.
Paz RM, Murer MG. Mechanisms of antiparkinsonian anticholinergic therapy revisited. Neuroscience. 2021;467:201–17.
Ward KM, Citrome L. Antipsychotic-related movement disorders: drug-induced parkinsonism vs. tardive dyskinesia - Key differences in pathophysiology and clinical management. Neurol Ther. 2018;7:233–48.
Kharkwal G, Brami-Cherrier K, Lizardi-Ortiz JE, Nelson AB, Ramos M, Del Barrio D, et al. Parkinsonism driven by antipsychotics originates from dopaminergic control of striatal cholinergic interneurons. Neuron. 2016;91:67–78.
Takeuchi H, Mori Y, Tsutsumi Y. Pathophysiology, prognosis and treatment of tardive dyskinesia. Ther Adv Psychopharmacol. 2022;12:20451253221117310.
Loonen AJ, Ivanova SA. New insights into the mechanism of drug-induced dyskinesia. CNS Spectr. 2013;18:15–20.
Bergman H, Soares-Weiser K. Anticholinergic medication for antipsychotic-induced tardive dyskinesia. Cochrane Database Syst Rev. 2018;1:Cd000204.
Bhidayasiri R, Fahn S, Weiner WJ, Gronseth GS, Sullivan KL, Zesiewicz TA, et al. Evidence-based guideline: treatment of tardive syndromes: report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology. 2013;81:463–9.
Klawans HL, Rubovits R. Effect of cholinergic and anticholinergic agents on tardive dyskinesia. J Neurol Neurosurg Psychiatry. 1974;37:941–7.
Greil W, Haag H, Rossnagl G, Rüther E. Effect of anticholinergics on tardive dyskinesia. A controlled discontinuation study. Br J Psychiatry. 1984;145:304–10.
Yassa R. Tardive dyskinesia and anticholinergic drugs. A critical review of the literature. L’Encephale. 1988;14 Spec No:233–9.
Egan MF, Apud J, Wyatt RJ. Treatment of tardive dyskinesia. Schizophr Bull. 1997;23:583–609.
Nishtala PS, Salahudeen MS, Hilmer SN. Anticholinergics: theoretical and clinical overview. Expert Opin Drug Saf. 2016;15:753–68.
Barrett MJ, Sargent L, Nawaz H, Weintraub D, Price ET, Willis AW. Antimuscarinic anticholinergic medications in Parkinson disease: to prescribe or deprescribe? Mov Disord Clin Pract. 2021;8:1181–8.
Caroff SN, Campbell EC. Drug-induced extrapyramidal syndromes: implications for contemporary practice. Psychiatric Clin North Am. 2016;39:391–411.
Caroff SN, Jain R, Morley JF. Revisiting amantadine as a treatment for drug-induced movement disorders. Ann Clin Psychiatry. 2020;32:198–208.
Bourbeau J, Bhutani M, Hernandez P, Aaron SD, Beauchesne MF, Kermelly SB, et al. 2023 Canadian Thoracic Society guideline on pharmacotherapy in patients with stable COPD. Chest. 2023;164:1159–83.
Fukuda N, Horita N, Kaneko A, Goto A, Kaneko T, Ota E, et al. Long-acting muscarinic antagonist (LAMA) plus long-acting beta-agonist (LABA) versus LABA plus inhaled corticosteroid (ICS) for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2023;6:CD012066.
Hsu FC, Weeks CE, Selph SS, Blazina I, Holmes RS, McDonagh MS. Updating the evidence on drugs to treat overactive bladder: a systematic review. Int Urogynecol J. 2019;30:1603–17.
Mostafaei H, Salehi-Pourmehr H, Jilch S, Carlin GL, Mori K, Quhal F, et al. Choosing the most efficacious and safe orl teatment for iiopathic oeractive badder: a systematic review and network meta-analysis. Eur Urol Focus. 2022;8:1072–89.
Gerretsen P, Pollock BG. Drugs with anticholinergic properties: a current perspective on use and safety. Expert Opin Drug Saf. 2011;10:751–65.
Brannan SK, Sawchak S, Miller AC, Lieberman JA, Paul SM, Breier A. Muscarinic cholinergic receptor agonist and peripheral antagonist for schizophrenia. N Engl J Med. 2021;384:717–26.
