Abstract
Background
Multiple switches (transitions) between biosimilars of the same reference biologic are now a reality, and they are expected to become more common in the future as more biosimilars enter the market. Switching between two biosimilars of the same reference biologic is generally driven by affordability, formulary requirements, or the relocation/travel of the patient. Evidence of whether switching between biosimilars of the same reference biologic provides similar safety and efficacy profiles is reviewed here.
Methods
A systematic search was undertaken using electronic databases (to December 2021): Biosis, Embase, MEDLINE, and EBM Reviews/Cochrane Database of Systematic Reviews via Ovid. Publications were evaluated for effectiveness and/or safety data linked to switching from one biosimilar to another.
Results
The systematic search yielded 982 citations. After eliminating duplicates, 626 citations remained for the initial title/abstract screening phase. Following the initial screening, 240 records were chosen; more thorough examination yielded 35 citations. After comprehensive screening and expert advice, 23 studies were selected, of which 13 were published in peer-reviewed journals; the remainder have been published as abstracts. Overall, 3657 patients were included in these studies. All studies were observational in nature; no randomized clinical trials were identified. The studies were heterogeneous in size, design, and endpoints. Across the studies, data are provided on safety, effectiveness, immunogenicity, pharmacokinetics, patient retention, patient and physician perceptions, and drug-use patterns. The majority of studies examined switches between biosimilar infliximabs, although switches between biosimilar adalimumabs, etanercepts, and rituximabs were also identified. Two use-pattern studies and one case report were also detected and are discussed.
Conclusion
Within the limitations of this systematic review, available data suggests that biosimilar-to-biosimilar switching is a safe and effective clinical practice, although it is not covered by current health authority regulations or guidance. No reduction in effectiveness or increase in adverse events was detected in biosimilar-to-biosimilar switching studies conducted to date.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The choice to switch a patient's therapy from one biosimilar to another is becoming increasingly feasible as a growing number of biosimilars of the same reference biologic are introduced onto the market. | |
Even though physicians are becoming more confident about transitioning patients from reference biologics to biosimilars due to a growing body of encouraging evidence on the safety and effectiveness of single transitions, there are limited data on switching patients from one biosimilar to another biosimilar of the same reference biologic. | |
Switching from a reference biologic to a biosimilar may be explored in a randomized clinical setting, but switching from one biosimilar to another is more likely to be assessed in real-world settings including observational studies and registries. | |
This systematic review outlines studies conducted to date on switching between two biosimilars of the same reference biologic, suggesting that these switches are a safe and effective clinical practice that is not associated with loss of effectiveness or an increase in adverse effects, although the practice is not covered by current health authority regulations or guidance. |
1 Introduction
A biosimilar is a biologic medicine that is highly similar to a reference biologic (reference medicine) with no clinically meaningful differences in structure, biologic activity, efficacy, safety, or immunogenicity profiles [1, 2]. Establishing biosimilarity with a reference biologic entails extensive structural and functional characterization, in vivo non-clinical assessments (if required by the regulatory agency), and clinical pharmacology studies [3]. Following the approval of the first biosimilar in 2006 (somatropin [Omnitrope®, Sandoz GmbH, Kundl, Austria]), as of the end of December 2021, the European Medicines Agency has recommended the approval of 84 biosimilars [4]. By the end of December 2021, the United States (US) Food and Drug Administration (FDA) had approved 33 biosimilars since the approval of the first biosimilar in 2015 (filgrastim [Zarxio®, Sandoz, Princeton, NJ, USA]) [5,6,7]. The first biosimilars were small therapeutic proteins, such as hormones (somatropin and insulin glargine) and growth factors (filgrastim and epoetin). In recent years, complex biosimilars (monoclonal antibodies and fusion proteins) have been licensed globally for rheumatology, dermatology, gastroenterology, and hematology (cancer and nephrology) [5].
In 2019, 27% of all novel cancer therapies approved in the European Union (EU), US, and Japan were biologics [8], and numerous high-value biological drugs are coming off-patent within the next 5 years [7, 9], highlighting a growing potential for future biosimilar development. Currently, clinical experience with biosimilars exceeds 2 billion patient-days, indicating an increase in global biosimilar adoption [7, 10]. A biosimilar is commonly less expensive than the reference biologic, and therefore may increase patient access to effective treatments [10,11,12]. By substituting biosimilars for reference biologics, the cost of high-priced medications used to treat diseases, such as cancer and rheumatoid arthritis (RA), might be reduced by $38.4 billion, or 5.9% of predicted total US biologics expenditure from 2021 to 2025 [13]. Increasing real-world clinical data boost confidence in the use of biosimilars [5, 10, 14], although there may be concerns about biosimilar use. For instance, clinicians and patients may mistakenly perceive that biosimilars have more adverse effects and less effectiveness than reference biologics due to lower costs and a different clinical development paradigm [8]. Moreover, according to a US survey, 70% of cancer patients are unaware of biosimilars [8]. The scientific literature available on the topic and educational materials provided by organizations and health authorities, including the American Society of Clinical Oncology and the European Society of Medical Oncology, are addressing these perceptions and misinformation and hopefully will have a meaningful impact [8, 15,16,17,18,19].
Transitions from a reference biologic to a biosimilar, or from one biosimilar to another, are often the consequence of insurance mandates and/or pharmacy benefit managers implementing formulary adjustments or limitations to reduce costs [20]. Patients might also transition between products if they relocate to a new region with different drug coverage (or if they are traveling). In a review published in 2017 of post-approval safety data of biosimilars and in a subsequent review published in 2021 of the safety, immunogenicity, and interchangeability of biosimilar monoclonal antibodies and fusion proteins, several individual members of European health authorities concluded that EU-licensed biosimilars should be regarded as interchangeable [21, 22]. The authors also emphasized that transitioning between a reference biologic and its biosimilar will neither induce nor enhance immunogenicity [21]. Based on evidence from 90 studies, including seven molecular entities, 14 indications, and 14,225 participants, a systematic review demonstrated that patients switching from reference biologics to biosimilars had no change in safety, efficacy, or immunogenicity [14]. In another systematic analysis conducted 2.5 years later that analyzed data from 178 trials that collectively enrolled > 21,000 participants (randomized controlled studies and real-world evidence), Barbier and colleagues concluded that existing data do not indicate any major efficacy, safety, or immunogenicity concerns regarding switching from reference biologics to biosimilars [5].
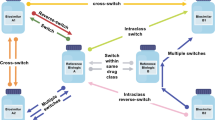
The possibility of multiple switches between biosimilars of the same reference biologic is already a reality, and these types of switches are expected to become more common in the future. Mysler and colleagues have reviewed all possible switching permutations in a multi-source environment when multiple biosimilars are available for a given reference biologic [23]. They propose using the term ‘cross-switching’ to describe switching between biosimilars to the same reference biologic. However, this is a new term that is not intuitive, and it does not have a regulatory basis. It is preferable instead to use the term ‘biosimilar-to-biosimilar switching’ as it is unambiguous and precisely describes the nature of the switch.
