Abstract
In Part One of this two-part series, we discussed skin physiology and anatomy as well as generalities concerning topical analgesics. This modality of therapy has lesser side effects and drug–drug interactions, and patients tolerate this form of therapy better than many oral options. Unfortunately, this modality is not used as often as it could be in chronic pain states, such as that from neuropathic pain. Part Two discusses specific therapies, local anesthetics, and other drugs, as well as how a clinician might use specific aspects of a patient’s neuropathic pain presentation to help guide them in the selection of a topical agent.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Topical agents do not have a large market share in the treatment of chronic pain. The total market for over-the-counter analgesics alone is estimated at $2.38 billion [1]. In 2007, the market for topical analgesics (TAs) was only $40 million [2]. These data suggest that these treatments are underutilized. There are a number of different potential therapies, other than diclofenac and lidocaine topical options. Multiple different medications and mechanisms are currently being evaluated by the pharmaceutical industry.
Indeed, some of these mechanisms can be taken advantage of today, by developing a working relationship with a pharmacy that can compound TAs with multiple mechanisms and different drugs. In line with the repeated observations that TA use is generally associated with a more desirable side effect profile than orally, transdermally, parenterally, or intrathecally administered analgesics, these observations should be especially considered when developing a pain management treatment regimen for an individual patient. Currently, only a few options are available in the market, and clinicians work in concert with compounding pharmacists to produce various compounded TAs. This is not a rare occurrence as an Internet survey of 120 physicians showed 27% of those surveyed used compounded topical agents as part of their practice [3]. The research approach of much of the compounding industry is the (n of 1) trial [4]. This is not unfamiliar to practicing clinicians who frequently “try” an agent in a given patient to evaluate efficacy and tolerability.
This review article is based on previously conducted studies, and does not involve any new studies of human or animal subjects performed by any of the authors.
Local Anesthetic Analgesics: an Overview of Topical Therapies
Sodium channels are highly expressed in axons and dendrites as well as centrally [5]. Furthermore, there are multiple channel subtypes, some of which are involved in the expression of pain. Although these transient channels appear very similar, such as voltage-gated sodium channel (Nav) 1.1–1.7, their actions are different [6]; Nav1.7, Nav1.8, Nav1.9 play roles in inflammatory pain [7]. The role for Nav1.3 is controversial and the channels Nav1.6, and Nav1.1 all warrant more in-depth investigation [8]. Local analgesic (LA) agents suppress the activity of peripheral sodium channels within sensory afferents and subsequent pain transmission; however, other mechanisms of action are still under investigation. There is decreased expression of messenger ribonucleic acid for specific sodium channel subtypes following use of LA [9, 10]. Furthermore, LAs preferentially block hyperexcitable cells [5]. LAs have been used by topical, intravenous, and subcutaneous routes in the treatment of chronic pain for years.
Several LAs used as TAs are currently available including the lidocaine 5% patch (L5P); the eutectic mixture of LAs, 2.5% lidocaine/2.5% prilocaine (EMLA); and a lidocaine 70 mg/tetracaine 70 mg patch (LTP). Of these three agents, only the L5P is associated with an analgesic effect without anesthetizing skin, whereas EMLA or LTP creates both analgesia and anesthesia. This difference may be useful in different clinical settings, for example, venipuncture and circumcision, for which creating both analgesia and anesthesia are helpful [10].
The use of specific TAs for the management of neuropathic pain has been supported by evidence from clinical trials and published reviews [11–13]. Among the currently available topical options, the L5P has been widely studied. The outcome of two studies assessed the tolerability and safety of 24-h use of up to four L5Ps for extended periods of time. There were no significant systemic side effects or dermal reactions, and plasma lidocaine levels remained low [14, 15]. Safety and tolerability were similar for either 12- or 24-h application [10, 14].
LAs have also been studied in conditions not typically considered responsive, such as low back pain. For example, a multicenter, open-label study involved 120 patients with acute (<6 weeks), subacute (<3 months), short-term chronic (3–12 months), or long-term chronic (>12 months) low back pain. During the 6-week study, a majority of patients experienced moderate or greater pain relief with four L5Ps applied to the most painful low back areas [16].
A randomized, double-blind, placebo-controlled trial conducted in Europe evaluated the analgesic efficacy of the L5P in the treatment of “focal” neuropathic pain syndromes, for example, mononeuropathies and ilioinguinal neuralgia [17]. Adding the patch to other pharmacotherapeutic regimens could reduce pain and allodynia as quickly as within 8 h of application [17]. A smaller open-label study of 16 patients with various neuropathic pain conditions, for example, complex regional pain syndrome (CRPS) or diabetic peripheral polyneuropathy (DPPN), demonstrated that the L5P provided pain relief without significant side effects in 81% of patients [18]. It is noteworthy that patients enrolled in this study, prior to the use of the L5P, had experienced suboptimal outcomes with other commonly prescribed agents. Several other noncontrolled studies using the L5P in patients with DPPN have been completed. These studies allowed patients to use as many as four L5Ps for as long as 18 h per day. Viewing these studies as a group, the majority of enrolled subjects reported pain reduction and good tolerability [19, 20]. An additional 3-week, single-center, open-label study of the L5P in patients with painful idiopathic sensory polyneuropathy noted significant improvements in both pain relief and quality of life [20]. In a phase IV study of 332 postherpetic neuralgia (PHN) patients using the L5P, 67% noted decreased pain intensity by the end of the first week. Further reductions were noted by the second week in more than 40% of those who had not experienced pain relief during the first study week. Overall, 70% of enrolled patients experienced improvement [21]. In another open-label study, treatment with the L5P was compared with pregabalin for PHN. The L5P was at least as effective as pregabalin in this study. Interestingly, for patients who had not responded to either treatment alone, these agents in combination resulted in greater benefit and tolerability [22].
Changes in PHN pain quality when using the L5P compared with placebo were evaluated in a multicenter, randomized, vehicle-controlled study of 150 patients [23]. Reduced pain symptoms, assessed using the Neuropathic Pain Scale, were more likely to occur in the L5P patients compared with placebo. Interestingly, neuropathic pain that was deep, sharp, and burning was previously assumed to be related to mechanisms of the central, and not peripheral, nervous system; however, L5P provided relief for these sensations as well. In the discussion of the study’s results, it was proposed that, given the localized primary peripheral nervous system (PNS) mechanism of action of the L5P, peripheral mechanisms might also be important for the development of other neuropathic pain qualities [23]. Functional brain magnetic resonance imaging studies of patients with PHN treated with the L5P demonstrated that brain activity appeared to be modulated in a manner related to the duration of L5P application, suggesting again that a peripherally acting agent may have an impact on central nervous system (CNS) pain mechanisms [24]. Central neuropathic itch has been treated successfully with the L5P in a single case report [25]. Treatment of focal neuropathic pain with the L5P has also been shown to be of some benefit in select patients with cancer [26].
Several studies of the use of EMLA preparations in treating PHN have shown mixed results. The results of a randomized, controlled study of patients with PHN using EMLA or placebo cream did not result in a significant difference in treatment outcome [27]. The results of two uncontrolled studies were more favorable suggesting that use of the EMLA could relieve the pain associated with PHN [28, 29]. Controlled studies have demonstrated the benefit of EMLA cream in reducing pain associated with circumcision and venipuncture as well as in surgery for breast cancer [10, 30].