Krystal JH, Kane JM, Correll CU, Walling DP, Leoni M, Duvvuri S, et al. Emraclidine, a novel positive allosteric modulator of cholinergic M4 receptors, for the treatment of schizophrenia: a two-part, randomised, double-blind, placebo-controlled, phase 1b trial. Lancet. 2022;400:2210–20.
Stahl SM. Stahl’s essential psychopharmacology: neuroscientific basis and practical applications. 5th ed. Cambridge: Cambridge University Press; 2021.
Dawson AH, Buckley NA. Pharmacological management of anticholinergic delirium - theory, evidence and practice. Br J Clin Pharmacol. 2016;81:516–24.
Wang GS, Baker K, Ng P, Janis GC, Leonard J, Mistry RD, et al. A randomized trial comparing physostigmine vs lorazepam for treatment of antimuscarinic (anticholinergic) toxidrome. Clin Toxicol (Phila). 2021;59:698–704.
Tune LE. Anticholinergic effects of medication in elderly patients. J Clin Psychiatry. 2001;62(Suppl 21):11–4.
Georgiou R, Lamnisos D, Giannakou K. Anticholinergic burden and cognitive performance in patients with schizophrenia: a systematic literature review. Front Psychiatry. 2021;12: 779607.
Kim SJ, Jung D, Shim JC, Moon JJ, Jeon DW, Kim YN, et al. The effect of anticholinergic burden on cognitive and daily living functions in patients with schizophrenia. Asian J Psychiatr. 2019;46:111–7.
Vinogradov S, Fisher M, Warm H, Holland C, Kirshner MA, Pollock BG. The cognitive cost of anticholinergic burden: decreased response to cognitive training in schizophrenia. Am J Psychiatry. 2009;166:1055–62.
Risacher SL, McDonald BC, Tallman EF, West JD, Farlow MR, Unverzagt FW, et al. Association between anticholinergic medication use and cognition, brain metabolism, and brain atrophy in cognitively normal older adults. JAMA Neurol. 2016;73:721–32.
The American Psychiatric Association practice guideline for the treatment of patients with schizophrenia. 3rd ed. Washington, DC: American Psychiatric Association; 2021.
Osmolex ER (amantadine) extended-release tablets. Prescribing information. Emeryville, CA: Adamas Pharma, LLC; 2021.
Satterthwaite TD, Wolf DH, Rosenheck RA, Gur RE, Caroff SN. A meta-analysis of the risk of acute extrapyramidal symptoms with intramuscular antipsychotics for the treatment of agitation. J Clin Psychiatry. 2008;69:1869–79.
Citrome L. Comparison of intramuscular ziprasidone, olanzapine, or aripiprazole for agitation: a quantitative review of efficacy and safety. J Clin Psychiatry. 2007;68:1876–85.
Citrome L, Risinger R, Rajachandran L, Robison H. Sublingual dexmedetomidine for agitation associated with schizophrenia or bipolar disorder: a post hoc analysis of number needed to treat, number needed to harm, and likelihood to be helped or harmed. Adv Ther. 2022;39:4821–35.
Burke RE, Fahn S, Jankovic J, Marsden CD, Lang AE, Gollomp S, et al. Tardive dystonia: late-onset and persistent dystonia caused by antipsychotic drugs. Neurology. 1982;32:1335–46.
Kiriakakis V, Bhatia KP, Quinn NP, Marsden CD. The natural history of tardive dystonia. A long-term follow-up study of 107 cases. Brain. 1998;121:2053–66.
Adityanjee, Aderibigbe YA, Jampala VC, Mathews T. The current status of tardive dystonia. Biol Psychiatry. 1999;45:715–30.
Grabel M, Merola A. Pallidal deep brain stimulation for tardive dystonia: meta-analysis of clinical outcomes. Neurol Sci. 2023;44:827–33.
Salem H, Nagpal C, Pigott T, Teixeira AL. Revisiting antipsychotic-induced akathisia: current issues and prospective challenges. Curr Neuropharmacol. 2017;15:789–98.
Rathbone J, Soares-Weiser K. Anticholinergics for neuroleptic-induced acute akathisia. Cochrane Database Syst Rev. 2006:CD003727.
Miller CH, Fleischhacker WW. Managing antipsychotic-induced acute and chronic akathisia. Drug Saf. 2000;22:73–81.
Burke RE, Kang UJ, Jankovic J, Miller LG, Fahn S. Tardive akathisia: an analysis of clinical features and response to open therapeutic trials. Mov Disord. 1989;4:157–75.
Van Putten T. The many faces of akathisia. Compr Psychiatry. 1975;16:43–7.
