Abstract
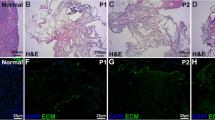
The present study makes assessments by analyzing the efficacy of combined decellularization protocol for human ovarian fragments. Tissues were decellularized by freeze–thaw cycles, and treated with Triton X-100 and four concentrations (0.1, 0.5, 1 and 1.5%) of sodium dodecyl sulfate (SDS) at two exposure times. The morphology and DNA content of decellularized tissues were analyzed, and the group with better morphology and lower DNA content was selected for further assessments. The Acridine orange, Masson’s trichrome, Alcian blue, and Periodic Acid-Schiff staining were used for extracellular matrix (ECM) evaluation. The amount of collagen types I and IV, glycosaminoglycans (GAGs), and elastin was quantified by Raman spectroscopy. The fine structure of the scaffold by scanning electron microscopy was studied. The endometrial mesenchymal cells were seeded onto decellularized scaffold by centrifugal method and cultured for 7 days. After 72 h the treated group with 0.5% SDS showed well-preserved ECM morphology with the minimum level of DNA (2.23% ± 0.08). Raman spectroscopy analysis confirmed that, the amount of ECM components was not significantly decreased in the decellularized group (P < 0.001) in comparison with native control. The electron micrographs demonstrated that the porosity and structure of ECM fibers in the decellularized group was similar to native ovary. The endometrial mesenchymal cells were attached and penetrated into the decellularized scaffold. In conclusion this combined protocol was an effective method to decellularize human ovarian tissue with high preservation of ECM contents, and human endometrial mesenchymal cells which successfully interacted with this created scaffold.







Similar content being viewed by others
Change history
03 December 2021
A Correction to this paper has been published: https://doi.org/10.1007/s40204-021-00175-2
References
Alshaikh AB, Padma AM, Dehlin M, Akouri R, Song MJ, Brännström M, Hellström M (2019) Decellularization of the mouse ovary: comparison of different scaffold generation protocols for future ovarian bioengineering. J Ovarian Res 12:1–9. https://doi.org/10.1186/s13048-019-0531-3
Alshaikh AB, Padma AM, Dehlin M, Akouri R, Song MJ, Brännström M, Hellström M (2020) Decellularization and recellularization of the ovary for bioengineering applications; studies in the mouse. Reprod Biol Endocrinol 18:1–10. https://doi.org/10.1186/s12958-020-00630-y
Bergholt MS, Serio A, Albro MB (2016) Raman spectroscopy reveals new insights into the zonal organization of native and tissue-engineered articular cartilage. ACS Cent Sci 2:885–895. https://doi.org/10.1021/acscentsci.6b00222
Bergholt MS, Serio A, Albro MB (2019) Raman spectroscopy: guiding light for the extracellular matrix. Front Bioeng Biotechnol 7:303. https://doi.org/10.3389/fbioe.2019.00303
Borel S, Prikryl EA, Vuong NH, Jonkman J, Vanderhyden B, Wilson BC, Murugkar S (2015) Discrimination of normal and malignant mouse ovarian surface epithelial cells in vitro using Raman microspectroscopy. Anal Methods 7:9520–9528. https://doi.org/10.1039/C5AY02462E
Eivazkhani F, Abtahi NS, Tavana S, Mirzaeian L, Abedi F, Ebrahimi B, Montazeri L, Valojerdi MR, Fathi R (2019) Evaluating two ovarian decellularization methods in three species. Mater Sci Eng C 102:670–682. https://doi.org/10.1016/j.msec.2019.04.092
Faulk DM, Carruthers CA, Warner HJ, Kramer CR, Reing JE, Zhang L, D’Amore A, Badylak SF (2014) The effect of detergents on the basement membrane complex of a biologic scaffold material. Acta Biomater 10:183–193. https://doi.org/10.1016/j.actbio.2013.09.006
Fayazi M, Salehnia M, Ziaei S (2015) Differentiation of human CD146-positive endometrial stem cells to adipogenic-, osteogenic-, neural progenitor-, and glial-like cells. Vitro Cell Dev Biol Anim 51(4):408–414. https://doi.org/10.1007/s11626-014-9842-2
Fazeli Z, Abedindo A, Omrani MD, Ghaderian SMH (2018) Mesenchymal stem cells (MSCs) therapy for recovery of fertility: a systematic review. Stem Cell Rev Rep 14:1–12. https://doi.org/10.1007/s12015-017-9765-x