While switching from a reference biologic to a biosimilar may be studied in a randomized clinical trial environment, biosimilar-to-biosimilar switching is most likely to be evaluated in real-world settings, including observational studies and registries [23,24,25]. Based on the structural similarities of existing biosimilars, transitioning from one biosimilar to another of the same reference biologic would not be expected to lead to changes in clinical or immunologic responses. Nonetheless, evidence is required to support this expectation. A single literature review on the topic has been published, but it was not conducted in a systematic manner consistent with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) methodology; in addition, this prior review identified only seven studies to assess [23]. Here, an extensive systematic review was performed of published biosimilar-to-biosimilar switch studies, identifying 23 studies that met criteria for inclusion and analysis; two use-pattern studies and one case report are also discussed because it is believed that they also provide useful information.
2 Methods
2.1 Data Sources and Search Strategy
The search strategy was developed based on the research question (biosimilar-to-biosimilar switching), employing controlled vocabulary specific to each database and free text. As a starting point, hand searching was performed for biosimilar-to-biosimilar switching studies, and after reading the identified articles, the following free-text terms based on the research question were also incorporated in the search string: switching between two different biosimilars, biosimilar-to-biosimilar switch(es), cross‑switch, double biosimilar switch, double-switching, multiple switches, single- or double-switch, successive switches to different biosimilars, switching from one biosimilar to another, second switch, switching between adalimumab/etanercept/filgrastim/infliximab/insulin/pegfilgrastim/rituximab/somatropin/terapatide biosimilars.
A systematic search was undertaken using the following electronic databases (to the end of December 2021): Biosis, Embase, MEDLINE, and EBM Reviews/Cochrane Database of Systematic Reviews via Ovid. The Medical Subject Heading (MeSH) ‘biosimilar pharmaceuticals’ OR ‘biologic factors’ was used. A biologic medication was included if a biosimilar version of the reference medicine was approved in the US or the EU. In addition, MeSH keywords were included for all smaller and bigger protein biologics (erythropoietin, human growth hormone, filgrastim, etanercept, adalimumab, infliximab, and rituximab).
2.2 Study Selection
Publications were evaluated for effectiveness or safety data linked to switching from one biosimilar to another. The inclusion criteria were kept wide to collect as much accessible data on biosimilar-to-biosimilar switching as possible. Non-English publications, non-human research, editorials, comments, and brief surveys were excluded. Literature specialists conducted the literature search and screened the results retrieved via the electronic databases. Screening was based on reading the titles and abstracts of publications and included all results that mentioned biosimilar-to-biosimilar switching. Screening results were reviewed by a subject matter expert (HC) who recommended exclusion or inclusion of a publication based on the research question. Based on his knowledge of the literature, he also had the ability to recommend inclusion of additional studies. These recommendations were then checked by another subject matter expert (SH). In the event that the two experts disagreed, a third subject matter expert (WB) reviewed the conflict and made the final decision regarding whether or not to include/exclude a publication. Peer-reviewed and published case reports were excluded from consideration as a ‘study,’ but were included in the discussion because they provided useful information. Studies that were published in multiple abstracts were only considered once, so a given data set was not duplicated. Likewise, if a study was published as an abstract and subsequently published as a full peer-reviewed publication, only the peer-reviewed publication was included.
2.3 Data Extraction, Quality Assessment, and Analysis
Data were collected from the full-text publications relevant to biosimilar-to-biosimilar switch, including trial design, patient demographics, safety, effectiveness, immunogenicity, and adverse events (AEs). Data were then imported into MS Excel, which was then validated by other researchers (SH and WB). Citation of a full-text publication that included all study data was favored over previously published abstracts. A cross-study quality evaluation was not performed due to the diversity of individual studies.
The objective of these analyses was to evaluate clinical outcomes from studies relevant to biosimilar-to-biosimilar switching. All endpoints were reported in a descriptive manner; no meta-analysis was performed because of varying study designs, endpoints, and statistical methodologies used in the selected studies.
3 Results
3.1 Studies Included
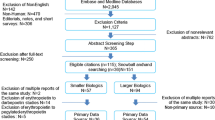
A systematic search yielded 982 citations. After eliminating duplicates, 626 citations remained for the initial title/abstract screening phase (Fig. 1). Following an initial screening, 240 records were chosen, and a more thorough examination yielded 35 citations. After assessing the selected citations, one expert (HC) (1) excluded 18 citations because, even though the prespecified terms were mentioned in the citations, they reported reference biologic-to-biosimilar transitions, and not biosimilar-to-biosimilar switches; (2) recommended exclusion of an abstract because it was subsequently published as a full article in a peer-reviewed journal; (3) recommended inclusion of six additional peer-reviewed publications based on his knowledge of the literature; and (4) recommended inclusion of three abstracts. The primary reason for inclusion of new citations that were not identified during the screening process was that the data provided within the citation described a biosimilar-to-biosimilar switching event, even though the pre-specified terms were not included within the text of the citation. Two other experts (SH and WB) reviewed and agreed with the recommendations of the first reviewer, except for the recommended deletion of one abstract, because the abstract contained some information that was not included in the subsequent peer-reviewed publication of the same study.
All citations chosen for inclusion were from published observational studies. Because of considerable inter-study variability of patient demographics, baseline characteristics, dosage regimens, and follow-up durations, it was not possible to conduct a meta-analysis. It was often difficult to perform and interpret a meaningful comparison of individual published observational biosimilar-to-biosimilar switch studies, especially when they evaluated different molecules. Because biosimilar-to-biosimilar switching is a relatively novel concept, all articles included in this study are from 2019 or later. The majority of studies investigated switches between one biosimilar infliximab (CT-P13) to another biosimilar infliximab (SB2). Other biosimilar-to-biosimilar switch scenarios discussed include adalimumab biosimilars (ABP 501 and SB5), etanercept biosimilars (SB4 and GP2015), and rituximab biosimilars (CT-P10 and GP2013). Of the 23 studies, 13 were published as full articles in peer-reviewed journals, the remainder were published as conference abstracts. A total of 3657 patients were included in these studies (Table 1). The studies were heterogeneous in size, design, and endpoints, providing data on safety, effectiveness, immunogenicity, pharmacokinetics, patient retention, patient and physician perceptions, and drug-use patterns (Fig. 2). An evidence table (see the Electronic Supplementary Material, supplemental Table S1) was developed based on the data synthesis and analysis that provides indications studied, number of patients, study design including endpoints, follow-up period, a summary of clinical outcomes, and the conclusions of the authors.
3.2 Biosimilar-to-Biosimilar Switch Studies of Infliximab Biosimilars (CT-P13 to SB2 and CT-P13 to GP1111)
Several studies evaluated patients who had switched from CT-P13 to SB2, from CT-P13 to GP1111, or from infliximab reference biologic to CT-P13 and subsequently to SB2. Infliximab biosimilar-to-biosimilar switch studies were most often conducted in the inflammatory bowel disease (IBD) indication; additional indications studied included psoriasis (PsO) and other immune-mediated inflammatory disorders.