Capsaicin
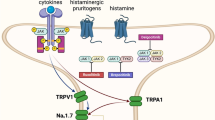
Capsaicin, a capsinoid, is a principal pungent component in the fruits of plants from the genus Capsicum, which are members of the nightshade family, Solanaceae [31]. Capsaicin was first isolated in 1816 in partially purified crystalline form by Bucholz and in pure crystalline form in 1876 by Thresh, who named it capsaicin [32, 33]. Topical capsaicin appears to act by agonist activity at the TRPV1 receptor on Aδ and C fibers, thus causing the release of substance P and calcitonin gene-related peptide [34–37]. TRPV1 receptors are deregulated by inflammation, heat, and acidification [38]. The results of both human and animal nerve biopsy studies demonstrate nerve fiber degeneration beneath the capsaicin application site. Such degeneration may be one of the mechanisms of pain relief and has been termed “dysfunctionalization” [39, 40].
Capsaicin, when applied topically, does not result in significant systemic accumulation and the incidence of burning may decrease with repeated use; however, the frequent occurrence of this side effect may negatively affect treatment adherence and consequently the patient’s ability to benefit from its use [34]. The analgesic effect of capsaicin is dose dependent and may last for several weeks. When capsaicin is compounded in a topical preparation at high concentrations and administered as a single application, the analgesic benefit appears to last for several weeks. Currently, topical liquid preparations of 10% and 20% capsaicin are in phased clinical trials [41, 42].
There has been interest in using capsaicin in a number of neuropathic pain disorders such as DPPN, human immunodeficiency virus neuropathy (HIVN), and PHN; however, many of the older studies yielded disappointing results, perhaps due in part to weakness, intolerability, efficacy, and adherence of previously available strengths of capsaicin: 0.025% and 0.075% [43]. For example, 0.025% capsaicin cream was determined to be no better than the vehicle (not active) cream in a randomized, double-blind study of 30 patients with temporomandibular joint pain [44]. A review of the published randomized trials involving low-dose topical capsaicin concluded that “although topically applied capsaicin has moderate to poor efficacy in the treatment of chronic musculoskeletal or neuropathic pain, it may be useful as an adjunct … for a small number of patients who are unresponsive to, or intolerant of, other treatments” [45].
However, higher strength capsaicin preparations have demonstrated better analgesia. In one study on HIVN, 7.5% topical capsaicin cream was applied. The patients experienced notable pain relief, but required epidural anesthesia to tolerate the application [46]. In studies leading to approval of the capsaicin 8% patch (C8P), it was more efficacious in reducing pain than an active, lower concentration capsaicin formulation serving as an active comparator and was well tolerated. In addition, the C8P has been studied in other neuropathic pain states with favorable outcome [47–50]. Efficacy shown from a single 60-min application in patients with neuropathic pain produced effective pain relief for up to 12 weeks [51]. Two open-label studies using the C8P in PHN and HIVN showed a majority of patients reported pain relief following a single application, up to 48 weeks [27, 36]. In a Cochrane review of higher dose capsaicin, the authors concluded “High-concentration topical capsaicin used to treat PHN and HIV neuropathy generates more participants with high levels of pain relief than does control treatment using a much lower concentration of capsaicin” [52].
Substantially fewer studies examining the use of capsaicin for low back pain or myofascial pain have been published. A randomized, double-blind, placebo-controlled study compared the use of topical capsaicin to placebo in 154 patients with chronic low back pain; of these, 60.8% of capsaicin-treated patients experienced 30% pain relief after 3 weeks of treatment (P < 0.02) versus 42.1% of patients treated with placebo [53].
Cyclooxygenase Inhibitors
The mechanism of action of a topical nonsteroidal anti-inflammatory drug (NSAID) is likely related to anti-inflammatory inhibition through prostaglandin synthesis via its adenosine triphosphate-sensitive K+ channel opening property [54, 55]. However, the extent of anti-inflammatory effect is not proportional to pain relief, and other mechanisms of action might be involved [55]. Diclofenac is available in dermal and transdermal formulations, gels, patches, creams, and lotions. Since 2007, three topical NSAIDs have been approved, including diclofenac sodium gel 1% for treating osteoarthritis (OA) pain, the diclofenac epolamine 1.3% topical patch for acute musculoskeletal pain, and the diclofenac 1.5% topical solution for OA of the knee [56].
The use of other topical NSAIDs has been studied most notably outside of the United States. For example, a topical ketoprofen patch (100 mg) was found superior to placebo in reducing pain following 7 days of treatment in a randomized, placebo-controlled study of 163 patients with ankle sprains [57]. In a separate study, patients with tendonitis were randomized in a double-blind, placebo-controlled study. The NSAID group fared better than placebo with only minor skin irritation and was well tolerated [58]. A small controlled study showed that a topical mixture of aspirin and diethyl ether was a significantly more effective analgesic than placebo in patients with acute herpes zoster and with PHN [59].
The use of TAs for the treatment of OA has also been studied and multiple recent reviews have been published recently [16, 60–62]. A diclofenac patch preparation demonstrated safety and efficacy in a randomized, double-blind controlled study in patients with chronic knee OA pain [63]. A randomized controlled study comparing topical diclofenac solution to oral diclofenac for knee OA concluded that the solution produced symptom relief equivalent to oral diclofenac, but with decreased incidence of gastrointestinal complaints [64]. Gastrointestinal side effects from oral diclofenac included abdominal pain, nausea, dyspepsia, and diarrhea [64]. A recently published long-term study with the same topical solution confirmed the safety parameters of the study [65]. In a pooled safety analysis of two large studies (one of which was [64]) comparing oral diclofenac with a topical diclofenac solution for the treatment of OA, although the rate of adverse events (AEs) between the two preparations was similar, patients treated with the oral diclofenac experienced more gastrointestinal (39.0% vs 25.4%, P < 0.0001) and cardiovascular (3.5% vs 1.5%, P = 0.055) AEs [66]. The oral diclofenac in the two studies was also associated with significantly greater increases in liver enzymes and creatinine, as well as greater decreases in creatinine clearance and hemoglobin (P < 0.001 for all). By contrast, patients receiving the topical solution had more dry skin at the application site (24.1% vs 1.9%, P < 0.0001) [66].
Diclofenac solution was compared with oral diclofenac in a parallel-arm study of individuals with temporomandibular joint pain. Although there was no significant difference in analgesic benefit, there were significantly fewer gastrointestinal side effects with the topical solution [67].
Two other trials demonstrated the efficacy of topical 1.16% diclofenac gel and topical solution compared with placebo, showing benefit and safety [68, 69]. In one meta-analysis, there was evidence to suggest that topical NSAIDs were more effective than placebo [70]. However, the authors concluded that available evidence indicated that topical NSAIDs were inferior to oral NSAIDs during the first week of treatment [70]. A separate meta-analysis examined the use of topical NSAIDs for chronic musculoskeletal pain and concluded they are effective and safe [71]. Interestingly, paraffin baths have been used to alleviate pain from OA and to improve function. When TAs were added to the paraffin bath, at least in one study using essential oils, this increased the analgesic effect of the bath [72].
A randomized controlled study of a diclofenac patch in 120 individuals experiencing acute pain after “blunt” injury demonstrated that the diclofenac patch was well tolerated and reduced pain intensity more than placebo [73]. Two studies, one noncontrolled and the other a multicenter, randomized, controlled study, noted that a diclofenac patch was found to be well tolerated and effective, with 60% pain relief in acute sports injuries [74, 75]. In another controlled study, topical ibuprofen cream was found to be superior in reducing pain from acute ankle sprains [76]. In a controlled study of ketoprofen gel in acute soft tissue pain, the gel was more effective than placebo in providing pain relief [77]. A topical formulation of ibuprofen 5% gel was examined in a 7-day placebo-controlled study in patients with soft tissue pain. Patients received either the ibuprofen 5% gel or placebo gel. A significant difference (P < 0.001) in pain reduction and improved physical activity was experienced in the study group [78]. A second similar study completed by the same investigators demonstrated similar results [60]. A further study using topical flurbiprofen for soft tissue pain reported greater pain reduction than oral diclofenac and fewer adverse effects [79]. This study is one of very few that actually compared a topical NSAID to an oral agent.