American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 5th ed., text revision. Washington, DC: American Psychiatric Association Publishing; 2022.
Waln O, Jankovic J. An update on tardive dyskinesia: from phenomenology to treatment. Tremor Other Hyperkinet Mov (NY). 2013;3:tre-03-161-4138-1.
Caroff SN. A new era in the diagnosis and treatment of tardive dyskinesia. CNS Spectr. 2022;28:4–14.
Caroff SN, Citrome L, Meyer J, Sajatovic M, Goldberg JF, Jain R, et al. A modified Delphi consensus study of the screening, diagnosis, and treatment of tardive dyskinesia. J Clin Psychiatry. 2020;81:19cs12983.
Bhidayasiri R, Jitkritsadakul O, Friedman JH, Fahn S. Updating the recommendations for treatment of tardive syndromes: a systematic review of new evidence and practical treatment algorithm. J Neurol Sci. 2018;389:67–75.
Caroff SN. Overcoming barriers to effective management of tardive dyskinesia. Neuropsychiatr Dis Treat. 2019;15:785–94.
Meltzer HY. Clozapine: balancing safety with superior antipsychotic efficacy. Clin Schizophr Relat Psychoses. 2012;6:134–44.
Joshi YB, Thomas ML, Braff DL, Green MF, Gur RC, Gur RE, et al. Anticholinergic medication burden-associated cognitive impairment in schizophrenia. Am J Psychiatry. 2021;178:838–47.
Trihexyphenidyl hydrochloride tablets. Prescribing information. Kothur-509 228, AP India: Natco Pharma Ltd; 2010.
Benztropine tablets. Prescribing information. Livonia: Major Pharmaceuticals; 2017.
Grigoriadis DE, Smith E, Hoare SRJ, Madan A, Bozigian H. Pharmacologic characterization of valbenazine (NBI-98854) and its metabolites. J Pharmacol Exp Ther. 2017;361:454–61.
INGREZZA® (valbenazine) capsules. Prescribing information. San Diego: Neurocrine Biosciences, Inc.; 2023.
Hauser RA, Factor SA, Marder SR, Knesevich MA, Ramirez PM, Jimenez R, et al. KINECT 3: a phase 3 randomized, double-blind, placebo-controlled trial of valbenazine for tardive dyskinesia. Am J Psychiatry. 2017;174:476–84.
Marder SR, Singer C, Lindenmayer JP, Tanner CM, Comella CL, Verghese C, et al. A phase 3, 1-year, open-label trial of valbenazine in adults with tardive dyskinesia. J Clin Psychopharmacol. 2019;39:620–7.
Austedo (deutetrabenazine) tablets and Austedo XR (deutetrabenazine) extended-release tablets. Prescribing information. Parsippany: Teva Pharmaceuticals USA, Inc.; 2023.
Anderson KE, Stamler D, Davis MD, Factor SA, Hauser RA, Isojarvi J, et al. Deutetrabenazine for treatment of involuntary movements in patients with tardive dyskinesia (AIM-TD): a double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Psychiatry. 2017;4:595–604.
Fernandez HH, Factor SA, Hauser RA, Jimenez-Shahed J, Ondo WG, Jarskog LF, et al. Randomized controlled trial of deutetrabenazine for tardive dyskinesia: the ARM-TD study. Neurology. 2017;88:2003–10.
Fernandez HH, Stamler D, Davis MD, Factor SA, Hauser RA, Jimenez-Shahed J, et al. Long-term safety and efficacy of deutetrabenazine for the treatment of tardive dyskinesia. J Neurol Neurosurg Psychiatry. 2019;90:1317–23.
Nielsen RE, Wallenstein Jensen SO, Nielsen J. Neuroleptic malignant syndrome-an 11-year longitudinal case-control study. Can J Psychiatry. 2012;57:512–8.
Caroff SN, Mann SC, Sullivan KA, Campbell EC. Neuroleptic malignant syndrome. In: Frucht SJ, editor. Movement disorder emergencies: diagnosis and treatment. 3rd ed. Cham: Humana Press; 2022.
Caroff SN, Mann SC, Campbell EC. Risk of fatal heatstroke after hospitalization. Psychiatr Serv. 2000;51:938-a.
Davariashtiyani A, Taherkhani M, Fattahpour S, Vitousek S. Exponential increases in high-temperature extremes in North America. Sci Rep. 2023;13:19177.
