Abstract
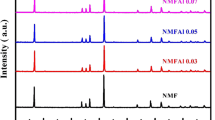
P2-type layered oxide Na0.67Fe0.5Mn0.5O2 is recognized as a very promising cathode material for sodium-ion batteries due to the merits of high capacity, high voltage, low cost, and easy preparation. However, its unsatisfactory cycle and rate performances remain huge obstacles for practical applications. Here, we report a strategy of SnO2 modification on P2-type Na0.67Fe0.5Mn0.5O2 to improve the cycle and rate performance. Scanning electron microscope(SEM) and transmission electron microscope(TEM) images indicate that an insular thin layer SnO2 is coated on the surface of Na0.67Fe0.5Mn0.5O2 after medication. The coating layer of SnO2 can protect Na0.67Fe0.5Mn0.5O2 from corrosion by electrolyte and the cycle performance is well enhanced. After 100 cycles at 1 C rate(1 C=200 mA/g), the capacity of SnO2 modified Na0.67Fe0.5Mn0.5O2 retains 83 mA·h/g(64% to the initial capacity), while the capacity for the pristine Na0.67Fe0.5Mn0.5O2 is only 38 mA·h/g(33.5% to the initial capacity). X-Ray photoelectron spectroscopy reveals that the ratio of Mn4+ increases after SnO2 modification, leading to less oxygen vacancy and expanded lattice. As a result, the capacity of Na0.67Fe0.5Mn0.5O2 increases from 178 mA·h/g to 197 mA·h/g after SnO2 modification. Furthermore, the rate performance of Na0.67Fe0.5Mn0.5O2 is enhanced with SnO2 coating, due to high electronic conductivity of SnO2 and expanded lattice after SnO2 coating. The capacity of SnO2 modified Na0.67Fe0.5Mn0.5O2 at 5 C increases from 21 mA·h/g(pristine Na0.67Fe0.5Mn0.5O2) to 35 mA·h/g.
Similar content being viewed by others
References
Crabtree G., Kócs E., Trahey L., Materials Research Society, 2015, 40, 1067
Etacheri V., Marom R., Elazari R., Salitra G., Aurbach D., Energy & Environmental Science, 2011, 4, 12
Wei L., Zhao S. X., Wu X., Zhao S. J., Nan C. W., Journal of Materiomics, 2018, 4, 179
Lei P., Liu K., Wan X., Luo D., Xiang X., Chemical Communications, 2019, 55, 509
Hwang J. Y., Myung S. T., Sun Y. K., Chemical Society Reviews, 2017, 46, 3529
Lee W., Muhammad S., Sergey C., Lee H., Yoon J., Kang Y. M., Yoon W. S., Angewandte Chemie International Edition, 2020, 59, 2578
Zheng X., Liu W., Qu Q., Zheng H., Huang Y., Journal of Materiomics, 2019, 5, 156
Zhao J., Zhang X., Wang J., Yang X., Deng J., Wang Y., Journal of Solid State Electrochemistry, 2020, 24, 1349
Ma P., Kang W., Wang Y., Cao D., Fan L., Sun D., Applied Surface Science, 2020, 5, 29
Zhao L. N., Zhang T., Zhao H. L., Hou Y. L., Materials Today Nano, 2020, 10, 439
Lu Y., Wang L., Cheng J., Goodenough J. B., Chemical Communications, 2012, 48, 6544
Jiang W., Qi W., Pan Q., Jia Q., Yang C., Cao B., International Journal of Hydrogen Energy, 2021, 46, 4252
Zhao Y., Fu Q., Wang D., Pang Q., Gao Y., Missiul A., Nemausat R., Sarapulova A., Ehrenberg H., Wei Y., Chen G., Energy Storage Materials, 2019, 18, 51
Chen M., Liu Q., Wang S. W., Wang E., Guo X., Chou S. L., Advanced Energy Materials, 2019, 9, 14
Lei Y.-J., Yan Z.-C., Lai W.-H., Chou S.-L., Wang Y.-X., Liu H.-K., Dou S.-X., Electrochemical Energy Reviews, 2020, 3, 766
Yabuuchi N., Kajiyama M., Iwatate J., Nishikawa H., Hitomi S., Okuyama R., Usui R., Yamada Y., Komaba S., Nature Materials, 2012, 11, 512
Li M., Wood D. L., Bai Y., Essehli R., Amin M. R., Jafta C., Muralidharan N., Li J., Belharouak I., ACS Appllied Materials Interfaces, 2020, 12, 23951
Sun W., Tang X., Wang Y., Electrochemical Energy Reviews, 2019, 3, 127
Yi T. F., Li Y., Fang Z., Cui P., Luo S., Xie Y., Journal of Materiomics, 2020, 6, 33
Fu J., Huang H., Shi K., Chen F., Yang Z., Zhang W., Electrochimica Acta, 2020, 3, 49
Chen T., Liu W., Zhuo Y., Hu H., Zhu M., Cai R., Chen X., Yan J., Liu K., Journal of Energy Chemistry, 2020, 43, 148
