Abstract
Introduction
Cataract extraction could improve visual acuity (VA) for patients with retinitis pigmentosa (RP), while the surgery may increase photoreceptor degeneration through light damage. In this study, we conducted a systematic review and meta-analysis to investigate the effectiveness and prediction of VA after cataract surgery in patients with RP.
Methods
We comprehensively extracted data from literature of available studies with quality control processing. Improvement of VA before and after cataract surgery of different durations of follow-up and different structural integrity of the preoperative macular ellipsoid zone (EZ) in patients with RP were compared. VA was measured by the logarithm of the minimum angle of resolution (logMAR).
Results
Sixteen studies were subjected to analysis. Postoperative VA was significantly improved versus preoperative, with a mean difference (MD) of 0.57 [95% confidence interval (CI) 0.45, 0.69], and a fixed-effect model was applied during follow-up durations of 1 day to 1 month (I2 = 0%). Similarly, for follow-up durations of 1–3 months, 3–6 months, and 6–12 months, postoperative VAs were all better than preoperative values, with MDs of 0.36 (95% CI 0.31, 0.41), 0.35 (95% CI 0.23, 0.46), and 0.22 (95% CI 0.14, 0.30) (I2 < 50%). For follow-up duration of 1–5 years, the random-effect model was applied for higher heterogeneity (I2 = 81%), with an MD of 0.26 (95% CI 0.09, 0.43). There was no significant difference in the improvement of the EZ-invisible group, with an MD of 0.27 (95% CI − 0.17, 0.70) (I2 = 82%). There were significant differences between EZ-abnormal and EZ-normal groups in preoperative and postoperative VA, with MDs of 0.56 (95% CI 0.27, 0.85) and 0.46 (95% CI 0.27, 0.65) (I2 > 50%).
Conclusions
Cataract surgery could improve VA for patients with RP during long-term follow-up, and the surgery is not recommended for patients with invisible preoperative macular EZ. However, further studies are required to address the problem of excessive light exposure to the degenerated retina in patients with RP with the cataract removed.
The study protocol was registered on the International Prospective Register of Systematic Reviews (PROSPERO) (CRD42022340165).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Previous studies have reported that for patients with retinitis pigmentosa (RP), cataract extraction may increase photoreceptor degeneration through light damage, which could accelerate the deterioration of the patient's visual acuity (VA) and increase complications postoperatively. |
In this study, we conducted a systematic review and meta-analysis to investigate the improvements in VA and different preoperative ellipsoid zone conditions to predict postoperative VA. |
What was learned from the study? |
These results suggest that cataract surgery could improve VA for patients with RP during long-term follow-up, and the surgery is not recommended for patients with invisible preoperative macular ellipsoid zone examined by optical coherence tomography. |
Introduction
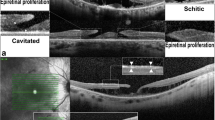
Retinitis pigmentosa (RP), the most common genetic ocular disease, is one of the leading causes of vision loss across the world [1]. The prevalence of RP varies from 1:13,121 in Spain [2], 1:9000 in Korea for all ages [3], 1:4000 in China for those over 55 years of age [4] to 1:750 in the adult population of rural Central India [5]. RP is characterized by progressive dysfunction of rod photoreceptors, followed by degeneration of cone photoreceptors. The inheritance patterns of RP are autosomal recessive, autosomal dominant, and X-linked. As for sporadic RP cases, causal autosomal dominant and X-linked variants were found in over 7.5% of cases [6]. The typical abnormalities in the fundus were bone spicule pigmentation, attenuation of retinal vessels, and waxy pallor of the optic nerve head, which result in the symptom of night blindness in the initial to central vision loss [1] (Fig. 1).
