Abstract
Background
Unprotected and fragile elderly people in nursing homes experienced the highest mortality rates during the initial coronavirus disease 2019 (COVID-19) pandemic.
Objective
Our aim was to study the role of two oral anti-inflammatory drugs, colchicine and prednisone, in elderly patients with COVID-19 in geriatric centers.
Methods
A phase II/III, randomized, controlled, multicenter clinical trial was performed in a geriatric population comparing the efficacy and safety of an oral combination of prednisone (60 mg/day for 3 days) and colchicine (at loading doses of 1–1.5 mg/day for 3 days, followed by 0.5 mg/day for 11 days) with the standard treatment, based on intravenous dexamethasone. Primary endpoints assessed the efficacy in reducing death or the modified endpoint death/therapeutic failure to the study drugs over a 28-day period, while secondary endpoints included safety, laboratory changes, and additional therapies used.
Results
Fifty-four patients (35 female/19 male) were enrolled, 25 (46.3%) of whom were allocated to the experimental arm and 29 (53.7%) to the control arm. At day 28, no differences in deaths were observed. The combination of mortality or therapeutic failure occurred in 12 (45.13%) patients receiving dexamethasone and 6 (28.13%) patients receiving colchicine/prednisone, resulting in a reduction of risk difference (RD) of − 17% (p = 0.17), with an average reduction of 39% (risk ratio [RR] 0.61) in patients receiving colchicine/prednisone (p = 0.25). Control patients received higher amounts of additional glucocorticoids (p = 0.0095) over a longer time frame (p = 0.0003). Colchicine/prednisone significantly reduced ferritin levels at day 14, as well as d-dimer and lactate dehydrogenase (LDH) levels at day 28. Adverse events were similar in both groups.
Conclusions
The combination colchicine/prednisone compared with intravenous dexamethasone has shown a remarkable trend to increase disease survival over a 28-day period in elderly patients requiring oxygen therapy in geriatric centers, without safety issues.
Clinical Trial Registry
Clinical Trials Registration Number: NCT04492358.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The FRAGILE-COLCOVID19 clinical trial was designed with the aim of reducing the excess mortality due to coronavirus disease 2019 (COVID-19) in fragile elderly patients admitted to nursing homes and other geriatric facilities with a simple oral combination of two anti-inflammatory drugs, colchicine and prednisone. |
Although not significant, the early administration of colchicine and prednisone in elderly patients with COVID-19 requiring oxygen therapy has shown a remarkable trend to reduce mortality or therapeutic failure over a 28-day period compared with intravenous dexamethasone, with no safety issues. |
In elderly patients, the oral combination of colchicine and prednisone has also shown positive results in terms of disease survival and other parameters of severity. |
1 Introduction
To June 2022, approximately 530 million people worldwide had been officially infected by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and more than 6 million people have died from coronavirus disease 2019 (COVID-19) [1]. By April 2020, the overall mortality rate for COVID-19 inpatients was reported to be 28% [2, 3]. Older age was identified as a risk factor associated with in-hospital death [2, 3] and more than 80% of COVID-19 deaths initially occurred in individuals over 60–65 years of age [3, 4]. Mortality was clearly higher in hospitalized patients with COVID-19 older than 65 years of age (reported to be from 27 to 37%) compared with patients younger than 65 years of age (reported to be from 2 to 10.6%) [3, 5, 6]. Indeed, the mortality rate was higher than 30 and 50% in COVID-19 patients older than 70 and 80 years of age, respectively [3, 5]. Furthermore, comparing the population aged > 65 years with patients aged < 30 years, those patients between 65 and 74, 75 and 84 and > 85 years of age experienced a mortality increase of 5, 8, and 13 times, respectively [4, 5]. During the first two waves of the COVID-19 pandemic in 2020, elderly people in nursing homes were considered an unprotected, fragile, and vulnerable population since they experienced the highest mortality rate, accounting for 30–60% of all COVID-19-related deaths (66% in Spain, 51% in Belgium, 50% in France, and 37% in Germany) [7, 8].
Patients with severe COVID-19 experience hyperinflammatory responses comparable with macrophage activation syndrome (MAS) [9,10,11]. Because of their anti-inflammatory properties, glucocorticoids, anti-interleukin (IL)-6 and anti-IL-1 therapies have already shown to be effective in MAS secondary to autoimmune diseases and chimeric antigen receptor (CAR) T-cell therapies [10]. Similarly, different types of glucocorticoids have been useful in reducing mortality in COVID-19 patients needing oxygen therapy [12,13,14,15,16,17], and several biologic agents, including tocilizumab, anakinra and baricitinib, have also proven efficacy in moderate-to-severe COVID-19 over time [18, 19]. Colchicine, a natural alkaloid orally used in different autoinflammatory diseases exhibits powerful and rapid anti-inflammatory effects [20,21,22]. Moreover, colchicine antiviral activity has also been described [23]. Because of their anti-inflammatory properties, colchicine and prednisone were postulated to be potential therapeutic candidates in SARS-CoV-2 infection. With regard to prednisone, this oral glucocorticoid was previously tested in two COVID-19 clinical trials prematurely terminated at the time of the formal approval of glucocorticoids for COVID-19 (NCT04344288 and NCT04451174 [24]), and no other clinical trials using prednisone for treating COVID-19 patients have been designed or published.
The global aim of this study was to contribute to reducing the excess mortality due to COVID-19 in elderly patients admitted to nursing homes and other geriatric facilities, with simple therapeutic strategies, easy to administer, and without the need for complex controls. The specific aim of this clinical trial was to study the utility of the early administration of a combination of two oral drugs with anti-inflammatory properties (colchicine and prednisone) in elderly patients with COVID-19 requiring oxygen therapy in geriatric centers.
2 Methods
2.1 Trial Design
A phase II/III, randomized, controlled, multicenter clinical trial was conducted to compare the efficacy and safety of an oral combination of colchicine and prednisone with the standard treatment approved by the participant centers, based on intravenous dexamethasone, for moderate/severe COVID-19 in elderly patients admitted to geriatric facilities. The two participating centers in the city of Barcelona (Spain) were the Geriatric Unit of the Department of Internal Medicine, Hospital Clínic of Barcelona, and the transitional care or nursing center Clínica Sant Antoni.
2.1.1 Inclusion and Exclusion Criteria
Patients’ inclusion criteria were: (1) ≥ 65 years of age admitted to a geriatric hospital unit or a transitional care center; (2) clinical diagnosis of SARS-CoV-2 infection microbiologically confirmed by real-time quantitative polymerase chain reaction (RT-PCR) or rapid antigen test (at any time during the current episode if clinical manifestations at admission were attributed to COVID-19), and a moderate (score of 4, requiring oxygen by mask or nasal prongs) to severe (score of 5, requiring non-invasive or high-flow oxygen ventilation) disease according to the 2020 World Health Organization (WHO) Ordinal Scale for Clinical Improvement [25]; (3) general condition allows swallowing of the oral medication, without risk of aspiration pneumonia; and (4) provided written informed consent to participate in the study (by the patient or responsible family member).