3.2.1 Studies in Patients with Inflammatory Bowel Disease
In patients who switched from CT-P13 to SB2, Trystram et al. reported a drug persistence (SB2 discontinuation-free survival) of 94.9% after 54 weeks [26]. In total, 17 patients (10.8%) experienced a loss of response to SB2, including ten who had been managed through dose-optimization and continued treatment. In infliximab-treated patients with IBD, up to 20% of them lose their response to the drug every year after the primary response [27, 28]. No changes were observed in clinical activity scores, fatigue, biologic activity, or pharmacokinetic parameters after the single- or double-switch. Overall, the authors concluded that after 54 weeks of follow-up, switching from infliximab reference biologic to CT-P13 and subsequently to SB2 did not affect efficacy, immunogenicity, or safety of anti-tumor necrosis factor (anti-TNF) treatment. Additionally, the authors found no link between multiple switching and patient views regarding the need for (and concerns about) biosimilars [26].
In addition, studies with comparable follow-up periods (~12 months) [26, 29,30,31] found no loss in clinical responses at the middle [26, 30, 31] or conclusion of the studies [26, 29,30,31]. Mazza et al. [31] found that the loss of response rate was ~15% at the conclusion of the trial, with no statistically significant differences between patients who switched from infliximab reference biologic to CT-P13 (n = 66) and those who switched from infliximab reference biologic to CT-P13 then to SB2 (n = 52); the observed loss of response was consistent with response loss with any infliximab treatment in patients with IBD [27, 28]. The double-switch group from infliximab reference biologic to CT-P13 to SB2 had no statistically significant changes in AEs compared with the single biosimilar-to-biosimilar switch group [31]. Additionally, in 186 patients who switched from CT-P13 to SB2 (of whom 99 had previously switched from infliximab reference biologic to CT-P13 to SB2), no adverse impact was observed on infliximab trough levels and clinical and biochemical disease activity, regardless of whether switching for the first or second time [30]. De novo antidrug antibodies (ADAs) were found in < 4% of patients in either the single- or double-switch group [32].
The French prospective/retrospective non-interventional cohort PERFUSE study analyzed data of patients (n = 1374) receiving the SB2 biosimilar for a period of 12 months [33]. Findings from the study suggested that patients with IBD can be successfully transitioned from reference or biosimilar infliximab to SB2 with no loss of disease control or safety concerns, and over 92% of patients who transitioned from reference infliximab or another biosimilar infliximab continued SB2 treatment at 12 months post-initiation [33]. A study with a follow-up period of < 1 year (6 months) showed similar clinical remission rates (58.3%) and normal C-reactive protein (CRP) values (94.4% and 91.7% of patients) prior to 6 months and after 3 months of treatment in 36 patients who switched from CT-P13 to SB2 (12 of them switched from reference infliximab to CT-P13 to SB2) [34]. Furthermore, in another study with a follow-up period of < 1 year (8 months, safety events in single-switch (n = 43; CT-P13 to SB2) and double-switch (n = 24, infliximab reference biologic to CT-P13 to SB2) groups did not differ from those of the reference-to-biologic switch group [35]. Similarly, a single-center study in the Edinburgh IBD unit examined IBD patients (n = 246) who were switched from CT-P13 to GP1111 (57/246, double-switch) [36]. No significant differences were observed between the Harvey-Bradshaw Index, Mayo Score, CRP, fecal calprotectin, and trough infliximab levels comparing before and 6 months after GP1111 switch in either CT-P13 to GP1111 (single biosimilar-to-biosimilar switch) or infliximab reference to CT-P13 to GP1111 groups (double switch) [36].
A study in a large teaching hospital reported findings from patients (n = 227) with IBD who switched from CT-P13 to SB2 using the same standard of care [37]. Overall, the switch was well received, patients were satisfied with the process, and 99.2% of patients did not report any AEs. Of minor note, two reports of AEs were attributable to changes in the rate of drug administration [37].
Findings from a study that measured outcomes of transitioning a cohort (n = 133) of IBD patients from CT-P13 to SB2 in a real-world setting showed that there was no detrimental effect on patient outcomes as assessed by disease activity scores, patient-reported outcome (PRO) measures, and drug persistence for at least 4 months [38].
A multicenter real-life IBD cohort study reported findings from switching between infliximab biosimilars in two IBD centers in the UK [39]. A total of 289 patients with IBD switched from biosimilar CT-P13 to GP1111. Outcomes reported included patient acceptability, infusion reactions, and quantity of ‘best value’ (brand with the lowest acquisition cost) infliximab administered over a 6-month period. The study concluded that switching between infliximab biosimilars was acceptable to patients and was associated with a low rate of infusion reactions. The proportion of patients who discontinued treatment due to loss of response over a 6-month period was consistent with the published loss-of-response rates [39].
3.2.2 Studies in Other Indications
Gisondi et al. reported the effectiveness and safety of switching from CT-P13 to SB2 in patients (n = 96) with chronic plaque PsO who had been regularly followed in an outpatient PsO clinic in Italy [40]. Findings showed stable PsO severity, as determined by Psoriasis Area and Severity Index (PASI), during 6 months of observation. Treatment was withdrawn in ten of 96 patients (10%) because of loss of response (n = 7) or acute infusion reactions (n = 3). The authors highlighted the reduction in healthcare costs with biosimilars and the need for controlled studies to build robust evidence to support biosimilar-to-biosimilar switching [40].
In a 3-year observational study, Lauret et al. assessed whether successive switches from innovator infliximab to an infliximab biosimilar (CT-P13), followed by a switch to a second infliximab biosimilar (SB2), increased the risk of immunogenicity in patients (n = 140) with chronic inflammatory diseases [41]. The study demonstrated that immunogenicity was low (3 per 100 patient-years), and that immunogenicity levels did not increase after the switch to infliximab biosimilars in treatment of chronic inflammatory diseases [41].
Fautrel et al. presented data of 496 rheumatic patients (ankylosing spondylitis [AxSpA], RA, and psoriatic arthritis [PsA]) receiving the SB2 infliximab biosimilar from the aforementioned French non-interventional PERFUSE study [42]. This 12-month analysis indicated that patients with AxSpA, RA, or PsA can be successfully treated with SB2 as initial infliximab therapy or transitioned from the reference biologic or another infliximab biosimilar without loss of disease control and without a dose penalty. At least 75% of patients who transitioned from a prior infliximab remained on SB2 at 12 months after initiation of treatment with SB2 [42].
A study analyzed biosimilar switching patterns in US Veterans Affairs (VA) patients using a real‐time infliximab safety dashboard comprising prescription, diagnosis, and switching-pattern data [43]. Overall, 607 patients switched from one biosimilar to another. A total of 138 patients had one or more adverse drug event; 22 had one or more hospitalization. The authors concluded that biosimilar-to-biosimilar switches did not lead to increased AEs or hospitalizations [43].
