Abstract
Collecting Sargassum spp. biomass and using it for the generation of renewable energy is a sustainable approach to mitigate the costs associated to this weed management. The biomethanation of this algal biomass with other organic waste to produce biogas promotes the integrated sustainable management of these materials while generating gaseous fuel for the tourism industry. The purpose of this work is to determine the percentage Sargassum spp. biomass during the anaerobic co-digestion of this brown algae with food waste that results in the best biogas composition, methane yield, production kinetics, and digestate. The biomethanation was conducted in 1200 L fed-batch and bench scale batch biodigestors and the kinetic parameters were estimated using the modified Gompertz model. The methane yield of Sargassum spp. and food waste combinations in 0.58 OLR at fed batch and 15 g/L organic load at batch were comparable. The methane yield produced in the 100% Sargassum spp. fed-batch anaerobic biodigester was 101.3 ± 23.6 N. L CH4/kg, but up to 615.5 ± 78.4 N. L CH4/kg in the 45% Sargassum spp. / 55% food waste biodigestor. The anaerobic co-digestion of Sargassum spp. and food waste in the batch system showed methane production rates as high as 14.6 ± 0.3 N.L CH4/kg.day. Higher H2S were detected in the biogas of the biodigesters fed with larger percentages of the Sargassum spp. with more than 5000 ppm during mono-digestion. Our results suggest that 55% Sargassum spp. and 45% food waste are the most promising feed combination under the studied conditions for the anaerobic co-digestion of these feedstock at larger scale.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Starting in 2011, coastal areas of the Caribbean Sea have experienced high yearly accumulation of pelagic Sargassum spp. The presence of these brown macroalgal mats in the Caribbean coasts is affecting the ecology, human health, and the economy of the countries in that region. The accumulation of this biomass on the beach has been linked to the dead of aquatic organisms and reduction in biodiversity due to the hypoxic conditions, and the high concentrations of ammonium (NH4+) and hydrogen sulfide (H2S) generated during Sargassum spp. decay [1]. Similarly, H2S and NH4+ are known to produce adverse conditions in human beings, such as acute respiratory distress syndrome, respiratory tract injury, acute lung injury, hypoxia, and cyanosis, which affect people living in the coastal areas [2,3,4]. Furthermore, the Sargassum spp. blooms have a negative impact on two of the main economic activities in the Caribbean countries: tourism, and fishing [5, 6]. Some of the negative impact of this weed in the fishery industry includes the obstruction of the operation of fishing boats, and the difficulty of catching some species that are physically protected by the seaweed mat, or that are driven away by it [7]. Similarly, the arrival of this algal biomass to the shores affects tourism by restricting the aesthetics, access, and use of the beaches, and by threatening the wellbeing of tourists that are exposed to the gases emitted during decomposition of the biomass [8]. Collecting the biomass before its arrival at the beach would alleviate their negative effects on the environment, the economy, and human wellbeing [6]. However, harvesting seaweed has been estimated to cost more than 2000 USD per hectare [9]. Using the collected Sargassum spp. biomass as feedstock to produce valuable products could mitigate the costs associated with harvesting.
The Sargassum spp. biomass has been used as feedstock for production of various goods, from furniture and construction materials to biofuels and bioactive compounds containing antioxidant, antifouling, antimicrobial, and/or antitumor properties [10, 11]. One of the most practical and simple applications for the use of this aquatic weed is anaerobic digestion. The energy potential of marine biomass derived from anaerobic digestion is estimated annually to be above 100 EJ, almost 5 times greater than that of the terrestrial biomass, and more than 10 times that of the municipal solid waste [12]. The biogas from the anaerobic digestion can be used as precursor for CHP technologies or transportation fuel, and the solid and liquid residues for energy and agricultural applications [13,14,15]. For instance, only 0.22 ha of grass land is required annually to fuel a car operating on biomethane produced from anaerobic digestion [14]. Experimental and simulated scale up systems based on anaerobic digestion and CHP produced 163.90 kWh and 74.80 kWh of electricity at 500 kg of food waste per day, respectively [13]. Similarly, the biosolids can be used as feedstock for gasification and pyrolysis applications, as well as soil amendment and crop production after being checked for pathogens and heavy metals prior to use [15]. The anaerobic digestion of Sargassum spp. has the potential to generate valuable products within the energy and agricultural industry while mitigating algal pollution on the coasts.
The use of biomass from Sargassum spp. as feedstock for biogas production has been studied by various researchers. For instance, the biochemical methane potential (BMP) of S. fulvellum was 142.91 ± 0.004 mL CH4/g VS after particle-size reduction, and that of S. muticum silage was ≤ 110 mL CH4/g VS [16, 17]. These results show that the BMP of pelagic Sargassum spp. biomass is roughly like other algal species and below invasive aquatic macrophytes. The methane yields of many algae species are typically ~ 200 L CH4/g VS, which is less than 50% of that from the commercially exploited feedstock [12]. For instance, the reported BMP yield for cast brown seaweed was 342 mL CH4/ g VS for Saccharina latissima and 166 mL CH4 g/VS for Ascophyllum nodosum [18]. However, the BMP of Pontederia crassipes, commonly known as water hyacinth, was found to be 399 mL CH4/ g VS [19]. The low BMP of pelagic Sargassum spp. might be due to the presence of inhibitory compounds like ammonia, sulfide, light metal ions, and heavy metals [20]. The inclusion of other feedstock helps to mitigate the inhibition during the anaerobic digestion and improve the process efficiency. For instance, co-digestion of microalgae and microalgae residues resulted in the increase of biodegradability and the reduction of ammonia inhibition risk associated with mono-digestion [21]. Suitable C/N ratio and higher degradation rate of lignocellulose were the result of the addition of cow manure as co-substrate in the anaerobic digestion of oat straw [22]. Similarly, co-digesting beet-molasses syrup residue (BMSr) with manure resulted in the reduction of the sodium and potassium inhibition occurring during the mono-digestion of BMSr [23]. Thus, co-digestion of Sargassum spp. with another feasible waste should be explored to improve the characteristics of the biomass and the biogas produced.