The exclusion criteria were (1) patients who were critically ill or in a terminal situation with very little or no expectancy to respond to specific treatment and with a vital prognosis limited to weeks or months; (2) unable to swallow safely; (c) taking the study drugs (colchicine or prednisone) chronically or 7 days before enrollment in the study (before inclusion in the study, one or two doses of any type of glucocorticoid, usually administered at the Emergency Department, were permitted and these doses were accounted for as concomitant medication); (d) concomitant treatment with macrolides (e.g. clarithromycin, erythromycin, telithromycin, azithromycin), antifungals (e.g. itraconazole, ketoconazole), cyclosporine and antiviral agents (e.g. lopinavir/ritonavir, indinavir, nelfinavir, saquinavir) because of potential drug interactions through the cytochrome P450 family 3 subfamily A member 4 (CYP3A4); (4) previous known hypersensitivity to any of the study drugs or their excipients; and (5) experiencing severe renal insufficiency (glomerular filtration rate [GFR] < 30 mL/min or undergoing hemodialysis), severe liver failure (defined as the development of severe acute liver injury with encephalopathy and impaired synthetic function, with an international normalized ratio [INR] of 1.5 or higher, in a patient without cirrhosis or pre-existing liver disease), digestive disorders (including gastrointestinal bleeding, severe diarrhea, and abdominal pain) or blood dyscrasias.
Intravenous medications without formal approval by drug regulatory authorities at the time the study was designed and conducted, such as remdesivir, anticytokine agents (e.g. tocilizumab, anakinra, or baricitinib), or convalescent plasma, were not initially permitted. If any clinical deterioration occurred during the study period and these medications were subsequently administered, together with escalation of ventilatory measures (e.g. from low-flow oxygen therapy to laminar air flow [LAF] ventilation or non-invasive/invasive ventilatory support), these clinical worsening situations would be considered a therapeutic failure.
2.1.2 Therapeutic Arms and Follow-Up Visits
The two therapeutic strategies were (1) experimental arm: a short course of oral prednisone (60 mg/day, for 3 consecutive days) coadministered with colchicine (at initial loading doses of 1–1.5 mg/day over 3 days, adjusted for renal function and body weight [electronic supplementary Table 1], followed by 0.5 mg/day for 14 days in total); and (2) control arm: standard treatment based on intravenous dexamethasone, since dexamethasone at 6 mg/day had demonstrated to decrease mortality in COVID-19 patients on oxygen or mechanical ventilation [14]; its use was supported by the WHO by June 2020 [26] and formally approved by the European Medicines Agency (EMA) by October 2020 [27]. A previous dose of any type of glucocorticoid was permitted. Patients were followed over a 28-day period and visited daily during the first 14 days of hospitalization or until discharge. After hospital discharge, daily telephone visits were undertaken until day 28. If possible, a last on-site visit was scheduled on day 28 or was undertaken by telephone when patients could not physically attend medical facilities.
2.1.3 Ethical Considerations
This study was approved by the Clinical Research Ethics Committee at the Hospital Clínic of Barcelona (EudraCT Number 2020-002462-14) and registered in the US Clinical Trials Registry (ClinicalTrials.gov identifier: NCT04492358). All patients or their legally authorized representatives signed the informed consent form.
2.2 Study Endpoints
The primary endpoints were to assess efficacy in terms of reduction in mortality and the extended combination of death or therapeutic failure to the experimental drugs compared with the standard treatment over a 28-day period after randomization. Therapeutic failure was modified as a primary outcome variable in the study protocol together with death during the study time by consensus of all investigators after realizing that some patients needed to be rescued from life‐threatening situations leading to a fatal outcome, by using measures not contemplated in the initial protocol design. As mentioned in the inclusion and exclusion criteria, hospitalized patients requiring the use of intravenous or oral medications without formal approval by drug regulatory authorities at that time and/or escalation from low-flow oxygen therapy to LAF ventilation or non-invasive/invasive ventilatory support, as well as patients requiring to be transferred to an intensive care unit (ICU) for any reason and those patients admitted at the transitional care center experiencing any clinical deterioration tributary of being treated in a tertiary hospital, were all considered patients experiencing therapeutic failure.
Secondary endpoints included the analysis of safety (adverse events), duration, and severity of symptoms, laboratory changes, and additional therapies used until day 28 in both groups.
2.3 Data Collection
Data on clinical manifestations, oxygen requirements and saturation (SpO2) levels, patients’ respiratory condition according to the 2020 WHO-8 Ordinal Scale for Clinical Improvement of COVID-19 [25], physical examination, vital signs, concomitant medications administered, and adverse effects were collected every day during admission and at the day 28 visit (when possible). The time elapsed from the onset of COVID-19 symptoms and time from SARS-CoV-2 microbiological confirmation until inclusion in the study, as well as previous comorbidities, were recorded at the screening visit. Blood tests and a chest X-ray were performed at baseline in all patients and at days 14 and 28 (when possible). Recorded laboratory parameters were C-reactive protein (CRP), ferritin, d-dimer, lactate dehydrogenase (LDH), alanine transaminase (ALT), aspartate aminotransferase (AST), GFR and creatinine levels, and hemoglobin, platelet, and total lymphocyte counts.
2.4 Outcome Measures
Death of any cause, therapeutic failure, and adverse effects occurring during the 28-day period were compared between groups. All clinical, laboratory, and oxygen therapy variables as well as chest X-ray results were compared between groups at days 14 and 28. Concomitant medications administered during the study were recorded, with special interest in glucocorticoids, from which all different types and doses were converted to prednisone-equivalent doses according to the Steroid Conversion Calculator (Harvard Medical School/Massachusetts General Hospital) [28], for comparison purposes. The concomitant use of low-molecular-weight heparin and antibiotics were also analyzed.
2.5 Statistical Analyses
The trial was designed for superiority, to determine if the experimental treatment was well tolerated and reduced mortality by 20% in vulnerable and fragile adult patients with COVID-19 admitted to geriatric facilities, compared with the standard approved treatment. Patients were centrally assigned to one group, at a ratio of 1:1, and the random assignment was performed in blocks. The investigators were blinded to the size of the randomization blocks and also to the allocation of each patient. The initial estimated sample size of 144 individuals was determined sufficient to detect a 20% decrease in mortality and death/therapeutic failure rates at day 28, with 80% power. The 20% rate represented the difference between the expected mortality rates of 30% and 10% in the control and experimental groups, respectively. The statistical test used was the bilateral Fisher’s exact test with a significance level of 5%.
