Abstract
Background and Objective
Pharmaceutical policies are generally based on the assumption that involved stakeholders make rational decisions. However, behavioral economics has taught us that this is not always the case as people deviate from rational behavior in rather predictable patterns. This scoping review examined to what extent behavioral concepts have already been applied in the pharmaceutical domain and what evidence exists about their effectiveness, with the aim of formulating future applications and research hypotheses on policymaking for best-value biologicals.
Methods
A scoping literature review was conducted on the evidence of behavioral applications to pharmaceuticals. Scientific databases (Embase, MEDLINE, APA PsycArticles, and Scopus) were searched up to 20 October, 2021.
Results
Forty-four full-text scientific articles were identified and narratively described in this article. Pharmaceutical domains where behavioral concepts have been investigated relate to influencing prescribing behavior, improving medication adherence, and increasing vaccination uptake. Multiple behavioral concepts were examined in the identified studies, such as social norms, defaults, framing, loss aversion, availability, and present bias. The effectiveness of the applied interventions was generally positive, but depended on the context. Some of the examined interventions can easily be translated into effective policy interventions for best-value biological medicines. However, some applications require further investigation in a research context.
Conclusions
Applications of behavioral economics to pharmaceutical policymaking are promising. However, further research is required to investigate the effect of behavioral applications on policy interventions for a more sustainable market environment for best-value biological medicines.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
A competitive and sustainable market for off-patent biologicals and biosimilars implies the usage of best-value biologicals, thereby ensuring high-quality and affordable pharmaceutical care. |
The implementation of insights from behavioral economics holds promise for increasing the impact and effectiveness of existing policy frameworks for best-value biologicals. |
Different behavioral concepts can be translated into policy interventions to promote the use of best-value biologicals, such as loss aversion, defaults, social norms, framing, availability, and present bias. Some of these can be implemented with evidence of their impact, while others need further investigation in a research context in the future. |
1 Introduction
Biological medicines have revolutionized the treatment outcomes for patients with several debilitating disorders such as cancer, diabetes mellitus, rheumatoid arthritis, and psoriasis. However, because of a costly research and development process, biologicals have placed a substantial financial burden on national healthcare systems worldwide [1, 2]. Once the market exclusivities of biological therapies have expired, follow-on products can introduce competition into the market. In this way, costs are reduced and more uniform access to biological medicines can be obtained [1, 3, 4]. Competition into the off-patent biologicals market is achieved through the market access of biosimilar medicines, which are similar versions of already authorized biological medicines [5]. European member states have therefore formulated a wide range of policy measures to support the uptake of biosimilars into clinical practice, thereby trying to promote competition in the market [6,7,8]. However, competition induced by biosimilar medicines may also reduce the prices of reference biologicals and competing products within the same or different therapeutic classes [9, 10]. As a result, both biosimilar and originator biological products may contribute to the benefits of a more sustainable healthcare system. Therefore, the term best-value biological is preferred and introduced here. It emphasizes that the main focus should be on reaching a healthy competition between off-patent and patented biological medicines, thereby guaranteeing high-quality care while maintaining an affordable medicines bill [11].
Most policy interventions in healthcare rely on the assumption that people behave rationally or strive to maximize their utilities or profits [12,13,14]. Moreover, they assume that humans will act in their own long-term interests. These assumptions emerge from neoclassical economic theory, meaning individuals act to maximize their long-term best interests, are consistently rational, and their preferences remain stable [15]. Yet, this is not always the case as our behavior may deviate from these concepts [14, 15]. This realization has led to the emergence of behavioral economics (BE) in the mid-20th century, which is a mixed discipline covering economics, psychology, and neuroscience. It thereby considers that our rationality, willpower, and self-interest may be bounded and influenced by several external factors [15,16,17]. Research in this field has elicited that our behavior deviates from rationality in rather predictable patterns, leading to cognitive biases in our behavior [18]. Considering that our behavior may differ from rationality, policymakers can use insights from BE to increase the impact of current policy frameworks. In addition, BE might also help explain why people’s short-term choices undermine their long-term interests [12, 19]. Table 1 provides an overview of the most relevant biases or behavioral concepts with applications in healthcare.
In the context of best-value biologicals, policymakers often rely on rational choices being made by decision makers when implementing policy measures to support best-value biological usage. For instance, governments provide information to healthcare providers about the need for biosimilars for a more sustainable healthcare system [20]. They thereby assume decision makers, having understood all potential benefits of biosimilar medicines, will use biosimilars whenever possible. Another example are prescriber incentives to stimulate biosimilar prescribing. Policymakers hereby assume that healthcare providers will seek to maximize their individual profits and therefore prescribe more biosimilars. Notwithstanding the positive experiences with some of these policy interventions based on neoclassical economics [21,22,23], their impact can be further increased when considering BE principles [14].
Principles of BE are not new, although they have gained attention, particularly in the last decades. BE principles and the idea that psychological aspects influence monetary choice even find their origin in neoclassical economics [19, 24]. Until recently, BE mainly had commercial applications in finance and marketing. However, because of the shortcomings of traditional economic assumptions in policy implementations, policymakers have increasingly adopted elements from BE in their policy strategies. Examples are the Behavioural Insights Team in the United Kingdom (UK) and the United States of Amerika (USA) White House Social and Behavioral Science Team. Both were founded to advise the government on how to implement insights of BE on policymaking [25, 26]. Such policy institutions are often called nudging units, referring to the behavioral concept of nudging. Nudges have the purpose of subtly steering humans in a particular direction of interest, without imposing a certain choice or limiting the available options [27]. They often imply small changes to the choice architecture without changing the options or imposing a particular choice [28]. Nudges have the potential to make established healthcare policy frameworks more effective by simulating behavior that is aligned with what is rational from a societal point of view, without jeopardizing freedom of choice. This idea is also referred to as libertarian paternalism, as described by Thaler and Sunstein [29].
The applicability of BE to best-value biological policy frameworks is an interesting area that has not been explored to date. Therefore, the authors of this paper aimed to provide a review of the evidence of healthcare interventions emerging from BE insights. In addition, policy strategies to promote the usage of best-value biologicals are examined on how insights from BE can be applied to them to increase their impact. The ultimate goal of this article is to introduce promising behavioral applications for a sustainable policy framework for best-value biologicals, so further investigation and future implementation may follow.
2 Methods
2.1 Aims and Research Question
A scoping literature review was carried out to map the evidence of behavioral interventions that have been applied in the pharmaceutical domain. Scoping reviews are the appropriate way to map available evidence around a specific topic and formulate hypotheses for further research [38,39,40]. As described in the theoretical framework for scoping reviews by Arksey and O’Malley, this scoping review aimed to summarize the extent, range, and nature of the existing research within this domain [38].
2.2 Literature Search Strategy
A scoping review protocol with clearly defined inclusion and exclusion criteria was determined in advance by the researchers (see Electronic Supplementary Material [ESM]). The review included scientific literature that was searched for in relevant scientific databases (i.e., Embase, MEDLINE, APA PsycArticles, Scopus). English full-text articles published between 2000 and October 2021 could be included. Only studies where interventions based on behavioral concepts were tested or evaluated in a pharmaceutical context were eligible for inclusion. It was desirable to formulate a broad search strategy to generate future hypotheses and research questions in a field where behavioral insights have not (or limited) been researched to date. Therefore, search terms were related to BE, healthcare, pharmaceuticals, and policy. The complete search query with the different identified concepts can be found in the ESM. Literature was searched up to 20 October, 2021.