Punta Cana, the most known all-inclusive touristic destination in the Dominican Republic, is affected by Sargassum spp. arrivals to the beaches, but also deals with food waste management. For instance, Sargassum Ocean Sequestration of Carbon (SOS Carbon), a spinoff sargassum-harvesting organization from the Mechanical Engineering department at the Massachusetts Institute of Technology (MIT), received a monthly average of 95 tons of biomass per km of coastline during Summer and Fall 2021. On the other hand, 3 kg of food waste is generated daily per guest at an all-inclusive hotel according to Ecoservices Dominicana, a waste management and recycling company that operates in Bávaro and Punta Cana. Therefore, at 80% capacity, the 45,000 hotel rooms in the area could generate in total more than 200 ton of food waste daily [24]. Food waste disposed to open dumpsites is responsible of at least 6% of the greenhouse gas emissions globally and almost 9% of that generated by food industry [25, 26]. In developed countries like United States, anaerobic digestion is a successful method for the management and transformation of food waste into valuable products at large scale [27]. The anaerobic co-digestion of Sargassum spp. biomass and food waste could be promising in mitigating possible inhibitory compounds and increasing the systems performance. The anaerobic co-digestion of Sargassum spp. biomass and food waste can contribute to the sustainable management of these two unwanted materials in Punta Cana.
The purpose of this paper is to determine the ratio of Sargassum spp. and food waste most suitable for the anaerobic co-digestion of these materials in touristic areas using batch and fed-batch systems at bench and pilot scale, respectively. The BMP and the kinetics of Sargassum spp. biomass and food waste combinations at 5 g/L and 15 g/L loading was determined in the batch system using the modified Gompertz model. The production rate of methane from Sargassum spp. and food waste combinations at 0.7 kg/day was estimated in the fed-batch system. For the first time, the co-digestion of Sargassum spp. biomass and food waste in the Dominican Republic and the Caribbean is demonstrated at pilot scale using commercial 1200 L biodigestors. The results of this work are intended to aid the governmental and the touristic institutions on the decision-making for the sustainable management of the pelagic Sargassum spp. in the countries affected by this weed.
Materials and methods
Sargassum spp. biomass
The biomass was provided by Fundación Grupo Punta Cana. About 10 tons of fresh biomass, mainly S. fluitans and S. natans [28], was collected in the sea using boats equipped with the SOS Carbon technology. The biomass was not washed after collection, because preliminary studies showed no difference (p = 0.98) between the salt yield (g salt/g biomassadded) of Sargassum spp. water extractives at washing periods ranging from 0 to 3.5 h. Similarly, previous work showed no difference between the BMP of wash and unwashed Sargassum spp. biomass [29]. The fresh Sargassum spp. biomass was spread on rubber, plastic, or zinc surfaces in layers below 5 inches thick to air dry under ambient outdoors’ conditions.
Food waste
The food waste on this study was provided by Fundación Grupo Punta Cana and consisted in the lunch’s leftovers of the Punta Cana International Airport’s employees’ restaurant. The lunch menu was not controlled, but consisted in carbohydrates, i.e., rice, legumes, rooted vegetables, and protein, i.e., beef, poultry, fish, and/or pork. The food was processed on the same day prior to storage. The food was classified, milled, packed, and stored before it was used. The non-degradable plastics and inert materials (e.g., seashell, bones) were discarded. The organic portion of the food waste was separated into two groups, protein or meat, and carbohydrates or rice. Daily food packages for the fed-batch and batch biodigestors were prepared containing 60% rice and 40% of milled meat, vacuumed, and stored in a fridge at 4 °C, up to 10 days prior to use.
Mechanical pretreatment
The dried Sargassum spp. biomass and the food waste meat were mechanically pretreated using a shredder–chipper hammer mill (Shreddemin LC 19–20, Santo Domingo, DR), designed and constructed by UNAPEC. The particle size of the dried biomass was reduced to 0.1 inches by the action of 24 free-swinging stainless-steel hammers. The food waste was chipped by a pair of knives at the primary flywheel before going to the shedding chamber to achieve a uniform consistency.
Chemical composition
Samples of Sargassum spp. biomass and food waste taken in 2020 were analyzed for organic matter, fat, fiber, and protein by Anaergia Lab (Treviglio, Italy). Samples of Sargassum spp. taken in 2021 and liquid residue of anaerobic digestion after 60 days of terminated were analyzed for heavy metals, salinity, nitrogen, and phosphorus by LAMENER (Santo Domingo, Dominican Republic). The analysis of the digestate during the anaerobic digestion for the determination of COD, NH3, P, TN, N-NH3, and TSS was conducted by the water laboratory of PQI (Santo Domingo, Dominican Republic).