Prophylactic use of anticholinergics in patients on long-term neuroleptic treatment. A consensus statement. World Health Organization heads of centres collaborating in WHO co-ordinated studies on biological aspects of mental illness. Br J Psychiatry. 1990;156:412.
Desmarais JE, Beauclair L, Margolese HC. Anticholinergics in the era of atypical antipsychotics: short-term or long-term treatment? J Psychopharmacol (Oxford, England). 2012;26:1167–74.
Stern TA, Anderson WH. Benztropine prophylaxis of dystonic reactions. Psychopharmacology. 1979;61:261–2.
Winslow RS, Stillner V, Coons DJ, Robison MW. Prevention of acute dystonic reactions in patients beginning high-potency neuroleptics. Am J Psychiatry. 1986;143:706–10.
Boyer WF, Bakalar NH, Lake CR. Anticholinergic prophylaxis of acute haloperidol-induced acute dystonic reactions. J Clin Psychopharmacol. 1987;7:164–6.
Carnahan RM, Lund BC, Perry PJ, Pollock BG, Culp KR. The Anticholinergic Drug Scale as a measure of drug-related anticholinergic burden: associations with serum anticholinergic activity. J Clin Pharmacol. 2006;46:1481–6.
Rudolph JL, Salow MJ, Angelini MC, McGlinchey RE. The anticholinergic risk scale and anticholinergic adverse effects in older persons. Arch Intern Med. 2008;168:508–13.
Ali S, Peterson GM, Bereznicki LR, Salahudeen MS. Association between anticholinergic drug burden and mortality in older people: a systematic review. Eur J Clin Pharmacol. 2020;76:319–35.
Graves-Morris K, Stewart C, Soiza RL, Taylor-Rowan M, Quinn TJ, Loke YK, et al. The prognostic value of anticholinergic burden measures in relation to mortality in older individuals: a systematic review and meta-analysis. Front Pharmacol. 2020;11:570.
Mehdizadeh D, Hale M, Todd O, Zaman H, Marques I, Petty D, et al. Associations between anticholinergic medication exposure and adverse health outcomes in older people with frailty: a systematic review and meta-analysis. Drugs Real World Outcomes. 2021;8:431–58.
Stewart C, Taylor-Rowan M, Soiza RL, Quinn TJ, Loke YK, Myint PK. Anticholinergic burden measures and older people’s falls risk: a systematic prognostic review. Ther Adv Drug Saf. 2021;12:20420986211016644.
Reinold J, Schafer W, Christianson L, Barone-Adesi F, Riedel O, Pisa FE. Anticholinergic burden and fractures: a systematic review with methodological appraisal. Drugs Aging. 2020;37:885–97.
Egberts A, Moreno-Gonzalez R, Alan H, Ziere G, Mattace-Raso FUS. Anticholinergic drug burden and delirium: a systematic review. J Am Med Dir Assoc. 2021;22(65–73): e4.
Taylor-Rowan M, Edwards S, Noel-Storr AH, McCleery J, Myint PK, Soiza R, et al. Anticholinergic burden (prognostic factor) for prediction of dementia or cognitive decline in older adults with no known cognitive syndrome. Cochrane Database Syst Rev. 2021;5:CD013540.
Richardson K, Fox C, Maidment I, Steel N, Loke YK, Arthur A, et al. Anticholinergic drugs and risk of dementia: case-control study. BMJ. 2018;361: k1315.
Lupu AM, MacCamy KL, Gannon JM, Brar JS, Chengappa KNR. Less is more: deprescribing anticholinergic medications in persons with severe mental illness. Ann Clin Psychiatry. 2021;33:80–92.
Desmarais JE, Beauclair L, Annable L, Bélanger MC, Kolivakis TT, Margolese HC. Effects of discontinuing anticholinergic treatment on movement disorders, cognition and psychopathology in patients with schizophrenia. Ther Adv Psychopharmacol. 2014;4:257–67.
Ailabouni N, Mangin D, Nishtala PS. DEFEAT-polypharmacy: deprescribing anticholinergic and sedative medicines feasibility trial in residential aged care facilities. Int J Clin Pharm. 2019;41:167–78.
Howland RH. Potential adverse effects of discontinuing psychotropic drugs. Part 1: adrenergic, cholinergic, and histamine drugs. J Psychosoc Nurs Ment Health Serv. 2010;48:11–4.
de Leon J, Canuso C, White AO, Simpson GM. A pilot effort to determine benztropine equivalents of anticholinergic medications. Hosp Community Psychiatry. 1994;45:606–7.