2.3 Selection of Studies and Data Extraction
All identified records were imported into Mendeley software and duplicates were removed. Second, two independent reviewers screened all records on title and abstract for relevance using Rayyan (Qatar Computing Research Institute, Doha, Qatar). Third, the remaining records were screened on their full texts based on the predefined inclusion criteria by one reviewer. Additionally, other relevant scientific papers were found through snowballing by looking at the reference lists of identified papers. The selected articles were summarized using Excel software. In doing so, all key characteristics per study (i.e., title, author, year, location, behavioral concepts tested, sample size, study design, outcome measures, objectives, and main results) were summarized (Table S2 of the ESM). Afterward, the identified studies were summarized narratively by the researchers.
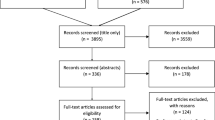
The researchers did not perform a formal methodological quality assessment, including a risk of bias evaluation, of the identified studies. This is in line with the existing guidelines for scoping literature reviews [38,39,40]. The results of the scoping review were reported according to the Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) [Fig. 1] [41].
3 Results
3.1 Literature Review
The structured literature search resulted in 6705 records in total. After removing duplicates and screening for relevance on title and abstract, 51 English full-text articles remained. The full texts of these identified records were assessed on their eligibility. Additional records were identified through reference list screening, after which 44 articles were included in the final analysis described in the paper (Fig. 1).
3.2 Evidence from BE Interventions
Of all identified studies, three main areas were found where they were conducted. The largest group was related to influencing prescribing behavior among physicians [42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61] (n = 20), a second group aimed to evaluate ways to stimulate vaccine uptake [62,63,64,65,66,67,68,69,70,71,72,73,74,75] (n = 14), and a last group examined methods to increase medication adherence [76,77,78,79,80,81,82,83,84,85] (n = 10). Identified studies were conducted in the USA (n = 32) [42, 44,45,46,47,48,49,50,51,52,53, 56, 58,59,60,61, 64, 66, 68,69,70,71,72, 74, 75, 78,79,80,81,82,83,84], the UK (n = 6) [43, 54, 55, 57, 63, 85], Singapore (n = 1) [76], Tanzania (n = 1) [77], Germany (n = 1) [62], France (n = 1) [73], Australia (n = 1) [67], and the Netherlands (n = 1) [65].
Multiple behavioral concepts were tested in the identified studies, but the majority involved social norms [44,45,46, 48,49,50, 54, 56, 57, 59, 62,63,64, 69, 77, 81] (n = 16), defaults [42, 43, 47, 50, 51, 53, 55,56,57,58, 65, 68, 82] (n = 13), availability [44, 45, 50, 52, 60, 71, 72, 75] (n = 8), framing [44, 47, 56, 61] (n = 4), loss aversion [76, 79, 80] (n = 3), and present bias [76, 79] (n = 1). A variety of study designs was observed among all studies, but most were randomized controlled trials [44, 46, 47, 49, 50, 54, 59, 60, 63,64,65, 67,68,69,70, 73,74,75,76, 78,79,80,81,82,83,84,85] (n = 27). Other study designs included pre-post longitudinal studies [42, 43, 51, 52, 55, 56, 61, 72, 77] (n = 9), randomized trials without a control group [45, 48, 62] (n = 3), retrospective [58, 71] (n = 2), quasi-experimental [53, 66] (n = 2), or qualitative multi-method studies [57] (n = 1).
Overall, 20 studies found a significant effect of the behavioral intervention on the desired outcome [43, 47, 48, 53,54,55,56,57,58,59,60,61, 66,67,68, 71, 72, 76, 78, 83], 15 found partially significant effects [42, 44,45,46, 49, 51, 52, 62, 64, 65, 69, 70, 74, 77, 82], and nine did not observe a significant effect [50, 63, 73, 75, 79,80,81, 84, 85]. A partial effect was found in studies in which several behavioral concepts or different situations were examined, and only one or a few resulted in a significant effect. A summary of the study characteristics related to the identified articles can be found in Table S2 of the ESM.
3.3 Prescribing Behavior
All but one of the identified studies related to prescribing behavior showed at least partially significant improvements in prescribing behavior or prescribing choices under the influence of an applied BE intervention [42,43,44,45,46,47,48,49, 51,52,53,54,55,56,57,58,59,60,61]. Different behavioral concepts were examined and applied to prescribing choices, such as defaults [42, 43, 47, 51, 53, 55, 58], social norms [46, 48, 54], availability [52, 60], and framing [61].
3.3.1 Defaults
In total, seven different studies in varying contexts examined the effect of default options to influence prescribing choices [42, 43, 47, 51, 53, 55, 58]. Three different studies looked at the effect of defaulting a lower opioid dose in the electronic prescribing software on opioid prescribing. In all three studies, such an intervention led to a decrease in the number of opioid prescriptions [42, 51, 55]. Another study defaulted chlorhexidine for intensive care patients in the prescribing software, leading to a sustained and predictable increase in chlorhexidine prescriptions [43]. Similar findings were also found for antibiotic prescribing when changing the order set in the electronic prescribing software in a simulation context [47].
Another possible area to influence prescribing behavior is to prescribe generics instead of branded drugs. Two studies investigated the effect of defaulting the generic alternative instead of the branded version [53, 58]. Redesigning the prescribing interface led to significant increases in generic prescriptions in both studies.
3.3.2 Social Norms
Researchers examined the effect of social norms on antibiotic prescribing through physicians receiving a letter from their chief medical officer that their antibiotic prescribing rates were among the highest within their region. This resulted in an sharp and significant decrease in antibiotic prescribing [54]. Another randomized controlled trial assessed the effect of peer comparison feedback to reduce opioid prescribing. Intervention groups received feedback on their opioid prescribing, with one group receiving additional peer comparison feedback. Although not significantly different from the control group, more physicians reduced their opioid prescribing in the intervention group with peer comparison information [46]. A third study evaluated the impact of a behavioral nudge that included peer comparison feedback on inappropriate antibiotic prescribing. The interventions led to a statistically significant drop in antibiotic prescribing. However, no significant isolated effect of social norm feedback was observed [48].
3.3.3 Availability
Availability insights were tested via indicating more expensive medications with cost transparency [60] or color alerts [52] in the electronic prescribing software. Both interventions were found to be effective in decreasing the prescribing of high-cost medication.
3.3.4 Framing
The effect of framing on antibiotic prescribing was studied among hospitalized patients treated with anti-methicillin-resistant Staphylococcus aureus and antipseudomonal antibiotics [61]. The intervention included a microbiology comment highlighting the absence of methicillin-resistant Staphylococcus aureus and Pseudomonas aeruginosa in the respiratory culture, after which a significant reduction in broad-spectrum antibiotic prescribing was observed.
3.3.5 Mixed or Non-Specific Concepts
Several studies were found that combined different concepts from BE to enhance the effect on the desired behavior [44, 45, 50, 56, 57, 59]. To promote appropriate diabetes management, a behavioral electronic health record technology was developed and found effective among adult patients [56]. The intervention included a combination of six different nudges, using framing, social norms, accountable justification, defaults, affirmation, and gamification.
The simultaneous effect of active choice framing (availability) and social norms feedback to increase guideline-concordant statin prescribing was also evaluated [44]. This combined intervention was found effective in increasing guideline-concordant statin prescribing. However, the effect of the intervention of active choice on its own did not reach statistical significance. Another trial examined the isolated effects of active choice, peer comparison, and accountable justification on more appropriate antibiotic prescribing [45]. Peer comparison and the active choice group caused a significant decrease in antibiotic prescribing. However, reductions were observed regardless of whether the researchers applied behavioral interventions. This study served as a precursor for a larger randomized controlled trial by the same researchers [49]. This larger trial partly confirmed the results of the pilot study. However, the researchers only found significant reductions in antibiotic prescribing rates for accountable justification and peer comparison. Different behavioral concepts were also combined to prevent prescribing errors and optimize prescribing behavior, such as social norms and defaults [57]. After implementing the combined intervention, the occurrence of medication errors significantly decreased. Another behavioral nudge based on the principle of time inconsistency and social norms was effective in reducing inappropriate antibiotic prescribing, by putting up commitment posters for 12 weeks in the doctor’s office [59].