An observational cohort study analyzed a nationwide switch from originator infliximab to biosimilar CT-P13, and subsequently to biosimilar GP1111, in real-world patients with inflammatory arthritis followed in the Danish DANBIO registry [44]. Patients with RA, PsA, or AxSpA were studied. In 2015, a total of 780 patients completed the first switch; patients were treated with reference biologic for a median of 7 years. At 1 year, 83% maintained CT-P13 treatment. In 2019, 52% of CT-P13-treated patients were still on treatment and performed a second, biosimilar-to-biosimilar switch to GP1111. At 1 year after the biosimilar-to-biosimilar switch, 91% maintained GP1111 treatment. For both rounds of switching, withdrawals were associated with worse baseline PROs, worse health assessment questionnaire scores, and less frequent patient acceptable symptom state (PASS), whereas objective markers (CRP, physician global assessment) were similar [44].
3.3 Biosimilar-to-Biosimilar Switch Studies of Adalimumab Biosimilars (ABP 501 and SB5)
Ribaldone and colleagues enrolled 61 patients with Crohn's disease who had switched from the adalimumab biosimilar ABP 501 to adalimumab biosimilar SB5, of whom 43 (70.5%) were initially treated with the reference biologic (a multiple switch from adalimumab reference to ABP 501 then SB5) [45]. After 6 months, 88.5% (54/61) of patients remained on SB5. The switch was successful in 82.0% (50/61) of patients (no systemic corticosteroids for 6 months, no cessation of SB5, no dosage increase). Seven patients (11.5%) had AEs compared with one (1.6%) in the 6 months before the switch (P = 0.03). The increase in AEs was notable. Nonetheless, the authors concluded that switching between adalimumab biosimilars was not associated with new safety or effectiveness concerns beyond those previously documented for adalimumab [45].
An observational cohort study in a tertiary IBD referral center examined patients (n = 35) with IBD who underwent a double-switch from the adalimumab reference biologic to adalimumab SB5 and subsequently to adalimumab biosimilar ABP 501. Patients were treated for a median of 30 months [interquartile range (IQR) 18–50] with the reference biologic. After a median of 28 weeks [IQR 15–43], patients were either switched from SB5 to ABP 501 (n = 31) or back to the reference biologic (n = 4). During ABP 501 treatment, no patients developed new detectable antibodies. None of the 35 patients who switched from SB5 to ABP 501 discontinued ADAs; trough levels remained stable over time [46].
The PERCEPTION study investigated outcomes of mono- and multi-switching scenarios in real-world settings in terms of patient attitudes towards switching from one adalimumab biosimilar to another [47]. Patients (n = 90) with chronic inflammatory rheumatic diseases, including RA, AxSpA, and PsA, were categorized into either mono-switch (reference biologic to biosimilar; n = 42) or multi-switch (reference biologic to first biosimilar and a second; n = 48) groups. The study showed that multi-switching did not result in decreased patient satisfaction in patients receiving biosimilar therapy. The number of switches, in particular, had no effect on patient satisfaction [47].
3.4 Biosimilar-to-Biosimilar Switch Studies of Etanercept Biosimilars (SB4 and GP2015)
Two studies were included in the current systematic review that investigated switching between etanercept biosimilars with a follow-up of 12 months [48] and 21.1 months, respectively [49]. In the study with the 12-month follow-up [48], patients (n = 76) with PsO switched from reference etanercept to etanercept biosimilar SB4 and subsequently to biosimilar etanercept GP2015. In terms of efficacy, median PASI was 1 (0–2) after 3 months and 0.5 (0–1) after 12 months of therapy. Two patients experienced psoriatic flares during the study. No treatment-emergent serious AEs were reported. Efficacy and safety were maintained after switching between etanercept biosimilars [48]. Kiltz et al. studied 100 patients (54 RA, 27 AxSpA, 19 PsA) who had switched etanercept biosimilar SB4 to etanercept biosimilar GP2015 with a mean follow-up of 21.1 ± 7.4 months [49]. The authors concluded that after multiple switches from the original etanercept to SB4 and then to GP2015, the retention rate was close to 90% 6 months after the final switch [49]. No changes in disease activity or function were found in any indication.
3.5 Biosimilar-to-Biosimilar Switch Study of Rituximab Biosimilars (CT-P10 and GP2013)
Urru and colleagues assessed the safety of switching between rituximab biosimilars (CT-P10 and GP2013) in a prospective observational study in patients with non-Hodgkin’s lymphoma (NHL) and chronic lymphocytic leukemia (CLL) [50]. Overall, 83 (NHL, n = 72; CLL, n = 11) patients were included in the study; 60% experienced one or more switch (reference biologic to biosimilar or biosimilar to biosimilar); 32% were switched from one biosimilar to another; the median follow-up was 10.5 months. Disease activity was assessed after the third cycle and at the end-of-treatment period; 93.1% (67 of 72) and 36.4% (4 of 11) of patients with NHL and CLL, respectively, achieved complete or partial responses. AEs were reported in 71 patients (85.5%) and were similar in patients who had received one or two biosimilar formulations (32/33 in the no-switch group vs 25/26 with one of the switches during the study period, P = 0.86). Findings from this publication support the position that switching between rituximab biosimilars is safe and does not lead to loss of efficacy. No safety signals emerged in association with the use of a specific biosimilar or with the practice of switching [50].
4 Discussion
Because of the availability of encouraging evidence on the safety and effectiveness of transitioning patients from reference biologics to biosimilars [5, 14, 26, 51], clinicians are becoming more confident about this scenario. There is a paucity of data, however, on switching between biosimilars of the same reference biologic. The primary sources of information for understanding biosimilar-to-biosimilar switches are observational studies and registry data. Randomized controlled trials (RCTs) are not practical; devices would have to be comparable to avoid a nocebo effect, and such research would be costly [52, 53]. Fortunately, data from a wide variety of settings on biosimilar-to-biosimilar switching are beginning to emerge and are the focus of this systematic review. The current examination of literature on this topic revealed 23 observational studies that included 3657 patients; two use-pattern studies and one case report have also been reported on the topic. The present systematic analysis indicates there were no clinically meaningful differences in safety or effectiveness up to 12 months between two biosimilars of the same reference biologic for infliximab, adalimumab, etanercept, or rituximab in studies conducted to date. These findings may help clinicians feel more confident about switching patients between two biosimilars of the same reference biologic.
Existing regulatory frameworks for development and approval of biosimilars all focus on comparisons of the proposed biosimilar and their corresponding reference biologic. To the best of our knowledge, there are no regulations or guidance anywhere in the world that discuss the option of biosimilar-to-biosimilar switching. Indeed, it would not be practical to mandate such studies, and if such studies were conducted the sheer number of studies would strain regulatory resources. Further considering the value of randomized clinical trials in the development of biosimilars, evidence has accumulated that phase 3-style efficacy and safety studies rarely contribute pivotal data to the totality of data supporting initial approval [54,55,56]. Likewise, analysis of the data from the studies identified in this systematic review clearly reveals that meaningful data on biosimilar-to-biosimilar switching can be obtained without the need for RCTs.