Feng P, Li P, Tan J (2019) Human menstrual blood-derived stromal cells promote recovery of premature ovarian insufficiency via regulating the ECM-dependent FAK/AKT signaling. Stem Cell Rev Rep 15:241–255. https://doi.org/10.1007/s12015-018-9867-0
Gandolfi F, Ghiringhelli M, Brevini TA (2019) Bioengineering the ovary to preserve and reestablish female fertility. Anim Reprod 16:45. https://doi.org/10.21451/1984-3143-AR2018-0099
Gargus ES, Rogers HB, McKinnon KE, Edmonds ME, Woodruff TK (2020) Engineered reproductive tissues. Nat Biomed Eng 4:381–393. https://doi.org/10.1038/s41551-020-0525-x
Gilpin A, Yang Y (2017) Decellularization strategies for regenerative medicine: from processing techniques to applications. Biomed Res Int. https://doi.org/10.1155/2017/9831534
Gullekson C, Lucas L, Hewitt K, Kreplak L (2011) Surface-sensitive Raman spectroscopy of collagen I fibrils. Biophys J 100(7):1837–1845. https://doi.org/10.1016/j.bpj.2011.02.026
Hassanpour A, Talaei-Khozani T, Kargar-Abarghouei E, Razban V, Vojdani Z (2018) Decellularized human ovarian scaffold based on a sodium lauryl ester sulfate (SLES)-treated protocol, as a natural three-dimensional scaffold for construction of bioengineered ovaries. Stem Cell Res Ther 9:1–13. https://doi.org/10.1186/s13287-018-0971-5
Huang B, Lu J, Ding C, Zou Q, Wang W, Li H (2018) Exosomes derived from human adipose mesenchymal stem cells improve ovary function of premature ovarian insufficiency by targeting SMAD. Stem Cell Res Ther 9:1–12. https://doi.org/10.1186/s13287-018-0953-7
Janko M, Davydovskaya P, Bauer M, Zink A, Stark RW (2010) Anisotropic Raman scattering in collagen bundles. Opt Lett 35:2765–2767. https://doi.org/10.1364/OL.35.002765
Kawecki M, Łabuś W, Klama-Baryla A, Kitala D, Kraut M, Glik J, Misiuga M, Nowak M, Bielecki T, Kasperczyk A (2018) A review of decellurization methods caused by an urgent need for quality control of cell-free extracellular matrix’scaffolds and their role in regenerative medicine. J Biomed Mater Res B Appl Biomater 106:909–923. https://doi.org/10.1002/jbm.b.33865
Kim BS, Choi JS, Kim JD, Choi YC, Cho YW (2012) Recellularization of decellularized human adipose-tissue-derived extracellular matrix sheets with other human cell types. Cell Tissue Res 348:559–567. https://doi.org/10.1007/s00441-012-1391-y
Laronda MM, Jakus AE, Whelan KA, Wertheim JA, Shah RN, Woodruff TK (2015) Initiation of puberty in mice following decellularized ovary transplant. Biomaterials 50:20–29. https://doi.org/10.1016/j.biomaterials.2015.01.051
Lim NSJ, Hamed Z, Yeow CH, Chan C, Huang Z (2011) Early detection of biomolecular changes in disrupted porcine cartilage using polarized Raman spectroscopy. J Biomed Optics 16(1):017003. https://doi.org/10.1117/1.3528006
Lim LJ, Ling LH, Neo YP, Chung AYF, Goh BKP, Chow PKH, Chan CY, Cheow PC, Lee SY, Lim TKH, Chong SS, Ooi LLPJ, Lee CG (2019) Human amnion-derived mesenchymal stem cell (hAD-MSC) transplantation improves ovarian function in rats with premature ovarian insufficiency (POI) at least partly through a paracrine mechanism. Stem Cell Res Ther 10:1–18. https://doi.org/10.1186/s13287-019-1136-x
Liu WY, Lin SG, Zhuo RY, Xie YY, Pan W, Lin XF, Shen FX (2017) Xenogeneic decellularized scaffold: a novel platform for ovary regeneration. Tissue Eng Part C Methods 23:61–71. https://doi.org/10.1089/ten.TEC.2016.0410
Lv XG, Feng C, Fu Q, Xie H, Wang Y, Huang JW, Xie MK, Atala A, Xu YM, Zhao WX (2016) Comparative study of different seeding methods based on a multilayer SIS scaffold: Which is the optimal procedure for urethral tissue engineering? J Biomed Mater Res B Appl Biomater 104:1098–1108. https://doi.org/10.1002/jbm.b.33460
Mirzaeian L, Eivazkhani F, Hezavehei M, Moini A, Esfandiari F, Valojerdi MR, Fathi R (2020) Optimizing the cell seeding protocol to human decellularized ovarian scaffold: application of dynamic system for bio-engineering. Cell J 22(2):227–235. https://doi.org/10.22074/cellj.2020.6604
Movasaghi Z, Rehman S, Rehman IU (2007) Raman spectroscopy of biological tissues. Appl Spectrosc Rev 42:493–541. https://doi.org/10.1080/05704920701551530