One study did not conclude any significant effect on prescribing behavior. This multi-component intervention, including social norms, priming (i.e., our behavior is subconsciously influenced by external stimuli such as sounds, smells, or views), availability, and defaults, did not result in significant differences in anticholinergic medication usage [50].
3.4 Vaccination Uptake
The second set of studies aimed to increase vaccination uptake through behavioral interventions. This topic has currently gained importance given the coronavirus disease 2019 (COVID-19) pandemic and low vaccination rates in certain countries or regions [86]. Out of 14 studies, 11 found (partial) significant positive effects [62, 64,65,66,67,68,69,70,71,72, 74] of behavioral interventions or nudges on vaccination uptake. Only three did not find any positive effects [63, 73, 75].
3.4.1 Social Norms
Three different studies assessed the effect of peer comparison or social norm feedback on improving vaccination rates [62,63,64]. During a randomized controlled trial, a vaccination game was developed to model the direct and indirect effects of vaccinations on disease elimination. Researchers hypothesized that rewarding the attainment goal (i.e., eliminating disease) and comparing vaccination rates of peers will increase the chances of getting vaccinated [62]. The study found a positive and significant effect of the reward intervention, but not of the peer comparison intervention. In contrast to most other experiments discussed in this review, this study was carried out in a laboratory environment. A second study did not find any significant difference in vaccination coverage between groups after a social norm-applied intervention was implemented, including letters comparing vaccination rates with peers [63]. A third study found significant effects on improving the quality of care using performance feedback and benchmarking [64]. Several quality indicators were used, including influenza vaccination rates, which increased significantly because of the intervention.
3.4.2 Defaults
Two different randomized controlled trials looked at the effect of making the influenza vaccination the default option on vaccination uptake [65, 68]. In both studies, participants were randomized to either the opt-in or the opt-out group. In the opt-out intervention group, vaccination was the default choice by sending a pre-scheduled appointment for vaccination. The control condition, or the opt-in group, asked participants to make an appointment actively. Both studies found positive effects on vaccination rates or vaccination appointments due to the default intervention. However, only in the larger trial did this result in significantly higher vaccination rates compared with the control group [68].
3.4.3 Availability
In two different studies, the effect of availability was examined in the patient’s electronic health record through offering an active choice. This included the electronic software automatically displaying a pop-up asking the clinician if a vaccine should be ordered. Both studies found large and significant increases in vaccination rates because of the active choice implementation [71, 72]. Electronic health record alerts to remind physicians to propose an influenza vaccination among children with asthma did not significantly increase vaccination rates, despite the large sample size of over 10,000 children [75].
3.4.4 Mixed or Non-Specific Concepts
Similar to one of the abovementioned studies looking at the effect of behavioral commitment posters on prescribing behavior [59], the effect of commitment posters was assessed to increase human papillomavirus vaccinations [69]. The researchers found no isolated effect of the commitment posters. However, this intervention was combined in both the control and intervention groups with other proven strategies to enhance vaccination uptake (e.g., feedback). The fact that vaccination uptake increased in both control and intervention groups could be attributed to the effect of these proven strategies.
Multiple behavioral concepts were also examined in a large study to increase COVID-19 vaccination uptake [74]. A positive and significant effect of text reminders on vaccination uptake was found for both a first and second reminder. A message to make participants feel ownership of the vaccine further enlarged this effect. The effect of text messages to nudge people towards getting vaccinated was also examined in other studies [66, 67, 70, 73]. Two large trials, with over 50,000 participants in total, showed a significant increase in vaccination uptake after text message-based nudges [67, 70]. In two other studies with smaller sample sizes, one found a positive effect of the behavioral nudge [66] and one did not [73].
3.5 Medication Adherence
Ten studies that examined the effects of interventions based on behavioral concepts to improve medication adherence were found [76,77,78,79,80,81,82,83,84,85]. Mixed effects of the different behavioral interventions in these ten different studies were found. Only five studies found (partial) significant effects on medication adherence [76,77,78, 82, 83], the remaining five did not [79,80,81, 84, 85].
A loss aversion-based intervention was found successful, where participants could lower the cost of their therapy by rebates depending on meeting the target adherence [76]. Improvements in medication adherence were also found in three other studies, with differing designs and behavioral concepts examined [77, 78, 83]. Human immunodeficiency virus (HIV) medication adherence was higher in the experimental group after introducing an intervention publicly recognizing their adherence via posters being publicly displayed at the clinic [77]. Positive results were also found when feedback reports were provided to non-adherent patients taking statin medication [78]. Significant effects were found on HIV medication adherence as well by texting reminder messages to patients [83]. The effect of defaults was also assessed by making the pharmacist automatically contact non-adherent patients by default [82]. Pharmacists contacting patients did not lead to higher adherence rates. Nonetheless, the pharmacist’s intervention was more likely to occur when it was triggered by default instead of by active choice.
An incentive based on loss aversion and present bias did not lead to higher antihypertensive or antihyperlipidemic medication adherence [79]. The intervention involved paying out an incentive immediately to adherent patients rather than deferring it, and reducing the incentive when doses were missed. A different loss aversion-based intervention to improve medication adherence was also tested in a trial among patients with acute coronary syndrome [80]. After the intervention period, adherence in the intervention group was higher than in the control group. However, this result was not statistically significant. Other behavioral nudges included automated text reminders and electronic monitoring containers, both not resulting in a significant increase in medication adherence [81, 84, 85].
4 Discussion
4.1 Evidence from BE Interventions
This scoping review presents an overview of the available evidence regarding the applications of behavioral sciences to various pharmaceutical fields. Within this context, BE insights have so far been mainly investigated to influence physician prescribing, increase medication adherence, and stimulate vaccination uptake. These fields have in common that they have pressing needs that require new ways to solve the existing issues. For example, the global problem of inappropriate and unrestrained antibiotic prescribing, leading to resistant bacteria [87]. There is also the problem of low medication adherence in several chronic conditions, leading to therapy failure over time [88, 89]. Even more applicable to the present is the importance of high vaccination coverage to contain the spread of infectious diseases (e.g., flu or coronavirus disease 2019) [86, 90]. This list should include rising medication costs, a problem that many countries are struggling with today. Competition through generics has proven to effectively contain spending on off-patent small molecules [91]. This is also the case for competition in the biological medicines market, where the use of biosimilars mitigates the rising costs of these expensive therapies after their patent expiry [1]. Moreover, these three fields have in common that they aim to increase the health status of individuals or society. Increasing the usage of best-value biologicals also offers advantages from a societal healthcare perspective. By increasing the competitiveness of the off-patent biologicals market, the cost of off-patent biologicals decreases and more patients can be treated with biological therapies [3, 4, 92,93,94]. Therefore, as with increasing vaccination uptake, governments aim to increase the usage of best-value biologicals as this is in the best interests of society.
In this review, we showed that several studies have already looked at the potential of BE to address a variety of societal issues in the pharmaceutical domain. The applications of behavioral insights have generally proven to be effective in the identified scientific articles. Nevertheless, a significant result was not always found. In nine of the 44 identified studies this was not the case [50, 63, 73, 75, 79,80,81, 84, 85]. Only partially significant effects were found in 15 studies [42, 44,45,46, 49, 51, 52, 62, 64, 65, 69, 70, 74, 77, 82]. Often, these studies examined multiple behavioral interventions or several outcome measures simultaneously, and not all of them proved effective.