Continuous or ordinal variables were presented as means and standard deviations (SDs) or medians and interquartile ranges (percentiles 25th–75th), as appropriate, and were compared using Students t test or Mann–Whitney U test. Categorical variables were expressed as absolute numbers and frequencies and compared using Fisher’s exact test. The primary endpoints (reduction of mortality or death/therapeutic failure at day 28) and the secondary endpoints were estimated using a binomial regression model including the stratification variable (type of center), and expressed as risk differences (RDs). As additional sensitivity analysis, risk ratios (RRs) were also calculated setting the link to log (log-binomial model). Longitudinal continuous variables were analyzed using the mixed models of repeated measures (MMRMs) approach. MMRM analyses included the fixed, categorical effects of treatment, type of center, time, and treatment-by-time interaction, as well as the continuous, fixed covariates of baseline score and baseline score-by-time interaction.
All analyses were performed according to the intention-to-treat (ITT) principle. A per-protocol analysis was also carried out. Two-sided p values < 0.05 were considered statistically significant. Calculation of the sample size was carried out using PASS 15 Power Analysis and Sample Size Software (2017; NCSS LLC, Kaysville, UT, USA), and all statistical analyses were performed using SAS v9.4 software (SAS Institute, Cary, NC, USA).
3 Results
3.1 Flow of Participants
Among the 56 individuals who underwent screening, 54 (35 females and 19 males) met the inclusion criteria and were enrolled in the study for ITT and safety analyses. Two patients were excluded from the per-protocol population. After randomization, 29 (53.7%) patients were allocated to the control arm (intravenous dexamethasone) and 25 (46.3%) were allocated to the experimental arm (oral colchicine/prednisone). Figure 1 shows the study flow diagram and the data regarding completion of the study.
Study flow diagram. aPatients excluded from the per-protocol population: one patient was taking prednisone 5 mg/day before enrollment in the study (met an exclusion criteria), and one patient stopped taking the study medications at day 3 for a decision from the patient’s treating physician. In the ITT/safety population, 42 (77.8%) patients completed the study [23 (79.3%) and 19 (76%) cases in the control and experimental groups, respectively]. Thirty-five (66%) patients completed the study according to the protocol [17 (73.9%) in the control group and 18 (94.7%) in the experimental group]. ITT intention-to-treat, PP per-protocol
3.2 Baseline Patient Characteristics and Differences Between Groups
Mean ± SD age of all patients was 83.8 ± 8.1 years. The remaining demographic data, time elapsed from COVID-19 symptoms and a positive SARS-CoV-2 test to randomization, COVID-19 clinical manifestations, and baseline SpO2 are illustrated in Table 1, while laboratory results are reported in Table 2. Medical comorbidities are included in electronic supplementary Table 2.
All hospitalized patients received oxygen and anti-inflammatory therapy in the early phase, since all required low-flow oxygen by mask or nasal prongs, which corresponds with mild/moderate disease (score 4) according to the 2020 WHO-8 Scale [25]. Chest X-ray was performed in 45 (83.3%) patients, and unilateral and bilateral pneumonia was observed in 7 (15.6%) and 37 (82.2%) of these patients, respectively.
Baseline heart and respiratory rates, blood pressure, and body temperature values did not differ between groups. Furthermore, no differences were observed at the screening visit with regard to age, sex, comorbidities, time from the initial symptoms to study inclusion, clinical manifestations, oxygen and laboratory parameters, and chest X-ray results (Tables 1, 2).
3.3 Outcomes
3.3.1 Mortality (Death and Therapeutic Failure)
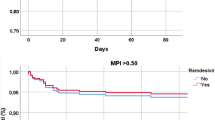
At day 28, 20.97% (95% confidence interval [CI] 3.67–38.37%; n = 5) of patients in the control arm and 22.64% (95% CI 4.56–40.71%; n = 5) of patients in the experimental arm died, with an RD of 1.67% (95% CI −19.01 to 22.35%; p = 0.87). However, 7 (24.1%) patients in the control group and 1 (4%) patient receiving colchicine/prednisone experienced therapeutic failure, requiring rescue measures to avoid death, during the study period. Therefore, the combination of mortality or therapeutic failure was 45.13% (95% CI 25.64–64.62%; n = 12) in controls (dexamethasone) and 28.13% (95% CI 8.24–48.02%; n = 6) in the experimental (colchicine/prednisone) group. The RD in mortality/therapeutic failure was − 17% (95% CI − 41.35 to 7.35%; p = 0.17), with a mortality/therapeutic failure reduction of 39% in patients receiving the experimental treatment (RR 0.61, 95% CI 0.26–1.41; p = 0.25).
3.3.2 Safety Analysis
Adverse events occurred without significant differences between groups. At least one adverse effect was similarly developed in the control and experimental groups (n = 20 [69%] vs. n = 15 [60%]; p = 0.58), while serious adverse events tended to occur more often in control patients (n = 12 [41.4%] vs. n = 7 [28%]; p = 0.40). Treatment-related adverse effects were only observed in patients receiving colchicine/prednisone (n = 6; 24%), and consisted of mild diarrhea (n = 5) and reversible palmar erythema (n = 1). No patients developed treatment-related severe adverse effects. Finally, 6 (20%) control patients and 5 (20%) experimental patients developed adverse events with an outcome of death during or after the study period (p > 0.99).
Although adverse events classified by etiology did not differ between groups, serious vascular thrombotic complications occurred in 4 (13.8%) patients receiving dexamethasone (Table 3).
3.3.3 Additional Therapeutic Measures and Medications Administered
Among the 16 patients experiencing therapeutic failure or dying (12 in the control group and 6 in the experimental group), different additional measures (one or more per patient) were employed. Two patients from the transitional care center were transferred to a tertiary hospital and four hospitalized patients were admitted to the ICU. High-flow oxygen therapy was administered to nine patients, two were intubated, and eight patients concomitantly received non-permitted medications (per protocol): anakinra (n = 2), tocilizumab (n = 1), remdesivir alone or with tocilizumab (n = 3), convalescent plasma (n = 1) and baricitinib (n = 1). Among patients treated with these medications and escalation of respiratory support measures, six improved and two died.
Patients taking dexamethasone received a higher additional amount of any type of glucocorticoid (438 [280–744] vs. 243 [180–360] mg of prednisone; p = 0.0095) and over a longer time period (10 [8–17] vs. 4 [3–6] days; p = 0.0003) than patients treated with colchicine/prednisone (Table 4). Among individuals completing the study in the same center (26 and 22 in the control and experimental groups, respectively), 5 (19.2%) and 6 (27.2%) patients, respectively, received intravenous glucocorticoid pulses at equivalent prednisone doses of 60 mg/day or higher. Sixteen (64%) patients treated with colchicine/prednisone received only the 3-day prednisone doses of the study or an extra single glucocorticoid dose (usually intravenous methylprednisolone or dexamethasone, administered at the Emergency Department before enrollment). Low-weight heparin was similarly administered in both groups, and antibiotics tended to be more often received by control patients (Table 4).