Na J, Kim GJ (2020) Recent trends in stem cell therapy for premature ovarian insufficiency and its therapeutic potential: a review. J Ovarian Res 13:1–10. https://doi.org/10.1186/s13048-020-00671-2
Nguyen T, Gobinet C, Feru J, Brassart-Pasco S, Manfait M, Piot O (2012) Characterization of type I and IV collagens by Raman microspectroscopy: Identification of spectral markers of the dermo-epidermal junction. Spectrosc an Int J 27:421–427. https://doi.org/10.1155/2012/686183
Nikniaz H, Zandieh Z, Nouri M, Daei-Farshbaf N, Aflatoonian R, Gholipourmalekabadi M, Jameie SB (2021) Comparing various protocols of human and bovine ovarian tissue decellularization to prepare extracellular matrix-alginate scaffold for better follicle development in vitro. BMC Biotechnol 21:1–8. https://doi.org/10.1186/s12896-020-00658-3
Pennarossa G, Ghiringhelli M, Gandolfi F, Brevini TAL (2020) Whole-ovary decellularization generates an effective 3D bioscaffold for ovarian bioengineering. J Assist Reprod Genet 37:1329–1339. https://doi.org/10.1007/s10815-020-01784-9
Pomin VH, Mulloy B (2018) Glycosaminoglycans and proteoglycans. Pharmaceuticals (basel) 11(1):27. https://doi.org/10.3390/ph11010027
Pors SE, Ramløse M, Nikiforov D, Lundsgaard K, Cheng J, Andersen CY, Kristensen SG (2019) Initial steps in reconstruction of the human ovary: survival of pre-antral stage follicles in a decellularized human ovarian scaffold. Hum Reprod 34:1523–1535. https://doi.org/10.1093/humrep/dez077
Porzionato A, Stocco E, Barbon S, Grandi F, Macchi V, De Caro R (2018) Tissue-engineered grafts from human decellularized extracellular matrices: a systematic review and future perspectives. Int J Mol Sci 19:4117. https://doi.org/10.3390/ijms19124117
Rajabi Z, Yazdekhasti H, Noori Mugahi SMH, Abbasi M, Kazemnejad S, Shirazi A, Majidi M, Zarnani AH (2018) Mouse preantral follicle growth in 3D co-culture system using human menstrual blood mesenchymal stem cell. Reprod Biol 18(1):122–131. https://doi.org/10.1016/j.repbio.2018.02.001
Simsa R, Padma AM, Heher P, Hellström M, Teuschl A, Jenndahl L, Bergh N, Fogelstrand P (2018) Systematic in vitro comparison of decellularization protocols for blood vessels. PLoS ONE. https://doi.org/10.1371/journal.pone.0209269
White DM, Carruthers CA, Warner HJ, Kramer CR, Reing JE, Zhang L, D’Amore A, Badylak SF (2014) The effect of detergents on the basement membrane complex of a biologic scaffold material. Acta Biomater 10:183–193. https://doi.org/10.1016/j.actbio.2013.09.006
White LJ, Taylor AJ, Faulk DM, Keane TJ, Saldin LT, Reing JE, Swinehart IT, Turner NJ, Ratner BD, Badylak SF (2017) The impact of detergents on the tissue decellularization process: a ToF-SIMS study. Acta Biomater 50:207–219. https://doi.org/10.1016/j.actbio.2016.12.033
Xia X, Wang T, Yin T, Yan L, Yan J, Lu C, Zhao L, Li M, Zhang Y, Jin H, Zhu X, Liu P, Li R, Qiao J (2015) Mesenchymal stem cells facilitate in vitro development of human preantral follicle. Reprod Sci 22(11):1367–1376. https://doi.org/10.1177/1933719115578922
Acknowledgements
This work was supported by Tarbiat Modares University of Medical Sciences as a Ph.D. thesis. Special thanks to Mr. Pour Bayranvand for technical assistance.
Author information
Authors and Affiliations
Contributions
MNS performed the experiments, analyzed the data, and contributed to writing the draft of the manuscript. MRV involved in protocol development. MS and SZ supervised the study and contributed to writing the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors clarify there is no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the ethical guidelines of Tarbiat Modares University, Tehran, Iran (IR.MODARES.REC.1398.006) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sistani, M.N., Zavareh, S., Valujerdi, M.R. et al. Characteristics of a decellularized human ovarian tissue created by combined protocols and its interaction with human endometrial mesenchymal cells. Prog Biomater 10, 195–206 (2021). https://doi.org/10.1007/s40204-021-00163-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40204-021-00163-6