Based on this review, the effect of influencing prescribing behavior appears to be larger than for vaccination uptake and medication adherence. However, such a conclusion would require a larger scale quantitative evaluation of the existing evidence, which was not the purpose of this scoping review. Moreover, as most studies were conducted in the USA, it remains uncertain to what extent these findings could be extrapolated to other healthcare systems. In some studies where researchers could not demonstrate the effectiveness of the behavioral intervention, this was attributed by the authors to a too small and underpowered study sample or ceiling effects (i.e., where one already had a high adherence or vaccine uptake at baseline, thus incremental effects are limited) [73, 79,80,81]. However, in most cases, the authors provided no specific explanation.
Even though this review provides an overview of the existing evidence on applications of behavioral sciences in the pharmaceutical domain, the applications to best-value biologicals have not been reported to date. Based on previous research in adjacent fields, policymakers and researchers should be optimistic about the outcomes of behavioral science-based interventions to existing policy measures. For this reason, one should not be reluctant to explore further and implement these concepts to promote the usage of best-value biologicals. When implementing behavioral concepts in the future to achieve a more sustainable market environment for best-value biologicals, one will need to carefully evaluate the effectiveness of such interventions in a research context [16, 95].
When creating behaviorally informed policy interventions, it is primarily necessary to thoroughly understand the underlying decision-making process of the intended behavior [16]. For instance, when looking at increasing the prescribing of best-value biologicals, it is required to understand the underlying drivers behind these prescribing choices. In this regard, a distinction has to be made between behavior deviating from individual and collective rationality due to cognitive biases, and when individual rational preferences are misaligned with societal interests [14]. In pharmaceutical decision making, individual interests are not always in line with what is rational from a societal point of view (e.g., a sustainable healthcare system) [96]. For example, related to best-value biologicals, hospitals try to maximize their profits by using expensive medicines [97], which is an individually rational choice. Yet, hospitals thereby undermine the sustainability of healthcare systems and act against what is collectively rational. These mechanisms are widespread across healthcare systems and may be underlying drivers for healthcare decision makers. Policymakers have therefore attempted to align individual and collective interests, using neoclassical economic theory (i.e., prescriber incentives). Behaviorally informed interventions can further improve the impact of such measures by further aligning individual with collective or societal interests.
The additional cost of applying BE principles to policy is relatively low, while the impact of existing measures can be increased [14, 16, 96, 98]. Therefore, the absence of state-of-the-art science-led pharmaceutical policy interventions creating effective nudges would be a missed opportunity. Certain elements could be efficiently applied to best-value biological policy interventions while preserving the nature of the original measure (Fig. 2). BE principles are therefore not intended to replace currently proven effective strategies for sustaining the off-patent biologicals market. Insights from BE should be seen as an extra layer on existing initiatives to make them more successful and increase their impact. It should be emphasized that certain existing policy measures to improve biosimilar competition based on neoclassical economics do have their value and impact [8, 9, 21, 23]. Therefore, future policy frameworks ideally include existing effective policy strategies based on neoclassical economics, accompanied or strengthened by policies based on BE insights. Both policy strategies should go hand in hand, not as separate strategies or as a substitute for one another [14].
The ways in which policymakers can apply BE insights to best-value biologicals are not country specific. However, as for all pharmaceutical policy interventions, they should be adapted and tailored to the features of the respective national healthcare system. A tailored and holistic approach regarding best-value biological policy frameworks should therefore be the standard. This includes a multifaceted approach, considering the characteristics of the off-patent biologicals market in a given healthcare setting.
4.2 Applying Concepts of BE to Promote the Use of Best-Value Biologicals
In the section below, we discuss how behavioral concepts could be applied to policymaking to increase the usage of best-value biological medicines. According to the authors, these concepts have the most potential and relevance within the context of policy interventions to support the use of best-value biologicals. Some behavioral insights have led to newly proposed policy measures, others are applications to established policy strategies (Fig. 2).
4.3 Loss Aversion
Interventions based on loss aversion can be applied in several ways to best-value biological policymaking, especially in incentive design or when creating gainsharing schemes. Gainsharing or benefit-sharing schemes refer to an incentive to encourage biosimilar usage by reinvesting savings back into patient care, while additional efforts of healthcare providers when transitioning from product A to product B are compensated [6, 99]. Without going into the modalities of such incentives, they usually assume that prescribers act rationally and wish to maximize their utilities. However, research from behavioral economists has taught us that the context or environment in which these incentives are provided strongly influences their success [100]. Altering the conditions in which prescriber incentives are given, without changing the nature of the incentive itself, might have considerable impact on the effectiveness of the incentive. A possible avenue for improvement for prescriber incentives is presenting the incentive as a loss rather than as a gain. An often-mentioned example of a tangible incentive is the financing of a nurse guiding the transitioning process to a biosimilar [101]. An alternative design could be that one already (partly) offers this support, but withdraws or reduces it after a certain period if one does not prescribe a certain proportion of biosimilars or best-value biologicals within their hospital. Future research could examine the effectiveness of such an alternative approach for gainsharing schemes.
In addition, the concept of loss aversion might also help explain the reluctance among prescribers to transition their patients from the originator biological to a biosimilar. Physicians often fear a decreasing efficacy after transitioning to a biosimilar, as well as a loss of possible informal benefits linked to the original biological (i.e., scientific support, clinical trial involvement) [102,103,104]. Pharmaceutical promotion is known to be a strong driver to prescribe new medicines [105]. Therefore, prescribing biosimilars could be linked to losing certain benefits offered by pharmaceutical companies. When designing policy interventions to promote best-value biologicals, policymakers or companies should consider ways to replace these existing informal benefits and emphasize with adequate information the equal clinical outcomes.
4.4 Framing
Options can be framed as losses or gains (i.e., negative or positive framing), while these options are equivalent. The European Medicines Agency refers to biosimilar medicines as having “no clinically meaningful differences compared with their reference medicine” [20]. Yet, this is a rather negatively framed statement about biosimilars. One could also phrase this as biosimilars having equivalent clinical outcomes compared with their reference medicine, or even as providing additional benefits for healthcare systems. National competent authorities or patient organizations often mention in their position statements that biosimilars are not inferior in terms of safety and efficacy to their reference product [106, 107]. Although such statements are scientifically correct, one can expect that trust among clinicians and patients would improve if these organizations would state that biosimilars are as safe and effective as their reference medicine [103]. Research has already proven that positively framed communication about biosimilars toward patients positively influences its acceptance [108].
4.5 Status Quo or Defaults
Important lessons can be drawn from previous healthcare applications of status quo or default biases for policymakers [109]. An important opportunity lies in prescribing software, used in most European countries. Suppose the most cost-effective medicine (i.e., best-value biological) of a particular molecule (or therapeutic class) is positioned as the first option to appear in the prescribing software. In that case, it can be assumed that physicians will preferably choose the more cost-effective product. This was already proven to be an effective strategy for generic medicines and is therefore a promising way to stimulate cost-conscious prescribing of biological medicines as well [58].
Status quo biases may also play a role in physicians’ reluctance to prescribe biosimilars [103, 110]. Sticking with what one is familiar with or has good experiences with, leads to prescriber inertia that suppresses the use of more cost-effective alternatives such as biosimilars.