Pilot scale-fed-batch system
The study was conducted using Homebiogas 2.0 biodigestors (Tel-Aviv, Israel) with 1200L capacity. The biodigesters were activated for 3 weeks with water and cattle manure and acclimatized for 1 month adding the organic load in the feeding profile (Table 1) and cattle manure when pH dropped below 7. The 30-day acclimatization was conducted to account for the anaerobic digestion lag phase [30]. After acclimatization, the daily organic load from the feeding profile (Table 1) was added to the experimental units for an OLR of 0.58 g/L-day on dry weight basis. During the 30-day fed-batch biodigestion, the pH of the experimental units was 7.0 ± 0.2, and the temperature 34.7 ± 3.3 °C. The control biodigestor was fed with cattle manure exclusively and had a pH equal to 7.0 ± 0.1, and temperature of 32.8 ± 2.6 °C throughout the experimental period.
Bench batch system
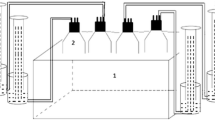
For the bench scale batch system, low-cost anaerobic digestion technology (Biobuckets) was designed and constructed by UNAPEC (Fig. 1). A full factorial experiment was designed to determine the organic load (5 g/L and 15 g/L), and the concentration of Sargassum spp. in the feed (i.e., 100%, 85%, 70%, 55%, and 45%) most suitable during the anaerobic co-digestion of this macroalgae with food waste. The experimental and control (cattle manure) units were prepared in triplicate for a total of 33 measurement units. For the experimental units at 5 g/L and 15 g/L, 35 g, and 105 g dw of feedstock were, respectively, added to 7 L of inoculum in the biobuckets before sealing them. The inoculum was taken from the 1200L biodigester control unit that was fed solely with cattle manure and had a pH of 8.55 ± 0.07, and 4.80 ± 0.41 g/L TDS. The F/I ratio of the systems was roughly 1.0 and 3.1 for the 5 g/L and 15 g/L samples, in that order. The feedstock was Sargassum spp. and food waste at percentages ranging from 45 to 100% w/w of the brown algae. After adding the feedstock to the biobuckets, the pH of the experimental units was in average 8.52 ± 0.11, and the TDS was 4.89 ± 0.40 g/L and 5.55 ± 0.38 g/L in the 5 g/L and 15 g/L units, respectively. During the anaerobic digestion, the volume and composition of the biogas were measured, and the gas storage bag emptied periodically. The anaerobic digestion at bench scale was carried out at ambient conditions and average temperatures of 28.7 ± 0.7 °C during the daytime and 21.2 ± 0.6 °C at night for 60 days.
Process control
The pH, temperature, and total dissolved solids (TDS) of the batch and fed-batch anaerobic digestion were periodically monitored. The pH and temperature were measured using an HM Digital PH-80 (Redondo Beach, CA, USA), and the TDS was measured with an EXTECH ExStik II EC400 Salinometer (Nashua, NH, USA). For the measurement of the composition (CH4, CO2, O2, and H2S) and volume of the biogas, a GEOTECH BIOGAS 5000 (Denver, CO, USA) meter and an Alicat Scientific Whisper mass flow meter (Tucson, AZ, USA) were used, respectively.
Calculations
For estimating the yields of biogas (\({Y}_{Biogas})\) and methane \({Y}_{methane}\) at the pilot scale fed-batch systems, Eqs. 1 and 2 were used. In this formula, the volume (Vi) produced within a short period (ti) is extrapolated considering the total mass digested (mT) throughout the total time \(\left({t}_{T}\right)\) in which the experiment was conducted (30 days). For the batch bench scale system, the measured volume (V) was converted to normal volume (V0) through Eq. 3, where T0 = 273.15 K and P0 = 101,325 Pa. The barometric pressure (P) and temperature (T) during the gas measurements were 101, 384 Pa and 300 K, on average. The daily average of the normalized volume of methane produced by the negative control units during the batch system was subtracted from the daily volume of each experimental unit to account for the methane volume due to inoculum substrate residues
Modified Gompertz model
The modified Gompertz model for the batch anaerobic digestion assumes that methane production follows the microbial growth pattern and is appropriate for batch systems [31]. In the model (Eq. 4), W [N.L CH4/kg VS added] is the accumulated methane produced as a function of time, A [N. L CH4/kg VS added] is the maximum methane produced, Kz [N. L CH4/Kg VS added × day] is the absolute growth rate, and Tlag [days] is the lag time. The doubling time (Td) was calculated from the model. The W(t) curves of each replicate were fitted using the data analysis add on “Solver” in Microsoft Excel 2010. The resulting kinetic parameters of the replicates were analyzed statistically (see Sect. 2.10).