4.1 Immunogenicity
At present, immunogenicity is an important consideration when switching from a reference biologic to a biosimilar or between two biosimilars of the same reference biologic [41]. It is well established that the presence of ADAs to biologic therapies may result in a reduction in trough-serum drug levels, a decrease in therapeutic response, and at times an increase in AEs. Because infliximab is one of the most immunogenic biologic treatments, preventing immunogenicity and managing its side effects is a major concern for physicians. However, consecutive use of two biosimilars of infliximab did not result in an increased risk of immunogenicity in comparison with that observed in trials that focused on the use of a single biosimilar alone [41].
Studies of the antigenic response to CT-P13 and infliximab originator in IBD patients showed similar epitope recognition [57]. Another study of epitope recognition revealed that the adalimumab biosimilar SB5 showed cross-immunogenicity to the adalimumab reference biologic but not to infliximab in IBD and RA patients [58].
In addition, cross-immunogenicity among infliximab biosimilars was observed in two studies, both of which examined immunogenicity and cross-reactivity across biosimilars [59, 60]. In the first study, serum samples taken from IBD patients receiving infliximab reference biologic or biosimilar CT-P13 were examined for ADAs to the reference biologic, CT-P13, or SB2 [23, 60]. The anti–reference-infliximab and anti–CT-P13 sera were tested for cross-reactivity using an enzyme-linked immunosorbent assay customized for reference infliximab [23, 60]. Anti–reference-infliximab and anti–CT-P13 sera cross-reacted with reference infliximab, as well as the biosimilars CT-P13 or SB2, demonstrating immunogenic similarities [23, 60]. In the event of an immune response to reference infliximab or an infliximab biosimilar, the study underlined the ineffectiveness of biosimilar cross-switching [23]. In vitro evidence of complete cross-reactivity also supports the safety of cross-switching [23]. The second study in patients with IBD showed that antibodies to biosimilar infliximab reacted identically to the reference infliximab and the two approved biosimilar molecules; these findings support safe switching between CT-P13 and SB2 biosimilars, and that switching between biosimilars and reference biologics will not result in differences in the production of antibodies to infliximab [59].
4.2 Case Report
Pagnini and colleagues described an IBD patient who developed PsO after an infliximab biosimilar-to-infliximab biosimilar switch, which then resolved after reverting to the first biosimilar [61]. A close examination of this case raises questions about the attribution of PsO exacerbation to the second infliximab biosimilar, because the induction or exacerbation of PsO during anti-TNFα therapy in IBD patients is well known and is already described as a common AE in product labeling. A systematic literature review of such cases based on 222 cases revealed that the mean latency time between initiation of anti-TNFα therapy and onset of the psoriatic lesions was 13.8 months [62], which is close to that observed by Pagnini et al. in their case report [61].
4.3 Use-Pattern Studies
Many healthcare systems, at both country and commercial organization levels (e.g., a healthcare maintenance organization), utilize tender processes for selection of biologic drugs, whereby the reference biologic and biosimilar companies submit competitive bids for a given contract that is in place for a set period of time. Healthcare systems have used tenders to select infliximabs, adalimumabs, etanercepts, filgrastims, and other biologics. When multiple biosimilars are available for a given reference biologic, it is possible that patients may be switched from one biosimilar to another based on the sequence of biologic drugs selected. Use-pattern studies can document these occurrences. We found two examples of use-pattern studies in the literature, but it is likely that this has occurred elsewhere as well. Use-pattern studies were not included in our results because they do not provide effectiveness or safety data, although these two publications provide the first published documentation that wide-scale biosimilar-to-biosimilar switching is already occurring in some real-world settings.
Kozlowski et al. described the uptake and use patterns of biosimilar infliximab in the US Medicare system from July 1, 2016 to December 31, 2018 [63]. While most patients during this time interval received reference infliximab, the authors found that 201 patients who received infliximab-abda during this interval had previously received infliximab-dyyb. This study did not include safety or effectiveness data [63]. Johnson et al. reported infliximab use patterns in the US VA healthcare system from January 1, 2016 to December 31, 2019 [64]. The VA is among the largest fully integrated healthcare system in the US and selects drugs for inclusion in a national VA formulary [64]. One biosimilar, infliximab-dyyb, was preferred by the VA national formulary from May 2017 to September 2018, after which the VA national formulary switched to preferring a second biosimilar infliximab (infliximab-abda) [64]. During the time window of May 2017 to August 2018, 30–50% of patients received infliximab-dyyb [64]. At the next time interval, from September 2018 to December 2019, 60% of patients already treated with either reference infliximab or infliximab-dyyb received infliximab-abda. While exact numbers are not provided in this publication, in a separate analysis of the same database, Cunningham et al. identified 607 patients who were switched from infliximab-dyyb to infliximab-abda [43].
These studies illustrate that biosimilar-to-biosimilar switches are already happening on a large scale. However, further conclusions regarding safety or effectiveness cannot be drawn from these use-pattern data or case reports without further epidemiological and statistically validated investigation.
4.4 Education
Patient education continues to be critical in helping patients build a strong relationship with their physician [25]. Improved medication adherence in patients with IBD has been shown to benefit from shared decision-making, demonstrating the importance of providing quality information to patients [25]. Many, if not most, patients would consider the use of a biosimilar if they were educated in a systematic way, and if biosimilars had the same clinical profile as reference medicines [25]. Indeed, patient education is also key to help minimize a potential nocebo effect. The nocebo effect is described as the incitation or exacerbation of symptoms generated by any unfavorable attitude resulting from non-pharmacologic therapeutic intervention, sham treatment, or active therapy [53]. When a patient expects a negative impact connected with an intervention, medicine, or medication change, they may either suffer an increase in the severity of the effect or experience the effect for the first time, depending on the situation [53]. Given the crucial role that biosimilars play in delivering cost-effective alternatives to reference biologics, expanding physician treatment choices, and providing patients with access to effective biologic therapy, it is critical that a nocebo effect is recognized and measures are taken to decrease it wherever feasible [53].
In addition, nurses play an important role in the community as the healthcare professionals (HCPs) primarily delivering therapies to patients, monitoring them frequently while they are undergoing treatment, and spending the most time with patients [65]. Nurses regularly rank as the most trusted group of HCPs. As a result, they are often asked questions regarding medicines by patients and therefore play a unique role in offering essential information to patients. Nurses also have the potential to influence biosimilar adoption, especially in terms of patient education, as a result of their frequent contact with patients. It is therefore essential for nurses to understand biosimilar medicines and their regulatory status in order to convey their effectiveness, safety, and possible advantages to patients correctly [65].