3.3.4 Assessment of the Disease Course
During the study period, 45 (83.3%) patients improved their baseline score of 4 on the WHO-8 Scale, with no differences between controls (n = 24, 82.8%) and experimental subjects (n = 21, 84%). Thirty-five (64.8%) patients experienced a deterioration in the WHO-8 Scale—14 (48.3%) in the control arm and 21 (32%) in the experimental arm (p = 0.27). Patients taking dexamethasone tended to remain hospitalized more frequently at the end of the study than patients taking colchicine/prednisone (7/23 [30.4%] vs. 2/20 [10%]; p = 0.14).
Regarding laboratory changes during the study, compared with control patients those receiving colchicine/prednisone significantly reduced ferritin levels and raised platelet counts at day 14 and decreased d-dimer and LDH levels at day 28. CRP and ferritin levels also tended to be lower at day 28 in patients taking colchicine/prednisone (Table 5). With regard to oxygen requirements, patients taking dexamethasone received higher fractions of inspired oxygen (FiO2) at day 14 than those taking colchicine/prednisone (Table 5). At day 28, in patients in whom a chest X-ray was repeated, 71.4% in the control group and 83.3% in the experimental group evidenced radiologic resolution of the pulmonary infiltrates (p = 0.47).
4 Discussion
Colchicine and glucocorticoids were initially considered two potential anti-inflammatory drugs for COVID-19 since they had previously demonstrated efficacy in other inflammatory and hyperinflammatory syndromes. The mechanism of action of the combination of colchicine and prednisone in patients with COVID-19 was assumed to be the sum of the different anti-inflammatory effects of both oral drugs. On the one hand, colchicine has demonstrated powerful and rapid anti-inflammatory effects (e.g. in gout and other autoinflammatory diseases), mainly by producing microtubule disruption in inflammatory cells, hindering their migration to inflammatory sites. Colchicine also contributes to attenuation of the inflammatory response by interfering with the activation of NLRP3 inflammasome and the tumor necrosis factor (TNF)-α-induced nuclear factor κΒ (NF-κΒ) pathway [20,21,22]. In addition, colchicine has evidenced antiviral effects in animal and ex vivo models of respiratory syncytial virus bronchiolitis [23]. On the other hand, glucocorticoids display anti-inflammatory, immunosuppressive, and antineoplastic properties, and their well-known anti-inflammatory effects are the consequence of the ability to inhibit the expression of multiple inflammatory genes of molecules involved in the systemic inflammatory response, mainly by direct inhibitory interaction between activated glucocorticoid receptors and activated transcription factors involved in the inflammatory gene expression, such as NF-κΒ and activator protein-1 [29]. In fact, glucocorticoids at medium-to-high doses are used as first-line therapy in inflammatory and autoimmune diseases [30], as well as hyperinflammatory processes (e.g. MAS and CAR T-cell therapies) [10]. Moreover, different types of glucocorticoids (including dexamethasone, methylprednisolone, and hydrocortisone) have clearly demonstrated to be effective in reducing mortality in COVID-19 patients needing oxygen therapy [12,13,14,15,16,17]. Besides their anti-inflammatory properties and their previous utility in other immune-inflammatory conditions, the combination of colchicine and prednisone, despite its oral delivery, was also considered a potential anti-inflammatory therapy for patients with moderate COVID-19 since the pharmacokinetics of both drugs ensure a quick effect. Colchicine and prednisone are rapidly absorbed after oral administration, reaching peak plasma concentrations after 0.5–1.5 h and 1–3 h, respectively [31, 32].
Results from the FRAGILE-COLCOVID19 clinical trial point out that compared with intravenous dexamethasone, an oral combination of colchicine and prednisone administered early in hospitalized elderly patients requiring oxygen therapy with a proven infection by SARS-CoV-2 is associated with better outcomes in terms of mortality or therapeutic failure needing additional measures to prevent death. Better survival results were achieved by almost two-thirds of patients receiving colchicine/prednisone without additional treatment other than a single glucocorticoid dose in some cases. In addition, patients taking colchicine/prednisone also needed lower amounts and shorter duration of additional glucocorticoids of any type, and showed a trend towards shorter hospitalization times than patients taking dexamethasone. Compared with dexamethasone, colchicine/prednisone also tended to decrease oxygen requirements at day 14 and assisted in controlling COVID-19-related hyperinflammatory responses by reducing several inflammatory biomarkers at day 28, including d-dimer levels.
With regard to safety, adverse effects were similarly developed in both groups. Although mild diarrhea occurred in the two arms, it was the only adverse event attributed to colchicine. Among other adverse events, serious vascular thrombotic complications remarkably only occurred in patients not receiving colchicine.
Retrospective studies of colchicine in patients with COVID-19 have already shown good results, with no major adverse effects in non-hospitalized [33] and hospitalized patients in terms of reduction in hospitalization and recovery time [34,35,36], progression to intubation [37], and mortality [34, 35, 37,38,39].
The favorable effects of colchicine on COVID-19 have also been probed in seven randomized clinical trials. (1) COLCORONA, a pivotal international clinical trial that showed that colchicine in 4159 outpatients with microbiologically-proven SARS-CoV-2 infection was associated with a 25% reduction in the composite of death or hospital admission rate compared with placebo [40]. (2) The GRECCO-19 trial compared colchicine with the standard treatment (with no glucocorticoids) in 105 hospitalized Greek patients with mild-to-moderate disease (34% without and 63% with oxygen requirements). Colchicine significantly reduced the clinical deterioration rate and time to clinical recovery compared with standard therapy [41]. (3) A Brazilian trial including 76 patients with mild (79%) and severe (21%) COVID-19 demonstrated that the addition of colchicine to standard treatment (with two-thirds of all patients receiving methylprednisolone for 5 days) significantly reduced the need for supplemental oxygen and hospitalization time [42]. (4) A Spanish study including 103 hospitalized patients with mild COVID-19 (33% without and 67% with oxygen therapy) disclosed that colchicine added to standard therapy did not improve clinical status or inflammatory markers. However, after adjustment by baseline risk factors and concomitant therapies, colchicine was significantly associated with a lower risk of clinical deterioration [43]. (5) A Syrian study of 49 patients with moderate-to-severe COVID-19 investigated the efficacy of adding colchicine to standard therapy based on dexamethasone. Patients taking colchicine had significant reductions in mortality and hospitalization time [44]. (6) The Russian COLORIT clinical trial compared colchicine with an anti-inflammatory therapy without glucocorticoids in 43 hospitalized patients requiring oxygen therapy. Colchicine was associated with improvement of disease severity and inflammatory markers, reduction of oxygen support, and a trend in reducing hospitalization time [45]. (7) An Iranian trial using colchicine analyzed the data of 153 subjects with moderate-to-severe COVID-19 completing the study (with 15% of ambulatory patients). Colchicine significantly improved respiratory symptoms and chest computed tomography (CT) compared with control therapy. Of note, 4.7% of patients taking colchicine and 37% of control patients were lost to follow-up because of clinical worsening. However, these differences favoring a possible beneficial effect of colchicine were not analyzed [46].