Besides the continuous progressive degeneration of photoreceptors and retinal pigment epithelium, complications such as cataracts and cystoid macular edema (CME) also could damage the remaining sight. It was reported that CME and cataracts were present in 58.6% and 23.4% of patients with RP, respectively [7]. Visual acuity (VA) could be improved for the complications that receive effective treatment, especially with the improvement of the cataract surgery pattern, which has been regarded as an efficient and safe surgery. Cataract surgery could improve the VA in patients with RP; however, it is still controversial whether or not performing surgery is necessary for patients with RP, as cataract extraction may increase photoreceptor degeneration through light damage [8], which could accelerate the deterioration of the patient's VA and increase complications such as posterior capsule opacification postoperatively.
The aim of this study is to evaluate the changes in VA before and after cataract surgery in patients with RP and whether the preoperative ellipsoid zone (EZ) examined by optical coherence tomography (OCT) could predict the final VA after cataract surgery in patients with RP. In this context, we investigate the improvements of VA during different durations of follow-up and different preoperative EZ conditions.
Methods
Study Protocol
The systematic review and meta-analysis were conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines [9]. The study protocol was registered on the International Prospective Register of Systematic Reviews (PROSPERO) (CRD42022340165), on 27 June 2022.
Inclusion Criteria
Retrospective, observational studies, case series, cross-sectional, cohort, case–control studies, and randomized controlled trials could all be included. The target population was patients with RP who underwent cataract surgery (phacoemulsification and intraocular lens implantation or extracapsular cataract extraction for those complicated cases).
Exclusion Criteria
The exclusion criteria were (1) non-English or Chinese articles; (2) studies without full-text articles or studies that do not contain primary data in their published articles, and we were unsuccessful in obtaining the original data from the authors; (3) animal and experimental studies; and (4) unpublished records such as conference papers, theses, and patents; (5) patients with uveitis or any disease that could cause RP-like fundus improvements or combined other ocular diseases, like glaucoma and complete luxation of the lens.
Databases and Search Strategy
A literature search was implemented in public databases, PubMed, Web of Science, Medline, the Chinese National Knowledge Infrastructure (CNKI), the Wanfang database, and the VIP database, from January 1, 2000 to May 31, 2022, using key and MeSH terms: retinitis pigmentosa; RP; cataract. The search strategy was ((retinitis pigmentosa) OR (RP)) AND (cataract), (“retinitis pigmentosa” [MeSH Terms] OR (“retinitis” [All Fields] AND “pigmentosa” [All Fields]) OR “retinitis pigmentosa” [All Fields] OR “RP” [All Fields]) AND (“cataract” [MeSH Terms] OR “cataract” [All Fields] OR “cataracts” [All Fields] OR “cataractic” [All Fields] OR “cataractous” [All Fields]).
Outcomes and Outcome Measurement
Improvement of VA During Different Durations of Follow-Up
We compared preoperative and postoperative VA in patients with RP and cataract according to different durations of follow-up after cataract surgery: (1) 0 months < postoperative ≤ 1 month; (2) 1 month < postoperative ≤ 3 months; (3) 3 months < postoperative ≤ 6 months; (4) 6 months < postoperative ≤ 12 months; and (5) 12 months < postoperative ≤ 60 months.
Changes of VA in Preoperative and Postoperative of Each EZ Grade
According to the OCT images, the preoperative conditions of the EZ were graded into the EZ-invisible group (invisible EZ in the macular region), the EZ-abnormal group (visible but discontinuous EZ in the macular region), and the EZ-normal group (complete EZ in the macular region).
Postoperative Complication
We reviewed posterior capsular opacification, CME, and other rare postoperative complications.
For the measurement of VA, 13 studies used the logarithm of the minimum angle of resolution (logMAR) for a statistical presentation and three studies used decimal acuities for statistical evaluation, which were converted into logMAR by the formula of logMAR = − log (decimal acuity) [10].
Data Extraction
Study Selection
Two authors (HLH, XDM) searched and screened the titles and abstracts independently for all articles from the aforementioned databases on the basis of a predefined selection protocol. Decisions were recorded using the PRISMA flowchart.
Data Extraction
Data extraction was done independently by two team members; reappraisal and checking of disagreements was done by them together (HLH, XDM). If disagreements were not resolved, a third reviewer (HS) was consulted. The following data were extracted: (1) author and year of the study; (2) design of the study; (3) the number of participants and eyes included in the study; and (4) main (VA) and additional (CME and posterior capsular opacification) outcome measures with their follow-up time.