Statistical analysis
The characteristics of the biogas (%CH4, %CO2, and H2S), the liquid media and residue (TS, VS, COD, N, P, TSS, salinity, and minerals), and the kinetic parameters of the Modified Gompertz model for the anaerobic co-digestion of Sargassum spp. food waste at different concentrations of Sargassum spp. were compared using ‘aov’ and ‘TukeyHSD’ functions in RStudio (version 2022.07.1), or the unpaired t test in QuickCalcs (www.graphpad.com). The populations were assumed to be independent, normally distributed, and unequal variances. The variability of the data was reported as the standard deviation of the mean (mean ± SD).
Results
Feedstock composition
Sargassum spp. The Sargassum spp. biomass had 16.1 ± 0.6 w/w % of total solids, of which 69.6 ± 3.4% were volatiles, and 6.9 ± 0.9 g of fat, 10.5 ± 0.6 g of fiber, and 11.6 ± 0.6 g of protein in 100 g on dry weight basis. These values are comparable to those of the pelagic Sargassum spp. from other regions. The macronutrients were 4.8 ± 1.9 g/kg N, 0.56 ± 0.16 g/kg P, and 39.5 ± 3.4 g/kg K. the sodium was 120 ± 18.4 mg/kg Na and the salinity was 13.8 ± 4.4 g/kg. Other minerals present in the biomass were 339.5 ± 147.8 mg/kg Fe, 24.1 0 ± 0.8 mg/kg Zn, 241.0 0 ± 8.5 mg/kg Mn, 18.1 ± 2.3 mg/kg Ni, 0.2 ± 0 mg/kg Pb, 96.4 ± 13.6 mg/kg Co, 58.9 ± 0.4 mg/kg S, and 7.6 ± 1.1 g/kg B. However, minerals below the limit of detection were Cr (< 23.5), Cu (< 23.5), As, (< 1.2), Ba (< 46.9), Cd (< 11.7), Mo (< 20.0), and Se (< 20.0) in mg/kg.
Food waste. The food waste biomass had ~ 13 w/w % of total solids, of which 84% were volatiles. The waste had ~ 21.5 g of fat, ~ 3.8 g of fiber, and ~ 33.8 g of protein in 100 g of biomass.
Biogas composition
Fed-batch system. The percentage of methane in the biogas generated from the anaerobic digestion of manure (56.6 ± 3.1% CH4) was significantly higher (p < 0.0290) than those from the Sargassum spp. mono-digestion and co-digestion of Sargassum spp. and food-waste (see Table 2). In contrast, the H2S of the control (57.0 ± 27.3 ppm) was significantly lower (p < 0.0001) than the experimental units. There is a positive correlation between the concentration of H2S in the biogas and the percentage of Sargassum spp. in the organic load added to the biodigestors.
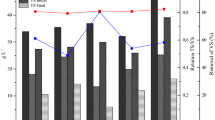
Batch system. The methane percentage (% CH4) in the biogas generated in the batch system was higher (p < 0.001) at 15 g/L (61.12 ± 7.8%) than that at 5 g/L (51.02 ± 9.6%), and 30.9 ± 9.4 to 63.8 ± 4.0 at (Table 2). However, the methane percentage in the biogas generated during the anaerobic digestion of 100% Sargassum spp. at 5 g/L (38.92 ± 7.9) and 15 g/L (36.81 ± 5.6) was not significantly different (p = 0.875) and lower (p < 0.001) than the biogas generated during co-digestion with food waste. There is a trend of reduction in the methane percentage in the biogas with the increase of the Sargassum spp. concentration in the feed during the co-digestion with food waste (Fig. 2A). Similarly, at lower Sargassum spp. percentage in the feed, the maximum %CH4 in the biogas occurred at digestion range 20–35 days, and at 100% Sargassum spp., the %CH4 increased with time, reaching the maximum peak at 60 days (Fig. 2B). On the other hand, the concentration of hydrogen sulfide in the gas generated from batch system does not show correlation with the Sargassum spp. percentage in the feed but does with the organic loading. The highest organic load (15 g/L) used on the anaerobic co-digestion of food waste and Sargassum spp. generated more H2S than that at 5 g/L (Table 2).
Interaction plots of the methane percentage in the biogas (%CH4) generated during the co-digestion of Sargassum spp. with food waste at different concentrations in batch system at bench scale. On the top (A) is the interaction plot of the organic load and the Sargassum spp. % in the feed. On the bottom (B), the behavior of %CH4 throughout the digestion time (60 days) at different Sargassum spp. % in the feed. The plots were extracted from R-Studio
Characteristics of biodigestion fed-batch media
During the 30-day anaerobic co-digestion of Sargassum spp. and food waste, the COD of the media in the inlet (3506 ± 389 ppm) was slightly higher than (p = 0.049) in the outlet (2510 ± 338 ppm). This difference represents a 28.4 ± 9.0% reduction in COD due to the HRT (0.58 g/L · day) in the biodigester. There was not significant difference (p = 0.8575) between the BOD’s inlet (29–171 ppm) and outlet (14–114 ppm) because of the percentage of Sargassum spp. in the organic load. The BOD/COD ratio is below 0.09. The biodigesters media have 150 ± 28 ppm P, 378–1418 ppm N, and 164–996 ppm N-NH3. As expected, N and NH3 tend to reduce at higher Sargassum spp. percentage in the feed, since the protein content is higher in the food waste than in the Sargassum spp. (p = 0.012, see Sect. 3.1).