4.6 Social Norms
Policymakers have already tried to nudge people’s behavior towards what is desirable using social norms in several ways [16, 96]. This is established by giving information to individuals about the behavior of peers, whereby one wishes to achieve the behavior of the others in the individual. Policymakers can apply insights from social norms to stimulate the use of best-value biologicals among both prescribers and purchasers as well. For example, health insurers can provide prescribing feedback to physicians, such as their proportion of best-value biological prescriptions. This should be compared with fellow physicians within their discipline. In this way, physicians who prescribe fewer best-value biologicals are encouraged to lean more towards the prescribing behavior of their colleagues. Displaying the cost of different products in the prescribing software has a further awareness-raising effect (see Sect. 4.8 on availability), especially when compared with peers [54]. Social norms have so far provided mixed results on influencing prescribing behavior. Therefore, further research is required in this regard.
4.7 Present Bias or Hyperbolic Discounting
Generally speaking, people will estimate the value of an immediate reward higher than the same reward further in time. This principle could also be applied to best-value biologicals. For example, in the context of prescriber incentives to compensate for the additional efforts made when transitioning to a biosimilar [101]. Such incentives are often awarded to clinicians after a certain period of time (e.g., at the end of the year). One could say that an incentive to prescribe biosimilars loses its value as it is given to the physician at a later date [111]. For that purpose, again without speaking about the nature of the incentive, the timing when it is awarded could determine its impact on physicians’ prescribing behavior. Therefore, it would be desirable to monitor prescribing behavior at more regular intervals (i.e., more regular feedback in comparison to peers), followed by timely provision of the incentive (e.g., quarterly instead of at the end of the year). As discussed earlier, such an alternative gainsharing design would benefit from further investigation in a research context.
Moreover, present bias could help clarify why clinicians, hospitals, and patients do not always understand the added value of best-value biologicals or biosimilars. The message that biosimilars provide more sustainable healthcare is too abstract and not tangible in the short term. After all, the benefits will only be visible in the long term (i.e., more and earlier access to biologicals), while the extra efforts (i.e., writing tenders, explanation to the patient when transitioning) are immediate drawbacks for stakeholders involved. It is therefore important that policymakers also ensure the implementation of short-term benefits for clinicians and patients, as the longer term value alone has less influence on their behavior. Such short-term benefits could be implemented as regular prescribing feedback, alongside regular provision of benefit-sharing initiatives (Fig. 2). Instead, longer term benefits should be part of the overall information package that policymakers provide.
4.8 Availability
People estimate the probability of a particular event based on how easily one can remember such an event. Often, people tend to overestimate the likelihood of events that are notable or salient [18, 37]. This refers to the behavioral principle of salience, meaning that people are influenced in their decisions by new and visible events [25]. Two studies discussed in this review aimed to increase the prescribing of cost-effective medicines by presenting the more cost-effective alternative or showing cost alerts when lower value medicines are prescribed [52, 60]. In doing so, they use information salience, steering prescribers in the direction of a higher value medicine by making this option particularly visible. One could apply this to best-value biologicals by showing cost alerts when a product is being prescribed for which a more cost-effective alternative (i.e., best-value biological) is available. In addition, active choices can be offered to physicians in the electronic prescribing software in such cases, where higher value biological alternatives are suggested to the physician.
Availability heuristics might also help explain why and how misinformation about biosimilars is one of the reasons for mistrust among physicians and patients. One of the most well-known misconceptions among physicians and patients is that biosimilars could lead to more side effects or decreased efficacy after transitioning [103]. The spread of stories of patients experiencing decreased efficacy after switching to the biosimilar, without a scientific basis, may have fueled this mistrust. These stories are more likely to be recalled, especially if they are repeated many times by the pharmaceutical industry [112, 113]. This could have led to physicians and patients overestimating the likelihood of such events, resulting in low confidence in and use of biosimilars. The most important way to prevent this is by continuously providing correct and repeated information about biosimilars, including positive experiences with biosimilars.
4.9 Study Strengths and Limitations
This literature review provides an overview of available evidence regarding applications of BE to pharmaceutical policy. A scoping review is an appropriate technique for summarizing the existing evidence and searching for hypotheses on future applications to policy interventions to increase the usage of best-value biologicals. A systematic approach was taken based on a predefined search protocol using broad search terms. In this way, as much as possible of the existing evidence in the scientific literature could be identified. This is the first scientific article that sheds light on potential applications of BE to future policy interventions for best-value biologicals, based on an existing body of evidence within the pharmaceutical domain.
However, there are some disadvantages inherent to scoping reviews. First, the full-text articles were not screened by two independent investigators, thus there is a certain risk that relevant articles have been missed. However, this probability was reduced by the structured search approach and screening of the reference lists of identified articles. Second, a scoping review does not include a quality assessment of the identified articles, as is the case for systematic literature reviews [40]. This review only included articles indexed in MEDLINE, Embase, APA PsycArticles, and Scopus. Hence, potentially relevant studies not included in these databases could not be identified. Because the researchers used a broad search strategy using general search terms related to healthcare and BE, relevant articles including more narrow search terms related to the discussed behavioral concepts (e.g., loss aversion, framing, status quo, defaults) might have been missed.
The applications of BE insights discussed in this article were mainly looked at from a European point of view. Therefore, some of the mentioned examples may not be generalizable to other healthcare systems.
5 Conclusions
Applications of BE to pharmaceutical policy are promising. A large body of evidence already exists on insights that can successfully be applied to medication adherence, vaccination uptake, and prescription behavior. As a result, one can be optimistic about applying concepts such as social norms, defaults, loss aversion, present bias, availability, and framing to policy interventions to promote the use of best-value biologicals. In return, this will support a sustainable market for off-patent biologicals. There are multiple entry points for applying these concepts to best-value biological policies, such as gainsharing, prescribing quota or feedback, electronic prescribing software, patient communication, and healthcare provider education or information. Some of the examined applications can easily be extrapolated to best-value biological policies. Others need to be further explored in this particular context. Continued research with concrete behavioral applications based on a behavioral model is therefore required. This scoping review provided an initial impetus by developing future research questions and exploring hypotheses to increase the use of best-value biological medicines.
References
IQVIA. The impact of biosimilar competition in Europe. 2020. Available from: https://www.iqvia.com/library/white-papers/the-impact-of-biosimilar-competition-in-europe. [Accessed 16 Jan 2021].
IQVIA. The global use of medicines 2022: outlook to 2026. 2021. Available from: https://www.iqvia.com/-/media/iqvia/pdfs/institute-reports/the-global-use-of-medicines-2022/global-use-of-medicines-2022-outlook-to-2026-12-21-forweb.pdf. [Accessed 16 Dec 2021].
IQVIA. The impact of biosimilar competition in Europe. 2021. Available from: https://www.iqvia.com/-/media/iqvia/pdfs/emea/the-impact-of-biosimilar-competition-in-europe-iqvia.pdf. [Accessed 3 Jan 2022].
Dutta B, Huys I, Vulto AG, Simoens S. Identifying key benefits in European off-patent biologics and biosimilar markets: it is not only about price! BioDrugs. 2020;34:159–70.
European Medicines Agency. Biosimilars in the EU: information guide for healthcare professionals. 2017. Available from: https://www.ema.europa.eu/en/documents/leaflet/biosimilars-eu-information-guide-healthcare-professionals_en.pdf. [Accessed 28 Jul 2022].
Moorkens E, Vulto AG, Huys I, Dylst P, Godman B, Keuerleber S, et al. Policies for biosimilar uptake in Europe: an overview. PLoS ONE. 2017;12: e0190147.
Remuzat C, Kapusniak A, Caban A, Ionescu D, Radiere G, Mendoza C, et al. Supply-side and demand-side policies for biosimilars: an overview in 10 European member states. J Mark Access Health Policy. 2017;5:1307315.
Vogler S, Schneider P, Zuba M, Busse R, Panteli D. Policies to encourage the use of biosimilars in European countries and their potential impact on pharmaceutical expenditure. Front Pharmacol. 2021;21: 625296.