Assessment of Risk of Bias and Paper Quality
The quality of the studies was assessed by two researchers (HLH, XDM) using the modified Newcastle–Ottawa scale (NOS) reported by Modesti and colleagues [11], which considers the selection (maximum 5 stars): (1) representativeness of the sample; (2) sample size; (3) non-respondents; (4) ascertainment of the exposure (risk factor); comparability (maximum 2 stars); outcome (maximum 3 stars): (1) assessment of the outcome; (2) statistical test. A study with a score higher than 6 could be included for meta-analysis; a study scoring more than 7 is considered as being of high quality. Consensus was achieved through discussion with the third reviewer (HS) in the event of any disagreements between the researchers regarding individual ratings. Publication bias was assessed and checked by Egger’s test [12].
Data Synthesis and Statistical Analysis
We used a fixed-effects model or a random-effects model to calculate the mean difference (MD) and 95% confidence interval (95% CI). Heterogeneity was checked using the Q test and I2 test. A P value for the Q test of less than 0.05 and/or I2 greater than 50% suggested high heterogeneity, in which case a random-effects model was used; otherwise, the fixed-effects model was used. All analyses will be performed using RevMan 5.3 and GraphPad Prism 9. Egger’s test was further tested for publication bias by the R program (version 3.4). P value less than 0.05 is considered statistically significant.
Ethics
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors. The patient whose eyes are shown in Fig. 1 provided signed informed consent.
Results
More than 4500 records were identified through database searching (Fig. 2). We included 19 articles assessed for eligibility, there were 5 Chinese studies and 14 English studies. One Chinese article [13] was excluded because of insufficient data (no duration of follow-up) and two English articles were excluded because the results had been repeatedly reported by the same research group [14, 15]. The remaining 16 studies [16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31] were included for data analysis. The flowchart of the selection process is shown in Fig. 2. All studies were retrospective and observational studies. VA in three studies was converted from decimal acuity into logMAR by the formula in “Postoperative Complication”. In the meta-analysis, a total of 796 patients (1133 eyes) were included. Characteristics of include 16 studies are shown in Table 1. The follow-up time ranged from 1 day to 60 months.
Risk of Bias Assessment
As shown in Table 2, the average number of stars of included studies scored by modified NOS was 8.5, and all included studies were of high quality with modified NOS ≥ 7 stars. VA and complications of the included 16 studies were self-reported and not obtained by independent blind assessment or record linkage with an assessment of outcome and therefore scored only 1 star. The sample size of studies by De Rojas et al. and Wang et al. was less than 20 eyes, and therefore scored no stars. VA in three studies was recorded by decimal acuity which needs to be converted into logMAR; thus,1 star was deducted from the ascertainment of exposure score. Chatterjee and colleagues’ study scored 1 star in comparability for the different surgery methods with phacoemulsification, manual small incision cataract surgery, and extracapsular cataract extraction, and Nakamura’s study scored 1 star for including patients from previous studies.
The results of Egger’s test shown in Table 3 indicate that there is no publication bias for the included studies.
Improvement of VA During Different Durations of Follow-Up
We found that VA during postoperative follow-up duration of 1 day to 1 month was better than preoperative VA, with an MD of 0.57 (95% CI 0.45, 0.69), with no heterogeneity among studies (I2 = 0% and P < 0.05); hence, we applied a fixed-effects model. Similarly, for follow-up durations of 1 month to 3 months, 3 months to 6 months, and 6 months to 1 year, postoperative VAs were all better than preoperative VAs, with MDs of 0.36 (95% CI 0.31, 0.41), 0.35 (95% CI 0.23, 0.46), and 0.22 (95% CI 0.14, 0.30), with quite small heterogeneity (I2 = 0%, 0%, and 37%), and there was no publication bias for all aforementioned outcomes by Egger’s test. As for follow-up duration of 1–5 years, we applied the random-effect model for higher heterogeneity for a larger span of follow-up, with I2 = 81%, MD was 0.26 (95% CI 0.09, 0.43) (Figs. 3, 4, 5, 6, 7).