Composition of fed-batch digestates
After 30 days of maturation, the liquid residue derived from the anaerobic digestion of Sargassum spp. and co-digestion of Sargassum spp. and food waste had 9.6 ± 1.6 g/L of total solids which only 6.5% (0.63 ± 0.13 g/L) were volatile solids. The total solids and the salinity (8.4 ± 1.7 g/L) in the liquid residue tended to decrease with the increase of Sargassum spp. percentage in the biodigesters feed (Fig. 3A). Regarding macronutrients (Fig. 3B), there was no correlation between Sargassum spp. concentration and phosphorus content (27.2 ± 4.8 mg/L P); however, the content of total nitrogen (47–1696 mg/L N) and potassium (648–2119 mg/L K) in the liquid residue tended to be higher when the percentage of Sargassum spp. was lower in the biodigesters feed load. Similarly, a negative correlation was observed between the content of Sargassum spp. in the biodigesters feed, and the content of copper (0.09–0.33 mg/L Cu), chromium (0.02—0.15 mg/L Cr), aluminum (0.018–0.337 mg/L Al), boron (1.3–3.0 mg/L B), and sulfur (0.73–2.6 mg/L S) in the liquid residue of the anaerobic co-digestion of Sargassum spp. and food waste (Fig. 3C, D).
Trends of the total solids, salinity, macronutrients (N, K, P), and minerals (Cr, Al, Cu, Bo, S) of the liquid residue derived from the anaerobic co-digestion of Sargassum spp. biomass and food waste as function of the algal biomass percentage in the feed. The linear regression lines are shown in the plots
Biogas and biomethane yield
The biogas and methane yields during the 1200 L fed-batch and 7 L batch anaerobic co-digestion of Sargassum spp. and food waste, differed based on the percentage of Sargassum spp. in the feed (Table 3). The biogas yield in the batch and fed-batch systems ranged from 100 to 1200 N.L/kg, while the methane yield ranged from 50 to 615 N.L CH4/kg. The biodigester fed with 100% Sargassum spp. produced 101.3 ± 23.6 N. L CH4/kg in the fed-batch system. In the 7L batch, the methane yields were larger than (p < 0.0001) in the 1200 L fed-batch system (Fig. 4). However, in both systems, methane increased when higher percentages of food waste and lower percentages of Sargassum spp. biomass were added to the biodigestors.
Biomethanation kinetics
According to the modified Gompertz model fitted to the batch system, the higher the Sargassum spp. content in the organic feed, the lower the estimated maximum methane produced (A) see Table 4. The methane yield of each experimental unit in batch system (Table 3) is not statistically different (p > 0.05) to the estimated equivalent parameter, A (Table 4). Similarly, the methane production rate (Kz) of the system fed with 45%–70% of Sargassum spp. and 55%—30% of food waste was 13.7 ± 2.3 N.L CH4/kg • day in average (p > 0.1016), and higher than (p > 0.05) that fed with 85% Sargassum spp. and 15% food waste. The systems doubling time was below 3 days. The R2 is above 0.96 and the RSME around 5% of the maximum methane produced except for the experimental unit fed with 85% Sargassum spp.
Discussion
Feedstock composition and co-digestion
The chemical and elemental composition of Sargassum spp. from Punta Cana coasts show similarities and discrepancies with previous results from pelagic Sargassum spp. of other areas, which confirms the spatial and temporal variability on the characteristics of the biomass [32]. For instance, the protein content in our feedstock was comparable to that harvested in Mexico and the Caribbean, and the lipids were like the biomass from Turks and Caicos Islands, but it had three times lower fiber content than the Sargassum spp. from Punta Cana [33, 34]. Regarding toxic elements, our results show that the Sargassum spp. collected from Punta Cana during Summer in 2021 did not have detectable arsenic (As) or cadmium (Cd), in discrepancy with results published previously by other authors [32].
Having the Sargassum spp. as part of a feedstock matrix with food waste can provide the versatility required for feedstocks that are not stable on their composition. Higher protein and fat content in food waste compared to Sargassum spp. biomass enriches the media for more balanced C/N in a carbohydrate-rich feedstock like Sargassum spp. and the separation of carbohydrate-rich from protein-rich food waste can add flexibility to the system to provide with the biodigesters feed appropriate to enhance the methanogenesis process.
Fed-batch media and digestates
When conducting the anaerobic co-digestion of Sargassum spp. and food waste under the study conditions, the digestates will require maturation before use on agricultural applications. During the 30 days of the experiment, the digestates that were collected from the biodigestors showed less than 30% of reduction in COD and no reduction in BOD compared to the inlet (Sect. 3.3), which suggest incompletion in the biodigestion process. Similarly, COD was more than 20 times higher than the BOD. A BOD5/COD ratio of less than 0.3 corresponds to the low biodegradability of organic material in wastewater [35]. After 30 days of maturation, the digestates showed higher salinity, N, K, Cr, Al, Cu, Bo, and S when feed at higher food waste and lower Sargassum spp. percentages, which indicates that these elements are present in the food waste and are minimal in Sargassum spp. biomass. Thus, when the biofertilizer is desired to be used on agricultural land requiring high N, and K, adding food waste to the anaerobic digestion system could make it possible.