Ogino S, Miyamoto S, Tenjin T, Kitajima R, Ojima K, Miyake N, et al. Effects of discontinuation of long-term biperiden use on cognitive function and quality of life in schizophrenia. Prog Neuropsychopharmacol Biol Psychiatry. 2011;35:78–83.
Lupu AM, Clinebell K, Gannon JM, Ellison JC, Chengappa KNR. Reducing anticholinergic medication burden in patients with psychotic or bipolar disorders. J Clin Psychiatry. 2017;78:e1270–5.
Corre KA, Niemann JT, Bessen HA. Extended therapy for acute dystonic reactions. Ann Emerg Med. 1984;13:194–7.
Kowalski JM. Medication-induced dystonic reactions. 2022. https://emedicine.medscape.com/article/814632-overview. Accessed 4 Apr 2023.
Trenaman SC, Bowles SK, Andrew MK, Goralski K. The role of sex, age and genetic polymorphisms of CYP enzymes on the pharmacokinetics of anticholinergic drugs. Pharmacol Res Perspect. 2021;9: e00775.
Roh E, Cota E, Lee JP, Madievsky R, Eskildsen MA. Polypharmacy in nursing homes. Clin Geriatr Med. 2022;38:653–66.
Young EH, Pan S, Yap AG, Reveles KR, Bhakta K. Polypharmacy prevalence in older adults seen in United States physician offices from 2009 to 2016. PLoS ONE. 2021;16: e0255642.
2023 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2023 updated AGS Beers Criteria® for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2023;2023(71):2052–81.
Edjolo A, Dartigues JF, Peres K, Proust-Lima C. Heterogeneous long-term trajectories of dependency in older adults: the PAQUID cohort, a population-based study over 22 years. J Gerontol A Biol Sci Med Sci. 2020;75:2396–403.
Ward LM, Cooper SA, Henderson A, Stanley B, Greenlaw N, Pacitti C, et al. A study on prescriptions contributing to the risk of high anticholinergic burden in adults with intellectual disabilities: retrospective record linkage study. Ann Gen Psychiatry. 2022;21:41.
Thayer N, White S, Islam J, Jones W, Kenzie S, Kullu R. Reducing risks associated with medicines and lifestyle in a residential care population with intellectual disabilities: evaluation of a pharmacy review initiative in England. BMJ Open. 2021;11: e046630.
Maltais A, Simard M, Vedel I, Sirois C. Changes in polypharmacy and psychotropic medication use after diagnosis of major neurocognitive disorders: a population-based study in Quebec, Canada. Alzheimer Dis Assoc Disord. 2022;36:222–9.
Wang K, Alan J, Page AT, Dimopoulos E, Etherton-Beer C. Anticholinergics and clinical outcomes amongst people with pre-existing dementia: a systematic review. Maturitas. 2021;151:1–14.
Land W, Pinsky D, Salzman C. Abuse and misuse of anticholinergic medications. Hosp Community Psychiatry. 1991;42:580–1.
Collins TN, Avondoglio JB, Terry LM. Correctional psychopharmacology: pitfalls, challenges, and victories of prescribing in a correctional setting. Int Rev Psychiatry. 2017;29:34–44.
Acknowledgements
The authors would like to recognize the contributions of our colleague, Evelyn Parrish, who was the Director of the Graduate Nursing Programs at the University of Kentucky College of Nursing. Dr. Parrish’s valuable insights from the roundtable are reflected in this article. They would also like to acknowledge Prescott Medical Communications Group for assistance with drafting and revising the manuscript, providing research and graphics support, and formatting the final approved manuscript for submission.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The expert roundtable referenced in this article was supported by Neurocrine Biosciences, Inc. (San Diego, CA). Writing and editorial assistance from Prescott Medical Communications Group (Chicago, IL) and open access funding were also provided by Neurocrine Biosciences.