We found with the extended follow-up, improvement of VA between preoperative and postoperative periods showed a downward trend until the follow-up duration of 12 months. Surprisingly, the downward trend was reversed with follow-ups of more than 1 year (Fig. 8).
Changes of Preoperative and Postoperative VA of Each EZ Grade
There was no significant difference in the improvement of the EZ-invisible group (P = 0.23), with an MD of 0.27 (95% CI − 0.17, 0.70), with an I2 = 82%, a random-effects model was applied. Changes in preoperative and postoperative VA in EZ-abnormal and EZ-normal groups were significant, with MDs of 0.56 (95% CI 0.27, 0.85) and 0.46 (95% CI 0.27, 0.65), a random-effects model was applied for I2 > 50% (66% and 69%) (Figs. 9, 10, 11, 12).
The result of Egger’s test showed that there is no publication bias for the included studies (P > 0.05).
Postoperative Complications
As shown in Table 4, among the 16 included articles, 11 reported complications after surgery in patients with RP and cataract. Posterior capsule opacification was the most common complication, with a prevalence of 42.63% (347/814). CME was observed in three studies (28 eyes). Postoperative complications include zonular dialysis in 20 eyes, capsule contraction syndrome in 12 eyes, and an increase in intraocular pressure noted in 5 eyes. Other rare complications were reported by Chatterjee and colleagues (two eyes suffered posterior capsule rupture, 33 eyes foveal thinning, 58 eyes optic atrophy, and 5 eyes postoperative uveitis) and by Nakamura and colleagues (two eyes suffered epiretinal membrane, one eye macular hole, and one eye vitreomacular traction syndrome).
Discussion
Cataract extraction for patients with RP combined with cataracts could help most patients enhance VA and improve their quality of life. As a result of the progressive degeneration of photoreceptors, postoperative complications, inflammatory response following surgery, and retinal damage during light exposure, where photopigment, retinoids, and bisretinoids may play important roles in photochemical damage, conclusions from studies on the long-term effects after cataract surgery in patients with RP gave contradictory answers [22, 32]. Moreover, not all patients are suitable for cataract surgery because it is difficult to improve VA as a result of deteriorated preoperative retinal conditions. Hence, we conducted this systematic review and meta-analysis to investigate the improvements of VA during different durations of follow-up and different preoperative EZ conditions to predict postoperative VA.
In the present systemic meta-analysis, the evidence is clear that cataract surgery could help improve VA after cataract surgery in patients with RP during all durations of follow-up. Our meta-analysis showed that postoperative VA could be up 0.22 to 0.57 log units higher than preoperative VA. And we found that the improvement of VA showed a downward trend with the extended follow-up of 1 day to 12 months. This indicated that postoperative VA would decrease with the passage of time, and there are several factors mentioned above that contribute to this result. However, the postoperative VA is still better than preoperative VA. Surprisingly, the downward trend in VA reversed with a follow-up of more than 1 year; one contribution to this observation may be that the postoperative complication of posterior capsule opacification usually occurs 6 months to 1 year after surgery, for which patients prefer to seek treatment by neodymium:yttrium–aluminum–garnet (Nd:YAG) laser capsulotomy to regain part of their vision [33].
In this meta-analysis, we also evaluated the VA outcomes after cataract surgery in patients with RP with different structural integrity of the preoperative macular EZ examined by OCT. We found no difference between preoperative and postoperative VA in the EZ-invisible group. As for the EZ-abnormal group and EZ-normal group, the difference between preoperative and postoperative VA was statistically significant and up to 0.56 and 0.46 log units for those patients with RP. Greater improvement of VA in the EZ-abnormal group than the EZ-normal group may in part be due to the baseline VA in the EZ-abnormal group being worse and combined with more severe cataracts. Hence, we do not recommend cataract surgery for patients with EZ-invisible RP, who will not improve their VA after the surgery and OCT should be examined for patients with RP before cataract surgery.