Regarding the salinity of the digestate or liquid residue, it could be inferred that the exposure of the Sargassum spp. to the rain during the drying period could have reduced the salinity that is conventionally associated with this biomass. On the other hand, the lower the Sargassum spp. percentage, the higher the content of typical food waste, which has high NaCl concentration. The anaerobic digestion of Sargassum spp. biomass has resulted in improvement of the methane production rate at 4.4 g/L of sodium salts but inhibition at concentrations above 10 g/L[36]. This might suggest that the sodium salt content due to the addition of Sargassum spp. is minimum compared to the food waste contributions, and the Sargassum spp. inhibition on the biodigestion might be due to the presence of elements different from sodium salts or the heavy metals in the biomass.
Biomethane yield and kinetics
The anaerobic co-digestion of Sargassum spp. and food waste showed best methane yield and methane production rate at lower Sargassum spp. and higher food waste percentages in the feed. However, as a proposed solution to the problem of Sargassum spp. arrival in the coasts of the Dominican Republic, the maximum possible percentage of Sargassum spp. in the feed added to an anaerobic digestion matrix is desirable. Since 45% and 55% of Sargassum spp. in the feed showed no significant difference in the methane yield (Table 3) and the kinetics parameters including methane rate (Kz, Table 4), the use of 55% of Sargassum spp. and 45% of food waste is preferable during high Sargassum spp. seasons and the former during low Sargassum spp. seasons.
Biogas characteristics
Our study showed that increasing the Sargassum spp. percentage on the load at fed-batch and the organic load from 5 to 15 g/L at the batch mode can rise the amount of H2S present in the biogas generated. This is a relevant point to consider when using Sargassum spp. as feedstock for biogas production. The use of techniques and methods for the input control, process regulation, and post-treatment should be considered. For instance, chemical pretreatment of substrate reduces sulfur input into AD via sulfur precipitation, microaeration can regulate AD to control H2S formation, and biotrickling filters and scrubbers can remove more than 95% of H2S but are not effective on preventing sulfur inhibition of methanogens [37]. Pretreatment of the biomass prior to its used is an approach that might be beneficial to increase the biodegradability of Sargassum spp. and reduce the presence of H2S in the biogas.
The biogas is above 50% CH4 for all the experimental units except for the biodigestor fed with 100% Sargassum spp. which did not reach 50% CH4 during 60 days of digestion. This suggests low conversion of acetic acid and CO2 by the methanogens because of a possible inhibition due to Sargassum spp. Unless pretreatment of Sargassum spp. is attained with the purpose of increasing the % CH4 in the biogas, the mono-digestion of Sargassum spp. should be avoided. For instance, when internal combustion engines fueled with biogas containing high CO2 were used instead of that with lower CO2, the flame initiation and combustion duration increased, and the thermal efficiency decreased [38]. Studies on how to use efficiently biogas from Sargassum spp. and food waste anaerobic co-digestion in internal combustion engines are some of the next steps that the authors are planning to attain with the collaboration of our partners in industry.
Conclusions
The anaerobic co-digestion of Sargassum spp. and food waste at pilot scale in fed-batch and bench scale in batch mode were demonstrated at different Sargassum spp. to feedstock ratio. The biogas and methane production tended to increase the higher the food waste and the lower the Sargassum spp. percentages in the feed. The biogas yield in the 100% Sargassum spp. fed-batch anaerobic biodigestors was 101.3 ± 23.6 N. L /kg and as high as 1110 ± 134.7 N. L /kg when fed with 45% Sargassum spp. and 55% food waste. Similarly, more H2S was detected in the biogas of the biodigesters fed with higher percentages of the Sargassum spp. The BMP of Sargassum spp. and food waste combinations in batch system at 15 g/L loading were aligned to the fed-batch results, with values of 536.1 ± 58.9 N.L CH4/kg feed, 411.6 ± 14.4 N.L CH4/kg feed, 275.0 ± 49.7 N.L CH4/kg feed, and 161.6 ± 32.8 N.L CH4/kg feed at 45%, 55%, 70%, and 85% of Sargassum spp. as food waste co-substrate, respectively. The anaerobic co-digestion of Sargassum spp. and food waste at batch system showed methane production rates as high as 14.6 ± 0.3 N.L CH4/kg.day according to the modified Gompertz model. Our results suggest that the percentage of Sargassum spp. to be used as feedstock for the anaerobic co-digestion must be 55% w/w. To use higher Sargassum spp. percentages, the use pretreatments techniques to enhance the biomethanation process should be explored.