A meta-analysis reviewed the limited data and concluded that the effects of topically applied rubefacients containing salicylates resulted in moderate to poor efficacy. The authors emphasized that efficacy estimates were currently unreliable because of poorly designed clinical trials [80]. A randomized, controlled study with the topical NSAID eltenac examined its effect compared with oral diclofenac and placebo in 290 patients with knee OA. The TA, compared with placebo, showed greater safety and efficacy [81]. Each active treatment, in patients with severe symptoms, resulted in superior analgesia compared with placebo, with the incidence of gastrointestinal side effects lower in the eltenac gel group compared with that of the oral diclofenac group [81]. A double-blind placebo-controlled study in patients with OA of the knee demonstrated efficacy with a topical diclofenac patch in reducing pain [82].
In conclusion, the 2007 Cochrane Database review suggested that topical NSAIDs can provide good levels of pain relief without significant systemic adverse effects associated with oral NSAIDs, for the treatment of acute musculoskeletal pain [78].
Topical Opioids
Although a large variety of topical transdermal opioid preparations exist (many for the treatment of chronic cancer pain), because these are absorbed systemically (unlike topical opioid preparations that are not, or not designed to be, absorbed systemically), they will not be discussed here. Instead we focus solely on topical opioid preparations. A published case series reported the potential benefit of “topical” morphine in the management of chronic OA-related pain; however, the report emphasized that morphine and/or its metabolites were identifiable in the urine of treated patients, thus calling into question how truly “topical” this preparation was, given its systemic effects [83]. Preclinical studies indicate that opioids such as morphine, methadone, and buprenorphine can be analgesics when administered topically [84–86]. Morphine gel can be effective in relieving pain of skin ulcers and calciphylaxis in dialysis patients [87, 88]. A morphine mouthwash was shown to be safe and effective in decreasing the severity and duration of pain in patients with head and neck carcinomas receiving concomitant chemoradiotherapy [89]. Loperamide is a peripherally acting μ-opioid agonist. It is commercially available for the symptomatic management of diarrhea. It does not accumulate within the CNS and, when used topically, may have a LA effect [90]. Topical opiates have been reported to reduce pain for two children with epidermolysis bullosa [91].
Other Drug Options and Combinations
Topical clonidine 0.1% gel significantly decreases pain in patients with DPPN who had previously experienced burning pain from capsaicin applied to the affected skin, an interesting finding in light of the discussion in Sect. “Potential predictors of response to neuropathic pain treatments”, below [92]. It was found to be safe, with application site reactions only observed in the placebo group [92]. By contrast, intrathecal clonidine for the treatment of chronic pain typically results in hypotension [93]. Topical clonidine can also relieve hyperalgesia in patients suffering from sympathetically maintained pain [94].
Amitriptyline and doxepin are known to have multiple mechanisms, including local anesthetic properties, and these drugs may be analgesic when used topically. Notably, doxepin is currently available as a cream for the symptomatic treatment of pruritus [95]. The results of a novel study comparing the pain-reducing effect of a topical preparation containing 3.3% doxepin alone or a topical preparation containing 3.3% doxepin combined with 0.075% capsaicin to placebo in patients with a variety of chronic neuropathic pain problems indicated that each treatment provided similar pain-reducing effects and both were superior to placebo [95]. Although two clinical trials of amitriptyline, ketamine, and the combination failed to show efficacy in patients with neuropathic pain, these have been used empirically [96, 97]. Observations of effectiveness of topical amitriptyline alone or in combination with other drugs, such as ketamine, have been reported mostly in the management of painful disorders, for example, proctodynia [98]. Two open-label trials, the first involving 28 patients with neuropathic pain who were treated with the combination amitriptyline 2%/ketamine 1%, showed pain reduction and encouraging results [96, 99]. Amitriptyline and ketamine topical combinations have also been used in patients suffering from cancer pain, such as in a study showing that a topical amitriptyline, ketamine, and lidocaine preparation was safe and effective in alleviating neuropathic pain caused by radiation dermatitis [100].
Noncontrolled trials evaluating the use of topical ketamine, one in patients with PHN and one in patients with CRPS type 1 have each concluded that topical ketamine may be effective; however, serum ketamine levels were not measured in either study [101]. This would be instructive to know given the difference in side effects ketamine has been shown to have between topical and oral formulations. For example, topical ketamine at 50 mg/mL was shown to not elicit any local or systemic side effects [102], whereas intravenous ketamine for use in chronic non-cancer pain has been shown to involve a number of undesirable side effects that can limit its use, such as tachycardia, intracranial hypertension, vivid dreams and hallucinations, and the potential for urinary tract toxicity [103]. The results of an open-label, enriched, enrollment study followed by a randomization to placebo-controlled arms using either 4% amitriptyline/2% ketamine cream, 2% amitriptyline/1% ketamine cream, or placebo for patients with PHN demonstrated that higher concentrations of study drug yielded the lowest daily pain intensity compared with the lower concentration or placebo [104]. Plasma levels of either drug were detected in fewer than 10% of study patients. Topical ketamine has also been shown to be effective for mucositis pain in a patient with squamous carcinoma of the tongue undergoing radiation therapy [105].
Baclofen appears to have peripheral analgesic properties. Although a GABAergic agent orally, its hypothesized topical mechanism of action may be related to its K+ channel opening property [106]. Topical baclofen has been used empirically in combination with amitriptyline for vulvodynia [107], and has been studied as a single agent for chemotherapy-induced painful neuropathy [108]. Topical gabapentin has been empirically used as a single agent or in combination with amitriptyline and other drugs for neuropathic pain, with a retrospective study suggesting benefit of topical gabapentin for vulvodynia [109].
Botulinum toxin type A is an oral poison, which means that the molecule must have an efficient mechanism of endocytosis to penetrate and cross not only axon terminals but also epithelial cells. It is a zinc metalloendoprotease that cleaves the SNAP-25 protein required for synaptic vesicle fusion to release transmitters. It has been approved for the treatment of chronic migraine. When injected intradermally, a LA effect for peripheral neuropathic pain was reported in a small randomized clinical trial [110].
Dimethyl sulfoxide (DMSO) is a powerful antioxidant. Antioxidants can prevent or reduce reactive oxygen species-induced cell damage and other free radicals known to promote a neuroinflammatory cascade that, in some instances, may play a role in the genesis of chronic pain disorders, such as CRPS. When used at the concentration of 50% in a cream base, DSMO appears to be beneficial for CRPS [111–113]. Palmitoylethanolamide (PEA) is an endogenous lipid—specifically, a fatty acid ethanolamide. PEA is an agonist of the peroxisome proliferator-activated receptor-α and has been found to possess anti-inflammatory and anti-nociceptive effects [114]. In addition, PEA may inhibit the enzyme that catalyzes the endocannabinoid anandamide. PEA is available in Europe as an oral nutraceutical. Some dermatologic creams for atopic dermatitis contain PEA. It has empirically been used as a topical treatment for neuropathic pain [115]. There is one report that suggests that the topical application of geranium oil may provide temporary relief from PHN [116].