We also reviewed the postoperative complications which could affect VA after cataract surgery. Posterior capsule opacification and CME were the most common complications. Strong et al. [34] concluded that the mechanisms for RP-associated CME include breakdown of the blood—retinal barrier, dysfunction of retinal pigment epithelial in pumping, vitreous traction, and edema of Müller cells and antiretinal antibodies. CME remains one of the most frequent complications of cataract surgery whose procedure could increase the risks for patients with RP [35]. And CME has been found to cause the formation of macular holes, and surgery of vitrectomy could significantly improve VA [36]. Zonular dialysis or dislocation of intraocular lens, posterior capsule rupture, postoperative uveitis, and epiretinal membrane were also reported. Some studies focused on capsule contraction syndrome, especially investigating the effect of with or without lens capsular tension ring on patients with RP. And capsular tension ring could reduce the complications caused by capsule contraction as usual in patients with cataracts. Dikopf and colleagues [17] found that 18.8% of eyes had some degree of zonular insufficiency and 10% of eyes had phacodonesis during surgery in patients with RP, which was higher than the 0.1% of senile cataract eyes that had developed zonular dialysis intraoperatively [37]. Hence, for patients with RP, the capsular tension ring, suitable size, and haptic geometry intraocular lens are highly recommended [38], while aggressive polishing of the capsule is suboptimal for preventing PCO in patients with zonular weakness RP [17].
However, further studies are required to address the problem of excessive light exposure to the degenerated retina in patients with RP with the cataract removed. As for late-stage patients with RP, the replacement of dead cells with diverse retinal cell types derived from pluripotent stem cells, including RPE cells and photoreceptors, may be a strategy to rescue the visual function of retinal degeneration [39, 40]. Moreover, the identification of novel genotype–phenotype correlations and the mutation spectrum should also be studied to assist in both the clinical diagnosis and the development of treatments for inherited retinal dystrophy, like RP. Our team has discovered AHI1 as a novel candidate gene for nonsyndromic RP by developing a targeted panel, which was previously reported to cause a congenital systemic disease called Joubert syndrome [41].
In conclusion, cataract surgery could improve VA for patients with RP and cataract during the long-term follow-up, although changes in VA showed a downward trend. Moreover, cataract surgery is not recommended for patients with EZ-invisible RP, and OCT should be examined for patients with RP before cataract surgery.
References
Verbakel SK, van Huet RAC, Boon CJF, et al. Non-syndromic retinitis pigmentosa. Prog Retin Eye Res. 2018;66:157–86.
Perea-Romero I, Gordo G, Iancu IF, et al. Genetic landscape of 6089 inherited retinal dystrophies affected cases in Spain and their therapeutic and extended epidemiological implications. Sci Rep. 2021;11(1):1526.
Na KH, Kim HJ, Kim KH, et al. Age at diagnosis, mortality, and cause of death in retinitis pigmentosa in Korea—a nationwide population-based study. Am J Ophthalmol. 2017;176:157–65.
You QS, Xu L, Wang YX, et al. Prevalence of retinitis pigmentosa in North China: the Beijing Eye Public Health Care Project. Acta Ophthalmol. 2013;91(6):e499-500.
Nangia V, Jonas JB, Khare A, Sinha A. Prevalence of retinitis pigmentosa in India: the Central India Eye and Medical Study. Acta Ophthalmol. 2012;90(8):e649–50.
Martin-Merida I, Avila-Fernandez A, Del Pozo-Valero M, et al. Genomic landscape of sporadic retinitis pigmentosa: findings from 877 Spanish cases. Ophthalmology. 2019;126(8):1181–8.
Liew G, Strong S, Bradley P, et al. Prevalence of cystoid macular oedema, epiretinal membrane and cataract in retinitis pigmentosa. Br J Ophthalmol. 2019;103(8):1163–6.