Vulto AG. Biologicals and biosimilars in hematology: the case of rituximab. HemaSphere. 2019;3: e322.
Autoriteit Consument & Markt. Sectoronderzoek concurrentie voor en na toetreding van biosimilars. 2019; September. Available from: https://www.acm.nl/nl/publicaties/sectoronderzoek-tnf-alfaremmers. [Accessed 17 Jun 2020].
NHS England and NHS Improvement. What is a biosimilar? 2019. Available from: https://www.england.nhs.uk/wp-content/uploads/2019/05/what-is-a-biosimilar-medicine-guide-v2.pdf. [Accessed 3 Dec 2021].
Soofi M, Najafi F, Karami-Matin B. Using insights from behavioral economics to mitigate the spread of COVID-19. Appl Health Econ Health Policy. 2020;18:345–50.
Ogdie A, Asch DA. Changing health behaviours in rheumatology: an introduction to behavioural economics. Nat Rev Rheumatol. 2020;16:53–60.
Connelly LB, Birch S. Sustainability of publicly funded health care systems: what does behavioural economics offer? Pharmacoeconomics. 2020;38:1289–95.
Kahneman D. A psychological perspective on economics. Am Econon Rev. 2003;93:162–8.
Matjasko JL, Cawley JH, Baker-Goering MM, Yokum DV. Applying behavioral economics to public health policy: illustrative examples and promising directions. Am J Prev Med. 2016;50:S13–9.
Tversky A, Kahneman D. Judgment under uncertainty: heuristics and biases. Science. 1974;185:1124–31.
Kahneman D. Thinking, fast and slow. New York: Farrar, Straus and Giroux; 2011.
Thaler RH. Behavioral economics: Past, present, and future. Am Econ Rev. 2016;106:1577–600.
European Medicines Agency (EMA). Biosimilars in the EU: information guide for healthcare professionals. 2017. Available from: https://www.ema.europa.eu/en/documents/leaflet/biosimilars-eu-information-guide-healthcare-professionals_en.pdf. [Accessed 12 Apr 2020].
Remuzat C, Dorey J, Cristeau O, Ionescu D, Radiere G, Toumi M, et al. Key drivers for market penetration of biosimilars in Europe. J Mark Access Health Policy. 2017;5:1272308.
Moorkens E, Simoens S, Troein P, Declerck P, Vulto AG, Huys I, et al. Different policy measures and practices between Swedish counties influence market dynamics: part 1-biosimilar and originator infliximab in the hospital setting. BioDrugs. 2019;33:285–97.
Moorkens E, Barcina Lacosta T, Vulto AG, Schulz M, Gradl G, Enners S, et al. Learnings from regional market dynamics of originator and biosimilar infliximab and etanercept in Germany. Pharmaceuticals (Basel). 2020;13:324.
ur Rehman T. Historical context of behavioral economics. Intellect Econ. 2016;10:128–32.
Cabinet Office and Behavioural Insights Team. Applying behavioural insight to health: behavioural insights team paper. 2010. Available from: https://www.gov.uk/government/publications/applying-behavioural-insight-to-health-behavioural-insights-team-paper. [Accessed 1 Jul 2021].
US Government. Social and Behavioral Science Team. 2014. Available from: https://sbst.gov/. [Accessed 1 Jul 2021].
Bhattacharya J, Garber AM, Goldhaber-Fiebert JD. Nudges in exercise commitment contracts: a randomized trial. National Bureau of Economic Research, Inc.; 2015.
Vallgårda S. Nudge: a new and better way to improve health? Health Policy (New York). 2012;104:200–3.
Thaler RH, Sunstein CR. Libertarian paternalism. Am Econ Rev. 2003;93:175–9.
Kahneman D, Tversky A. Prospect theory: an analysis of decision under risk. Econometrica. 1979;47:263–91.
Samuelson W, Zeckhauser R. Status quo bias in decision making. J Risk Uncertain. 1988;1:7–59.
Tversky A, Kahneman D. The framing of decisions and the psychology of choice. Science. 1981;211:453–8.
Levin IP, Schneider SL, Gaeth GJ. All frames are not created equal: a typology and critical analysis of framing effects. Organ Behav Hum Decis Process. 1998;76:149–88.
Schultz PW, Nolan JM, Cialdini RB, Goldstein NJ, Griskevicius V. The constructive, destructive, and reconstructive power of social norms: reprise. Perspect Psychol Sci. 2018;13:249–54.
Laibson D. Golden eggs and hyperbolic discounting. Q J Econ. 1997;112:443–78.
Hepburn C, Duncan S, Papachristodoulou A. Behavioural economics, hyperbolic discounting and environmental policy. Environ Resour Econ. 2010;46:189–206.
Noggle R. Manipulation, salience, and nudges. Bioethics. 2018;32:164–70.
Arksey H, O’Malley L. Scoping studies: towards a methodological framework. Int J Soc Res Methodol. 2005;8:19–32.
Peters MDJ, Godfrey CM, Khalil H, McInerney P, Parker D, Soares CB. Guidance for conducting systematic scoping reviews. Int J Evid Based Healthc. 2015;13(3):141–6.
Munn Z, Peters MDJ, Stern C, Tufanaru C, McArthur A, Aromataris E. Systematic review or scoping review? Guidance for authors when choosing between a systematic or scoping review approach. BMC Med Res Methodol. 2018;18:143.
Tricco AC, Lillie E, Zarin W, O’Brien KK, Colquhoun H, Levac D, et al. PRISMA extension for scoping reviews (PRISMA-ScR): checklist and explanation. Ann Intern Med. 2018;169:467–73.
Ancker JS, Gossey JT, Nosal S, Xu C, Banerjee S, Wang Y, et al. Effect of an electronic health record “nudge” on opioid prescribing and electronic health record keystrokes in ambulatory care. J Gen Intern Med. 2021;36:430–7.
Bourdeaux CP, Davies KJ, Thomas MJC, Bewley JS, Gould TH. Using “nudge” principles for order set design: a before and after evaluation of an electronic prescribing template in critical care. BMJ Qual Saf. 2014;23:382–8.
Patel MS, Kurtzman GW, Kannan S, Small DS, Morris A, Honeywell SJ, et al. Effect of an automated patient dashboard using active choice and peer comparison performance feedback to physicians on statin prescribing: the PRESCRIBE cluster randomized clinical trial. JAMA Netw Open. 2018;1: e180818.
Persell SD, Doctor JN, Friedberg MW, Meeker D, Friesema E, Cooper A, et al. Behavioral interventions to reduce inappropriate antibiotic prescribing: a randomized pilot trial. BMC Infect Dis. 2016;16:373.
Suffoletto B, Landau A. Nudging emergency care providers to reduce opioid prescribing using peer norm comparison feedback: a pilot randomized trial. Pain Med. 2020;21:1393–9.
Tannenbaum D, Doctor JN, Persell SD, Friedberg MW, Meeker D, Friesema EM, et al. Nudging physician prescription decisions by partitioning the order set: results of a vignette-based study. J Gen Intern Med. 2015;30(3):298–304.
Yadav K, Meeker D, Mistry RD, Doctor JN, Fleming-Dutra KE, Fleischman RJ, et al. A multifaceted intervention improves prescribing for acute respiratory infection for adults and children in emergency department and urgent care settings. Acad Emerg Med. 2019;26:719–31.
Meeker D, Linder JA, Fox CR, Friedberg MW, Persell SD, Goldstein NJ, et al. Effect of behavioral interventions on inappropriate antibiotic prescribing among primary care practices: a randomized clinical trial. JAMA. 2016;315:562–70.