The analgesic effect of menthol, an ingredient common to many over-the-counter analgesic preparations, was hypothesized to be the result of activation of κ-opioid receptors [117]. Burn pain has been reported to be treated effectively with a topical loperamide preparation [118]. Two randomized controlled studies—one involving postoperative pain (diclofenac patch) and one involving wound pain treatment (capsicum plaster topically applied at acupuncture sites)—have been published as well [119, 120].
An open-label study using a 0.25% capsaicin topical agent in a lidocaine vehicle in 25 DPPN patients and seven PHN patients demonstrated pain relief in a majority of patients [121]. In a noncontrolled study of 23 patients with acute migraine headache, a topically applied 0.1% capsaicin gel was helpful in reducing mild or moderate pain [122]. In a randomized, double-blind study assessing 154 patients with chronic pain due to lateral epicondylitis, topical glyceryl trinitrate (0.72 mg/day), compared with placebo, was found to provide statistically significant greater pain relief after 8 weeks [123]. In a study of 52 patients with chronic pain due to a chronic Achilles tendinopathy, patients who had been treated with topical glyceryl trinitrate for 6 months were more likely 3 years after treatment ended to have less pain and more function than those who had been treated with placebo [124]. Published reports of the use of topical phenytoin in the treatment of pain due to superficial burns or chronic leg ulcers are noted as well [125, 126].
The use of topical “essential oxygen oil” was reviewed in a 2010 paper [127]. This essential oil is a hyperoxygenated, peroxidized, triglycerol-oxyester-rich oil. Although the published studies using this approach were open label or case reports and series, 100% of the patients felt that their pain was dramatically reduced.
A randomized controlled study of a topical cream containing glucosamine sulfate, chondroitin sulfate, and camphor for knee OA showed a significant pain reduction in the treatment group compared with the placebo group [128].
An interesting study using a homeopathic gel compared with piroxicam gel in 184 patients with OA found that “the homeopathic gel was at least as effective and as well tolerated as the NSAID gel” [129]. A Cochrane review from 2006 evaluating herbal therapies for low back pain found “three low quality trials on Capsicum Frutescens (Cayenne)” used as TAs, finding, “moderate evidence that Capsicum Frutescens produced more favorable results than placebo” [130].
Table 1 lists topical non-LAs.
Potential Predictors of Response to Neuropathic Pain Treatments
Neuropathic pain is accompanied by somatosensory signs, which include contributions from peripheral and CNS structures [131]. As has been discussed in previous sections of this two-part series, topical treatments are becoming an alternative to systemic treatment for peripheral neuropathic pain syndromes. However, as with most treatments for chronic pain, the response cannot be predicted. The variable responses seen in the previously discussed clinical trials of L5P and C8P illustrate the difficulty in predicting individual patient response. Obviously, if response could be predicted, patient care would be dramatically improved. Fortunately, there are data to suggest that responses using certain topical agents may be related to certain somatosensory patterns [47, 48, 132–135]. So far, in peripheral neuropathic pain, no predictors of treatment response have yet been identified. If certain sensory characteristics can be identified and associated with a specific treatment response, this could lead to improved treatment targeting.
Recent volunteer studies analyzed the analgesic efficacy of the L5P in two randomized, double-blind, placebo-controlled, crossover studies in healthy volunteers using a capsaicin and a sunburn pain model [136]. Lidocaine and placebo patches were simultaneously applied to forearms and thighs at contralateral body sites for three alternating 12-h “on/off” periods. Between the second and third “on” period, circular spots on both pretreated thighs were irradiated with ultraviolet B light to induce inflammatory skin hyperalgesia. In addition, on the forearms after the last “on” period, 20 µL of 0.1% capsaicin was injected intradermally to induce experimental pain and skin hyperalgesia. The area of pinprick hyperalgesia was diminished by 53% (P < 0.003) in the capsaicin model and by 84% (P < 0.0001) in the sunburn model; the intensity of mechanical hyperalgesia to rigid filaments (8–512 mN) was reduced in both models. The threshold of cold pain perception was reduced as well (19.7 °C ± 8.0 °C vs. 21.8 °C ± 6.8 °C for placebo, P < 0.05, sunburn). No effect was seen on capsaicin-induced spontaneous pain, flare size, blood flow in the sunburn area, or heat hyperalgesia in either study. These data suggest that relief of mechanical hyperalgesia is the predominant effect of the L5P.
A prospective, observational, open-label study was undertaken at two pain clinics in Austria to determine whether any patient characteristics, including Quantitative Sensory Testing (QST), can predict response to treatment with the C8P in patients suffering from peripheral neuropathic pain [137]. Patients assessed their pain at baseline and thereafter on days 1, 7, 28, and 84 posttreatment. QST was undertaken at the same time points on the painful area at the region of maximum peripheral neuropathic pain and on a control area. In addition, the size of the painful area was assessed at baseline and days 7, 28, and 84. Of the 57 patients, a total of 35.2% achieved a 30% or more reduction in the visual analog scale pain score at day 7 posttreatment compared with baseline; these were defined as ‘responders’. Responders were found to have had a significantly longer mean duration of peripheral neuropathic pain than nonresponders. Analysis of the QST data showed that the peripheral neuropathic pain area in responders, but not in nonresponders, had a significantly lower pressure pain threshold compared with the control area at baseline. Furthermore, nonresponders appeared to have significantly worse allodynia at baseline than responders, with an approximately three times greater degree of allodynia in nonresponders compared with responders across all tests.
Following treatment with the C8P, responders showed a significant reduction in allodynia and preexisting mechanical hypersensitivity and also appeared to show normalization of pinprick hyperalgesia at some stimulus levels. Responders to therapy had significantly greater reductions than nonresponders in the size of the painful area at days 28 and 84 following treatment. However, all patients, including the nonresponders, had meaningful reductions in the size of the painful area compared with baseline values [137]. It can be concluded that differences can be identified in the sensory profiles of patients with peripheral neuropathic pain who respond to the C8P and those who do not, specifically with regard to pressure pain threshold and degree of allodynia. Notably, both responders and nonresponders experienced meaningful reduction in the size of the painful area following treatment.
In summary, topical treatments that are actually available reduce spontaneous neuropathic pain and predominantly affect mechanical hyperalgesia, which may serve as a key marker to predict outcome. Clinical predictors such as pain intensity and the etiology or type of pain do not serve as predictors of treatment response. However, other characteristics that seem to suggest response, as discussed earlier, are intriguing and research continues to refine this diagnostic approach.
Conclusion
Of great interest is the recent observation that although a TA’s primary mechanism of action may be locally within the PNS, effects can be detected in the CNS using functional neuroimaging [138]. Although pain cannot be experienced without brain activation, a TA may interrupt pain transmission. Given these results and the previous discussions, TAs can have a significant role in patient care. Furthermore, empirically—although this needs more study—the use of two or more different compounds in a given topical gel or cream could target pain through different mechanisms. This concept of rational polypharmacy is used in the oral treatment of neuropathic pain. From the perspective of this review, this approach could involve “rational topical polypharmacy”, that is, an attempt to include two or more TAs to affect multiple and different pain mechanisms.
In conclusion, although TAs appear to be underused in clinical practice and have only a few options that are well studied, they are effective and may offer clinicians a number of distinct benefits compared with oral medications, including fewer side effects and no drug–drug interactions. Their use in neuropathic and other chronic pain syndromes may represent an important addition to the primary care physician’s and specialist’s armamentarium for patients of any pain level.