Four clinical trials using colchicine in COVID-19 patients disclosed negative results. (1) The PRINCIPLE trial included 156 ambulatory patients treated with colchicine 0.5 mg/day over a 14-day period, of 2755 patients receiving the usual care (1145 of whom were only receiving antipyretics). Compared with the usual therapy, the addition of colchicine did not influence recovery time, hospitalizations, or mortality [47]. Since colchicine was administered (without initial higher loading doses) at doses considered infratherapeutic for adult patients with inflammatory conditions [21], these negative results should be interpreted with caution. (2) The international RECOVERY trial (in which up to 12 different drugs could be concomitantly and indistinctly used with every study medication) analyzed the efficacy of adding colchicine to 5730 of 11,340 hospitalized patients (68.5% without or with oxygen therapy, 26.5% on non-invasive ventilation, and 5% on invasive mechanical ventilation). Colchicine was not associated with reductions in mortality, hospitalization time, or risk of progression to invasive mechanical ventilation or death at day 28 [48]. (3) The Argentinian COLCOVID trial assessed the efficacy of adding colchicine to the standard of care (91.5% of patients received glucocorticoids) in 1279 hospitalized patients with severe COVID-19 (15.3% oxygen-free, 77.8% on supplemental oxygen, and 6.9% on non-invasive/invasive mechanical ventilation). Colchicine was not associated with improvement of the composite of new mechanical ventilation requirement or death at day 28 [49]. However, analysis of the secondary combined outcome of intubation/mechanical ventilation or mortality at day 28 in non-intubated patients at baseline found that it occurred more frequently in control patients than in those receiving colchicine (27.1 vs. 22.3%; hazard ratio [HR] 0.79, 95% CI 0.63–0.99; p = 0.04) [49]. In order to investigate the subset of COVID-19 patients in whom colchicine may have had a discriminative impact in these two large clinical trials (RECOVERY and COLCOVID), it would have been interesting to perform separate additional outcome analyses according to the initial disease severity. In this sense, as detected in the secondary outcome analysis of the COLCOVID trial, colchicine seemed to protect from intubation or death in patients with overall less severe COVID-19 at baseline [49]. (4) The Mexican COLCHIVID trial analyzed the safety and efficacy of colchicine in 116 hospitalized patients with severe COVID-19 (based on the NutriCoV severity score), without observing any impact on progression to critical disease or death. However, patients treated with placebo received dexamethasone at baseline more frequently than patients in the colchicine arm (45 vs. 23.2%; p = 0.01), and perhaps the potential effect of colchicine was neutralized by the imbalanced positive effect of glucocorticoids in the placebo group [50]. The main characteristics of the randomized clinical trials using colchicine in COVID-19 patients (including colchicine loading and maintenance doses) are summarized in Table 6.
Of note, a Colombian open-label clinical trial in 633 hospitalized individuals with mild-to-severe COVID-19 (17% oxygen-free, 53% mild disease, and 30% in the ICU) used colchicine with rosuvastatin in 153 patients, or a combination of both plus emtricitabine/tenofovir in 159 patients, and compared the results with those receiving dexamethasone. The role of colchicine is difficult to interpret since no benefit was observed at day 28 with the colchicine/rosuvastatin therapy and only the combination of the four drugs was associated with a reduction of mortality and a lower need for invasive mechanical ventilation compared with dexamethasone [51].
With regard to the data from systematic reviews and meta-analyses, including randomized clinical trials and observational studies, regarding the use of colchicine in COVID-19, controversial results have been reported, mostly due to the number and type of articles included at different times during the COVID-19 pandemic. While several meta-analyses of randomized clinical trials [52,53,54,55] and observational studies [56, 57] have provided negative or inconclusive results regarding the role of colchicine in COVID-19 mortality, a higher number of these studies found positive effects of colchicine in terms of reduction of hospital admissions [52], hospitalization time [58], and mortality [53, 54, 59,60,61,62,63,64].
As observed in the present study, previous clinical trials analyzing the safety of colchicine in COVID-19 showed similar serious adverse events as placebo [40] or standard therapy [46, 49, 50]. Mild diarrhea associated with colchicine occurred in 28% of our patients, which was in accordance with the 3–45.5% reported in previous COVID-19 clinical trials [40,41,42, 46, 49].
Our study also evidenced the ability of colchicine to control several abnormal circulating COVID-19-related biomarkers. This positive effect has been similarly reported in a previous meta-analysis of six COVID-19 studies, in which CRP, LDH, and d-dimer serum levels were significantly lower at the end of treatment compared with those levels from patients not receiving colchicine [65]. The contrasted d-dimer reduction by using colchicine has been previously demonstrated in clinical trials [65, 66] and in vitro studies [66], suggesting that colchicine attenuates prothrombotic pathways by decreasing d-dimer production. In this sense, no thrombotic events occurred in patients taking colchicine in our study.
Dexamethasone was the first drug proving mortality reduction in hospitalized COVID-19 patients requiring any type of oxygen therapy [14]. Other glucocorticoids, such as methylprednisolone, also demonstrated similar results in severe COVID-19 [15], or even better results than dexamethasone in terms of recovery and hospitalization time, need of ICU admission/mechanical ventilation, and improvement in biomarkers [16, 17]. In the current study, prednisone has reported results for the first time in a randomized clinical trial in COVID-19. Such results suggest that prednisone administered early in a limited dose (for only 3 days) contributes to counterbalance the contrasted anti-inflammatory effect of dexamethasone in the control group, and therefore colchicine appears to provide the additional positive effects found in the study.
As a result, all types of glucocorticoids (including prednisone) have demonstrated to be useful in reducing mortality in COVID-19 patients requiring oxygen therapy. Furthermore, colchicine has shown to exert positive effects in the early phases in patients with mild COVID-19. Overall, based on previous clinical trials, patients with severe COVID-19 do not seem to have the benefit of being treated with colchicine.
4.1 Study Limitations
As the clinical trial had to be terminated prematurely, the statistical power of this study was considerably reduced, resulting in an increase in the established type II error. Although secondary endpoints clearly favored the experimental therapy, the lower number of patients finally enrolled in the study (initially calculated for 144 patients) could have influenced the results, which, although positive, did not obtain statistical significance for the primary endpoint. The reasons for an incomplete enrollment were as follows: (1) during the initial devastating peak of the COVID-19 outbreak, professionals involved in the management and administration of nursing homes, as well as hospital physicians, were reluctant to acknowledge the usefulness of the oral drugs proposed in the study, and initiation of the study was delayed from March to October 2020; (2) during the study period, the number of eligible patients clearly decreased because of the abundance of clinical trials with other potential drugs and the discretional use of medications not contemplated in the current studywith no scientific evidence of effectiveness in COVID-19 at that time (their use as standard therapy could have consequently biased the final results); and (3) the advent of vaccines against SARS-CoV-2 acted as a positive but ultimate limiting factor to enroll more patients. In consequence, the clinical trial was prematurely ended in May 2021.