Cideciyan AV, Jacobson SG, Aleman TS, et al. In vivo dynamics of retinal injury and repair in the rhodopsin mutant dog model of human retinitis pigmentosa. Proc Natl Acad Sci U S A. 2005;102(14):5233–8.
Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372: n71.
Holladay JT. Visual acuity measurements. J Cataract Refract Surg. 2004;30(2):287–90.
Modesti PA, Reboldi G, Cappuccio FP, et al. Panethnic differences in blood pressure in Europe: a systematic review and meta-analysis. PLoS ONE. 2016;11(1):e0147601.
Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–34.
Guo G. Efficacy analysis of phacoemulsification combined with intraocular lens implantation in the treatment of retinitis pigmentosa complicated with cataract. J Front Med. 2018;8(23):198.
Yoshida N, Ikeda Y, Murakami Y, et al. Factors affecting visual acuity after cataract surgery in patients with retinitis pigmentosa. Ophthalmology. 2015;122(5):903–8.
Miura G, Baba T, Tatsumi T, Yokouchi H, Yamamoto S. Effects of cataract surgery on vision-related quality of life in patients with retinitis pigmentosa and the predictive factors of quality of life improvement. Biomed Res Int. 2021;2021:3846867.
Jackson H, Garway-Heath D, Rosen P, Bird AC, Tuft SJ. Outcome of cataract surgery in patients with retinitis pigmentosa. Br J Ophthalmol. 2001;85(8):936–8.
Dikopf MS, Chow CC, Mieler WF, Tu EY. Cataract extraction outcomes and the prevalence of zonular insufficiency in retinitis pigmentosa. Am J Ophthalmol. 2013;156(1):82-88.e2.
Bayyoud T, Bartz-Schmidt KU, Yoeruek E. Long-term clinical results after cataract surgery with and without capsular tension ring in patients with retinitis pigmentosa: a retrospective study. BMJ Open. 2013;3(4): e002616.
Garcia-Martin E, Rodriguez-Mena D, Dolz I, et al. Influence of cataract surgery on optical coherence tomography and neurophysiology measurements in patients with retinitis pigmentosa. Am J Ophthalmol. 2013;156(2):293-303.e2.
Nakamura Y, Mitamura Y, Hagiwara A, et al. Relationship between retinal microstructures and visual acuity after cataract surgery in patients with retinitis pigmentosa. Br J Ophthalmol. 2015;99(4):508–11.
Chan TCY, Lam SC, Mohamed S, Wong RLM. Survival analysis of visual improvement after cataract surgery in advanced retinitis pigmentosa. Eye (Lond). 2017;31(12):1747–8.
De Rojas JO, Schuerch K, Mathews PM, et al. Evaluating structural progression of retinitis pigmentosa after cataract surgery. Am J Ophthalmol. 2017;180:117–23.
Lu JQ, Bi J, Du HB, Wang D, Liu Q. Efficacy analysis of phacoemulsification combined with intraocular lens implantation in the treatment of retinitis pigmentosa complicated with cataract. Rec Adv Ophthalmol. 2017;37(11):1064–7.
Mao J, Fang D, Chen Y, et al. Prediction of visual acuity after cataract surgery using optical coherence tomography findings in eyes with retinitis pigmentosa. Ophthalmic Surg Lasers Imaging Retina. 2018;49(8):587–94.
Fan W, Yuan RD. Effects of optical coherence tomography on the visual outcomes of cataract surgery in patients with retinitis pigmentosa. Rec Adv Ophthalmol. 2019;39(10):961–3.
Wang JD, Zhang JS, Xiong Y, et al. Clinical observation of the effect of surgery on retinitis pigmentosa patients with cataract. J Clin Ophthalmol. 2022;28(4):330–2.
Zhang M, Dou GR, Wang YS. Evaluating visual prognosis of cataract patients with primary retinitis pigmentosa after phacoemulsification and IOL implantation by SD-OCT. Rec Adv Ophthalmol. 2020;40(2):136–40.
Chatterjee S, Agrawal D, Agrawal D, Parchand SM, Sahu A. Cataract surgery in retinitis pigmentosa. Indian J Ophthalmol. 2021;69(7):1753–7.