References
The Changing US Analgesia Market. 27 June 1994. Available from: http://www.thepharmaletter.com/file/55010/the-changing-us-analgesia-market.html. Accessed April 21, 2013.
Topical Analgesics. Pharmaceuticals: market research reports. Available from: http://www.marketresearch.com/Millennium-Research-Group-v648/Topical-Analgesics-6355096/. Accessed April 21, 2013. Available by purchase only.
Ness TJ, Jones L, Smith H. Use of compounded topical analgesics-Results of an internet survey. Reg Anes Pain Med. 2002;27:309–12.
Gabler NB, Duan N, Vohra S, Kravitz RL. N-of-1 trials in the medical literature: a systematic review. Med Care. 2011;49:761–8.
Bloms-Funke P. Sodium Channels. In: Buschmann H, Christoph T, Friderichs E, Maul C, Sundermann B, editors. Analgesics: from chemistry and pharmacology to clinical application. Weinheim: Wiley; 2002. p. 297–331.
Eijkelkanp N, Linley JE, Baker MD, et al. Neurological perspectives on voltage-gated sodium channels. Brain. 2012;135:2585–612.
Yoemans DC, Levinson SR, Peters MC, et al. Decrease in inflammatory hyperalgesia by herpes vector-mediated knockdown of Nav1.7 sodium channels in primary afferents. Hum Gene Ther. 2005;16:271–7.
Cummins TR, Sheets PL, Waxman SG. The roles of sodium channels in nociception: implications for mechanisms of pain. Pain. 2007;131:243–57.
Argoff CE. New analgesics for neuropathic pain: the lidocaine patch. Clin J Pain. 2000; 16:S62–S65.
Galer BS. Topical Medications. In: Loeser JD, editor. Bonica’s Management of Pain. Philadelphia: Lippincott-Williams and Wilkins; 2001. p. 1736–41.
Sawynok J. Topical analgesics in neuropathic pain. Curr Pharm Des. 2005;11(23):2995–3004.
Attal N, Crucci G, Haanpaa M, et al. EFNS guidelines on pharmacological treatment of neuropathic pain. Eur J Neurol. 2006;13:1153–69.
Rowbotham MC. Pharmacologic management of complex regional pain syndrome. Clin J Pain. 2006;22:425–9.
Gammaitoni AR, Alvarez NA. 24-h application of the lidocaine patch 5% for 3 consecutive days is safe and well tolerated in healthy adult men and women. Abstract PO6.20. Presented at the 54th Annual American Academy of Neurology Meeting, April 13–20, 2002, Denver.
Argoff C, Nicholson B, Moskowitz M, et al. Effectiveness of lidocaine patch 5% (Lidoderm®) in the treatment of low back pain. Presented at the 10th World Congress on Pain, August 17–22, 2002, San Diego.
Haroutiunian S, Drennan DA, Lipman AG. Topical NSAID therapy for musculoskeletal pain. Pain Med. 2010;11:535–49.
Meier T, Wasner G, Faust M, et al. Efficacy of lidocaine patch 5% in the treatment of focal peripheral neuropathic pain syndromes: a randomized, double-blind, placebo-controlled study. Pain. 2003;106:151–8.
Devers A, Galer BS. Topical lidocaine patch relieves a variety of neuropathic pain conditions: an open-label study. Clin J Pain. 2000;16:205–8.
Galer BS, Jensen MP. Development and preliminary validation of a pain measure specific to neuropathic pain: the Neuropathic Pain Scale. Neurology. 1997;48:332–8.
Herrmann DN, Barbano RL, Hart-Gouleau S, Pennella-Vaughan J, Dworkin RH. An open-label study of the lidocaine patch 5% in painful polyneuropathy. Pain Med. 2005;6:379–84.
Katz NP, Davis MW, Dworkin RH. Topical lidocaine patch produces a significant improvement in mean pain scores and pain relief in treated PHN patients: results of a multicenter open-label trial. J Pain. 2001;2:9–18.
Rehm S, Binder A, Baron R. Post-herpetic neuralgia: 5% lidocaine medicated plaster, pregabalin or a combination of both? A randomized, open clinical effectiveness study. Curr Med Res Opin. 2010;26:1607–19.
Galer BS, Jensen MP, Ma T, Davies PS, Rowbotham MC. The lidocaine patch 5% effectively treats all Neuropathic Pain Qualities: results of a randomized, double-blind, vehicle-controlled, 3-week efficacy study with use of the Neuropathic Pain Scale. Clin J Pain. 2002;18:297–301.
Geha PY, Baliki MN, Chialvo DR, Harden RN, Paice JA, Apkarian AV. Brain activity for spontaneous pain of post herpetic neuralgia and its modulation by lidocaine patch therapy. Pain. 2007;128:88–100.
Sandroni P. Central neuropathic itch: a new treatment option? Neurology. 2002;59:778–9.
López Ramírez E. Treatment of acute and chronic focal neuropathic pain in cancer patients with lidocaine 5% patches. A radiation and oncology department experience. Support Care Cancer. 2013;21:1329–34.
Lycka BA, Watson CP, Nevin K, et al. EMLA® cream for the treatment of pain caused by post-herpetic neuralgia: a double-blind, placebo-controlled study. In: Proceedings of the Annual Meeting of the American Pain Society 1996;A111(abstract).
Attal N, Brasseur L, Chauvin M, Bouhassira D. Effects of single and repeated applications of a eutectic mixture of local anesthetics (EMLA®) cream on spontaneous and evoked pain in post-herpetic neuralgia. Pain. 1999;81:203–9.
Litman SJ, Vitkun SA, Poppers PJ. Use of EMLA® cream in the treatment of post-herpetic neuralgia. J Clin Anesth. 1996;8:54–7.
Fassoulaki A, Sarantopoulos C, Melemeni A, Hogan Q. EMLA reduces acute and chronic pain after breast surgery for cancer. Reg Anesth Pain Med. 2000;25:350–5.
Bode AM, Dong Z. The two faces of capsaicin. Cancer Res. 2011;71:2809–14.
Pereira J. Capsicum annuum. In: Carson J, editor. The elements of materia medica and therapeutics. 3rd ed. Philadelphia: Blanchard and Lea; 1854. p. 505–7.
Thresh JC. Isolation of capsaicin. Pharmaceut J Trans. 1876;6:941–7.
Watson CPN. Topical capsaicin as an adjuvant analgesic. J Pain Symptom Manag. 1994;9:425–33.
Robbins W. Clinical applications of capsaicinoids. Clin J Pain. 2000;16:S86–S89.
Backonja M, Malan P, Brady S. One-hour high concentration trans-capsaicin applications provide durable pain relief in initial and repeat treatment of postherpetic neuralgia. Presented at the 2004 Annual Scientific Meeting of the American Academy of Neurology, San Francisco.
Bley KR. Recent developments in transient receptor potential vanilloid receptor 1 agonist-based therapies. Expert Opin Investig Drugs. 2004;13:1445–56.
Knotkova H, Pappagallo M, Szallasi A. Capsaicin (TRPV1 agonist) therapy for pain relief: farewell or revival? Clin J Pain. 2008;24:142–54.
Kennedy WR, Vanhove GF, Lu SP, et al. A randomized, controlled, open-label study of the long-term effects of NGX-4010, a high-concentration capsaicin patch, on epidermal nerve fiber density and sensory function in healthy volunteers. J Pain. 2010;11:579–87.
Solcsanyi J. Turning up the heat on pain. TRPV1 receptors in pain and inflammation. In: Malmberg AB, Bley KR, eds. Birkhauser: Basel Switzerland; 2005.