References
Rodríguez-Martínez, R., Medina-Valmaseda, A., Blanchon, P., Monroy-Velázquez, L., Almazán-Becerril, A., Delgado-Pech, B., Vásquez-Yeomans, L., Francisco, V., García-Rivas, M.: Faunal mortality associated with massive beaching and decomposition of pelagic Sargassum. Mar. Pollut. Bull. 146, 201–205 (2019)
Padappayil, R., Borger, J.: Ammonia toxicity. StatPearls Publishing, Treasure Island (2021)
George, A., Goetz, D.: A case of sulfhemoglobinemia in a child with chronic constipation. Respir. Med. case reports. 21, 21–24 (2017). https://doi.org/10.1016/j.rmcr.2017.03.009
Mousa, H.A.L.: Short-term effects of subchronic low-level hydrogen sulfide exposure on oil field workers. Environ. Health Prev. Med. 20, 12–17 (2015). https://doi.org/10.1007/s12199-014-0415-5
Semeleer, E., Lamers, M., Reviewer, S., Student, J.: Framing environmental change and adaptation governance : The case of Sargassum seaweed in the Caribbean, (2021)
Hu, C., Murch, B., Barnes, B., Wang, M., Maréchal, J., Franks, J., Johnson, D., Lapointe, B., Goodwin, D., Schell, J.: Sargassum watch warns of incoming seaweed, (2016)
Ramlogan, N.R., Mcconney, P., Oxenford, H.A.: Socio-economic impacts of Sargassum influx events on the fishery sector of Barbados. CERMES Technical Report No 81. (2017)
Taylor, A.: Sargassum is strangling tourism in the Caribbean. Can scientists find a use for it? C&EN (Chemical Eng. News). 97, (2019)
van den Burg, S.W.K., van Duijn, A.P., Bartelings, H., van Krimpen, M.M., Poelman, M.: The economic feasibility of seaweed production in the North Sea. Aquac. Econ. Manag. 20, 235–252 (2016). https://doi.org/10.1080/13657305.2016.1177859
Pinteus, S., Lemos, M.F.L., Alves, C., Neugebauer, A., Silva, J., Thomas, O.P., Botana, L.M., Gaspar, H., Pedrosa, R.: Marine invasive macroalgae: Turning a real threat into a major opportunity—the biotechnological potential of Sargassum muticum and Asparagopsis armata. Algal Res. 34, 217–234 (2018). https://doi.org/10.1016/j.algal.2018.06.018
Desrochers, A., Cox, S.-A., Oxenford, H.A., van Tussenbroek, B.: Sargassum Uses Guide: A resource for caribbean researchers, entrepreneurs and policy makers. (2020)
Milledge, J.J., Nielsen, B. V, Maneein, S., Harvey, P.J.: A brief review of anaerobic digestion of algae for bioenergy, (2019)
Zhang, J., Gu, D., Chen, J., He, Y., Dai, Y., Loh, K.-C., Tong, Y.W.: Assessment and optimization of a decentralized food-waste-to-energy system with anaerobic digestion and CHP for energy utilization. Energy Convers. Manag. 228, 113654 (2021). https://doi.org/10.1016/j.enconman.2020.113654
Goulding, D., Power, N.: Which is the preferable biogas utilisation technology for anaerobic digestion of agricultural crops in Ireland: Biogas to CHP or biomethane as a transport fuel? Renew. Energy. 53, 121–131 (2013). https://doi.org/10.1016/j.renene.2012.11.001
Kumar, V., Chopra, A.K., Kumar, A.: A review on sewage sludge (biosolids) a resource for sustainable agriculture. Arch. Agric. Environ. Sci. 2, 340–347 (2017). https://doi.org/10.26832/24566632.2017.020417
Milledge, J.J., Harvey, P.J.: Ensilage and anaerobic digestion of Sargassum muticum. J. Appl. Phycol. 28, 3021–3030 (2016). https://doi.org/10.1007/s10811-016-0804-9
Farghali, M., Mohamed, I.M.A., Iwasaki, M., Tangtaweewipat, S., Ihara, I., Sakai, R., Umetsu, K.: Potential of biogas production from the anaerobic digestion of Sargassum fulvellum macroalgae: Influences of mechanical, chemical, and biological pretreatments. Biochem. Eng. J. 175, 108140 (2021). https://doi.org/10.1016/j.bej.2021.108140
Allen, E., Wall, D., Herrmann, C., Xia, A., Murphy, J.: What is the gross energy yield of third generation gaseous biofuel sourced from seaweed? Energy (2015). https://doi.org/10.1016/j.energy.2014.12.048
Castro, Y.A., Agblevor, F.A.: Biomethanation of invasive water hyacinth from eutrophic waters as a post weed management practice in the Dominican Republic, a developing country. Environ. Sci. Pollut. Res. 27, 14138–14149 (2020). https://doi.org/10.1007/s11356-020-07927-w
Chen, Y., Cheng, J.J., Creamer, K.S.: Inhibition of anaerobic digestion process: A review. Bioresour. Technol. 99, 4044–4064 (2008). https://doi.org/10.1016/j.biortech.2007.01.057
Solé-Bundó, M., Passos, F., Romero-Güiza, M.S., Ferrer, I., Astals, S.: Co-digestion strategies to enhance microalgae anaerobic digestion: A review. Renew. Sustain. Energy Rev. 112, 471–482 (2019). https://doi.org/10.1016/j.rser.2019.05.036
Zhao, Y., Sun, F., Yu, J., Cai, Y., Luo, X., Cui, Z., Hu, Y., Wang, X.: Co-digestion of oat straw and cow manure during anaerobic digestion: Stimulative and inhibitory effects on fermentation. Bioresour. Technol. 269, 143–152 (2018). https://doi.org/10.1016/j.biortech.2018.08.040