Conflicts of Interests/Competing Interests
N.V.A. has received consulting honoraria from Neurocrine Biosciences and research support from the National Institutes of Health and the Parkinson’s Disease Foundation. S.N.C. has served as a consultant to Neurocrine Biosciences, Teva Pharmaceuticals, Osmotica Pharmaceuticals, and Dispersol Technologies. S.N.C. has received separate research grants from Neurocrine Biosciences, Osmotica Pharmaceuticals, and Eagle Pharmaceuticals. L.C. has received consulting fees from: AbbVie/Allergan, Acadia, Adamas, Alkermes, Angelini, Astellas, Avanir, Axsome, Biogen, BioXcel, Boehringer Ingelheim, Cadent Therapeutics, Cerevel, Clinilabs, COMPASS, Delpor, Eisai, Enteris BioPharma, HLS Therapeutics, Idorsia, Inmune Bio, Impel, Intra-Cellular Therapies, Janssen, Karuna, Lundbeck, Luye, Lyndra, MapLight, Marvin, Medavante-ProPhase, Merck, Mitsubishi-Tanabe Pharma, Neumora, Neurocrine, Neurelis, Noema, Novartis, Noven, Otsuka, Ovid, Praxis, Recordati, Relmada, Reviva, Sage, Sumitomo/Sunovion, Supernus, Teva, University of Arizona, Vanda, Wells Fargo, and one-off ad hoc consulting for individuals/entities conducting marketing, commercial, or scientific scoping research; is a speaker for: AbbVie/Allergan, Acadia, Alkermes, Angelini, Axsome, BioXcel, Eisai, Idorsia, Intra-Cellular Therapies, Janssen, Lundbeck, Neurocrine, Noven, Otsuka, Recordati, Sage, Sunovion, Takeda, Teva, and CME activities organized by medical education companies such as Medscape, NACCME, NEI, Vindico, and universities and professional organizations/societies; owns stocks (small number of shares of common stock) in: Bristol-Myers Squibb, Eli Lilly, J & J, Merck, Pfizer purchased > 10 years ago, stock options: Reviva; and receives royalties/publishing income from: Taylor & Francis (Editor-in-Chief, Current Medical Research and Opinion, 2022-date), Wiley (Editor-in-Chief, International Journal of Clinical Practice, through end 2019), UpToDate (reviewer), Springer Healthcare (book), and Elsevier (Topic Editor, Psychiatry, Clinical Therapeutics) J.C. has been a consultant or advisor to Neurocrine Biosciences, Sunovion, Takeda, AstraZeneca, Otsuka, Janssen, Actavis (now AbbVie), Lundbeck, and Alkermes. R.S.M. has received research support from the Global Alliance for Chronic Diseases, Canadian Institutes of Health Research, National Natural Science Foundation of China-Mental Health Team, and the Milken Institute, and speaker/consultation fees from Lundbeck, Janssen, Alkermes, Neumora Therapeutics, Boehringer Ingelheim, Sage, Biogen, Mitsubishi Tanabe, Purdue, Pfizer, Otsuka, Takeda, Neurocrine Biosciences, Neurawell, Sunovion, Bausch Health, Axsome, Novo Nordisk, Kris Pharma, Sanofi, Eisai, Intra-Cellular Therapies, NewBridge Pharmaceuticals, Viatris, Abbvie, and Atai Life Sciences. R.S.M. is the CEO of Braxia Scientific Corp. J.M.M. has served as a speaker or advisor for Alkermes, AbbVie, Axsome, BioXcel, Cerevel, Intra-Cellular Therapies, Karuna, Neurocrine Biosciences, Noven Pharmaceuticals, Otsuka America, Inc., Relmada, Sunovion, and Teva Pharmaceuticals. A.P. has received consultant or speaker fees from Neurocrine Biosciences, Avanir Pharmaceuticals, Acadia Pharmaceuticals, Otsuka, Allergan (now AbbVie), Teva Pharmaceuticals, AstraZeneca, and Takeda. J.M.S. has received consulting fees from Neurocrine Biosciences. K.F., R.M., and S.A.C. are full-time employees of Neurocrine Biosciences, Inc. L.L. is a former full-time employee of Neurocrine Biosciences.
Author Contributions
N.V.A. and S.N.C. were involved in the conception and planning of the expert roundtable discussion. N.V.A., S.N.C., L.C., J.C., R.S.M., J.M.M., A.P., and J.M.S. participated in the expert roundtable featured in this article. All authors contributed to the development of the manuscript and provided critical reviews of multiple drafts. All authors approved the final draft for submission.
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent to Publish
Not applicable.
Availability of Data and Material
Not applicable.
Code Availability
Not applicable.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Vanegas-Arroyave, N., Caroff, S.N., Citrome, L. et al. An Evidence-Based Update on Anticholinergic Use for Drug-Induced Movement Disorders. CNS Drugs 38, 239–254 (2024). https://doi.org/10.1007/s40263-024-01078-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40263-024-01078-z