Campbell NL, Holden RJ, Tang Q, Boustani MA, Teal E, Hillstrom J, et al. Multicomponent behavioral intervention to reduce exposure to anticholinergics in primary care older adults. J Am Geriatr Soc. 2021;69:1490–9.
Delgado MK, Shofer FS, Patel MS, Halpern S, Edwards C, Meisel ZF, et al. Association between electronic medical record implementation of default opioid prescription quantities and prescribing behavior in two emergency departments. J Gen Intern Med. 2018;33:409–11.
Fischer MA, Vogeli C, Stedman M, Ferris T, Brookhart MA, Weissman JS. Effect of electronic prescribing with formulary decision support on medication use and cost. Arch Intern Med. 2008;168:2433–9.
Patel MS, Day S, Small DS, Howell JT 3rd, Lautenbach GL, Nierman EH, et al. Using default options within the electronic health record to increase the prescribing of generic-equivalent medications: a quasi-experimental study. Ann Intern Med. 2014;161(10 Suppl.):S44-52.
Hallsworth M, Chadborn T, Sallis A, Sanders M, Berry D, Greaves F, et al. Provision of social norm feedback to high prescribers of antibiotics in general practice: a pragmatic national randomised controlled trial. Lancet. 2016;387:1743–52.
Montoy JCC, Coralic Z, Herring AA, Clattenburg EJ, Raven MC. Association of default electronic medical record settings with health care professional patterns of opioid prescribing in emergency departments: a randomized quality improvement study. JAMA Intern Med. 2020;180:487–93.
Belli HM, Chokshi SK, Hegde R, Troxel AB, Blecker S, Testa PA, et al. Implementation of a behavioral economics electronic health record (BE-EHR) module to reduce overtreatment of diabetes in older adults. J Gen Intern Med. 2020;35:3254–61.
King D, Jabbar A, Charani E, Bicknell C, Wu Z, Miller G, et al. Redesigning the “choice architecture” of hospital prescription charts: a mixed methods study incorporating in situ simulation testing. BMJ Open. 2014;4: e005473.
Malhotra S, Cheriff AD, Gossey JT, Cole CL, Kaushal R, Ancker JS. Effects of an e-prescribing interface redesign on rates of generic drug prescribing: exploiting default options. J Am Med Inform Assoc. 2016;23:891–8.
Meeker D, Knight TK, Friedberg MW, Linder JA, Goldstein NJ, Fox CR, et al. Nudging guideline-concordant antibiotic prescribing: a randomized clinical trial. JAMA Intern Med. 2014;174:425–31.
Monsen CB, Liao JM, Gaster B, Flynn KJ, Payne TH. The effect of medication cost transparency alerts on prescriber behavior. J Am Med Inform Assoc. 2019;26:920–7.
Musgrove MA, Kenney RM, Kendall RE, Peters M, Tibbetts R, Samuel L, et al. Microbiology comment nudge improves pneumonia prescribing. Open Forum Infect Dis. 2018;5:162.
Korn L, Betsch C, Böhm R, Meier NW. Social nudging: the effect of social feedback interventions on vaccine uptake. Heal Psychol. 2018;37:1045–54.
Schmidtke KA, Nightingale PG, Reeves K, Gallier S, Vlaev I, Watson SI, et al. Randomised controlled trial of a theory-based intervention to prompt front-line staff to take up the seasonal influenza vaccine. BMJ Qual Saf. 2020;29:189–97.
Kiefe CI, Allison JJ, Williams OD, Person SD, Weaver MT, Weissman NW. Improving quality improvement using achievable benchmarks for physician feedback: a randomized controlled trial. JAMA. 2001;285:2871–9.
Lehmann BA, Chapman GB, Franssen FME, Kok G, Ruiter RAC. Changing the default to promote influenza vaccination among health care workers. Vaccine. 2016;34:1389–92.
Matheson EC, Derouin A, Gagliano M, Thompson JA, Blood-Siegfried J. Increasing HPV vaccination series completion rates via text message reminders. J Pediatr Health Care. 2014;28:e35–9.
Regan AK, Bloomfield L, Peters I, Effler PV. Randomized controlled trial of text message reminders for increasing influenza vaccination. Ann Fam Med. 2017;15:507–14.
Chapman GB, Li M, Colby H, Yoon H. Opting in vs opting out of influenza vaccination. JAMA. 2010;304:43–4.
Bradley-Ewing A, Lee BR, Doctor JN, Meredith G, Goggin K, Myers A. A pilot intervention combining assessment and feedback with communication training and behavioral nudges to increase HPV vaccine uptake. Hum Vaccin Immunother. 2022;18:1885968.
Milkman KL, Patel MS, Gandhi L, Graci HN, Gromet DM, Ho H, et al. A megastudy of text-based nudges encouraging patients to get vaccinated at an upcoming doctor’s appointment. Proc Natl Acad Sci U S A. 2021;118: e2101165118.
Kim RH, Day SC, Small DS, Snider CK, Rareshide CAL, Patel MS. Variations in influenza vaccination by clinic appointment time and an active choice intervention in the electronic health record to increase influenza vaccination. JAMA Netw Open. 2018;1: e181770.
Patel MS, Volpp KG, Small DS, Wynne C, Zhu J, Yang L, et al. Using active choice within the electronic health record to increase influenza vaccination rates. J Gen Intern Med. 2017;32:790–5.
Barbaroux A, Benoit L, Raymondie RA, Milhabet I. Nudging health care workers towards a flu shot: reminders are accepted but not necessarily effective: a randomized controlled study among residents in general practice in France. Fam Pract. 2021. https://doi.org/10.1093/fampra/cmab001.
Dai H, Saccardo S, Han MA, Roh L, Raja N, Vangala S, et al. Behavioural nudges increase COVID-19 vaccinations. Nature. 2021;597:404–9.
Fiks AG, Hunter KF, Localio AR, Grundmeier RW, Bryant-Stephens T, Luberti AA, et al. Impact of electronic health record-based alerts on influenza vaccination for children with asthma. Pediatrics. 2009;124:159–69.
Bilger M, Wong TT, Lee JY, Howard KL, Bundoc FG, Lamoureux EL, et al. Using adherence-contingent rebates on chronic disease treatment costs to promote medication adherence: results from a randomized controlled trial. Appl Health Econ Health Policy. 2019;17:841–55.
McCoy SI, Fahey C, Rao A, Kapologwe N, Njau PF, Bautista-Arredondo S. Pilot study of a multi-pronged intervention using social norms and priming to improve adherence to antiretroviral therapy and retention in care among adults living with HIV in Tanzania. PLoS ONE. 2017;12: e0177394.
Reddy A, Huseman TL, Canamucio A, Marcus SC, Asch DA, Volpp K, et al. Patient and partner feedback reports to improve statin medication adherence: a randomized control trial. J Gen Intern Med. 2017;32:256–61.
Garza KB, Owensby JK, Braxton Lloyd K, Wood EA, Hansen RA. Pilot study to test the effectiveness of different financial incentives to improve medication adherence. Ann Pharmacother. 2016;50:32–8.
Riegel B, Stephens-Shields A, Jaskowiak-Barr A, Daus M, Kimmel SE. A behavioral economics-based telehealth intervention to improve aspirin adherence following hospitalization for acute coronary syndrome. Pharmacoepidemiol Drug Saf. 2020;29:513–7.
Luong P, Glorioso TJ, Grunwald GK, Peterson P, Allen LA, Khanna A, et al. Text message medication adherence reminders automated and delivered at scale across two institutions: testing the nudge system: pilot study. Circ Cardiovasc Qual Outcomes. 2021;14: e007015.
McConnell M, Rogers W, Simeonova E, Wilson IB. Architecting process of care: a randomized controlled study evaluating the impact of providing nonadherence information and pharmacist assistance to physicians. Health Serv Res. 2020;55:136–45.