5 Conclusions
An oral therapy consisting of a combination of prednisone (60 mg daily for 3 days) and colchicine (at a loading dose of 1–1.5 mg daily for 3 days and 0.5 mg/day until completing 14 days), although not significant, has shown a remarkable trend to increase disease survival by reducing mortality or therapeutic failure over a 28-day period in elderly patients with COVID-19 requiring oxygen therapy, compared with the approved effective therapy (intravenous dexamethasone), with no safety issues. Colchicine/prednisone therapy was also associated with lower glucocorticoid requirements, a trend for shorter hospitalization times, and a pronounced reduction of inflammatory biomarkers, including ferritin and d-dimer levels, during the study time. No thrombotic complications were observed in patients receiving colchicine.
Based on ours and previous results from clinical trials, while colchicine does not provide clear benefit in severe COVID-19 patients, its administration in mild COVID-19 ambulatory subjects or its early use combined with glucocorticoids (including prednisone) in patients with mild-to-moderate COVID-19 has shown positive results in terms of disease survival and other parameters of severity. Although our results should be supported or confirmed by ongoing and future clinical trials, the early administration of colchicine and prednisone has shown to be a simple, safe, and effective first-line anti-inflammatory therapeutic option in elderly patients and could also be a potential opportunity for patients with COVID-19 in situations of vulnerability, including those individuals from regions or countries with fewer resources and not yet completely vaccinated.
References
COVID-19 Dashboard by the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University (JHU). 2020. Available at : https://www.arcgis.com/apps/dashboards/bda7594740fd40299423467b48e9ecf6. Accessed 1 June 2022.
Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054–62.
Geriatric Medicine Research C, Covid C, Welch C. Age and frailty are independently associated with increased COVID-19 mortality and increased care needs in survivors: results of an international multi-centre study. Age Ageing. 2021;50(3):617–30.
Zeng F, Deng G, Cui Y, et al. A predictive model for the severity of COVID-19 in elderly patients. Aging (Albany NY). 2020;12(21):20982–96.
Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA. 2020;323(20):2052–9.
Chen T, Dai Z, Mo P, et al. Clinical characteristics and outcomes of older patients with coronavirus disease 2019 (COVID-19) in Wuhan, China: a single-centered, retrospective study. J Gerontol A Biol Sci Med Sci. 2020;75(9):1788–95.
Infographic: COVID-19 in care homes—European Centre for Disease Prevention and Control. 2020. Available at: https://www.ecdceuropa.eu/en/publications-data/covid-19-care-homes-infographic. Accessed 11 Apr 2022.
Data on elderly people living in nursing homes or alone in the EU. 2020. Available at: https://www.europarleuropa.eu/doceo/document/E-9-2020-006697_ENhtml. Accessed 11 Apr 2022.
González-Gay MA, Mayo J, Castañeda S, Cifrián JM, Hernández-Rodríguez J. Tocilizumab: from the rheumatology practice to the fight against COVID-19, a virus infection with multiple faces. Expert Opin Biol Ther. 2020;20(7):717–23.
Mehta P, Cron RQ, Hartwell J, Manson JJ, Tattersall RS. Silencing the cytokine storm: the use of intravenous anakinra in haemophagocytic lymphohistiocytosis or macrophage activation syndrome. Lancet Rheumatol. 2020;2(6):e358–67.
Mehta P, McAuley DF, Brown M, et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395(10229):1033–4.
Callejas Rubio JL, Luna Del Castillo JD, de la Hera FJ, Guirao Arrabal E, Colmenero Ruiz M, Ortego CN. Effectiveness of corticoid pulses in patients with cytokine storm syndrome induced by SARS-CoV-2 infection. Med Clin (Barc). 2020;155(4):159–61.
Ruiz-Irastorza G, Pijoan JI, Bereciartua E, et al. Second week methyl-prednisolone pulses improve prognosis in patients with severe coronavirus disease 2019 pneumonia: an observational comparative study using routine care data. PLoS ONE. 2020;15(9): e0239401.
Group RC, Horby P, Lim WS, et al. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021;384(8):693–704.
WHO Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group, Sterne JAC, Murthy S, et al. Association between administration of systemic corticosteroids and mortality among critically ill patients with COVID-19: a meta-analysis. JAMA. 2020;324(13):1330–41.
Ranjbar K, Moghadami M, Mirahmadizadeh A, et al. Methylprednisolone or dexamethasone, which one is superior corticosteroid in the treatment of hospitalized COVID-19 patients: a triple-blinded randomized controlled trial. BMC Infect Dis. 2021;21(1):337.
Pinzon MA, Ortiz S, Holguin H, et al. Dexamethasone vs methylprednisolone high dose for Covid-19 pneumonia. PLoS ONE. 2021;16(5): e0252057.
Wang Y, Zhu K, Dai R, et al. Specific interleukin-1 inhibitors, specific interleukin-6 inhibitors, and GM-CSF blockades for COVID-19 (at the edge of sepsis): a systematic review. Front Pharmacol. 2021;12: 804250.
Zhang X, Shang L, Fan G, et al. The efficacy and safety of janus kinase inhibitors for patients with COVID-19: a living systematic review and meta-analysis. Front Med (Lausanne). 2021;8: 800492.
Angelidis C, Kotsialou Z, Kossyvakis C, et al. Colchicine pharmacokinetics and mechanism of action. Curr Pharm Des. 2018;24(6):659–63.
Soriano A, Soriano M, Espinosa G, et al. Current therapeutic options for the main monogenic autoinflammatory diseases and PFAPA syndrome: Evidence-based approach and proposal of a practical guide. Front Immunol. 2020;11:865.
Piantoni S, Patroni A, Toniati P, et al. Why not to use colchicine in COVID-19? An oldanti-inflammatory drug for a novel auto-inflammatory disease. Rheumatology (Oxford). 2020;59(7):1769–70.
Lu N, Yang Y, Liu H, et al. Inhibition of respiratory syncytial virus replication and suppression of RSV-induced airway inflammation in neonatal rats by colchicine. 3 Biotech. 2019;9(11):392.
Salinas M, Andino P, Palma L, Valencia J, Figueroa E, Ortega J. Early use of corticosteroids in non-critical patients with COVID-19 pneumonia (PREDCOVID): a structured summary of a study protocol for a randomised controlled trial. Trials. 2021;22(1):92.
WHO R&D Blueprint novel Coronavirus COVID-19 Therapeutic Trial Synopsis: Ordinal Scale for clinical improvement. 2020. Available at: https://www.who.int/publications/i/item/covid-19-therapeutic-trial-synopsis. Accessed 10 Apr 2022.
WHO welcomes preliminary results about dexamethasone use in treating critically ill COVID-19 patients. 2020. Available at: https://www.who.int/news/item/16-06-2020-who-welcomes-preliminary-results-about-dexamethasone-use-in-treating-critically-ill-covid-19-patients. Accessed 11 Apr 2022.