Lastly, when considering education, clinical pharmacists have a broad understanding of the clinical, logistical, and supply chain aspects of biosimilars; thus, they can also be valuable assets in educating stakeholders, such as prescribers, formulary committee members, other pharmacists, nurses, administrative personnel, and patients, around biosimilars in an effort to improve access and rational usage [66].
4.5 Limitations
The current systematic review has several limitations. Conclusions about biosimilar-to-biosimilar switches for those biosimilars for which there are currently only one or two such studies should be made with caution. Another limitation is that all studies were observational in nature, with varying outcome assessments, a lack of appropriate controls, and an absence of long-term effects of biosimilar-to-biosimilar switching. Additionally, the inclusion of conference abstracts may be seen as a constraint. Abstracts often do not provide sufficient detail about methodology and findings to allow assessment for scientific rigor. Also, conference abstracts may contain preliminary/interim data, and they do not always undergo peer review. Nonetheless, given that biosimilar-to-biosimilar switching is a relatively new phenomenon with a significant amount of meaningful data available in abstract form only, abstracts were included in this review in an attempt to gather all available data while being open about the source. Since observational studies are by nature subjective, there is risk of individual researcher bias. Finally, use-pattern studies do not provide safety or efficacy data related to biosimilar-to-biosimilar switching, but only document that this practice is occurring in a real-world setting.
It should be noted that since biosimilar-to-biosimilar switching is already occurring in some settings, additional data on this topic will likely become available in the future. Nonetheless, given that it is a current topic of interest to HCPs, their patients, and healthcare systems, it is useful to collate and evaluate existing data even while acknowledging that additional data will likely become available in the future.
5 Conclusions
Economic considerations, formulary decisions, and relocation or travel can motivate switching between two biosimilars of the same reference biologic. Lower medication costs can translate into increased access to high-cost therapies for a greater percentage of patients. Thus, proof that biosimilars from the same reference biologic are equivalent in terms of effectiveness and safety is required to support switching between biosimilars of the same reference biologic.
Data from the literature provided in this systematic review suggest that switching between two biosimilars of the same reference biologic is a safe and effective clinical practice, although it is not covered by current health authority regulations or guidance. No reduction in effectiveness or increase in AEs was detected in biosimilar-to-biosimilar switching studies conducted to date, although there are limitations in the individual studies included in the current analysis. As is the case with all biologic drugs, ongoing pharmacovigilance is essential to detect uncommon AEs and unanticipated changes in effectiveness or safety profiles that may arise as a result of modifications in the manufacturing process. Comprehensive pharmacovigilance of all biosimilars should improve patient, HCP, and public trust in biosimilars, resulting in increased adoption of these medications with the economic and access advantages that they can bring.
References
Biosimilar medicines: overview https://www.ema.europa.eu/en/human-regulatory/overview/biosimilar-medicines-overview
Biosimilar and interchangeable products https://www.fda.gov/drugs/biosimilars/biosimilar-and-interchangeable-products#biosimilar.
Kirchhoff CF, Wang XM, Conlon HD, et al. Biosimilars: key regulatory considerations and similarity assessment tools. Biotechnol Bioeng. 2017;114:2696–705.
Generics and biosimilar initiative: biosimilars approved in Europe https://gabionline.net/biosimilars/general/biosimilars-approved-in-europe 28 January 2022.
Barbier L, Ebbers HC, Declerck P, et al. The efficacy, safety, and immunogenicity of switching between reference biopharmaceuticals and biosimilars: a systematic review. Clin Pharmacol Ther. 2020;108:734–55.
Biosimilar product information: https://www.fda.gov/drugs/biosimilars/biosimilar-product-information. 27 Dec 2021.
CardinalHealth. 2022 Biosimilars Report: The US Journey and Path Ahead. 2022 Biosimilars Report (cardinalhealth.com) Accessed May 23, 2022.
Bennett CL, Schoen MW, Hoque S, et al. Improving oncology biosimilar launches in the EU, the USA, and Japan: an updated policy review from the Southern Network on Adverse Reactions. Lancet Oncol. 2020;21:e575–88.
IQVIA. The Impact of Biosimilar Competition in Europe. December 2021. The Impact of Biosimilar Competition in Europe 2021 - IQVIA Accessed May 23, 2022
Kim H, Alten R, Avedano L, Dignass A, et al. The future of biosimilars: maximizing benefits across immune-mediated inflammatory diseases. Drugs. 2020;80:99–113.
Huang HY, Liu CC, Yu Y, et al. Pharmacoeconomic evaluation of cancer biosimilars worldwide: a systematic review. Front Pharmacol. 2020;11: 572569.
Smolen JS, Caporali R, Doerner T, et al. 2021. Treatment journey in rheumatoid arthritis with biosimilars: from better access to good disease control through cost savings and prevention of nocebo effects. RMD Open 7.
Mulcahy Andrew BC, Finegold Kenneth, El-Kilani Zeid, et al. 2022. Projected US savings from biosimilars, 2021-2025. AJMC 28(7): In Press. Available on-line at: www.ajmc.com/view/projected-us-savings-from-biosimilars-2021-2025 (Accessed May 24, 2022)
Cohen HP, Blauvelt A, Rifkin RM, et al. Switching reference medicines to biosimilars: a systematic literature review of clinical outcomes. Drugs. 2018;78:463–78.
Cohen HP, McCabe D. The importance of countering biosimilar disparagement and misinformation. BioDrugs. 2020;34:407–14.
Biosimilars in the EU - Information guide for healthcare professionals https://www.ema.europa.eu/en/documents/leaflet/biosimilars-eu-information-guide-healthcare-professionals_en.pdf Prepared jointly by the European Medicines Agency and the European Commission.
Biosimilars | FDA https://www.fda.gov/drugs/therapeutic-biologics-applications-bla/biosimilars
Danese S, Fiorino G, Raine T, et al. ECCO position statement on the use of biosimilars for inflammatory bowel disease-an update. J Crohns Colitis. 2017;11:26–34.
ESMO education material to help stakeholders to better understand biosimilars: https://www.esmo.org/policy/biosimilarsportal/education
Coleman C, Salam T, Duhig A, et al. Impact of non-medical switching of prescription medications on health outcomes: an e-survey of high-volume medicare and medicaid physician providers. J Mark Access Health Policy 2020;8:1829883.
Kurki P, van Aerts L, Wolff-Holz E, et al. Interchangeability of biosimilars: a European perspective. BioDrugs. 2017;31:83–91.
Kurki P, Barry S, Bourges I, et al. Safety, immunogenicity and interchangeability of biosimilar monoclonal antibodies and fusion proteins: a regulatory perspective. Drugs. 2021;81:1881–96.
Mysler E, Azevedo VF, Danese S, et al. Biosimilar-to-biosimilar switching: what is the rationale and current experience? Drugs. 2021;81:1859–79.
Kay J, Schoels MM, Dörner T, et al. Consensus-based recommendations for the use of biosimilars to treat rheumatological diseases. Ann Rheum Dis. 2018;77:165–74.