Ingersoll KS, Dillingham RA, Hettema JE, Conaway M, Freeman J, Reynolds G, et al. Pilot RCT of bidirectional text messaging for ART adherence among nonurban substance users with HIV. Heal Psychol. 2015;34S:1305–15.
Hou MY, Hurwitz S, Kavanagh E, Fortin J, Goldberg AB. Using daily text-message reminders to improve adherence with oral contraceptives: a randomized controlled trial. Obstet Gynecol. 2010;116:633–40.
Sutton S, Kinmonth A-L, Hardeman W, Hughes D, Boase S, Prevost AT, et al. Does electronic monitoring influence adherence to medication? Randomized controlled trial of measurement reactivity. Ann Behav Med. 2014;48:293–9.
Our World in Data. Coronavirus (COVID-19) vaccinations. 2021. Available from: https://ourworldindata.org/covid-vaccinations. [Accessed 14 Oct 2021].
World Health Organization (WHO). Antibiotic resistance. 2020. Available from: http://www.who.int/en/news-room/fact-sheets/detail/antibiotic-resistance. [Accessed 14 Oct 2021].
Naderi SH, Bestwick JP, Wald DS. Adherence to drugs that prevent cardiovascular disease: meta-analysis on 376,162 patients. Am J Med. 2012;125:882-7.e1.
Vrijens B, Vincze G, Kristanto P, Urquhart J, Burnier M. Adherence to prescribed antihypertensive drug treatments: longitudinal study of electronically compiled dosing histories. BMJ. 2008;336:1114–7.
Razai MS, Chaudhry UAR, Doerholt K, Bauld L, Majeed A. Covid-19 vaccination hesitancy. BMJ. 2021;373: n1138.
Dylst P, Simoens S. Does the market share of generic medicines influence the price level? Pharmacoeconomics. 2011;29:875–82.
Gulácsi L, Brodszky V, Baji P, Rencz F, Péntek M. The rituximab biosimilar CT-P10 in rheumatology and cancer: a budget impact analysis in 28 European countries. Adv Ther. 2017;34:1128–44.
NICE. NICE recommends several treatment options to help thousands with moderate rheumatoid arthritis. 2021. Available from: https://www.nice.org.uk/news/article/nice-recommends-several-treatment-options-to-help-thousands-with-moderate-rheumatoid-arthritis. [Accessed 10 May 2021].
Simoens S, Vulto AG. A health economic guide to market access of biosimilars. Expert Opin Biol Ther. 2021;21:9–17.
Berg N, Gigerenzer G. As-if behavioral economics: neoclassical economics in disguise. Hist Econ Ideas. 2010;18:133–65.
Madrian BC. Applying insights from behavioral economics to policy design. Annu Rev Econ. 2014;6:663–88.
Vandenplas Y, Simoens S, Van Wilder P, Vulto AG, Huys I. Off-patent biological and biosimilar medicines in Belgium: a market landscape analysis. Front Pharmacol. 2021. https://doi.org/10.3389/fphar.2021.644187.
Li M, Chapman GB. Nudge to health: harnessing decision research to promote health behavior. Soc Personal Psychol Compass. 2013;7:187–98.
IQVIA. Advancing biosimilar sustainability in Europe. 2018. Available from: https://www.iqvia.com/insights/the-iqvia-institute/reports/advancing-biosimilar-sustainability-in-europe. [Accessed 2 Feb 2020].
Vlaev I, King D, Darzi A, Dolan P. Changing health behaviors using financial incentives: a review from behavioral economics. BMC Public Health. 2019;19:1059.
Barbier L, Simoens S, Vulto AG, Huys I. European stakeholder learnings regarding biosimilars: part II: improving biosimilar use in clinical practice. BioDrugs. 2020;34(6):797–808.
Barbier L, Simoens S, Vulto AG, Huys I. European stakeholder learnings regarding biosimilars: part I: improving biosimilar understanding and adoption. BioDrugs. 2020;34:783–96.
Nabhan C, Feinberg BA. Behavioral economics and the future of biosimilars. J Natl Compr Cancer Netw. 2017;15:1449–51.
Barbier L, Vulto AG. Interchangeability of biosimilars: overcoming the final hurdles. Drugs. 2021;81:1897–903.
Lublóy Á. Factors affecting the uptake of new medicines: a systematic literature review. BMC Health Serv Res. 2014;14:469.
Barbier L, Mbuaki A, Simoens S, Vulto A, Huys I. The role of regulatory guidance and information dissemination for biosimilars medicines: the perspective of healthcare professrionals and industry. Value Health. 2019;22:S786–7.
Vandenplas Y, Simoens S, Van Wilder P, Vulto AG, Huys I. Informing patients about biosimilar medicines: the role of European patient associations. Pharmaceuticals (Basel). 2021;14:117.
Gasteiger C, Jones ASK, Kleinstauber M, Lobo M, Horne R, Dalbeth N, et al. The effects of message framing on patients’ perceptions and willingness to change to a biosimilar in a hypothetical drug switch. Arthritis Care Res (Hoboken). 2019. https://doi.org/10.1002/acr.24012.
Johnson EJ, Goldstein D. Do defaults save lives? Science. 2003;302:1338–9.
Oskouei ST, Kusmierczyk AR. Biosimilar uptake: the importance of healthcare provider education. Pharmaceut Med. 2021;35:215–24.
Odum AL. Delay discounting: I’m a k, you’re a k. J Exp Anal Behav. 2011;96:427–39.
Building a wall against biosimilars. Nat Biotechnol. 2013;31:264.
Cohen HP, McCabe D. The importance of countering biosimilar disparagement and misinformation. BioDrugs. 2020. https://doi.org/10.1007/s40259-020-00433-y.
Acknowledgments
The authors express their appreciation towards the Pharmaceutical Policy Department of the Belgian National Institute for Health and Disability Insurance for their financial support in this research project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This research project and manuscript are supported and funded by KU Leuven and the Belgian National Institute for Health and Disability Insurance.
Conflicts of Interest
SS, IH, and AGV have founded the KU Leuven Fund on Market Analysis of Biologics and Biosimilars following Loss of Exclusivity (MABEL). SS was involved in a stakeholder roundtable on biologics and biosimilars sponsored by Amgen, Pfizer, and MSD; he has participated in advisory board meetings for Pfizer, Sandoz, and Amgen; he has contributed to studies on biologics and biosimilars for Hospira (together with AGV and IH), Celltrion, Mundipharma, and Pfizer, and he has had speaking engagements for Amgen, Celltrion, and Sandoz. AGV is involved in consulting, advisory work, and speaking engagements for a number of companies, AbbVie, Accord, Amgen, Biogen, Effik Benelux, Pfizer/Hospira, Fresenius Kabi, Mundipharma, Medicines for Europe, Roche, and Sandoz. FT acted as an advisor to and consultant of several pharmaceutical organizations and has represented pharmaceutical organizations in professional associations. All other authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Availability of Data and Material
All data and material are available in the article and the supplementary material.
Code Availability
Not applicable.
Authors’ Contributions
IH, FT, AGV, SS, and YV developed the idea for and were involved in the design of the study. YV was involved in the data collection and drafted the initial version of the manuscript. IH, FT, AGV, and SS critically reviewed the manuscript. All authors read and approved the final manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Vandenplas, Y., Simoens, S., Turk, F. et al. Applications of Behavioral Economics to Pharmaceutical Policymaking: A Scoping Review with Implications for Best-Value Biological Medicines. Appl Health Econ Health Policy 20, 803–817 (2022). https://doi.org/10.1007/s40258-022-00751-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40258-022-00751-y