ClinicalTrials.gov. A study to investigate the tolerability and effects on epidermal nerve fiber density of multiple low-concentrations of NGX-1998 in healthy volunteers. Available from: http://www.clinicaltrials.gov/ct2/show/NCT00912262?term=capsaicin+and+liquid&rank=1. Accessed April 20, 2013.
ClinicalTrials.gov. ALGRX 4975 in the treatment of patients with Morton’s neuroma. http://www.clinicaltrials.gov/ct2/show/NCT00130962?term=capsaicin+and+mortons+neuroma&rank=1. NCT00130962. Accessed April 20, 2013.
Rains C, Bryson HM. Topical capsaicin: a review of its pharmacological properties and therapeutic potential in post-herpetic neuralgia, diabetic neuropathy, and osteoarthritis. Drugs Aging. 1995;7:317–28.
Winocur E, Gavish A, Halachmi M, Eli I, Gazit E. Topical application of capsaicin for the treatment of localized pain in the temporomandibular joint area. J Orofac Pain. 2000;14:31–6.
Mason L, Moore RA, Derry S, Edwards JE, McQuay HJ. Systematic review of topical capsaicin for the treatment of chronic pain. BMJ. 2004;24(328):991–6.
Robbins WR, Staats PS, Levine J, et al. Treatment of intractable pain with topical large-dose capsaicin: preliminary report. Anesth Analg. 1998;86:579–83.
Backonja MM, Malan TP, Vanhove GF. Tobias JK; C102/106 Study Group. NGX-4010, a high concentration patch, for the treatment of post herpetic neuralgia: a randomized, double-blind, controlled study with an open-label extension. Pain Med. 2010;11:600–8.
Backonja M, Wallace MS, Blonsky ER, et al. NGX-4010, a high-concentration capsaicin patch, for the treatment of post herpetic neuralgia: a randomised, double-blind study. Lancet Neurol. 2008;7:1106–12.
Wallace M, Pappagallo M. Qutenza®: a capsaicin 8% patch for the management of post herpetic neuralgia. Expert Rev Neurother. 2011;11:15–27.
Simpson DM, Brown S, Tobias J, NGX-4010 C107 Study Group. Controlled trial of high-concentration capsaicin patch for treatment of painful HIV neuropathy. Neurol. 2008;70:2305–13.
Anand P, Bley K. Topical capsaicin for pain management: therapeutic potential and mechanisms of action of the new high-concentration capsaicin 8% patch. Br J Anaesth. 2011;107:490–502.
Derry S, Sven-Rice A, Cole P, Tan T, Moore RA. Topical capsaicin (high concentration) for chronic neuropathic pain in adults. Cochrane Database Syst Rev. 2013. doi:10.1002/14651858.CD007393.pub3.
Keitel W, Frerick H, Kuhn U, Schmidt U, Kuhlmann M, Bredehorst A. Capsicum pain plaster in chronic non-specific low back pain. Arzneimittelforschung. 2001;51:896–903.
Ortiz MI, Torres-López JE, Castañeda-Hernández G, Rosas R, Vidal-Cantú GC, Granados-Soto V. Pharmacological evidence for the activation of K+ channels by diclofenac. Eur J Pharmacol. 2002;438:85–91.
Cashman JN. The mechanism of action of NSAIDs in analgesia. Drugs. 1996;52(Suppl 5):13–23.
Barthel HR, Axford-Gatley RA. Topical non-steroidal anti-inflammatory drugs for osteoarthritis. Postgrad Med. 2010;122:98–106.
Mazieres B, Rouanet S, Velicy J, Scarsi C, Reiner V. Topical ketoprofen patch (100 mg) for the treatment of ankle sprain: a randomized, double-blind, placebo-controlled study. Am J Sports Med. 2005;33:515–23.
Mazieres B, Rouanet S, Guillon Y, Scarsi C, Reiner V. Topical ketoprofen patch in the treatment of tendonitis: a randomized, double blind, placebo controlled study. J Rheumatol. 2005;32:1563–70.
De Benedittis G, Lorenzetti A. Topical aspirin/diethyl ether mixture versus indomethacin and diclofenac/diethyl ether mixtures for acute herpetic neuralgia and postherpetic neuralgia: a double-blind crossover placebo-controlled study. Pain. 1996;65:45–51.
Brewer AR, McCarberg B, Argoff CE. Update on the use of topical NSAIDs for the treatment of soft tissue and musculoskeletal pain: a review of recent data and current treatment options. Phys Sports Med. 2010;38:62–70.
Stanos SP. Topical agents for the management of musculoskeletal pain. J Pain Symp Manag. 2007;33:342–55.
Baraf HS, Gloth FM, Barthel HR, Gold MS, Altman RD. Safety and efficacy of topical diclofenac sodium gel for knee osteoarthritis in elderly and younger patients; pooled data from three randomized, double-blind, parallel-group, placebo-controlled, multicenter trials. Drugs Aging. 2011;28:27–40.
Bruhlmann P, Michel BA. Topical diclofenac patch in patients with knee osteoarthritis: a randomized, double-blind, controlled clinical trial. Clin Exp Rheumatol. 2003;21:193–8.
Tugwell PS, Wells GA, Shainhouse JZ. Equivalence study of a topical diclofenac solution (Pennsaid) compared with oral diclofenac in symptomatic treatment of osteoarthritis of the knee: a randomized, controlled trial. J Rheumtol. 2004;31:2002–12.
Shainhouse JZ, Grierson LM, Naseer Z. A long-term open label study to confirm the safety of topical diclofenac solution containing dimethyl sulfoxide in the treatment of the osteoarthritic knee. Am J Ther. 2010;17:566–76.
Roth SH, Fuller P. Diclofenac topical solution compared with oral diclofenac: a pooled safety analysis. J Pain Res. 2011;4:159–67.
Di Rienzo BL, Di Rienzo Businco A, D’Emilia M, Lauriello M, Coen Tirelli G. Topical versus systemic diclofenac in the treatment of temporo-mandibular joint dysfunction symptoms. Acta Otorhinolaryngol Ital. 2004;24:279–83.
Niethard FU, Gold MS, Solomon GS, et al. Efficacy of topical diclofenac diethylamine gel in osteoarthritis of the knee. J Rheumatol. 2005;32:2384–92.
Baer PA, Thomas LM, Shainhouse Z. Treatment of osteoarthritis of the knee with a topical diclofenac solution: a randomized, controlled, 6 week trial (ISRCTN53366886). BMC Musculoskelet Disord. 2005;6:44.
Lin J, Zhang W, Jones A, Doherty M. Efficacy of topical non-steroidal anti-inflammatory drugs in the treatment of osteoarthritis: meta-analysis of randomized controlled trials. BMJ. 2004;329:324–8.
Mason L, Moore RA, Edwards JE, Derry S, McQuay HJ. Topical NSAIDS for chronic musculoskeletal pain: a systematic review and meta-analysis. BMC Musculoskelet Disord. 2004;5:28.
Myrer JW, Johnson AW, Mitchell UH, Measom GJ, Fellingham GW. Topical analgesic added to paraffin enhances paraffin bath treatment of individuals with hand osteoarthritis. Disab Rehab. 2011;33:467–74.
Predel HG, Koll R, Pabst H, et al. Diclofenac patch for topical treatment of acute impact injuries: a randomized, double blind, placebo controlled, multicenter study. Br J Sports Med. 2004;38:318–23.
Galer BS, Rowbotham M, Perander J, Devers A, Friedman E. Topical diclofenac patch significantly reduces pain associated with minor sports injuries: results of a randomized, double-blind, placebo-controlled, multicenter study. J Pain Symptom Manag. 2000;19:287–94.