Han M. H., Gonzalo E., Sharma N., del Amo J. M. L., Armand M., Avdeev M., Garitaonandia J. J. S., Rojo T., Chemistry of Materials, 2015, 28, 106
Wang H., Gao R., Li Z., Sun L., Hu Z., Liu X., Inorganic Chemistry, 2018, 57, 5249
Chu S., Chen Y., Wang J., Dai J., Liao K., Zhou W., Shao Z., Composites Part B, 2019, 77, 383
Kong W., Wang H., Zhai Y., Sun L., Liu X., Journal of Physical Chemistry C, 2018, 122, 25909
Kong W., Wang H., Sun L., Su C., Liu X., Applied Surface Science, 2019, 4, 97
Joshua J. R., Lee Y. S., Maiyalagan T., Nallamuthu N., Yuvraj P., Sivakumar N., Journal of Electroanalytical Chemistry, 2020, 8, 56
Yu Y., Kong W., Li Q., Ning D., Schuck G., Schumacher G., Su C., Liu X., ACS Applied Energy Materials, 2020, 3, 933
Sun H. H., Hwang J. Y., Yoon C. S., Heller A., Mullins C. B., ACS Nano, 2018, 12, 12912
Zhang Y., Pei Y., Liu W., Zhang S., Xie J., Xia J., Nie S., Liu L., Wang X., Chemical Engineering Journal, 2020, 3, 82
Aragón M. J., Lavela P., Ortiz G., Alcántara R., Tirado J. L., Journal of Alloys and Compounds, 2017, 724, 465
Kalluri S., Seng K. H., Pang W. K., Guo Z., Chen Z., Liu H. K., Dou S. X., ACS Applied Materials & Interfaces, 2014, 68, 953
Kalluri S., Yoon M., Jo M., Park S., Myeong S., Kim J., Dou S. X., Guo Z., Cho J., Advanced Energy Materials, 2017, 7, 245
Idris M. S., Osman R. A. M., Advanced Materials Research, 2013, 795, 479
Wang F., Zhang Y., Zou J., Liu W., Chen Y., Journal of Alloys and Compounds, 2013, 55, 8172
Guignard M., Didier C., Darriet J., Bordet P., Elkaim E., Delmas C., Nature Materials, 2013, 12, 74
Zhao D., Clites M., Ying G., Kota S., Wang J., Natu V., Wang X., Pomerantseva E., Cao M., Barsoum M. W., Chemical Communications, 2018, 54, 4533
Berthelot R., Carlier D., Delmas C., Nature Materials, 2011, 10, 74
Zheng L., Li J., Obrovac M. N., Chemistry of Materials, 2017, 29, 1623
Dang R., Li Q., Chen M., Hu Z., Xiao X., Physical Chemistry Chemical Physics, 2018, 21, 314
Mo Y., Ong S. P., Ceder G., Chemistry of Materials, 2014, 26, 5208
Lan T., Wei W., Xiao S., He G., Hong J., Journal of Materials Science: Materials in Electronics, 2020, 31, 9423
Li B., Wang J., Cao Z., Zhang P., Zhao J., Journal of Power Sources, 2016, 325, 84
Zhang X., Xu G., Hu J., Lv J., Wang J., Wu Y., Royal Society of Chemistry Advances, 2016, 6, 63241
Zhao Y., Sun Y., Yue Y., Hu X., Xia M., Electrochimica Acta, 2014, 130, 66
Feng X. Y., Shen C., Fang X., Chen C. H., Journal of Alloys and Compounds, 2011, 50, 3623
Luo Z. M., Sun Y. G., Liu H. Y., Chinese Chemical Letters, 2015, 26, 1403
Wu X., Wang S., Lin X., Zhong G., Gong Z., Yang Y., Journal of Materials Chemistry A, 2014, 2, 1006
Hu G. R., Cao J. C., Peng Z. D., Cao Y. B., Du K., Electrochimica Acta, 2014, 149, 49
Chu S., Wei S., Chen Y., Cai R., Liao K., Zhou W., Shao Z., Ceramics International, 2018, 44, 5184
Li Z. Y., Zhang J., Gao R., Zhang H., Hu Z., Liu X., ACS Appllied Materias Interfaces, 2016, 8, 15439
Bai Y., Zhao L., Wu C., Li H., Li Y., Wu F., ACS Appllied Materrials Interfaces, 2016, 8, 2857
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No.21676067), the Natural Science Foundation of Anhui Province, China(No. 1908085QE178) and the Fundamental Research Funds for the Central Universities, China.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare no conflicts of interest.
Electronic Supplementary Material
40242_2021_1287_MOESM1_ESM.pdf
Enhanced electrochemical performance of Na0.67Fe0.5Mn0.5O2 cathode for sodium ion battery by the combination strategy of Sn4+ doping and SnO2 coating
Rights and permissions
About this article
Cite this article
Ye, P., Liu, Y., Ma, J. et al. Enhanced Electrochemical Performance of Na0.67Fe0.5Mn0.5O2 Cathode with SnO2 Modification. Chem. Res. Chin. Univ. 37, 1130–1136 (2021). https://doi.org/10.1007/s40242-021-1287-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-021-1287-z