EMA endorses use of dexamethasone in COVID-19 patients on oxygen or mechanical ventilation—European Medicines Agency. 2020. Available at: https://www.emaeuropa.eu/en/news/ema-endorses-use-dexamethasone-covid-19-patients-oxygen-mechanical-ventilation. Accessed 10 Apr 2022.
Steroid Conversion Calculator. 2005. Available at: https://www.mdcalc.com/calc/2040/steroid-conversion-calculator. Accessed 10 Apr 2022.
Barnes PJ. Anti-inflammatory actions of glucocorticoids: molecular mechanisms. Clin Sci (Lond). 1998;94(6):557–72.
Ronchetti S, Ayroldi E, Ricci E, Gentili M, Migliorati G, Riccardi C. A glance at the use of glucocorticoids in rare inflammatory and autoimmune diseases: Still an indispensable pharmacological tool? Front Immunol. 2020;11: 613435.
Ferron GM, Rochdi M, Jusko WJ, Scherrmann JM. Oral absorption characteristics and pharmacokinetics of colchicine in healthy volunteers after single and multiple doses. J Clin Pharmacol. 1996;36(10):874–83.
Pickup ME. Clinical pharmacokinetics of prednisone and prednisolone. Clin Pharmacokinet. 1979;4(2):111–28.
Della-Torre E, Della-Torre F, Kusanovic M, et al. Treating COVID-19 with colchicine in community healthcare setting. Clin Immunol. 2020;217: 108490.
Brunetti L, Diawara O, Tsai A, et al. Colchicine to weather the cytokine storm in hospitalized patients with COVID-19. J Clin Med. 2020;9(9):2961.
Manenti L, Maggiore U, Fiaccadori E, et al. Reduced mortality in COVID-19 patients treated with colchicine: results from a retrospective, observational study. PLoS ONE. 2021;16(3): e0248276.
Karakas O, Erden A, Guven SC, et al. Reducing length of hospital stay with colchicine. J Infect Dev Ctries. 2022;16(1):57–62.
Sandhu T, Tieng A, Chilimuri S, Franchin G. A case control study to evaluate the impact of colchicine on patients admitted to the hospital with moderate to severe COVID-19 infection. Can J Infect Dis Med Microbiol. 2020;2020:8865954.
Scarsi M, Piantoni S, Colombo E, et al. Association between treatment with colchicine and improved survival in a single-centre cohort of adult hospitalised patients with COVID-19 pneumonia and acute respiratory distress syndrome. Ann Rheum Dis. 2020;79(10):1286–9.
Pinzon MA, Cardona Arango D, Betancur JF, et al. Clinical outcome of patients with COVID-19 Pneumonia treated with corticosteroids and colchicine in Colombia. Ann Clin Microbiol Antimicrob. 2021;20(1):66.
Tardif JC, Bouabdallaoui N, L’Allier PL, et al. Colchicine for community-treated patients with COVID-19 (COLCORONA): a phase 3, randomised, double-blinded, adaptive, placebo-controlled, multicentre trial. Lancet Respir Med. 2021;9(8):924–32.
Deftereos SG, Giannopoulos G, Vrachatis DA, et al. Effect of colchicine vs standard care on cardiac and inflammatory biomarkers and clinical outcomes in patients hospitalized with coronavirus disease 2019: the GRECCO-19 randomized clinical trial. JAMA Netw Open. 2020;3(6): e2013136.
Lopes MI, Bonjorno LP, Giannini MC, et al. Beneficial effects of colchicine for moderate to severe COVID-19: a randomised, double-blinded, placebo-controlled clinical trial. RMD Open. 2021;7(1): e001455.
Pascual-Figal DA, Roura-Piloto AE, Moral-Escudero E, et al. Colchicine in recently hospitalized patients with COVID-19: a randomized controlled trial (COL-COVID). Int J Gen Med. 2021;14:5517–26.
Alsultan M, Obeid A, Alsamarrai O, et al. Efficacy of colchicine and budesonide in improvement outcomes of patients with coronavirus infection 2019 in Damascus, Syria: a randomized control trial. Interdiscip Perspect Infect Dis. 2021;2021:2129006.
Mareev VY, Orlova YA, Plisyk AG, et al. Proactive anti-inflammatory therapy with colchicine in the treatment of advanced stages of new coronavirus infection. The first results of the COLORIT study. Kardiologiia. 2021;61(2):15–27.
Pourdowlat G, Saghafi F, Mozafari A, et al. Efficacy and safety of colchicine treatment in patients with COVID-19: a prospective, multicenter, randomized clinical trial. Phytother Res. 2022;36(2):891–8.
Dorward J, Yu LM, Hayward G, et al. Colchicine for COVID-19 in adults in the community (PRINCIPLE): a randomised, controlled, adaptive platform trial. Br J Gen Pract. 2022;72(720):e446–55.
Recovery Collaborative Group. Colchicine in patients admitted to hospital with COVID-19 (RECOVERY): a randomised, controlled, open-label, platform trial. Lancet Respir Med. 2021;9(12):1419–26.
Diaz R, Orlandini A, Castellana N, et al. Effect of colchicine vs usual care alone on intubation and 28-day mortality in patients hospitalized with COVID-19: a randomized clinical trial. JAMA Netw Open. 2021;4(12): e2141328.
Absalon-Aguilar A, Rull-Gabayet M, Perez-Fragoso A, et al. Colchicine is safe though ineffective in the treatment of severe COVID-19: a randomized clinical trial (COLCHIVID). J Gen Intern Med. 2022;37(1):4–14.
Gaitan-Duarte HG, Alvarez-Moreno C, Rincon-Rodriguez CJ, et al. Effectiveness of rosuvastatin plus colchicine, emtricitabine/tenofovir and combinations thereof in hospitalized patients with COVID-19: a pragmatic, open-label randomized trial. EClinicalMedicine. 2022;43: 101242.
Mikolajewska A, Fischer AL, Piechotta V, et al. Colchicine for the treatment of COVID-19. Cochrane Database Syst Rev. 2021;10:CD015045.
Zein A, Raffaello WM. Effect of colchicine on mortality in patients with COVID-19—a systematic review and meta-analysis. Diabetes Metab Syndr. 2022;16(2): 102395.
Lien CH, Lee MD, Weng SL, et al. Repurposing colchicine in treating patients with COVID-19: a systematic review and meta-analysis. Life (Basel). 2021;11(8):864.
Mehta KG, Patel T, Chavda PD, Patel P. Efficacy and safety of colchicine in COVID-19: a meta-analysis of randomised controlled trials. RMD Open. 2021;7(3): e001746.
De-Miguel-Balsa E, Estevan-Ortega R, Sempere-Selva MT, et al. Can we still consider treatment with colchicine effective in SARS-COV-2 infection? Systematic review, meta-analysis, and trial sequential analysis. Eur Rev Med Pharmacol Sci. 2021;25(22):7151–61.
Sanghavi D, Bansal P, Kaur IP, et al. Impact of colchicine on mortality and morbidity in COVID-19: a systematic review. Ann Med. 2022;54(1):775–89.