Jenoure P, Segesser B, Lühti U, Gremion G. A trial with diclofenac HEP plaster as topical treatment in minor sports injuries. Drugs Exp Clin Res. 1993;19:125–31.
Machen J, Whitefield M. Efficacy of a proprietary ibuprofen gel in soft tissue injuries: a randomized, double-blind, placebo-controlled study. Intl J Clin Prac. 2002;56:102–6.
Whitefield M, O’Kane CJ, Anderson S. Comparative efficacy of a proprietary topical ibuprofen gel and oral ibuprofen in acute soft tissue injuries: a randomized, double-blind study. J Clin Pharm Ther. 2002;27:409–17.
Massey T, Derry S, Moore RA, McQuay HJ. Topical NSAIDs for acute pain in adults. Cochrane Database Syst Rev. 2010. doi:10.1002/14651858.CD007402.pub2.
Marten M. Efficacy and tolerability of a topical NSAID patch (local action transcutaneous flurbiprofen) and oral diclofenac in the treatment of soft-tissue rheumatism. Clin Rheumatol. 1997;16:25–31.
Matthews P, Derry S, Moore RA, McQuay HJ. Topical rubefacients for acute and chronic pain in adults. Cochrane Database Syst Rev. 2009;8:CD007403.
Sandelin J, Harilainen A, Crone H, Hamberg P, Forsskåhl B, Tamelander G. Local NSAID gel (eltenac) in the treatment of osteoarthritis of the knee. A double-blind study comparing eltenac with oral diclofenac and placebo gel. Scand J Rheumatol. 1997;26:287–92.
Dreiser RL, Tisne-Camus M. DHEP plasters as a topical treatment of knee osteoarthritis: a double-blind placebo-controlled study. Drugs Exp Clin Res. 1993;19:107–15.
Wilken M, Ineck JR, Rule AM. Chronic arthritis pain management with topical morphine:case series. J Pain Palliat Care Pharmacother. 2005;19:39–44.
Kolesnikov Y, Jain S, Wilson R, Pasternak GW. Peripheral kappa 1-opioid receptor-mediated analgesia in mice. Eur J Pharmacol. 1996;310:141–3.
Kolesnikov Y, Pasternak GW. Topical opioids in mice: analgesia and reversal of tolerance by a topical N-methyl-d-aspartate antagonist. J Pharmacol Exp Ther. 1999;290:247–52.
He L, Kim J, Ou C, McFadden W, van Rijn RM, Whistler JL. Methadone antinociception is dependent on peripheral opioid receptors. J Pain. 2009;10:369–79.
Zeppetella G, Paul J, Ribeiro MD. Analgesic efficacy of morphine applied topically to painful ulcers. J Pain Symptom Manag. 2003;25:555–8.
Bastami S, Frödin T, Ahlner J, Uppugunduri S. Topical morphine gel in the treatment of painful leg ulcers, a double-blind, placebo-controlled clinical trial: a pilot study. Int Wound J. 2012;9:419–27.
Cerchietti LC, Navigante AH, Bonomi MR, et al. Effect of topical morphine for mucositis-associated pain following concomitant chemoradiotherapy for head and neck carcinoma. Cancer. 2002;95:2230–6.
Nozaki-Taguchi N, Yaksh TL. Characterization of the antihyperalgesic action of a novel peripheral mu-opioid receptor agonist–loperamide. Anesthesiology. 1999;90:225–34.
Watterson G, Howard R, Goldman A. Peripheral opiates in inflammatory pain. Arch Dis Child. 2004;89:679–81.
Campbell CM, Kipnes MS, Stouch BC, et al. Randomized control trial of topical clonidine for treatment of painful diabetic neuropathy. Pain. 2012;153:1815–23.
Ackerman LL, Follett KA, Rosenquist RW. Long-term outcomes during treatment of chronic pain with intrathecal clonidine or clonidine/opioid combinations. J Pain Symptom Manag. 2003;26:668–77.
Davis KD, Treede RD, Raja SN, Meyer RA, Campbell JN. Topical application of clonidine relieves hyperalgesia in patients with sympathetically maintained pain. Pain. 1991;47:309–17.
McCleane G. Topical application of doxepin hydrochloride, capsaicin and a combination of both produces analgesia in chronic human neuropathic pain: a randomized, double-blind, placebo-controlled study. Br J Clin Pharmacol. 2000;49:574–9.
Lynch ME, Clark AJ, Sawynok J. A pilot study examining topical amitriptyline, ketamine, and a combination of both in the treatment of neuropathic pain. Clin J Pain. 2003;19:323–8.
Ho KY, Huh BK, White WD, Yeh CC, Miller EJ. Topical amitriptyline versus lidocaine in the treatment of neuropathic pain. Clin J Pain. 2008;24:51–5.
Lehman JS, Sciallis GF. Effective use of topical amitriptyline hydrochloride 2.5% and ketamine hydrochloride 0.5% for analgesia in refractory proctodynia. J Drugs Dermatol. 2008;7:887–9.
Lynch ME, Clark AJ, Sawynok J, Sullivan MJ. Topical amitriptyline and ketamine in neuropathic pain syndromes: an open-label study. J Pain. 2005;6:644–9.
Uzaraga I, Gerbis B, Holwerda E, Gillis D, Wai E. Amitriptyline, ketamine, and lidocaine in neuropathic pain caused by radiation skin reaction: a pilot study. Support Care Cancer. 2011;20:1515–24.
Quan D, Wellish M, Gilden DH. Topical ketamine treatment of postherpetic neuralgia. Neurology. 2003;60:1391–2.
Pöyhiä R, Vainio A. Topically administered ketamine reduces capsaicin-evoked mechanical hyperalgesia. Clin J Pain. 2006;22:32–6.
Quibell R, Prommer EE, Mihalyo M, Twycross R, Wilcock A. Ketamine. J Pain Symptom Manage. 2011;41:640–9.
Bernstein J, Phillips S, Group T. A new topical medication for the adjunctive relief of painful diabetic neuropathy and postherpetic neuralgia. J Pain. 2004;5(3 Suppl 1):82.
Slatkin NE, Rhiner M. Topical ketamine in the treatment of mucositis pain. Pain Med. 2003;4:298–303.
Reis GML, Duarte IDG. Baclofen, an agonist at peripheral GABAB receptors, induces antinociception via activation of TEA-sensitive potassium channels. Br J Pharmacol. 2006;149:733–9.
Nyirjesy P, Ahinoam LS, Leny M, Culhane JF. Topical amitriptyline-baclofen cream for the treatment of provoked vestibulodynia. J Low Gen Tract Dis. 2009;13:230–6.
Barton DL, Wos EJ, Qin R, et al. A double-blind, placebo-controlled trial of a topical treatment for chemotherapy-induced peripheral neuropathy: NCCTG trial N06CA. Support Care Cancer. 2011;19:833–41.
Boardman LA, Cooper AS, Blais LR, Raker CA. Topical gabapentin in the treatment of localized and generalized vulvodynia. Obstet Gynecol. 2008;112:579–85.
Ranoux D, Attal N, Morain F, Bouhassira D. Botulinum toxin type A induces direct analgesic effects in chronic neuropathic pain. Ann Neurol. 2008;64:274–83.
Perez RS, Zuurmond WW, Bezemer PD, et al. The treatment of complex regional pain syndrome type I with free radical scavengers: a randomized controlled study. Pain. 2003;102:297–307.