Jorgensen KK, Olsen IC, Goll GL, et al. Switching from originator infliximab to biosimilar CT-P13 compared with maintained treatment with originator infliximab (NOR-SWITCH): a 52-week, randomised, double-blind, non-inferiority trial. Lancet. 2017;89:2304–16.
Trystram N, Abitbol V, Tannoury J, Lecomte M, Assaraf J, Malamut G, et al. Outcomes after double switching from originator Infliximab to biosimilar CT-P13 and biosimilar SB2 in patients with inflammatory bowel disease: a 12-month prospective cohort study. Aliment Pharmacol Ther. 2021;53:887–99.
Ben-Horin S, Chowers Y. Review article: loss of response to anti-TNF treatments in Crohn’s disease. Aliment Pharmacol Ther. 2011;33:987–95.
Oussalah A, Chevaux JB, Fay R, et al. Predictors of infliximab failure after azathioprine withdrawal in Crohn’s disease treated with combination therapy. Am J Gastroenterol. 2010;105:1142–9.
Khan N, Patel D, Pernes T, Patel M, Trivedi C, Medvedeva E, et al. The efficacy and safety of switching from originator infliximab to single or double switch biosimilar among a nationwide cohort of inflammatory bowel disease patients. Crohn's & Colitis 360. 2021;3:otab022.
Luber RP, O’Neill R, Singh S, Sharma E, Cunningham G, Honap S, et al. An observational study of switching infliximab biosimilar: No adverse impact on inflammatory bowel disease control or drug levels with first or second switch. Aliment Pharmacol Ther. 2021;54:678–88.
Mazza S, Piazza O Sed N, Conforti FS, Fascì A, Rimondi A, Marinoni B, et al. Safety and clinical efficacy of the double switch from originator infliximab to biosimilars CT-P13 and SB2 in patients with inflammatory bowel diseases (SCESICS): A multicenter cohort study. Clin Transl Sci. 2022;15:172–81.
Hanzel J, Jansen JM, Ter Steege RWF, Gecse KB, D’Haens GR. Multiple switches from the originator infliximab to biosimilars is effective and safe in inflammatory bowel disease: a prospective multicenter cohort study. Inflamm Bowel Dis. 2022;28:495–501.
Bouhnik Y, Fautrel B, Desjeux G, Freudensprung U, Brigui A, Addison J, et al. PERFUSE: A French non-interventional cohort study of IFX-naïve and transitioned patients receiving IFX biosimilar SB2: An interim analysis. ECCO 2020 Annual Meeting. Vienna, Austria.
Lovero R, Losurdo G, La Fortezza RF, Terracciano F, Biscaglia G, Martino G, et al. Safety and efficacy of switching from infliximab biosimilar CT-P13 to infliximab biosimilar SB2 in patients with inflammatory bowel disease. Eur J Gastroenterol Hepatol. 2021;32:201–7.
Macaluso FS, Fries W, Viola A, Centritto A, Cappello M, Giuffrida E, et al. The SPOSIB SB2 Sicilian cohort: safety and effectiveness of infliximab biosimilar SB2 in inflammatory bowel diseases, including multiple switches. Inflamm Bowel Dis. 2021;27:182–9.
Siakavellas S, Barrett R, Plevris N, Derikx L, Gauci J, Lucaciu L, et al. 610 Both single and multiple switching between infliximab biosimilars can be safe and effective in inflammatory bowel disease (IBD): real world outcomes from the Edinburgh IBD unit. Gastroenterology. 2021;160:S-120.
O’neill R, Singh S, Luber R. 4CPS-108 A review of infliximab biosimilar to biosimilar switch: Remsima to Flixabi. Eur J Hosp Pharm. 2020;27:A98-A.
Harris C, Harris R, Young D, McDonnell M, Harvey J, Felwick R, et al. IBD biosimilar to biosimilar infliximab switching study: preliminary results. United European Gastroenterol J. 2019;7(8).
Mott A, Mott A, Taherzadeh N, Shah T, Whitley L, Murray C, et al. PMO-29 Switching between infliximab biosimilars: Experience from two inflammatory bowel disease centres. Gut. 2021;70:A92.
Gisondi P, Virga C, Piaserico S, Meneguzzo A, Odorici G, Conti A, et al. Switching from one infliximab biosimilar (CT-P13) to another infliximab biosimilar (SB2) in patients with chronic plaque psoriasis. Br J Dermatol. 2020;183:397–8.
Lauret A, Moltó A, Abitbol V, Gutermann L, Conort O, Chast F, et al. Effects of successive switches to different biosimilars infliximab on immunogenicity in chronic inflammatory diseases in daily clinical practice. Semin Arthritis Rheum. 2020;50:1449–56.
Fautrel B, Bouhnik Y, Dougados M, et al. POS0614 PERFUSE: A French Prospective/Retrospective Noninterventional Cohort Study Of Infliximab-Naïve And Transitioned Patients Receiving Infliximab Biosimilar Sb2; 12-Month Analysis. Ann Rheum Dis. 2021;80:544.
Cunningham F, Shurmunov A, Dong D, Salone C, Zacher J, Glassman P. Biosimilar safety dashboard to assess switching in veterans. Abstracts Pharmacoepidemiol Drug Saf. 2019;28:5–586.
Nabi H, Glintborg B, Loft AG, Hendricks O, Pedersen JK, Just SA,,et al. OP16. Multiple infliximab biosimilar switches: results following a nationwide switch from originatorinfliximab to biosimilar CT-P13, and subsequently to biosimilar GP1111 in real-world patients with inflammatory arthritis followed in the Danish DANBIO registry. Abstracts of the 38th Scandinavian Congress Of Rheumatology; Scandinavian Journal of Rheumatology. 2021;50:14.
Ribaldone DG, Tribocco E, Rosso C, Armandi A, Vernero M, Bugianesi E, et al. Switching from biosimilar to biosimilar adalimumab, including multiple switching, in Crohn’s disease: A prospective study. J Clin Med. 2021;10:3387.
Derikx LAAP, Dolby HW, Plevris N, Lucaciu L, Rees CS, Lyons M, et al. Effectiveness and safety of adalimumab biosimilar SB5 in IBD: Outcomes in originator to SB5 switch, double biosimilar switch and bio-naïve SB5 observational cohorts. J Crohns Colitis. 2021;15:2011–21.
Gall S, Kiltz U, Kobylinski T, Andreica I, Vaupel K, Baraliakos X, et al. POS0301 No major differences between patients with chronic inflammatory rheumatic disease who underwent mono- or multiswitching of biosimilars in routine care (PERCEPTION STUDY). Ann Rheum Dis. 2021;80:376.
Piaserico S, Conti A, Messina F, Meneguzzo A, Odorici G, Bellinato F, et al. Cross-switch from etanercept originator to biosimilar SB4 and to GP2015 in patients with chronic plaque psoriasis. BioDrugs. 2021;35:469–71.