Kow CS, Lee LH, Ramachandram DS, Hasan SS, Ming LC, Goh HP. The effect of colchicine on mortality outcome and duration of hospital stay in patients with COVID-19: A meta-analysis of randomized trials. Immun Inflamm Dis. 2022;10(2):255–64.
Nawangsih EN, Kusmala YY, Rakhmat II, et al. Colchicine and mortality in patients with coronavirus disease 2019 (COVID-19) pneumonia: a systematic review, meta-analysis, and meta-regression. Int Immunopharmacol. 2021;96: 107723.
Golpour M, Mousavi T, Alimohammadi M, et al. The effectiveness of colchicine as an anti-inflammatory drug in the treatment of coronavirus disease 2019: meta-analysis. Int J Immunopathol Pharmacol. 2021;35:20587384211031764.
Drosos AA, Pelechas E, Drossou V, Voulgari PV. Colchicine against SARS-CoV-2 infection: what is the evidence? Rheumatol Ther. 2022;9(2):379–89.
Elshafei MN, El-Bardissy A, Khalil A, et al. Colchicine use might be associated with lower mortality in COVID-19 patients: a meta-analysis. Eur J Clin Invest. 2021;51(9): e13645.
Hariyanto TI, Halim DA, Jodhinata C, Yanto TA, Kurniawan A. Colchicine treatment can improve outcomes of coronavirus disease 2019 (COVID-19): a systematic review and meta-analysis. Clin Exp Pharmacol Physiol. 2021;48(6):823–30.
Chiu L, Lo CH, Shen M, et al. Colchicine use in patients with COVID-19: a systematic review and meta-analysis. PLoS ONE. 2021;16(12): e0261358.
Sarwar M, Ali Z, Fatima M, Sarfraz Z, Sarfraz A, Cherrez-Ojeda I. Colchicine, COVID-19 and hematological parameters: a meta-analysis. J Clin Lab Anal. 2021;35(12): e24057.
Abdeen S, Abu-Fanne R, Bdeir K, et al. Divergent impacts of tocilizumab and colchicine in COVID-19-associated coagulopathy: the role of alpha-defensins. Br J Haematol. 2022;196(4):923–7.
Acknowledgements
FRAGILE-COLCOVID19 Study Group: Francisco J. Afonso, Jesús Aibar, Xavier Alemany, Luis Aparicio, Yolanda Asensio, Anna Aldea-Parés, Ana B. Azuaga, Giuseppe Barilaro, Nieves Benages, Katherine Cajiao, Júlia Calvo, Maria del Carme Cañueto, Aina Capdevila-Reniu, Irene Carbonell, Miriam Costafreda, Cecilia Cuzco, Anna de-Daniel-Bisbe, Gema Doménech, Mónica Doménech, Gerard Espinosa, Maria Feliu, Alberto Foncillas, Cristina Gabara, Gloria Gámez, Carlos García-Gutiérrez, Lucía García-Jarque, Andrea García-Ortega, Begoña Gómez, Verónica Gómez-Caverzaschi, Vanesa Gómez-Urbano, Tania Gonzalo, Ignacio Grafia, Neus Guasch, Mar Guillén, Ana M. Guió, Marta Illa, Marco Inzitari, Maria Joyera, Andrea Ladino, Irina S. Luzko-Scheid, Gema M. Lledó, Néstor López-Guerra, Daniel Marco, Guillem Masdeu, Ana Matas-García, Irene Macaya, Ferran Masanés, Miriam I. Mayor, José C. Milisenda, Montse Montero, Verónica Montes, Marc Montoya-Rodés, Núria Moragas, Alma Morancho, Pedro J. Moreno, José Naval, Josep Ortega, Anna Pahisa, Martina Pellicé, Sona Pilarcikova, Ester Pujol, Elisabet Rivero, Joan Ribot, Roberto Ríos-Garcés, Olga Rodríguez-Núñez, Omar M. Saavedra, Emilio Sacanella, Adelaido Salazar, Adriana San-Miguel, Amparo Sánchez-Buitrago, Víctor Sánchez-Palacios, Ángeles Sanz, Ferran Seguí, Adrià Tomé-Pérez, Manuel Torres-Elorza, Montse Tuset, Helena Ventosa, Roser Ventura, Esther Viñas-Esmel, Carles Zamora.
For details of the FRAGILE-COLCOVID19 Study Group members’ affiliations see the electronic supplementary material. The authors are thankful to all patients who participated in this study and their families for their contribution to increase the knowledge about COVID-19 and its therapeutic approaches in a very difficult and critical personal situation. The authors also thank Mrs. Montse Gómez Segura for her administrative support; Mr. Carlos Palmero, Mrs. Esther Ballester, and Mr. Joaquín Vila for their support of the project in difficult moments; and Mr. Guillem Masdeu for believing in this project and for his technical guidance during the entire process.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Funding
This study was sponsored by SEID S.A. and the Hospital Clínic of Barcelona Sponsorship Program, and partially by Alchemlife Iberia S.L. SEID S.A. (Lliçà de Vall, Barcelona, Spain) also provided the Colchicina SEID® tablets used in the study. None of the sponsors had any role in the study design or development, data analysis, or manuscript writing.
Conflict of interest
José Hernández-Rodríguez, Julio Durán-Sanclemente, Sergio Prieto-González, Olga Araújo, Teresa Hospital-Vidal, Georgina Casanovas, Víctor Sapena, José Luis Blanco, and Alfonso López-Soto declare no conflicts of interest in this work.
Ethics approval
This study was approved by the Clinical Research Ethics Committee at the Hospital Clínic of Barcelona (EudraCT Number 2020-002462-14).
Consent to participate
All patients or legally authorized representatives signed the informed consent form.
Consent for publication
Not applicable.
Availability of data and material
The data generated during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
Author contributions
JHR and JLB were involved in the conceptualization and design of the study protocol. ALS and JDS allowed the launch of the study by providing the geriatric facilities and encouraging the participation of their healthcare personnel. JDS, SPG, OA, THV, and all Members of the FRAGILE-COLCOVID19 Study Group recruited patients, collected data, and/or monitored patients enrolled in the study. VS and GC performed the statistical analysis and summarized the statistical data for the manuscript. JHR was responsible for funding acquisition, project administration and data analysis, and wrote the manuscript. All authors contributed to the critical revision and editing of the manuscript and approved its final version.
Additional information
The members of the FRAGILE-COLCOVID19 Study Group are listed in Acknowledgements section.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Hernández-Rodríguez, J., Durán-Sanclemente, J., Prieto-González, S. et al. FRAGILE-COLCOVID19: A Clinical Trial Based on Early Administration of an Oral Combination of Colchicine and Prednisone in Elderly Patients with COVID-19 in Geriatric Facilities. Clin Drug Investig 42, 949–964 (2022). https://doi.org/10.1007/s40261-022-01201-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-022-01201-2