Zuurmond WW, Langendijk PN, Bezemer PD, Brink HE, de Lange JJ, van loenen AC. Treatment of acute reflex sympathetic dystrophy with DMSO 50% in a fatty cream. Acta Anaesthesiol Scand. 1996;40:364–7.
Gaspar M, Bovaira M, Carrera-Hueso FJ, Querol M, Jiménez A, Moreno L. Efficacy of a topical treatment protocol with dimethyl sulfoxide 50% in type 1 complex regional pain syndrome. Farm Hosp. 2012;36:385–91.
Costa B, Comelli F, Bettoni I, Colleoni M, Giagnoni G. The endogenous fatty acid amide, palmitoylethanolamide, has anti-allodynic and anti-hyperalgesic effects in a murine model of neuropathic pain: involvement of CB(1), TRPV1 and PPARgamma receptors and neurotrophic factors. Pain. 2008;139:541–50.
Phan NQ, Siepmann D, Gralow I, Ständer S. Adjuvant topical therapy with a cannabinoid receptor agonist in facial postherpetic neuralgia. J Dtsch Dermatol Ges. 2010;8:88–91.
Greenway FL, Frome BM, Engels TM 3rd, McLellan A. Temporary relief of postherpetic neuralgia pain with topical geranium oil. Am J Med. 2003;115:586–7.
Galeotti N, Di Cesare Mannelli L, Mazzanti G, Bartolini A, Ghelardini C. Menthol: a natural analgesic compound. Neurosci Lett. 2002;322:145–8.
Ray SB. Loperamide: a potential topical analgesic for the treatment of burn pain. J Burn Care Res. 2006;27:121–2.
Alessandri F, Lijoi D, Mistrangelo E, Nicoletti A, Crosa M, Ragni N. Topical diclofenac patch for postoperative wound pain in laparoscopic gynecologic surgery: a randomized study. J Minim Invasive Gynecol. 2006;13:195–200.
Kim KS, Nam YM. The analgesic effects of capsicum plaster at the Zusanli point after abdominal hysterectomy. Anesth Analg. 2006;103:709–13.
Kolesnikov YA, Chereshnev I, Pasternak GW. Analgesic synergy between topical lidocaine and topical opioids. J Pharmacol Exp Ther. 2000;295:546–51.
Cianchetti C. Capsaicin jelly against migraine pain. Int J Clin Pract. 2010;64:457–9.
Paoloni JA, Murrell GA, Burch RM, Ang RY. Randomised, double-blind, placebo-controlled clinical trial of a new topical glyceryl trinitrate patch for chronic lateral epicondylosis. Br J Sports Med. 2009;43:299–302.
Paoloni JA, Murrell GA. Three-year followup study of topical glyceryl trinitrate treatment of chronic noninsertional Achilles tendinopathy. Foot Ankle Int. 2007;28:1064–8.
Carneiro PM, Rwanyuma LR, Mkony CA. A comparison of topical phenytoin with silverex in the treatment of superficial dermal burn wounds. Cent Afr J Med. 2002;48:105–8.
Carneiro PM, Nyawawa ET. Topical phenytoin versus EUSOL in the treatment of non-malignant chronic leg ulcers. East Afr Med J. 2003;80:124–9.
Pergolizzi JV, Pappagallo M, Raffa RB. Preliminary observations of a novel topical oil with analgesic properties for treatment of acute and chronic pain syndromes. Pain Pract. 2010;10:201–13.
Cohen M, Wolfe R, Mai T, Lewis D. A randomized, double blind placebo-controlled trial of a topical crème containing glucosamine sulfate, chondroitin sulfate and camphor for osteoarthritis of the knee. J Rheumatol. 2003;30:523–8.
Van Haselen RA, Fisher PAG. A randomized controlled trial comparing topical piroxicam gel with a homeopathic gel in osteoarthritis of the knee. Rheumatology. 2000;39:714–9.
Gagnier JJ, van Tulder M, Berman B, Bombardier C. Herbal medicine for low back pain. Cochrane Database Syst Rev. 2006;9(2):CD004504.
Maier C, Baron R, Tölle TR, et al. Quantitative sensory testing in the German Research Network on Neuropathic Pain (DFNS): somatosensory abnormalities in 1236 patients with different neuropathic pain syndromes. Pain. 2010;150:439–50.
Wasner G, Kleinert A, Binder A, Schattschneider J, Baron R. Postherpetic neuralgia: topical lidocaine is effective in nociceptor-deprived skin. J Neurol. 2005;252:677–86.
Barbano RL, Herrmann DN, Hart-Gouleau S, Pennella-Vaughan J, Lodewick PA, Dworkin RH. Effectiveness, tolerability, and impact on quality of life of the 5% lidocaine patch in diabetic polyneuropathy. Arch Neurol. 2004;61:914–8.
Finnerup NB, Sindrup SH, Jensen TS. The evidence for pharmacological treatment of neuropathic pain. Pain. 2010;150:573–81.
Baron R, Binder A, Wasner G. Neuropathic pain: diagnosis, pathophysiological mechanisms, and treatment. Lancet Neurol. 2010;9:807–19.
Gustorff B, Hauer D, Thaler J, Seis A, Draxler J. Antihyperalgesic efficacy of 5% lidocaine medicated plaster in capsaicin and sunburn pain models—two randomized, double-blinded, placebo-controlled crossover trials in healthy volunteers. Exp Opin Pharmacother. 2011;12:2781–90.
World Congress of Pain, Milano 2012 Poster PH 113: Gustorff et al. Treatment of peripheral neuropathic pain with the capsaicin 8% patch: is quantitative sensory testing a useful tool to predict response of treatment? 2013.
Abdulredaa MH, Faleoa G, Molano RD, et al. High-resolution, noninvasive longitudinal live imaging of immune responses. PNAS. 2011;108:12863–8.
Acknowledgments
All named authors meet the ICMJE criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval to the version to be published. This work did not receive funding from any source. The authors thank Steven Tresker and Dr. Smitha Mathews of Cactus Communications for editing assistance. Funding for the editing assistance was provided by Mallinckrodt Pharmaceuticals. The authors received no remuneration for this work and the paper was written before Dr. Peppin entered the employ of Mallinckrodt. Dr. Peppin was not with industry when this article was written and accepted.
Conflict of interest
J. Peppin holds consulting and advising roles with AIT Laboratories, Ameritox, Endo Pharmaceuticals, INSYS Therapeutics, Salix Pharmaceuticals, and Zogenix Pharmaceuticals. P.J. Albrecht has received investigator-initiated grants from Endo Pharmaceuticals, Forest Laboratories, and Eli Lilly, and has ownership equity in Integrated Tissue Dynamics, LLC. B. Gustorff received unrestricted commercial research grants from Astellas Pharma Europe Ltd. and Grünenthal, Germany, and is a consultant/advisor for Grünenthal, Pfizer, Mundipharma, Janssen-Cilag, Bayer, Astellas Pharma Europe Ltd., and Newron. F.L. Rice has received investigator-initiated grants from Endo Pharmaceuticals, Forest Laboratories, and Eli Lilly. C. Argoff, M. Pappagallo, and M. Wallace declare they have no potential conflicts of interest.
Compliance with Ethics Guidelines
This review article is based on previously conducted studies, and does not involve any new studies of human or animal subjects performed by any of the authors.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Peppin, J.F., Albrecht, P.J., Argoff, C. et al. Skin Matters: A Review of Topical Treatments for Chronic Pain. Part Two: Treatments and Applications. Pain Ther 4, 33–50 (2015). https://doi.org/10.1007/s40122-015-0032-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40122-015-0032-z