Kiltz U, Tsiami S, Baraliakos X, Braun J. AB1171 Effects of successive switches of two different biosimilars of etanercept on outcomes in inflammatory rheumatic diseases in daily practice. Ann Rheum Dis. 2020;79:1876.
Urru SAM, Spila Alegiani S, Guella A, et al. Safety of switching between rituximab biosimilars in onco-hematology. Sci Rep. 2021;11:5956.
Bente G, Anne GL, Emina O, et al. To switch or not to switch: results of a nationwide guideline of mandatory switching from originator to biosimilar etanercept. One-year treatment outcomes in 2061 patients with inflammatory arthritis from the DANBIO registry. Ann Rheum Dis. 2019;78:192–200.
Kvien TK, Patel K, Strand V. 2021. The cost savings of biosimilars can help increase patient access and lift the financial burden of health care systems. Semin Arthritis Rheum: 151939.
Kristensen LE, Alten R, Puig L, et al. Non-pharmacological effects in switching medication: the nocebo effect in switching from originator to biosimilar agent. BioDrugs. 2018;32:397–404.
Schiestl M, Ranganna G, Watson K, Jung B, Roth K, Capsius B, et al. The Path Toward a Tailored Clinical Biosimilar Development. BioDrugs. 2022;34:297–306.
Wolff-Holz E, Tiitso, K, Vleminckx, Weise M. Evolution of the EU Biosimilar Framework: Past and Future. Biodrugs. 2019;33:621-34.
Kurki P, Kang HN, Ekman N, Knezevic I, Weise M Wolff-Holz E. Regulatory Evaluation of Biosimilars: Refinement of Principles Based on Scientific Evidence and Clinical Experience. BioDrugs. 2022;36:359-71.
Goncalves J, Santos M, Acurcio R, Iria I, Gouveia L, Matos Brito P, et al. Antigenic response to CT-P13 and infliximab originator in inflammatory bowel disease patients shows similar epitope recognition. Aliment Pharmacol Ther. 2018;48:507–22.
Goncalves J, Myung G, Park M, Jeong D, Ghil J, et al. SB5 shows cross-immunogenicity to adalimumab but not infliximab: results in patients with inflammatory bowel disease or rheumatoid arthritis. Therap Adv Gastroenterol. 2019;12:1756284819891081.
Fiorino G, Ruiz-Argüello MB, Maguregui A, et al. Full interchangeability in regard to immunogenicity between the infliximab reference biologic and biosimilars CT-P13 and SB2 in Inflammatory Bowel Disease. Inflamm Bowel Dis. 2018;24:601–6.
Magro F, Rocha C, Vieira AI, Sousa HT, et al. 2018. The performance of Remicade®-optimized quantification assays in the assessment of Flixabi® levels. Therap Adv Gastroenterol 11: 1756284818796956.
Pagnini C, Di Paolo MC, De Angelis G, Torcolacci F, et al. Similar but not identical: plaque psoriasis exacerbation in a patient with Crohn’s disease after switching from CT-P13 to SB2 infliximab biosimilar. Inflamm Bowel Dis. 2020;26:e83–4.
Denadai R, Teixeira FV, Steinwurz F, et al. Induction or exacerbation of psoriatic lesions during anti-TNF-α therapy for inflammatory bowel disease: a systematic literature review based on 222 cases. J Crohns Colitis. 2013;7:517–24.
Kozlowski S, Flowers N, Birger N, et al. Uptake and usage patterns of biosimilar infliximab in the medicare population. J Gen Intern Med. 2021;36:2170–3.
Johnson J, Gawron A, Curtis J, et al. S0765 Infliximab biosimilar use for inflammatory bowel disease: a national Veterans Affairs experience. Am J Gastroenterol 2020;115: S389-S.
Waller CF, Friganović A. Biosimilars in oncology: key role of nurses in patient education. Future Oncol. 2020;16:1931–9.
Okoro RN. Biosimilar medicines uptake: The role of the clinical pharmacist. Explor Res Clin Soc Pharm. 2021;1: 100008.
Acknowledgments
Avinash Thakur (Novartis Healthcare, Hyderabad) performed the literature search and provided medical writing support. Divya Chandrasekhar and Nihal Maremanda (both Novartis Healthcare, Hyderabad, India) helped during the manuscript revisions. The writing support was funded by Novartis Pharma AG, Basel, Switzerland in accordance with the Good Publication Practice (GPP3) guidelines (http://www.ismpp.org/gpp3).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received by the authors for writing this paper. Medical writing support was funded by Novartis Pharma AG, Basel, Switzerland.
Conflict of interest
AB has served as a speaker for AbbVie, Leo, and UCB as well as a scientific adviser and/or clinical study investigator for AbbVie, Abcentra, Aligos, Almirall, Amgen, Arcutis, Arena, Aslan, Athenex, Boehringer Ingelheim, Bristol-Myers Squibb, Dermavant, EcoR1, Eli Lilly and Company, Evommune, Forte, Galderma, Incyte, Janssen, Landos, Leo, Novartis, Pfizer, Rapt, Regeneron, Sanofi Genzyme, Sun Pharma, UCB Pharma, Vibliome, and Xencor. SD has served as a speaker, consultant and an advisory board member for AbbVie, Allergan, Biogen, Boehringer Ingelheim, Celgene, Celltrion, Ferring, Hospira, Johnson & Johnson, Merck-Serono, Merck Sharp and Dohme, Takeda, Mundipharma, Pfizer, Sandoz, Tigenix, UCB Pharma, and Vifor. TKK has received fees for speaking from Amgen, Celltrion, Egis, Evapharma, Ewopharma, Hikma, Oktal, Sandoz, and Sanofi. He received consulting fees from AbbVie, Amgen, Biogen, Celltrion, Eli Lilly, Gilead, Janssen, Mylan, Novartis, Pfizer, Sandoz, and Sanofi. Research funding for Diakonhjemmet Hospital was provided from AbbVie, Amgen, BMS, MSD, Novartis, Pfizer, and UCB. HC is an employee of Sandoz Inc., a division of Novartis. He may own stock in Novartis. SH and WB are employees of Sandoz Biopharmaceuticals GmBH, a division of Novartis. They may own stock in Novartis. Sandoz manufactures and markets multiple biosimilars worldwide, including several discussed in this publication.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Availability of data and material
All publications included in this systematic review are available in the public domain.
Code availability
Not applicable.
Author contributions
HC, SH, and WB developed the concept for this publication and participated in the adjudication process for selection of studies included in this review. HC developed the initial outline and wrote portions of the manuscript. SH conducted the analysis of the case report that was identified. All authors participated in the editing of the manuscript and take responsibility for its contents.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Cohen, H.P., Hachaichi, S., Bodenmueller, W. et al. Switching from One Biosimilar to Another Biosimilar of the Same Reference Biologic: A Systematic Review of Studies. BioDrugs 36, 625–637 (2022). https://doi.org/10.1007/s40259-022-00546-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40259-022-00546-6