Fang, C., Boe, K., Angelidaki, I.: Anaerobic co-digestion of desugared molasses with cow manure; focusing on sodium and potassium inhibition. Bioresour. Technol. 102, 1005–1011 (2011). https://doi.org/10.1016/j.biortech.2010.09.077
Lopez, A.: Hotel rooms in the Dominican Republic 2019, by region, https://www.statista.com/statistics/1039083/dominican-republic-hotel-rooms-province/#statisticContainer
Ritchie, H.: Food waste is responsible for 6% of global greenhouse gas emissions, https://ourworldindata.org/food-waste-emissions
Tubiello, F.N., Rosenzweig, C., Conchedda, G., Karl, K., Gütschow, J., Xueyao, P., Obli-Laryea, G., Wanner, N., Qiu, S.Y., Barros, J.D., Flammini, A., Mencos-Contreras, E., Souza, L., Quadrelli, R., Heioarsdóttir, H.H., Benoit, P., Hayek, M., Sandalow, D.: Greenhouse gas emissions from food systems: Building the evidence base. Environ. Res. Lett. (2021). https://doi.org/10.1088/1748-9326/ac018e
Dalke, R., Demro, D., Khalid, Y., Wu, H., Urgun-Demirtas, M.: Current status of anaerobic digestion of food waste in the United States. Renew. Sustain. Energy Rev. 151, 111554 (2021). https://doi.org/10.1016/j.rser.2021.111554
Mengqiu, W., Chuanmin, H., Gary, M., Brian, L.: The great Atlantic Sargassum belt. Science 365, 83–87 (2019). https://doi.org/10.1126/science.aaw7912
Milledge, J.J., Nielsen, B.V., Sadek, M.S., Harvey, P.J.: Effect of freshwaterwashing pretreatment on sargassum muticum as a feedstock for biogas production. Energies (2018). https://doi.org/10.3390/en11071771
Castro, Y.A., Agblevor, F.A.: Interaction effect of high feed to inoculum ratio (F/I) and temperature on the biomethanation kinetics of water hyacinth. SN Appl. Sci. 2, 1–9 (2020). https://doi.org/10.1007/s42452-020-03626-w
Kafle, G.K., Chen, L.: Comparison on batch anaerobic digestion of five different livestock manures and prediction of biochemical methane potential (BMP) using different statistical models. Waste Manag. 48, 492–502 (2016). https://doi.org/10.1016/j.wasman.2015.10.021
Lopresto, C.G., Paletta, R., Filippelli, P., Galluccio, L., de la Rosa, C., Amaro, E., Jáuregui-Haza, U., de Frias, J.A.: Sargassum invasion in the caribbean: an opportunity for coastal communities to produce bioenergy based on biorefinery—an overview. Waste Biomass Valoriz. (2022). https://doi.org/10.1007/s12649-021-01669-7
Milledge, J.J., Maneein, S., Arribas-López, E., Bartlett, D.: Sargassum inundations in Turks and Caicos : methane. Energies 13, 1–27 (2020)
Vázquez-Delfín, E., Freile-Pelegrín, Y., Salazar-Garibay, A., Serviere-Zaragoza, E., Méndez-Rodríguez, L.C., Robledo, D.: Species composition and chemical characterization of Sargassum influx at six different locations along the Mexican Caribbean coast. Sci. Total Environ. 795, 148852 (2021). https://doi.org/10.1016/j.scitotenv.2021.148852
Dinçer, A.R.: Increasing BOD5/COD ratio of non-biodegradable compound (reactive black 5) with ozone and catalase enzyme combination. SN Appl. Sci. 2, 1–10 (2020). https://doi.org/10.1007/s42452-020-2557-y
Zhang, Y., Li, L., Kong, X., Zhen, F., Wang, Z., Sun, Y., Dong, P., Lv, P.: Inhibition effect of sodium concentrations on the anaerobic digestion performance of sargassum species. Energy Fuels 31, 7101–7109 (2017). https://doi.org/10.1021/acs.energyfuels.7b00557
Vu, H.P., Nguyen, L.N., Wang, Q., Ngo, H.H., Liu, Q., Zhang, X., Nghiem, L.D.: Hydrogen sulphide management in anaerobic digestion: A critical review on input control, process regulation, and post-treatment. Bioresour. Technol. 346, 126634 (2022). https://doi.org/10.1016/j.biortech.2021.126634
Gupta, S.K., Mittal, M.: Effect of biogas composition variations on engine characteristics including operational limits of a spark-ignition engine. J. Eng. Gas Turbines Power. (2019). https://doi.org/10.1115/1.4044195
Acknowledgements
The equipment used to conduct this research was donated to UNAPEC by Y.A. MAOF. The authors acknowledge the collaboration and support of Fundación Grupo Punta Cana (FGPC) during the execution of this work. We acknowledge PQI lab (Santo Domingo, DR) for their contribution on the analyses of laboratory. We are especially thankful to Cesar Pucheu, Operative Manager of Ecoservices, and Andrés Bisonó, CEO of SOS Carbon, for providing us with data related to the management activities of food waste and Sargassum spp. We acknowledge the work of the co-author Emin Rivera in the design and construction of the hammer mill Shreddemin LC 19-20, which made possible the execution of the experimental work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Castro, Y.A., Rodríguez, A. & Rivera, E. Biomethane production kinetics during the anaerobic co-digestion of Sargassum spp. and food waste using batch and fed-batch systems in Punta Cana, Dominican Republic. Mater Renew Sustain Energy 11, 287–297 (2022). https://doi.org/10.1007/s40243-022-00224-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40243-022-00224-1