Chen CX, Wang JD, Zhang JS, et al. Effect of lens capsular tension ring on preventing capsular contraction syndrome in the surgery of retinitis pigmentosa combined with cataract: retrospective case series. Int J Clin Pract. 2021;75(8): e14272.
Miura G, Baba T, Tatsumi T, Yokouchi H, Yamamoto S. The impact of cataract surgery on contrast visual acuity and retinal sensitivity in patients with retinitis pigmentosa. J Ophthalmol. 2021;2021:2281834.
Nakamura S, Fujiwara K, Yoshida N, et al. Long-term outcomes of cataract surgery in patients with retinitis pigmentosa. Ophthalmol Retina. 2022;6(4):268–72.
Hunter JJ, Morgan JI, Merigan WH, Sliney DH, Sparrow JR, Williams DR. The susceptibility of the retina to photochemical damage from visible light. Prog Retin Eye Res. 2012;31(1):28–42.
Wormstone IM, Wormstone YM, Smith AJO, Eldred JA. Posterior capsule opacification: what’s in the bag? Prog Retin Eye Res. 2020;82: 100905.
Strong S, Liew G, Michaelides M. Retinitis pigmentosa-associated cystoid macular oedema: pathogenesis and avenues of intervention. Br J Ophthalmol. 2017;101(1):31–7.
Holló G, Aung T, Cantor LB, Aihara M. Cystoid macular edema related to cataract surgery and topical prostaglandin analogs: mechanism, diagnosis, and management. Surv Ophthalmol. 2020;65(5):496–512.
Jin ZB, Gan DK, Xu GZ, Nao-I N. Macular hole formation in patients with retinitis pigmentosa and prognosis of pars plana vitrectomy. Retina. 2008;28(4):610–4.
Liu XW, Wang Z, Yu WH, et al. Idiopathic phacodonesis in senile cataract patients in Qinghai, China. Int J Ophthalmol. 2011;4(5):508–12.
Borkenstein AF, Borkenstein EM. Geometry of acrylic, hydrophobic IOLs and changes in haptic-capsular bag relationship according to compression and different well diameters: a bench study using computed tomography. Ophthalmol Ther. 2022;11(2):711–27.
Jin ZB, Gao ML, Deng WL, et al. Stemming retinal regeneration with pluripotent stem cells. Prog Retin Eye Res. 2019;69:38–56.
Zhang CJ, Ma Y, Jin ZB. The road to restore vision with photoreceptor regeneration. Exp Eye Res. 2021;202: 108283.
Huang XF, Huang F, Wu KC, et al. Genotype-phenotype correlation and mutation spectrum in a large cohort of patients with inherited retinal dystrophy revealed by next-generation sequencing. Genet Med. 2015;17(4):271–8.
Acknowledgements
The authors thank Fan Wei (Department of Ophthalmology, the Second Affiliated Hospital of Army Medical University, Chongqing 400037, China) for providing the original data.
Funding
This study is partly supported by the National Natural Science Foundation of China (82125007), and National Key R&D Program of China (2017YFA0105300). The journal’s Rapid Service Fee was funded by the corresponding author (Zi-Bing Jin).
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Hailong He, Hao Song, Xiaodie Meng, Kai Cao, and Yi-Xin Liu. The first draft of the manuscript was written by Hailong He, Zi-Bing Jin and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Disclosures
Hailong He, Hao Song, Xiaodie Meng, Kai Cao, Yi-Xin Liu, Jinda Wang, Xiu Hua Wan, and Zi-Bing Jin have nothing to disclose.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors. The patient whose eyes are shown in Fig. 1 provided signed informed consent.
Data Availability
The datasets generated during and analyzed during the current study are not publicly available due to the need for further research but are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
He, H., Song, H., Meng, X. et al. Effects and Prognosis of Cataract Surgery in Patients with Retinitis Pigmentosa. Ophthalmol Ther 11, 1975–1989 (2022). https://doi.org/10.1007/s40123-022-00563-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-022-00563-2